Abstract
Background
Musculoskeletal conditions are the leading cause of disability worldwide. Telerehabilitation may be a viable option in the management of these conditions, facilitating access and patient adherence. Nevertheless, the impact of biofeedback-assisted asynchronous telerehabilitation remains unknown.
Objective
To systematically review and assess the effectiveness of exercise-based asynchronous biofeedback-assisted telerehabilitation on pain and function in individuals with musculoskeletal conditions.
Methods
This systematic review followed Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines. The search was conducted using three databases: PubMed, Scopus, and PEDro. Study criteria included articles written in English and published from January 2017 to August 2022, reporting interventional trials evaluating exercise-based asynchronous telerehabilitation using biofeedback in adults with musculoskeletal disorders. The risks of bias and certainty of evidence were appraised using the Cochrane tool and Grading of Recommendations, Assessment, Development, and Evaluation (GRADE), respectively. The results are narratively summarized, and the effect sizes of the main outcomes were calculated.
Results
Fourteen trials were included: 10 using motion tracker technology (N = 1284) and four with camera-based biofeedback (N = 467). Telerehabilitation with motion trackers yields at least similar improvements in pain and function in people with musculoskeletal conditions (effect sizes: 0.19–1.45; low certainty of evidence). Uncertain evidence exists for the effectiveness of camera-based telerehabilitation (effect sizes: 0.11–0.13; very low evidence). No study found superior results in a control group.
Conclusions
Asynchronous telerehabilitation may be an option in the management of musculoskeletal conditions. Considering its potential for scalability and access democratization, additional high-quality research is needed to address long-term outcomes, comparativeness, and cost-effectiveness and identify treatment responders.
Introduction
Musculoskeletal (MSK) conditions are characterized by “impairments in the muscles, bones, joints, and adjacent connective tissues leading to temporary or lifelong limitations in functioning and participation,” 1 being the number one cause of disability worldwide 1 (nearly 150 million years lived with disability). 2 Although this concept encompasses several diagnosis, the present work will focus on conditions with non-rheumatoid, non-metabolic, and non-autoimmune etiology. These conditions can produce significant limitations in mobility and functionality, compromising the ability to perform daily activities and work productivity. This, compounding to the frequently associated mental health distress, contributes to the reduced reported quality of life. The subsequent economic expenditure driven by direct healthcare-related and indirect costs exceeds those for heart disease and cancer, 3 imposing a tremendous societal impact.
Adequate treatments may substantially reduce this burden, with exercise and behavior-change strategies being widely recommended as first-line interventions in the management of these conditions.4–7 Access to conservative care, namely, physical therapy, is frequently hampered by numerous barriers including geographic and travel constraints, lack of clinicians and healthcare facilities, and high costs associated with in-person care.8,9
Telerehabilitation, a subset of telemedicine, arose as an attempt to overcome these challenges and aims to facilitate access and improve adherence to treatment. 10 The recent COVID-19 pandemic highlighted the potential of telemedicine to ensure continued care delivery, in a situation where access to in-person care was severely limited.11,12 The Centers for Disease Control and Prevention reported a 50% increase in telehealth visits from January to March 2020 compared with the same period in 2019, with an astounding 154% increase from March 23 to March 28. 13
Previous systematic reviews assessed the effectiveness of telerehabilitation in patients with MSK conditions, addressing specifically pain and function improvement.14–16 Cottrell et al. 14 conducted a systematic review with meta-analysis and concluded that telerehabilitation is as effective and comparable to conventional care for both function and pain in a variety of MSK conditions (including shoulder, hip, and knee arthroplasties, low back pain, neck pain, and osteoarthritis). Similar findings were reported in two other systematic reviews supporting its use for non-acute 16 and chronic 15 MSK conditions. However, these systematic reviews considered only synchronous interventions (where patients are accompanied by therapists in real time through video conferencing or telephone) including those in a hybrid format (i.e., telerehabilitation combined with in-person care) and excluded asynchronous interventions (where sessions displayed in a digital format are performed independently by the patient). Synchronous telerehabilitation may pose some limitations, particularly regarding the scalability of treatment and scheduling constraints, which has generated interest in asynchronous telerehabilitation. 17 Gava et al. 18 conducted a systematic review focused on telerehabilitation in participants with shoulder pain, gathering evidence from six randomized controlled trials (RCTs) (1 synchronous + 5 asynchronous). The authors reported low to very low certainty of evidence supporting the use of telerehabilitation to improve pain and disability. 18
Recently, the development of innovative technologies has allowed the integration of important features to enable and optimize asynchronous care delivery. 12 Motion tracking systems have now been integrated into telerehabilitation interventions to provide real-time biofeedback during exercise. These encompass diverse technologies, such as wearable sensors with inertial measurement units, built-in smartphone sensors, and camera-based sensors. Such technologies guide patients during sessions and promote close remote monitoring on patient progress, thereby permitting individualized support and reinforcing accountability.19,20 Additionally, movement digitalization with respective data storage in web platforms may assist therapists in patient monitoring and intervention data-driven adjustments.
Despite advances in biofeedback technologies and the clinical applicability of such strategies, no systematic review has assessed the evidence on asynchronous telerehabilitation with biofeedback, particularly on stand-alone MSK telerehabilitation (i.e., without in-person sessions).
The aim of this systematic review is to summarize the evidence and assess the effectiveness of exercise-based asynchronous telerehabilitation incorporating biofeedback systems on pain and function in patients with MSK conditions, focusing on the last 5 years. The secondary objectives are to assess patient adherence and satisfaction with such programs.
Methods
Study design
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines 21 and the Cochrane Handbook for Systematic Reviews 22 (PRISMA checklist is provided in Appendix 1). Although the protocol of this systematic review was not registered, the review methodology was established prior to the conduct of the review and was not modified post hoc.
