Abstract
Background
Obstructive sleep apnea syndrome is a common sleep-breathing disorder associated with adverse health outcomes including excessive daytime sleepiness, impaired quality of life and is well-established as a cardiovascular risk factor. Continuous positive airway pressure is the reference treatment, but its cardiovascular and metabolic benefits are still debated. Combined interventions aiming at improving patient's lifestyle behaviours are recommended in guidelines management of obstructive sleep apnea syndrome but adherence decreases over time and access to rehabilitation programmes is limited. Telerehabilitation is a promising approach to address these issues, but data are scarce on obstructive sleep apnea syndrome.
Methods
The aim of this study is to assess the potential benefits of a telerehabilitation programme implemented at continuous positive airway pressure initiation, compared to continuous positive airway pressure alone and usual care, on symptoms and cardiometabolic risk factors of obstructive sleep apnea syndrome. This study is a 6-months multicentre randomized, parallel controlled trial during which 180 obese patients with severe obstructive sleep apnea syndrome will be included. We will use a sequential hierarchical criterion for major endpoints including sleepiness, quality of life, nocturnal systolic blood pressure and inflammation biological parameters.
Discussion
m-Rehab obstructive sleep apnea syndrome is the first multicentre randomized controlled trial to examine the effectiveness of a telerehabilitation lifestyle programme in obstructive sleep apnea syndrome. We hypothesize that a telerehabilitation lifestyle intervention associated with continuous positive airway pressure for 6 months will be more efficient than continuous positive airway pressure alone on symptoms, quality of life and cardiometabolic risk profile. Main secondary outcomes include continuous positive airway pressure adherence, usability and satisfaction with the telerehabilitation platform and medico-economic evaluation.
Trial registration
Clinicaltrials.gov Identifier: NCT05049928. Registration data: 20 September 2021
Keywords
Introduction
Obstructive sleep apnea syndrome(OSAS) is one of the most frequent sleep disorder associated with many disabilities including excessive daytime sleepiness (EDS), 1 impaired quality of life, 2 metabolic and cardiovascular complications. 3 There is a bidirectional relationship between OSAS and obesity. Obesity is the prominent risk factor for OSAS 4 and OSAS by itself can contribute to weight gain. 5 OSAS and obesity exert a synergistic deleterious effect on the cardiovascular system through various mechanisms including low-grade systemic inflammation.6,7
The first line treatment for OSAS is continuous positive airway pressure (CPAP), which normalizes the apnea-hypopnea index (AHI), improves sleep quality, daytime sleepiness and quality of life. 8 The effect of CPAP on blood pressure (BP) is limited with an average decrease of 2–2.5 mmHg of 24-h systolic or diastolic BP. 9 Overall, the cardiovascular benefit of CPAP treatment in OSAS remains still debated since randomized controlled trials (RCT) failed to demonstrate a protective effect of CPAP in secondary prevention of cardiovascular disease.10,11 These disappointing results could be partly explained by adherence to CPAP treatment which is often insufficient 12 while an average compliance greater than 4 h per night is necessary to achieve efficacy on BP 13 and for the reduction of some cardiovascular events.14,15 Telemonitoring and eHealth interventions could improve adherence to CPAP.16–18 The use of multimodal telemonitoring platforms at CPAP initiation improves sleepiness 19 and may enhance body weight reduction 20 without increasing the number of steps per day or decreasing the BP.19,20 Altogether, these data support the need for well-conducted lifestyle interventions combined to CPAP and supported by telemedicine for integrated care in OSA.
The improvement in cardiovascular health in OSAS should not be limited to target CPAP adherence 10 and the development of global strategies is necessary for the appropriate management of OSAS and associated comorbidities. 21 Exercise training reduces the severity of OSAS and improves daytime sleepiness 22,23 while weight loss interventions improve BP, insulin resistance, hypertriglyceridemia and inflammation in obese patients with severe OSAS.24,25
The technological advances of recent years, particularly in the field of information and communication technologies (ICT) have brought new tools dedicated to the integrated management of chronic diseases. We previously demonstrated in an RCT that telerehabilitation (TR) in 50 obese patients was feasible, providing good patient satisfaction, and improved quality of life. 26
To the best of our knowledge, no study has examined the impact of a holistic TR intervention combining physical activity, nutrition and patient engagement and educational tools in a population suffering from OSAS and obesity. We, therefore, propose to demonstrate the impact of a TR solution combined with CPAP on sleepiness, quality of life, nocturnal systolic BP and inflammation biological markers.
