Abstract
Objective
This open pilot study examines the feasibility, acceptability, and qualitative outcomes of an interactive web- and text message-delivered personalized feedback intervention aimed at cultivating motivation and tolerance of distress for adults initiating outpatient buprenorphine treatment.
Methods
Patients (n = 10) initiating buprenorphine within the past 8 weeks first completed a web-based intervention focused on enhancing motivation and providing psychoeducation on distress tolerance skills. Participants then received 8 weeks of daily personalized text messages that provided reminders of salient motivational factors and recommended distress tolerance-oriented coping skills. Participants completed self-report measures to assess intervention satisfaction, perceived usability, and preliminary efficacy. Additional perspectives were captured via qualitative exit interviews.
Results
In total, 100% of retained participants (n = 9) engaged with the text messages throughout the entire 8-week period. Mean scores of 27 (SD = 5.05) on the Client Satisfaction Questionnaire at the end of 8-week period indicated a high degree of satisfaction with the text-based intervention. The average rating on the System Usability Scale was 65.3 at the end of the 8-week program, suggesting that the intervention was relatively easy to use. Participants also endorsed positive experiences with the intervention during qualitative interviews. Clinical improvements were observed across the intervention period.
Conclusions
Preliminary findings from this pilot suggest that the content and delivery method of this combined web- and text message-based personalized feedback intervention is perceived by patients as feasible and acceptable. Leveraging digital health platforms to augment buprenorphine has potential for high scalability and impact to reduce opioid use, increase adherence/retention to treatment, and prevent future incidence of overdose. Future work will evaluate the efficacy of the intervention in a randomized clinical trial design.
Keywords
Introduction
The prevalence of opioid use disorder (OUD) has reached epidemic rates in the United States, 1 resulting in a dramatic rise in morbidity and mortality.2,3 In 2018, over 46,800 people died from an opioid-involved overdose, accounting for 70% of all drug overdose deaths.2,3 The emergence of coronavirus disease 2019 (COVID-19) has further exacerbated this issue with rates of fatal overdoses increasing substantially in 2020. 4
Medications for OUD (MOUD) such as buprenorphine-naloxone (suboxone), methadone, and naltrexone are currently the most efficacious treatments for OUD. 5 Due to its safety profile and flexible administration, buprenorphine has grown in popularity over the last decade. 6 Although buprenorphine is highly effective in reducing mortality and morbidity,7,9 maintaining adherence to and retention in treatment is a significant challenge. A substantial portion of patients receiving buprenorphine discontinue treatment prematurely and/or return to illicit opioid use.10,12 Recent studies suggest that approximately 50% of individuals who initiate buprenorphine will discontinue medication by 6-months. 13 This is notable as current recommendations highlight the importance of adhering to buprenorphine for at least 6 months to confer its full benefits and stabilize the overall functioning of the individual (e.g., return to work). 14 These findings emphasize the need to develop novel interventions aimed at improving engagement and retention in traditional medication-based treatments for OUD.
Prior research has examined factors that contribute to treatment non-adherence and reduced retention for buprenorphine and documented the role of numerous multi-level barriers such as transportation challenges, 15 family conflict, 16 stigma towards OUD and MOUD 17 and cost of MOUD 17 as contributing factors. Whereas addressing each of these factors requires a multi-level approach, addressing underlying factors may be one way to promote retention in face of these challenges. Negative affective experiences are a product of all these barriers. Consistent with the negative reinforcement model of substance use, individuals use opioids to alleviate aversive internal states, such as depression, anxiety, or frustration.11,18 Indeed, negative affect is a well-established precipitant of a return to opioid use. 19 Distress tolerance (DT), which reflects the actual or perceived ability to tolerate or manage negative affect, 20 has been linked to a variety of substance use outcomes, including frequency and severity of drug use, premature treatment discontinuation, and return to illicit substance use.21,24 According to the functional model of DT, it involves a behavioral inhibition in face of negative reinforcement and is understood as a propensity to respond to immediate (negative) reinforcement or reward. 25 Preliminary evidence also demonstrates that increasing DT is associated with decreased opioid use, 26 by promoting the ability to persist in goal-directed activity (e.g., medication adherence), even when experiencing negative affect. 27
Personalized feedback interventions, such as decisional balance frameworks, also show promising effects in reducing problematic substance use. 28 Consistent with a motivational enhancement perspective, decisional balance procedures involve an evaluation of the pros/cons of engaging in a certain behavior (e.g., opioid use), compared to the pros/cons of alternative behavior (e.g., recovery), and also offer strategies to support behavior change (e.g., DT skills training).29,30 Decisional balance procedures can be utilized as a part of multi-component interventions,31,32 but are also effective when used as a stand-alone intervention.29,33,34 Stand-alone decisional balance procedures have demonstrated effectiveness for reducing substance use 35 among various populations including non-treatment seeking college student drinkers 29 and non-treatment-seeking smokers.36,37 Whereas limited research has examined the effectiveness of decisional-balance procedures for reducing opioid use, one qualitative study on youth engaged in treatment for OUD noted that decisional balance was useful in realizing that help was needed for their opioid use. 38 Decisional-balance procedures, embedded with other motivation interviewing techniques, are recommended as best practices for OUD treatment. 35 However, no integrated protocol exists that seek to both enhance motivation and improve DT skills among individuals with OUD.