Search strategy
A systematic literature search was performed on PubMed, Scopus, and PEDro on published articles. A primary search was conducted on 27 April 2022, and subsequent searches were conducted on 29 April 2022 (Scopus), 2 May 2022 (PubMed), and 24 August 2022 (PEDro) to ensure a thorough appraisal and selection of relevant literature. In order to select studies with the most recent and innovative technological biofeedback solutions for MSK telerehabilitation, only articles published in the previous 5 years were considered (filter used: publication date after 1 January 2016). This time frame was chosen because of the dramatic changes in this technology that have occurred in the past several years; a subsequent search revealed only a single article published earlier. 23 The search strategy for each database consisted of free text words and Medical Subject Headings (MeSH) as reported in Appendix 2. All keywords were searched independently and then combined using relevant Boolean terms. Additionally, the reference list of included articles and of relevant previous systematic reviews were manually searched to guarantee that all relevant literature was included.
Selection criteria
The study selection criteria were defined based on the following PICOS:
Participants: Adult patients (>18 years old) with MSK-related conditions, defined as those causing MSK-related pain or disability in either acute or chronic stages. Studies including pregnant patients, as well as those comprising conditions related to metabolic diseases (e.g., osteoporosis and diabetes), neurologic disorders (e.g., post-stroke), chronic widespread pain (e.g., fibromyalgia), cancer, and autoimmune causes including inflammatory arthropathies (e.g., rheumatoid arthritis) were excluded. Intervention: Exercise-based asynchronous telerehabilitation, defined by remote interventions where exercise sessions were performed independently by the patient, delivered via telecommunication technologies incorporating biofeedback systems for MSK care, with a minimum treatment time of 4 weeks (considered a sufficient time frame to obtain consistent results on the defined outcomes24,25). Hybrid modalities (i.e., combining telerehabilitation with in-clinic treatment) were excluded. Comparison: Control groups from eligible studies should include one of the following: placebo, standard care, no treatment (waiting list), and other active treatments (conservative care, in solo or with adjunctive telerehabilitation). Studies without control groups (i.e., single-arm interventional) were eligible. Outcome: Pain intensity and self-reported function. Secondary outcomes were patient adherence and satisfaction. Setting/study design: Home-based/outpatients (any country, but had to be written in English). Eligible study designs included controlled trials (randomized and non-randomized), before-after trials, and interventional single-arm longitudinal studies. Clinical trials without a control group were included due to the expected limited body of evidence to gather insights on studied interventions details, feasibility, patient's acceptability and engagement, and preliminary observed results.
Inclusion and exclusion criteria are summarized in Table 1.
Inclusion and exclusion criteria.

Abbreviation: MSK, musculoskeletal.
Selection process and data extraction
Search results were examined by three individual researchers (D.J., B.W., and F.C.) based on the established inclusion and exclusion criteria listed above. Duplicates were removed before the studies were first screened using titles and abstracts. Full-text screening and quality assessment were performed by three authors independently (D.J., B.W., and F.C.). Any disagreements were resolved by consensus. Data extraction was performed into a Microsoft® Excel® template including first author, date of intervention, country, study design, industry funding/sponsorship, sample size, patient's demographics, MSK condition, type of telerehabilitation and comparator, intervention duration, follow-up period, adverse events and dropouts, outcome measures, and results. Outcome extracted data included post-intervention scores for each group and mean difference (MD) between groups, with respective SDs, and confidence intervals (CIs). When an included study did not report the aforementioned data in sufficient detail, the corresponding author was contacted via email to provide additional data.
Evidence synthesis
Results were narratively synthesized according to the Synthesis Without Meta-analysis (SWiM) reporting guideline. 26 The narrative synthesis was grouped based on the underlying biofeedback technology incorporated (inertial motion sensors and camera-based biofeedback) and reported by MSK condition. For the synthesis methods, please see the “Statistical analysis” section.
Quality and risk of bias assessment
To assess the risk of bias (ROB), the Cochrane ROB tool (version 2.0) 31 was used for randomized controlled trials (RCT), whereas non-randomized trials were assessed using the ROB in Nonrandomized Studies of Interventions (ROBINS) tool. 32 The ROB assessed by the Cochrane ROB tool was rated as high, low, or having some concerns, while ROB in ROBINS was rated as critical, serious, moderate, or low.
The Grading of Recommendations, Assessment, Development and Evaluation (GRADE) approach was used to appraise the effectiveness quality of evidence. 33 The quality of evidence was initially considered as high, and downgraded one level for serious concerns and two levels for very serious concerns, based on five criteria: high ROB, inconsistency of results, indirectness of evidence, imprecision, and publication bias. Finally, the quality of evidence for each outcome was assessed according to four categories: high, moderate, low, or very low.
Since single-arm studies lack a control group, these were not considered on the effectiveness assessment and therefore their ROB and quality of evidence were not assessed.
Statistical analysis
A summary of effect size estimates was performed in a table format. Considering the different measurement scales used to assess either pain or function across studies, data were converted into standardized mean differences (SMDs) with 95% CI considering the first post-intervention result as the time frame to assess outcomes (independent of intervention length). These were calculated for each study by subtracting the post-intervention mean of the control group from the post-intervention mean of the intervention group and dividing by the pooled SD for the sample. A positive value denotes superior results in the intervention group compared to the control group. Some studies did not note the primary outcome or used other primary outcomes besides pain and function. In such cases we prioritized numerical pain rating scales or visual analogue scales (VAS) for pain, as they are the most recommended metrics, including by IMMPACT, ICHOM, and other groups.27–30 For function, condition-specific patient-reported outcome measures were selected, as self-perception of functional limitations and disabilities is considered an important domain by the same groups mentioned above. Whenever available, intent-to-treat results were selected. Effect direction plots were produced to visually depict the range of the obtained effect estimates and CI.
A post hoc sensitivity analysis was performed to assess whether effect sizes for pain and function were different between industry-sponsored and non-industry-sponsored studies.
Results
Search results
The details on the literature search and screening/eligibility processes are depicted in Figure 1. From the initial screening, 141 papers were appraised in full text for eligibility, and 14 papers completely matched the inclusion/exclusion criteria.

Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flow chart of study selection process.
Characteristics of the included studies
The details of the included studies, namely, MSK condition, interventions, outcomes, and main findings are presented in Table 2. Further details on the intervention of the studies are presented in Appendix 3 and on adverse events and reasons for dropouts in Appendix 4.