Herein, we describe the rationale and the design of the multicentre, parallel m-Rehab OSAS RCT trial.
Objectives of the study
Primary objective
The aim of our RCT was to assess the potential benefits in severe obese OSAS patients of a 6-month TR programme implemented at CPAP initiation, compared to CPAP alone and usual care, on symptoms and cardiometabolic risk factors of OSAS. This primary outcome is evaluated by a hierarchical sequence test including, in the following order, the changes from baseline to 6 months of:
Sleepiness assessed by the Epworth Sleepiness Scale (ESS).
27
Quality of life assessed by the Functional Outcomes of Sleep Questionnaire (FOSQ).
28
Nocturnal systolic BP measured by 24-h ambulatory BP monitoring (ABPM). Inflammation assessed by the high-sensitivity C-reactive protein (hs-CRP) blood test.
Secondary objectives
The main secondary outcomes are the changes from baseline to 6 months between the 2 groups of adherence to CPAP, physical activity level assessed by the International Physical Activity Questionnaire (IPAQ) 29 and measured by actimetry, exercise tolerance evaluated by exercise test on a cycloergometer, anthropometric parameters, biological parameters of metabolic risk, BP profile parameters evaluated by 24h-ABPM and sleep architecture evaluated by polysomnography.
Other objectives are the evolution of fatigue (Pichot questionnaire 30 ), sleepiness (Idiopathic Hypersomnia Severity Scale 31 ), quality of life (EQ-5D 32 ), sleep quality (Pittsburgh Sleep Quality Index: PSQI 33 ), insomnia symptoms (Insomnia Severity Index: ISI 34 ) and anxiety and depression symptoms (Hospital Anxiety and Depression-HAD scale 35 ).
An assessment of motivational factors for physical activity is planned, based on self-administered questionnaires: Expectations of results, self-efficacy, perception of risks, intentions, planning, self-efficacy to barriers, habits, and social support.
Moreover, a medico-economic analysis will be carried out with an assessment of the incremental cost-utility ratio in the experimental and control group.
In the TR group only, we also aim to evaluate the usability of the TR digital solution with the System Usability Scale (SUS) 36 and the usage of the TR during the 6-month period by the extraction of data from the digital solution and with data-mining analysis to allow to study the relationships between the TR digital solution usage and the effects of TR programme in the TR group.
Methods
Study design
The present study is a multicentre, randomized, controlled, parallel, trial involving 13 centres in France with Montpellier as the coordinating centre.
180 newly diagnosed obese patients with severe OSAS will be randomly allocated to CPAP combined with TR or CPAP alone (control group). Data will be collected at baseline and after 4, 12, and 24 weeks (endpoint).
The study was approved by the ethics committee (CPP IDF IV – France. IDRCB : 2020-A02100-39). ClinicalTrials.gov identifier NCT05049928 This protocol follows the SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) 2013 37 and the reporting will respect the Consolidated Standards of Reporting Trials (CONSORT) statement for non-pharmacologic trials. 38
Patients randomized to the control group will be informed that, at the end of the 6-month protocol, they will be offered a 6-month period usage of the TR digital solution developed for the study. Patients in the intervention group will have the opportunity to continue the TR intervention for an additional 6-month period. Satisfaction, usability, and patient experience will be assessed to improve ergonomics and functionality of the digital solution. The flow chart of the study is detailed in Figure 1.

Study flow.
Study population
Newly diagnosed severe obstructive sleep apnea (AHI ≥30/hour, measured by polysomnography). Body mass index ≥ 30 kg/m². Age between 30 and 75 years’ old Written consent Presence of contraindications to perform exercise training (neuromuscular disease, orthopaedic cause, acute coronary syndrome or stroke in the previous 3 months). Active infection, chronic inflammatory disease, cancer under treatment. Systemic treatment with immunosuppressants or corticosteroids. Obstructive sleep apnea treated by CPAP or mandibular advancement device in the 3 months preceding inclusion. Participation in a rehabilitation programme within 6 months preceding inclusion. Predominantly central sleep apnea syndrome (AC > 50%). Heart failure with Left Ventricular Ejection Fraction <40%. Bariatric surgery in the previous 6 months or bariatric surgery planned in the 6 months following inclusion. Alcohol intake > 14 drinks per week. Inability to understand and/or answer questionnaires. Refusal to use a smartphone or digital device. Inability to access an internet connection at home. Subject in a period of relative exclusion with respect to another protocol or for which the maximum annual amount of compensation of € 4500 has been reached. Subject not affiliated with a social security scheme, or not beneficiary of such a scheme. Pregnant or breastfeeding woman, patient unable to give her protected adult consent, vulnerable people. Subject deprived of liberty by judicial or administrative decision.
Exclusion criteria are:
Randomization
In total, 180 obese patients with OSAS will be randomly allocated to a TR programme with CPAP (intervention group) or to a CPAP (control group).
The randomization sequence will be computer-generated using random blocks in an order unknown by the investigators. The list is established by an experienced statistician (NM) and is only accessible by personnel in charge of randomization. Although participants cannot be blinded to their treatment, both interventions are presented as active interventions.
Randomization between the 2 arms will be stratified based on the study centre and on the baseline ESS 22 value (>14 vs. ≤ 14), with a 1:1 ratio.
Statistical analysis plan
The number of patients contacted for the study and the number of subjects included and randomized in each group will be reported on a study flow chart. Premature stops (lost to follow-up, death, study withdrawals) and their reasons will be reported as well as any deviations from the protocol.
An initial descriptive analysis of each group will be carried out. For qualitative variables, this description will include the number as well as the frequency of the different modalities. Concerning the quantitative variables, the description will include the number, the mean, the standard deviation, the median as well as the extreme values according to the distribution.
The comparability of the 2 groups will be verified for all the initial characteristics likely to influence the results. In the event of non-comparability on one or more parameters, an adjustment will be made on this or these parameters for the between-groups endpoints comparisons.
Analysis of the primary outcome measure: The variation in the evolution of main criteria will be compared between the 2 groups using a nonparametric means comparison test (Mann–Whitney test) or if the distribution turns out to be Gaussian and the conditions application are verified, a parametric test (Student's T).
Analysis of secondary endpoints: The qualitative variables will be compared by a Chi-square test. Otherwise, if the conditions for performing this test are not met, Fisher's exact test will be used. The quantitative variables will be compared between the two groups. In the case of Gaussian variables, a student's
Longitudinal data (quality of life scores, physical activities) will be modelled by a mixed regression with the patient effect as a random variable. A linear regression model modelling the variation of the longitudinal endpoints will be fitted to the covariates having at least one trend (
Management of missing data: The Intuition for Inverse Probability of Treatment Weighting method will be used. The lost to follow-up will be considered using a weighting on the inverse of the probability of being censored.
Sample size calculation
The calculations are made by taking a power of 80% and alpha risk at 5%. Our assumptions are that:
For the ESS, the difference between the two arms will be 2 points ± 4.8,39 Thus, 80 subjects per study arm are needed For the FOSQ, the difference will be 1.5 points ±2.5.
40
Thus, 59 subjects per study arm are necessary, For nocturnal systolic BP, the difference will be 10 points ±20.
24
Thus, 63 subjects per study arm are required. For CRP, the difference will be 8 points ±16.
24
Thus, 63 subjects per study arm are necessary.
In order to consider a rate of participants lost to follow-up and unusable data of around 12.5%, we propose to include in this study
Description of the intervention
Patients in both groups will be treated with a single CPAP brand to reduce heterogeneity (PRISMA SMART max LÖWENSTEIN, Hamburg, Germany). The CPAP settings will be made by the doctor in charge of the patient. Patients will be followed at home by associated healthcare providers.