Characteristics of the included studies.

Abbreviations: CG, control group; DASH, disabilities of the arm, shoulder, and hand; HOOS, Hip Dysfunction and Osteoarthritis Outcome Score; IG, interventional group; KOOS, Knee injury and Osteoarthritis Outcome Score; KOOS-PS, Knee injury and Osteoarthritis Outcome Score for Physical Functioning short-form; MCID, Minimal Clinically Important Difference; MSK, musculoskeletal; MvK, modified von Korff; NDI, Neck Disability Index; NPRS, numerical pain rating scale; ODI, Oswestry Disability Index; OSS, Oxford Shoulder Score; QuickDASH, Quick Disabilities for the Arm, Shoulder and Hand; RCT, randomized controlled trial; ROM, range of motion; SPADI, Shoulder Pain and Disability Index; TKR, total knee replacement; THR, total hip replacement; VAS, visual analogue scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis.
Reported by mean (SD), median (IQR), or mean (95% confidence interval).
All studies were published between 10 October 2017 and 8 January 2022. A total of 8 RCTs, 3 non-randomized control trials, and 3 single-arm studies were included, with sample sizes ranging from 12 to 343. Half of the studies (N = 7/14) included cohorts with a majority of females,34–40 while four studies had a majority of males.41–44 The mean ages differed between studies with three studies reporting cohorts with mean ages of 30–40 years,39,42,43 eight with mean ages between 50 and 60 years,34,36,37,40,41,44–46 and three with a mean age ≥60 years.35,38,47
Regarding the technology interface used for care delivery, nine studies reported on app-based interventions,34–36,41–43,45–47 four were web-based interventions,37,38,40,44 and one focused on virtual reality. 39 The majority of studies (N = 10/14) used wearable inertial motion sensors to provide biofeedback during exercise sessions,34–36,39–43,46,47 and four studies used camera-based sensors.37,38,44,45 Eight studies focused on interventions consisting only of exercise,34,37–41,44,45 whereas the other six studies reported on multimodal programs combining exercise with education and cognitive-behavioral therapy.35,36,42,43,46,47 The majority of the assessed studies (10/14) included simultaneously pain and function as outcomes (either primary or secondary).35,38–43,45–47
Although all studies acknowledged the ability of telerehabilitation systems to register and monitor adherence, only 11 studies provided metrics of adherence.35,36,38,39,41–47 Most of these studies reported metrics solely about the intervention group (7/11),36,42–47 while the remaining four compared metrics between groups.35,38,39,41 Satisfaction scores were provided by seven studies.34–36,38,44,46,47
All studies included some form of communication system between health professionals and patients, a majority (7/14) through scheduled contacts35,36,38–40,46,47 and two focusing on ensuring adherence to exercise.34,42 Three studies reported unidirectional communication (when the healthcare professionals decided it was needed),37,41,44 while six also allowed the patients to reach out as needed.35,36,38,43,45,46
Methodological quality and risk of bias
The assessment of ROB for all studies is summarized in Figure 2. The main risks of bias found in the included studies were the lack of blinding in both participants and investigators, presence of missing data, and, in non-RCT, potential baseline confounding. It was also unclear if allocation was concealed in two RCTs.37,38 From the included studies, nine were sponsored by industry.35–38,42–44,46,47

Risk of bias (ROB) assessment for (a) randomized trials (ROB 2.0) and (b) non-randomized trials. (ROBINS-I: ROB in non-randomized studies of interventions).
Evaluation of the included studies suggested high heterogeneity, mostly related to the diversity of included MSK conditions, the type of intervention, comparison groups, and outcome measures. Visual inspection of the forest plots, combined with the I2 statistic (pain, 70%, P < .0001; function, 80%, P < .0001), confirmed this high heterogeneity, 22 precluding the performance of a meta-analysis.
Efficacy of the outcomes
The summary of findings and respective certainty of evidence are reported in Table 3. The reasons for downgrading the quality of evidence level are described in the table footnotes. Effect direction plots for pain and function are presented in Figure 3 to provide a visual display of the results.

Effect direction plots. Telerehabilitation versus comparison groups with two sub-groups: inertial motion trackers biofeedback and camera-based biofeedback: (a) pain (b) function.
Summary of findings on pain and function outcomes.