Control group
Patients will receive the usual advice on adapted physical activity and nutrition during the initial consultation. For physical activity, patients will be advised to achieve the World Health Organization (WHO) recommendations of physical activity (i.e., at least 150–300 min of moderate-intensity aerobic physical activity per week), to decrease sedentary time and increase the daily number of steps.
Intervention group
Patients will have access to the m-Rehab® TR programme, a digital solution designed et developed for the present study.
The organization and content of the TR programme is fully described in the supplementary information file with screenshots of the patient solution. Figure 2 shows the global model of m-Rehab digital solution and organization.

Multimodal interventions will be implemented and the “care manager” will play a key role by providing coordinating and tracking the home-based TR intervention. The objective being to promote behavioural changes et motivation to increase patient physical activity level, nutritional change behaviours adapted to obesity, sleep hygiene habits, and adherence to CPAP. Patients will have the possibility to request a consultation with their sleep specialists via medical teleconsultation, with the health-care provider (for CPAP follow-up) and with the m-Rehab technical support team for the use of the digital solution. The sequence of interventions is detailed in Figure 3.
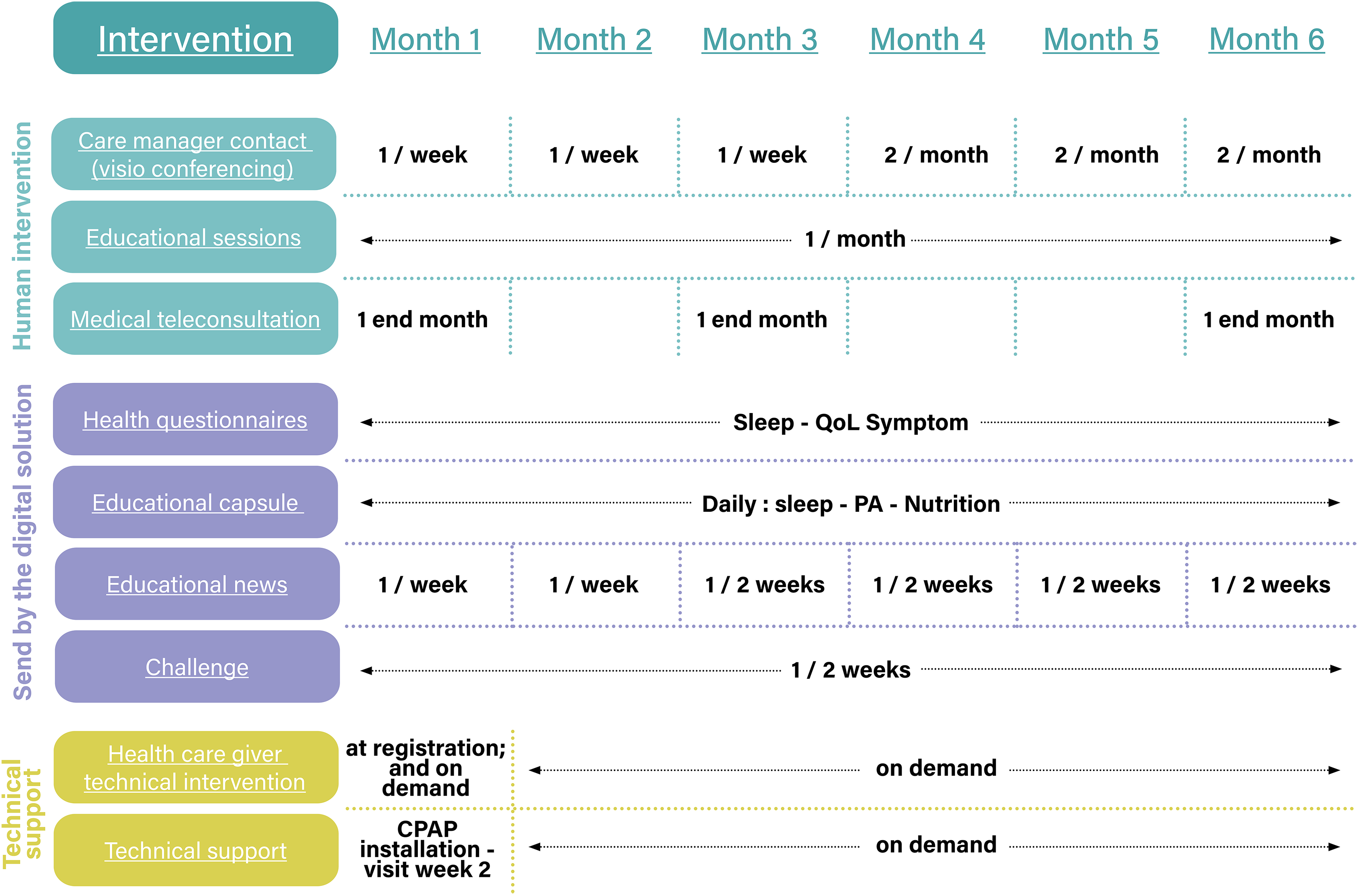
Sequences of interventions during the 6-month study, in the telerehabilitation group.