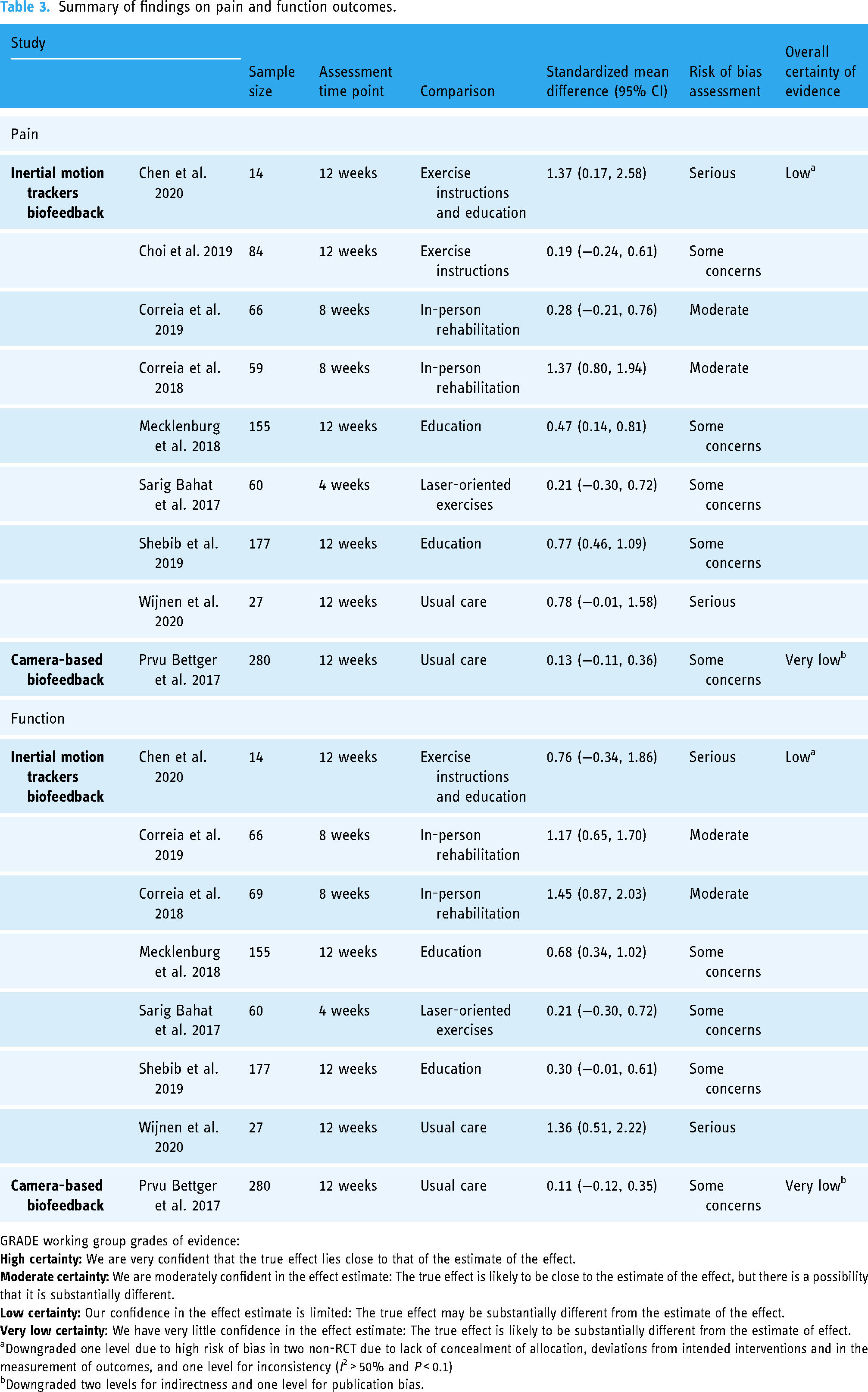
GRADE working group grades of evidence:
Downgraded one level due to high risk of bias in two non-RCT due to lack of concealment of allocation, deviations from intended interventions and in the measurement of outcomes, and one level for inconsistency (I2 > 50% and P < 0.1)
Downgraded two levels for indirectness and one level for publication bias.
Effect sizes were not calculated for three single-arm studies,36,44,46 for one study that did not report sufficient outcome statistical data 37 and another study that did not report results for pain intensity and function individually, 45 in which these parameters could not be retrieved from the authors. These studies were therefore not included in the “Summary of findings” table.
Telerehabilitation with inertial motion trackers biofeedback
Ten studies incorporated inertial motion trackers to provide biofeedback during exercise, including four RCTs,34,39,42,43 four non-RCT,35,40,41,47 and two single-arm interventional studies36,46 (total patient sample size: N = 1284). Intervention duration was similar across studies, ranging between 8 and 12 weeks, except for one study with a shorter treatment duration (4 weeks). 39
Three non-RCT studies35,40,47 focused on rehabilitation after
Two RCTs42,43 compared a multimodal telerehabilitation program to education in patients with
An exercise-based intervention with virtual reality was investigated in an RCT composed of 90 participants with
The remaining study incorporating inertial motion trackers investigated a multimodal telerehabilitation approach in individuals with
Overall, no study found superior results for pain or function in any control group compared to telerehabilitation. These results suggest that telerehabilitation delivered through devices incorporating inertial motion trackers can yield similar or better results than other interventions in improving pain and function, albeit with a low certainty of evidence.
Telerehabilitation with camera-based biofeedback
Biofeedback-assisted telerehabilitation programs supported by camera vision were evaluated in four studies (three RCTs and one single-arm study, N = 467).
In a large RCT (N = 280), Bettger et al.
38
assessed an exercise-based telerehabilitation program
The other RCT
37
compared an exercise program to standard physical therapy in 64 participants who underwent
Last, a small single-arm study
44
evaluated a telerehabilitation program based on exercise alone in 12 patients with
Overall, whether camera-based telerehabilitation is effective in improving pain compared to other interventions remains uncertain due to the observed very low certainty of evidence.
Sensitivity analysis
In order to verify if conclusions were affected by study sponsorship, a sensitivity analysis was performed stratifying the analysis per industry sponsorship (Appendix 5). The reasons supporting the GRADE appraisal are reported in Appendix 6. GRADE appraisal showed that non-industry-sponsored studies provided very low certainty of evidence, while industry-sponsored studies provided low certainty of evidence for pain and function improvements after asynchronous telerehabilitation, which do not impact the conclusions reported previously.
Discussion
Principal findings
To our knowledge, this is the first systematic review evaluating the effectiveness of telerehabilitation incorporating biofeedback to enable asynchronous care in individuals presenting MSK conditions.
The research gathered in this systematic review suggests that asynchronous telerehabilitation with biofeedback provided by inertial motion trackers may be effective in improving pain and function compared to other interventions based on low certainty of evidence. A large majority of the included digital interventions were supported by wearable motion sensors composed of accelerometers and gyroscopes. Very low certainty of evidence was obtained for camera-based telerehabilitation interventions. Importantly, none of the included studies reported superior results of in-person care when compared to telerehabilitation.
Comparison with literature
Compared to face-to-face rehabilitation, remote synchronous care has been touted to be more affordable, accessible,48,49 and as effective as in-person rehabilitation in several MSK conditions.16,18,19,50 While these interventions are useful to mitigate geographic barriers, they may be insufficient to address time availability and scheduling constraints and do not take into account the shortage of human resources.7,8 New care delivery models based on biofeedback-based asynchronous telerehabilitation have emerged with the ambition of being an alternative scalable solution to solve access constraints. However, to date, no systematic reviews have critically examined the potential of such interventions. The present systematic review reports similar findings to the previously reported in synchronous care, suggesting the effectiveness for improving pain and function of an asynchronous model.