During the course of the TR intervention, feedback will be available to the patient via various connected devices: smartwatch for the measurement of steps per day; connected weight scale; and a connected tensiometer. Data from the CPAP device will also be transmitted in the patient application and web site.
Outcome measures
All assessments made at baseline will be realized at the 6 months.
The data will be collected by personnel attached to the research group who have previously been instructed in the procedures to follow and who are not aware of patients’ randomization.
Outcome measures are detailed in Table 1.
Primary and secondary outcomes.

ABPM: ambulatory blood pressure monitoring; CPAP: continuous positive airway pressure; ESS: Epworth Sleepiness Scale; FOSQ: Functional Outcomes of Sleep Questionnaire.
* TR group-specific analysis
Primary outcome measures
The determination of hs-CRP will be performed by immunoturbidimetry on a Roche Cobas 8000.
Secondary outcomes measures
Actigraphy
Evaluation will be realized by an Actigraph Gt3X triaxial accelerometer worn for 7 days on the hip. The following data will be collected: duration of time in moderate to vigorous physical activity, sedentary time, number of daily steps, intensity and distribution of PA during the day/week and energy expenditure.
Cardiopulmonary exercise testing
Maximum incremental stress test on a bicycle ergometer. Using a12-lead electrocardiogram and a gas exchange analysis system. Maximum criteria will be determined in accordance with international stress testing standards. The variables of interest will be the maximum oxygen uptake at peak (maximum VO2, ml/min and ml/min/kg), the maximum power (watts) and the ventilatory threshold (% of theoretical maximal VO2).
Anthropometric parameters
Anthropometric measurements will be taken in the morning after 12 h of fasting. The following parameters will be collected: height (m), weight (kg), calculation of the body mass index (BMI) in kg/m², hip, waist and neck measurements (cm).
Polysomnography
Sleep recordings are made in the sleep laboratory or at the patient's home. PSG included several EEG leads right and left electrooculogram, electromyography of chin and tibialis anterior muscle, electrocardiogram, nasal cannula/pressure transducer, mouth thermistor, chest and abdominal bands, and pulse. The following data are collected: stages of sleep (minutes and %), sleep efficiency (%), micro-arousal index (events/hour), AHI, AHI in REM and NREM, desaturation index (events/hour), average SpO2 (%), time spent under a SpO2 of 90% (minutes and %).
Others self-reported questionnaires
Pichot Fatigue Scale, 30 Idiopathic Hypersomnia Severity Scale, 31 EQ-5D(32), PSQI, 33 ISI, 34 HAD scale, 35 IPAQ. 29
Adherence to CPAP
Data on the use (minutes and hours) and efficacy of CPAP will be collected by healthcare providers either during the visit to the patient's home or via remote monitoring. Data from CPAP at 1 month (residual AHI (events/hour), leaks (% and L/min), deep sleep indicator (minutes and %) will be collected to assess whether it is associated with adherence at 6 months.
Motivational self-reported questionnaires
Questionnaires on perceived risk, expectations of consequences, self-efficacy, intentions, planning, self-efficacy against barriers, social support and questionnaire relating to the automatic nature of behaviour. 41
Medico-economic endpoint
The medico-economic judgment criterion will be the incremental cost-utility ratio at 6 months. The utility will be derived from the EQ-5D (EuroQol five dimensions questionnaire). 32
Usability
Usability will be evaluated in the TR group with the SUS(36). It is a 10-item questionnaire with five response options for the respondents. A SUS score of >68 is considered above average, and anything <68 is below average.