Diverse MSK conditions were addressed in this systematic review, with almost all focused on chronic pain, except for one single-arm study targeting acute MSK conditions. Different results were observed depending on the chronic pain condition studied. For chronic knee and low back pain in particular, the results suggest better outcomes for pain after asynchronous telerehabilitation than usual care. This is consistent with findings of previous systematic reviews focusing on remote synchronous care. 15 Of note, a significant proportion of trials (43%, N = 6/14) were dedicated to post-surgical rehabilitation,35,37,38,45,47 with results also indicating comparable outcomes in pain and function compared to in-person rehabilitation,35,37,47 usual care,38,40 or no intervention. 45 This is in line with results reported in a previous systematic review and meta-analysis, 19 which found no difference between technology-assisted rehabilitation and in-person physical therapy following total hip/knee replacement, although including studies where intervention group combined telerehabilitation with in-person treatment. This finding is particularly auspicious considering the potential of this modality as an alternative avenue to provide scalable and equitable care. It may decrease waiting time between discharge and the initiation of outpatient rehabilitation, circumventing obstacles to access in-person care.
Technology in telerehabilitation
The digitalization of healthcare has been seen as a natural evolution of care delivery, considering that more than 63.5% of the global population now has internet access (Worldwide Digital Population as of October 2022 51 ). Initial developments involved telemedicine through synchronous telehealth appointments. Yet, despite being helpful, this may be insufficient for growing needs as it lacks scalability. 52 Asynchronous telerehabilitation may unlock the potential of democratizing access to care by reaching historically underserved populations. The asynchronous telerehabilitation interventions herein leveraged different technologies to provide real-time biofeedback to patients, resulting in effective and clinically supported care delivery. Most evidence supporting the effectiveness of such interventions in MSK care was gathered with programs whose biofeedback was supported by wearable motion sensors composed of accelerometers and gyroscopes. Historically, these were first applied in this context, and therefore most of the advanced programs are currently supported by this type of technology.35,42,43,47 Only four studies provided camera-based telerehabilitation, and these did not provide sufficient evidence regarding pain and disability.37,38,44,45 The potential of camera-based solutions has been showcased in other industries, 53 so a beneficial application to healthcare delivery might become more evident with improved technology and implementation and further research.
Patient-centered care
Digital interventions may offer highly scalable solutions to deliver evidence-based interdisciplinary interventions, 54 improving continuity of care in cases where trained health professionals may not be readily available 52 and promoting adherence to treatment.55,56 In line with a paradigm shift toward a biopsychosocial model, clinical guidelines increasingly recommend conservative multimodal interventions consisting not just a “one-size-fits-all” stand-alone exercise but also a tailored exercise program combined with patient education and behavior changes. The lack of adoption of multimodal interventions has been previously highlighted a shortcoming in care delivery. 57 In the present systematic review, less than half of the trials included multimodal digital programs (N = 6).35,36,42,43,46,47 Although these studies provided some evidence for pain management effectiveness, more research is needed to better understand the impact of each intervention component on outcomes. To foster the adoption of patient-centered approaches, interventions must be tailored and continuously adapted to patient's goals and needs. In the present study, although most studies reported some tailoring of exercise programs, not all reported how the exercise protocol progression was administered.34,37,40–42
Therapeutic alliance
For patient–therapist interactions, compelling evidence has found that the therapeutic alliance (defined as the interaction between participants during the therapeutic process) can influence patient's adherence and treatment outcomes, with communication being a key factor.58–60 The perception of impersonal care with telehealth has been a concern reported in previous published qualitative research embedded within clinical trials. 61 However, previous studies have challenged this notion with reports of patients stating that relationship with the healthcare provider was not affected with videoconferencing 62 and that the comfort of being at home may provide a more relaxed and personal encounter. 63 Elliott et al. 64 reported that communication, particularly the establishment of rapport and the development of patient-centered relationships, was frequently mentioned by highly satisfied patients with telemedicine. Improvements in communication channels (including simple phone calls, text messaging, emails, or videoconferencing) may provide easy access to healthcare professionals, and more timely feedback. This is particularly critical in asynchronous care, where communication needs to be more dynamic and interactive than with scheduled appointments. In the present systematic review, the great majority of studies reported some type of communication during interventions, through diverse strategies. Some interventions used a more simplistic approach, using communication channels solely for exercise reminders,34,42 or as a tool available only to healthcare professionals (who would reach out to the patient as needed).37,41,44 Others diversified and optimized communication channels, allowing bidirectional communication with patients.35,36,38,43,45,46 However, very few details on these parameters were disclosed in the included trials, which is a gap that should be narrowed in future research.
Adherence and satisfaction
One of the major concerns in rehabilitation is patient adherence.65–67 The reasons behind low adherence to rehabilitation interventions are multifactorial, including challenges in access (both geographical and time barriers), costs (e.g., travel costs, work time off, and potentially childcare costs), or, more recently, the perceived risk of contracting communicable infections. In patients prescribed home-based exercises, non-adherence is reported to be as high as 50%. 68 Reasons for this non-adherence levels include low self-efficacy and locus of control, patient beliefs, doubts regarding exercise execution, lack of feedback, and lack of accountability. 68
Telerehabilitation, allied with the development of motion sensing technology, can bring important advantages in this field. Although all studies acknowledge the ability of telerehabilitation systems to register participants’ exercise usability, only 11 studies provided metrics of adherence.35,36,38,39,41–47 A wide variety of adherence metrics were reported in these studies, highlighting the lack of standardization. Nevertheless, overall adherence was high, as indicated by the volume of exercise completed being similar to that initially prescribed.35,36,38,39,42–47 Future research and guidelines should seek to address the lack of consensus on proper outcome measures in this domain.
Satisfaction was assessed less frequently but, similarly to adherence, was high to very high for telerehabilitation in the seven studies in which it was measured.34–36,38,44,46,47 This is in accordance with literature that reports patients’ perception of telerehabilitation as generally positive,16,69,70 with the provision of feedback being one of the most desirable features.71,72
Strengths, limitations, and future directions
This review has important strengths. To our knowledge, this is the first systematic review specifically focused on the effectiveness of asynchronous telerehabilitation incorporating biofeedback technology in the management of MSK conditions. The search gathered evidence from several MSK conditions, including post-surgical telerehabilitation.35,37,38,40,45,47 The included trials were thoroughly assessed using validated reference-standard tools (i.e., the Cochrane ROB tool and GRADE approach), both for ROB and certainty of evidence appraisal. Communication with corresponding authors of the included trials was also conducted in cases where further details/data were needed.
However, this review also contains several limitations. A small number of RCTs were deemed eligible and three of the included studies had a high ROB.40,41,45 There was high heterogeneity among the included trials including for conditions studies, interventions, and outcome measures, precluding the conduct of a meta-analysis.
To attenuate this limitation, a narrative synthesis based on calculated SMD was conducted, which improved the standardization of outcomes and decreased bias. Outcomes were assessed using patient-reported outcome measures. Although these can be prone to be influenced by subjective factors, an important outcome to assess on MSK rehabilitation is the impact of the condition on a patient's activities of daily living. Therefore, the evaluation of patients’ perspectives about their condition is paramount, which can be conducted through patient-reported outcome measures.
Comparison groups were very heterogeneous, with only three studies35,37,47 comparing telerehabilitation with similar interventions provided in-person, limiting the conclusions regarding equivalence or non-inferiority of this innovative approach. Further studies investigating the clinical outcomes compared to in-person rehabilitation are needed.
Patient adherence and satisfaction were addressed, but diverse metrics were used, which hinders rigorous appraisal. Standardization of these outcomes is required.
It is noteworthy that the most studied MSK conditions involved the hip, low back, or proximal joints. No study included patients with distal joints conditions, which warrants caution regarding generalization of findings.
Further rigorous clinical trials are warranted to draw conclusions based on more solid certainty, besides including long-term outcomes to evaluate the consistency of results. It also remains unclear which patients may benefit most from this treatment delivery and if there are clusters of patients in which these are not recommended. Identifying treatment responders is a priority for pain medicine and can favorably alter the risk:benefit ratio. Finally, the cost-effectiveness of asynchronous telerehabilitation with biofeedback was not ascertained in this review and should be explored in future research.
Conclusions
In conclusion, low to very low certainty of evidence suggests that exercise-based asynchronous telerehabilitation which incorporates motion sensors biofeedback technology is effective in improving pain and function in patients with MSK conditions. There is still insufficient evidence to evaluate the utility of telerehabilitation involving camera-based motion tracking. Considering the scalability of interventions with asynchronous care and the potential to democratize care accessibility and promote adherence, further research and development is warranted. Future studies should also address long-term outcomes and cost-effectiveness.
Footnotes
Contributorship
F.D.C., V.Y., and F.C. conceived the idea. D.J., F.C., and B.W. carried out the study selection, data extraction, data analysis, and manuscript drafting. D.J., F.C., F.D.C., V.Y., and S.C. contributed for interpretation of data. A.A., M.M., J.K.S., J.L., V.B., S.C., F.D.C., and V.Y. contributed to the critical revision of the manuscript for important intellectual content. All authors read and approved the final manuscript.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.J, F.C., A.A., M.M., F.C., and V.Y. are employees at Sword Health, the study sponsor. F.D.C., V.Y., and V.B. also hold equity from Sword Health. B.W., J.S., S.P.C., and J.L. are independent scientific/clinical consultants who received advisor honorarium from Sword Health.
Ethical Approval
Institutional review board approval was not required as this article was a systematic review of the literature and not original research.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Author employees of Sword Health were involved in study design, data collection, interpretation, and writing of the manuscript.
Guarantor
V.Y.
Informed Consent
Patient consent was obtained in the studies included in this systematic review. No further consent was requested since this systematic review of the literature only used publicly available data.
Availability of Data
All relevant data is included in the review or available as supplementary material. Further data may be provided upon reasonable request to the corresponding author.
PRISMA checklist.
| Section and topic | Item # | Checklist item | Location where item is reported |
|---|---|---|---|
|
|
|||
| Title | 1 | Identify the report as a systematic review. | p. 1 |
|
|
|||
| Abstract | 2 | See the PRISMA 2020 for abstracts checklist. | p. 2–3 |
|
|
|||
| Rationale | 3 | Describe the rationale for the review in the context of existing knowledge. | p. 3–5 |
| Objectives | 4 | Provide an explicit statement of the objective(s) or question(s) the review addresses. | p. 5 |
|
|
|||
| Eligibility criteria | 5 | Specify the inclusion and exclusion criteria for the review and how studies were grouped for the syntheses. | p. 6 and 7 and Table 1 |
| Information sources | 6 | Specify all databases, registers, websites, organizations, reference lists, and other sources searched or consulted to identify studies. Specify the date when each source was last searched or consulted. | p. 6 |
| Search strategy | 7 | Present the full search strategies for all databases, registers and websites, including any filters and limits used. | p. 6 and Appendix 2 |