Discussion
We have designed a multicentre, randomized, controlled, parallel, clinical trial to study the effects of a comprehensive 6-month TR programme, in patients with obesity and severe OSAS, compared to CPAP alone and usual care. The current article presents a detailed description of the multicentre RCT.
CPAP and TR for the management of OSAS
Severe OSAS and obesity have common characteristics and comorbidities, such as sleep disturbances, increased cardiometabolic risks (metabolic syndrome, diabetes, hypertension and other cardiovascular diseases), reduced physical activity levels, unhealthy nutritional behaviours, and frequently poor self-management for their chronic diseases. In both conditions and
A TR solution guided by care managers
We have designed a set of digital solutions including a website for professionals and patients and an application on a smartphone for patients. We called it “m-Rehab” to summarize the concept of “mobile telerehabilitation”, in the same way as m-health or e-health: providing a rehabilitation course to patients via ICTs, allowing access to information to gain mobility anywhere, close to patient's living place. Our first experience in TR for obese patients, revealed the need for an individual human home care intervention to complete the programme. 26 Therefore, we have conceived a human-centred management by introducing remote coordination via a “care manager.” 49 Many organizational home-care services or telemonitoring interventions have been studied in chronic diseases,50,51 and case management seems to be effective on outcomes. 49 We believe that such a remote service in TR will play a facilitating role to develop individualized action plans for increasing physical activity levels, improving nutritional behaviours and CPAP adherence. Such an organization has the potential for solving problems or remove barriers allowing patients to adopt more favourable health behaviours.
The m-Rehab 6-month programme includes several interventions aimed at reaching patient-oriented outcomes via a rehabilitation model: increase patient health education, particularly for sleep and nutritional disorders, promote regular physical activity, and improve nutritional behaviours. Feed-back from connected medical devices (connected watch, scale, tensiometer) or from the CPAP device, will only be here to help the patient track each of these dimensions and to think about what he/she can/wants to do. m-Rehab course is not conceived as a “45-min 3 times per week exercise training program” or an “active weight loss diet program”. The m-Rehab digital solution and human care management aims to provide a behavioural and educative-oriented approach that can encourage the patient to engage beneficial health changes.
A robust methodology for a multicentre study
We have designed a RCT with hierarchical multiple evaluation criterion, a reference method in therapeutics evaluation in human disease. The effects of a new, original intervention have to be evaluated by a basic comparison with the usual care in a given disease. In our model, the blind modality is not conceivable because of two main challenges. The first was to consider patient deception when randomized in the control usual care arm of the study. This could cause higher volunteer dropouts in the control group or encourage them to adopt an alternative digital solution of exercise training, for example. To avoid these risks, we will propose the use of the digital solution during 6 months at the end of the 6-month control period, shortly after the randomization process. Second, is the lack of blindness in such a study design which could represent a bias in the evaluation procedures. To avoid this, the questionnaires used in the present study will only be self-administered for both groups via an online platform (MHLink) that is different from the TR digital solution. In addition, several primary outcomes of the study will be independent of the operator (24-h ABPM, hs-CRP dosage), therefore not based on any operator or patient influence. Finally, we have chosen a hierarchical testing strategy 52 based on 4 different dimensions including sleepiness, quality of life, BP and biological markers of low-grade inflammation. It is quite challenging to reach statistical significance in this type of interventional TR trial. However, we believe that to be convincing TR should take into account the multiple dimensions of TR in chronic disease.