| Selection process | 8 | Specify the methods used to decide whether a study met the inclusion criteria of the review, including how many reviewers screened each record and each report retrieved, whether they worked independently, and if applicable, details of automation tools used in the process. | p. 8 |
| Data collection process | 9 | Specify the methods used to collect data from reports, including how many reviewers collected data from each report, whether they worked independently, any processes for obtaining or confirming data from study investigators, and if applicable, details of automation tools used in the process. | p. 8 and 9 |
| Data items | 10a | List and define all outcomes for which data were sought. Specify whether all results that were compatible with each outcome domain in each study were sought (e.g., for all measures, time points, analyses), and if not, the methods used to decide which results to collect. | p. 8 and 9 |
| 10b | List and define all other variables for which data were sought (e.g., participant and intervention characteristics, funding sources). Describe any assumptions made about any missing or unclear information. | p. 8 and 9 | |
| Study risk of bias assessment | 11 | Specify the methods used to assess risk of bias in the included studies, including details of the tool(s) used, how many reviewers assessed each study and whether they worked independently, and if applicable, details of automation tools used in the process. | p. 9 |
| Effect measures | 12 | Specify for each outcome the effect measure(s) (e.g., risk ratio and mean difference) used in the synthesis or presentation of results. | p. 10 |
| Synthesis methods | 13a | Describe the processes used to decide which studies were eligible for each synthesis (e.g., tabulating the study intervention characteristics and comparing against the planned groups for each synthesis (item #5)). | p. 9 |
| 13b | Describe any methods required to prepare the data for presentation or synthesis, such as handling of missing summary statistics, or data conversions. | p. 9 and 10 | |
| 13c | Describe any methods used to tabulate or visually display results of individual studies and syntheses. | p. 10 | |
| 13d | Describe any methods used to synthesize results and provide a rationale for the choice(s). If meta-analysis was performed, describe the model(s), method(s) to identify the presence and extent of statistical heterogeneity, and software package(s) used. | p. 9 | |
| 13e | Describe any methods used to explore possible causes of heterogeneity among study results (e.g., subgroup analysis and meta-regression). | - | |
| 13f | Describe any sensitivity analyses conducted to assess robustness of the synthesized results. | p. 10 | |
| Reporting bias assessment | 14 | Describe any methods used to assess risk of bias due to missing results in a synthesis (arising from reporting biases). | p. 9 |
| Certainty assessment | 15 | Describe any methods used to assess certainty (or confidence) in the body of evidence for an outcome. | p. 9 |
|
|
|||
| Study selection | 16a | Describe the results of the search and selection process, from the number of records identified in the search to the number of studies included in the review, ideally using a flow diagram. | p. 10 and Fig. 1 |
| 16b | Cite studies that might appear to meet the inclusion criteria, but which were excluded, and explain why they were excluded. | p. 6 | |
| Study characteristics | 17 | Cite each included study and present its characteristics. | p. 11 and 12 and Table 2 and Appendix 3 and Appendix 4 |
| Risk of bias in studies | 18 | Present assessments of risk of bias for each included study. | p. 20 and Figure 2 |
| Results of individual studies | 19 | For all outcomes, present, for each study: (a) summary statistics for each group (where appropriate) and (b) an effect estimate and its precision (e.g., confidence/credible interval), ideally using structured tables or plots. | p. 23–26 and Table 3 and Figure 3 |
| Results of syntheses | 20a | For each synthesis, briefly summarize the characteristics and risk of bias among contributing studies. | p. 23–26 |
| 20b | Present results of all statistical syntheses conducted. If meta-analysis was done, present for each the summary estimate and its precision (e.g., confidence/credible interval) and measures of statistical heterogeneity. If comparing groups, describe the direction of the effect. | p. 23–26 and Table 3 and Figure 3 | |
| 20c | Present results of all investigations of possible causes of heterogeneity among study results. | - | |
| 20d | Present results of all sensitivity analyses conducted to assess the robustness of the synthesized results. | p. 26 and 27 and Appendix 5 and Appendix 6 | |
| Reporting biases | 21 | Present assessments of risk of bias due to missing results (arising from reporting biases) for each synthesis assessed. | - |
| Certainty of evidence | 22 | Present assessments of certainty (or confidence) in the body of evidence for each outcome assessed. | p. 25 and 26 and Table 3 |
|
|
|||
| Discussion | 23a | Provide a general interpretation of the results in the context of other evidence. | p. 27–32 |
| 23b | Discuss any limitations of the evidence included in the review. | p. 32 and 33 | |
| 23c | Discuss any limitations of the review processes used. | p. 32 and 33 | |
| 23d | Discuss implications of the results for practice, policy, and future research. | p. 32 and 33 | |
|
|
|||
| Registration and protocol | 24a | Provide registration information for the review, including register name and registration number, or state that the review was not registered. | p. 5 |
| 24b | Indicate where the review protocol can be accessed, or state that a protocol was not prepared. | p. 5 | |
| 24c | Describe and explain any amendments to information provided at registration or in the protocol. | - | |
| Support | 25 | Describe sources of financial or non-financial support for the review, and the role of the funders or sponsors in the review. | p. 34 |
| Competing interests | 26 | Declare any competing interests of review authors. | p. 35 |
| Availability of data, code, and other materials | 27 | Report which of the following are publicly available and where they can be found: template data collection forms, data extracted from included studies, data used for all analyses, analytic code, and any other materials used in the review. | p. 35 |
Adverse events and reasons for dropouts.

| Author, year of publication | Description |
|---|---|
| Inertial motion sensor-based interventions | |
| Y. P. Chen et al., 2020 | AE IG: 1 patient with progressive shoulder pain and weakness 1 month after rehabilitation (diagnosed with full-thickness rotator cuff tear) CG: no AE |
| Dropout reasons: no dropouts | |
| Y. Choi et al., 2019 | AE: did not report |
| Dropout reasons: no dropouts | |
| Correia et al., 2019 | AE IG: three patients developed pain during hip abduction (spontaneous recovery after 2 weeks); one patient with inflammatory signs in the surgical wound; one patient had a fall (not related to the intervention and no need of hospital assistance). CG: one patient had a surgical wound infection (with hospital readmission and procedure revision); one patient developed groin pain; two patients with inflammatory signs in the surgical wound; one patient had a thrombophlebitis; one patient with unilateral lower limb edema (spontaneous recovery); one patient had a fall (no need of hospital assistance) (no statistically significant differences between groups). |
| Dropout reasons IG: three patients were excluded due to developing pain during hip abduction; two patients did not adapt to the intervention. CG: two patients were excluded due to hospital readmission for a surgical wound infection and due to developing groin pain. |
|
| Correia et al., 2018 | AE IG: one patient had a thrombophlebitis CG: one patient had a thrombophlebitis; one patient had a surgical wound infection (with hospital readmission and procedure revision); one patient with alcohol abuse; three patients with inflammatory signs in the surgical wound (no differences between groups). |
| Dropout reasons IG: seven patients withdrew consent; one patient was excluded due to attending additional physical therapy outside the study. CG: two patients were excluded due to hospital readmission for a surgical wound infection and due to alcohol abuse. |
|
| Costa et al., 2022 | AE: did not report |
| Dropout reasons: 29 patients with low compliance, 3 patients referred for conventional physical therapy, 9 patients with other reasons | |
| Janela et al., 2022 | AE: did not report |
| Dropout reasons: 42 patients with low compliance, 8 patients with other reasons | |
| Mecklenburg et al., 2018 | AE: did not report |
| Dropout reasons IG: 14 patients did not respond to invitation; 1 patient had an accident; 6 patients due to personal reasons (time constraints or stress at work). CG: 7 patients entered the IG due to an administrative error. |
|
| Sarig Bahat et al., 2017 | AE IG: five patients with virtual reality-associated side effects (sickness) and headache. |
| Dropout reasons IG: three patients with virtual-reality side effects; one patient due to pain; one patient due to time CG1 (laser training group): one patient due to sickness with hospital admission; one patient due to headache from exercises; two patients due to time CG2 (waiting list): one patient due to side effects; four patients due to time |
|
| Shebib et al., 2019 | AE: did not report |
| Dropout reasons IG: 4 patients were unresponsive; 1 patient had unrelated surgery before study start; 17 patients did not respond to invitation CG: one patient entered the IG due to an administrative error; one patient discontinued intervention due to back surgery |
|
| A. Wijnen et al., 2020 | AE: did not report |
| Dropout reasons: no dropouts | |
| Camera-based interventions | |
| Eichler et al., 2019 | AE: did not report |
| Dropout reasons IG: seven patients due to personal reasons; one patient due to medical reasons CG: ten patients due to personal reasons; five patients due to medical reasons; one patient discontinued intervention |
|
| W. D. Marley et al. 2022 | AE IG: one patient developed biceps pain following shoulder surgery (had an injection; not related to the study intervention). CG: one patient with post-operative pain and stiffness |
| Dropout reasons IG: one patient was lost to follow-up CG: one patient withdrew from the study |
|
| Prvu Bettger et al., 2020 | AE IG: 12 patients had rehospitalizations in 12 weeks; 27/139 patients had a fall. CG: 30 patients had rehospitalizations in 12 weeks; 20/137 patients had a fall. (the difference on the number of rehospitalizations between groups was statistically significant; the difference on falls was not significant). |
| Dropout reasons IG: two patients withdrew from the study. CG: one patient withdrew from the study. |
|
| B. Steiner et al., 2020 | AE: did not report |
| Dropout reasons: two patients were excluded retrospectively as they underwent additional clinical exercise in parallel to the study. | |
Abbreviation: AE, adverse events.
Appendix 5. Sensitivity analysis: effect direction plots.
A. Telerehabilitation versus comparison groups with two sub-groups: industry-sponsored and non-industry-sponsored; outcome, pain.
B. Telerehabilitation versus comparison groups with two sub-groups: industry-sponsored and non-industry-sponsored; outcome, function.
Sensitivity analysis: GRADE appraisal.

| Study | Sample size | Assessment time point | Comparison | Standardized mean difference |
Risk of bias |
Overall |
Reasons for downgrade | |
|---|---|---|---|---|---|---|---|---|
| Pain | ||||||||
|
|
Correia et al. 2019 | 66 | 8 weeks | In-person rehabilitation | 0.28 (−0.21, 0.76) | Moderate | Low | Inconsistency due to high heterogeneity (one level); indirectness (half level); publication bias (half level) |
| Correia et al. 2018 | 59 | 8 weeks | In-person rehabilitation | 1.37 (0.80, 1.94) | Moderate | |||
| Mecklenburg et al. 2018 | 155 | 12 weeks | Education | 0.47 (0.14, 0.81) | Some concerns | |||
| Prvu Bettger et al. 2017 | 280 | 12 weeks | Usual care | 0.13 (−0.11, 0.36) | Some concerns | |||
| Shebib et al. 2019 | 177 | 12 weeks | Education | 0.77 (0.46, 1.09) | Some concerns | |||
|
|
Chen et al. 2020 | 14 | 12 weeks | Exercise instructions and education | 1.37 (0.17, 2.58) | Serious | Very Low | Serious risk of bias in two studies (two levels); indirectness (half level); imprecision due to sample size (one level) |
| Choi et al. 2019 | 84 | 12 weeks | Exercise instructions | 0.19 (−0.24, 0.61) | Some concerns | |||
| Sarig Bahat et al. 2017 | 60 | 4 weeks | Laser-oriented exercises | 0.21 (−0.30, 0.72) | Some concerns | |||
| Wijnen et al. 2020 | 27 | 12 weeks | Usual care | 0.78 (−0.01, 1.58) | Serious | |||
| Function | ||||||||
|
|
Correia et al. 2019 | 66 | 8 weeks | In-person rehabilitation | 1.17 (0.65, 1.70) | Moderate | Low | Inconsistency due to high heterogeneity (one level); indirectness (half level); publication bias (half level) |
| Correia et al. 2018 | 69 | 8 weeks | In-person rehabilitation | 1.45 (0.87, 2.03) | Moderate | |||
| Mecklenburg et al. 2018 | 155 | 12 weeks | Education | 0.68 (0.34, 1.02) | Some concerns | |||
| Prvu Bettger et al. 2017 | 280 | 12 weeks | Usual care | 0.11 (−0.12, 0.35) | Some concerns | |||
| Shebib et al. 2019 | 177 | 12 weeks | Education | 0.30 (−0.01, 0.61) | Some concerns | |||
|
|
Chen et al. 2020 | 14 | 12 weeks | Exercise instructions and education | 0.76 (−0.34, 1.86) | Serious | Very Low | Serious risk of bias in two studies (two levels); inconsistency due to high heterogeneity (one level); indirectness (half level); imprecision due to sample size (one level) |
| Sarig Bahat et al. 2017 | 60 | 4 weeks | Laser-oriented exercises | 0.21 (−0.30, 0.72) | Some concerns | |||
| Wijnen et al. 2020 | 27 | 12 weeks | Usual care | 1.36 (0.51, 2.22) | Serious | |||
| GRADE working group grades of evidence: |
||||||||