Conclusion
With the explosion of the obesity epidemic in industrialized countries, the prevalence of OSAS has increased in recent years. The benefits of CPAP are limited by poor adherence and limited effects on cardiovascular risk, making the development of a combination between CPAP and lifestyle interventions mandatory. The m-Rehab OSAS study is a rigorously designed multicentre RCT evaluating the effects of a multidimensional TR programme aimed at engaging patients with obesity and severe OSAS in a physically active lifestyle, and healthy diet behaviours, using an original and human-centred digital solution. The potential success of this programme will open a new area in patient daily and holistic care.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076231167009 - Supplemental material for Impact of a telerehabilitation programme combined with continuous positive airway pressure on symptoms and cardiometabolic risk factors in obstructive sleep apnea patients
Supplemental material, sj-docx-1-dhj-10.1177_20552076231167009 for Impact of a telerehabilitation programme combined with continuous positive airway pressure on symptoms and cardiometabolic risk factors in obstructive sleep apnea patients by François Bughin, Monique Mendelson, Dany Jaffuel, Jean-Louis Pépin, Frédéric Gagnadoux, Frédéric Goutorbe, Beatriz Abril, Bronia Ayoub, Alexandre Aranda, Khuder Alagha, Pascal Pomiès, François Roubille, Jacques Mercier, Nicolas Molinari, Yves Dauvilliers, Nelly Héraud and M Hayot in DIGITAL HEALTH
Footnotes
Declaration of conflicting interests
FB has received lecture fees or conference traveling grants from Lowenstein Medical, Bioprojet, Vitalaire, Sos Oxygene, Agir à dom, Bastide, ISIS Médical. DJ has received grants, lecture or consulting fees or conference traveling grants from Novartis, Sefam, Adene, Lowenstein, Nomics, GSK. JLP has received lecture or consulting or conference traveling grants from ResMed, Philips, Fisher and Paykel, AstraZeneca, Jazz Pharmaceutical, Agiradom, Mutualia, Vitalaire, Boehringer Ingelheim, Night Balance, Sefam. FG has received lecture or consulting or conference traveling grants from ResMed, Jazz Pharmaceutical, Sefam, Bioprojet, Philips Respironics, Cidelec, Asten Santé. KA has board engagements with Sanofi Genzyme, GSK, Astra Zeneca.
FR has speaking honoraria and board engagements with Abbott, Air Liquide, Bayer, Pfizer, Astra Zeneca, Servier, Boehringer, Vifor, Novartis, Novonordisk, YD has speaking honoraria and board engagements with UCB Pharma, JAZZ, Orexia, Bioprojet, Avadel, Idorsia and Takeda. MH has received research grants from Bastide Medical, which are not related to the current project; personal fees from AstraZeneca for participation to scientific lectures; financial support for congress participation from SOS Oxygène, Eole Santé, Boehringer Ingelheim, GlaxoSmithKline, and AstraZeneca; and hospitalities during local scientific meetings from ALK-Abelló, Actelion Pharmaceuticals France, Vifor Fresenius Medical Care Renal Pharma, Sanofi Aventis France, Novartis Pharma, LVL Medical Sud, Chiesi, and SOS Oxygene Mediterranee. Other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted in accordance with the CONSORT ethical guidelines and was approved by the ethics committee (CPP IDF IV – France. IDRCB: 2020-A02100-39). ClinicalTrials.gov identifier NCT05049928.
Funding
This project was funded by the Occitanie Region (south of France) and FEDER (European regional development fund). The University of Montpellier was the leader manager of the project
The sponsor played no part in the study design; collection, management, analysis, and interpretation of the data; writing of the report; and the decision to submit the report for publication
Informed consent
All participants gave their written informed consent.
Guarantor
FB
Contributorship
FB and MH researched literature and conceived the study. FB wrote the first draft of the manuscript. MM, DJ, JLP, YD and NH made a substantial contribution to the concept or design of the work. NM is responsible for designing the statistical procedures. FG, BA, BB, AA, PP and JL contributed to the protocol development and provided clinical expertise All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Acknowledgments
The authors would like to acknowledge the physicians involved in the recruitment of patients: Khuder Alagha, Caroline Alovisetti, Jean-François Le Calvez, Elisa Evangelista, Marie-Pierre Cadars, Pierre Oliver, Vincent Puel, Fabrice Thoin, Jean-Guillaume Weber and the members of the consortium “m-Rehab”: Julie Boiche, Blandine Chapel, Christophe Latrille, Catherine Miffre, Anne-Lise Courbis, Nathalie Jean and Jordan Michel.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