Technological platforms, such as text-based modalities, offer the advantage of delivering evidence-based strategies to participants’ natural environments, with individually tailored content, timing, and frequency, that may overcome challenges with drug cues and/or negative affect states. Indeed, text-based interventions are effective in promoting various health and behavior changes, with personalized text messaging showing the greatest benefit. 39 There is growing evidence that decisional balance procedures can be effectively delivered in digital format, 29 however, DT-based interventions designed to target substance use have predominately been delivered in a structured, in-person, format. 27 Thus, there is a greater need in research to explore the integration of DT-based interventions with decisional balance procedures (DT + Decisional Balance), and translation of these intervention procedures into a digital health platform to promote greater reach and access.
We developed and refined a web- and text message-based intervention that is grounded in DT theory and motivation enhancement theory. It combines DT skills training with decisional balance procedures to promote engagement in and adherence to buprenorphine treatment.40,41 The pilot phase of this intervention was conducted among individuals engaged in buprenorphine at two outpatient treatment centers. The primary objective of this pilot intervention was to examine preliminary indices of feasibility, acceptability, and qualitative outcomes of the developed intervention.
Methods
Participants and procedures
The current study is an open pilot development and trial that was conducted at Rhode Island Hospital. Ten adults were recruited to participate from two hospital-affiliated clinics in Rhode Island that offer buprenorphine to treat OUD. Data was collected from October 2020 to September 2021. Participants were eligible for the study if they were over 18 years of age, met the Diagnostic and Statistical Manual of Mental Health Disorders (Fifth Edition) 42 criteria for OUD per treating provider, initiated buprenorphine within the past 8 weeks, and endorsed having access to a cell phone with text message capability. Exclusion criteria included: active suicidality and/or psychosis that would interfere with the ability to participate in the intervention; not fluent in English; limited mental capacity or inability to provide informed written consent. Participants were between the ages of 25 and 68 (Mage = 40.2, SD = 13.5), predominately male (n = 8), non-Hispanic (n = 7), White (n = 7), and living with a spouse or partner (n = 5). Four of the participants had engaged in treatment prior to the current episode, with detox (n = 3) and residential (n = 3) endorsed as the most attended programs. One participant reported previously using online support for OUD. All participants reported having access to a smartphone with an unlimited text messaging plan and provided informed written consent. See Table 1 for an overview of sample characteristics.
Participant demographics at baseline.

N = 10. Participants were on average 40.2 years old (SD = 13.5).
As a result of the COVID-19 pandemic, “routine clinic visits” involved virtual and in-person appointments, thus the majority of recruitment and enrollment procedures were conducted remotely. To be in compliance with the recruitment site's COVID-19 safety measures, study recruitment efforts relied on provider referrals and limited contact with the few individuals being seen in-person, posing a challenge to enrollment numbers and attrition from initial contact (socially-distanced, in person) to the baseline assessment (remotely). See Figure 1 for an overview of study recruitment, enrollment, and follow-up procedures. The acceptability, feasibility, and preliminary efficacy results presented below are based on 10 participants who completed the intervention.

Overview of study procedures.
Enhancing distress tolerance to uplift motivation in recovery intervention
The two-part enhancing distress tolerance to uplift motivation in recovery (iENDURE) intervention focused on enhancing motivational processes in combination with strategies for tolerating distress through a single, brief, web-based session followed by 8 weeks of personalized text messages.
Web-based intervention
The initial intervention phase was delivered online via Qualtrics, a web-based survey platform. The goals of this phase were as follows: (a) engage participants in a decisional balance exercise to evaluate the perceived advantages/disadvantages of making a behavior change, related to their opioid use, to enhance motivation and (b) provide concrete strategies to better tolerate emotional and physical discomfort to persist with behavioral goals while initiating treatment. Throughout the web-based intervention, participants were presented with predetermined lists of ‘pros’ and ‘cons’ for discontinuing opioid use and initiating treatment, from which they self-selected personally relevant items or wrote in personal motivators. Additionally, individuals were prompted by the intervention to enter their short-term and long-term recovery goals; examples were provided for guidance. Lastly, participants were presented with psychoeducation on DT-based coping strategies, including distraction, self-soothing, changing body chemistry, evaluating pros/cons, and improving the moment. Similarly, participants were encouraged to select strategies that were most useful/relevant to their recovery. Participant responses were then summarized and embedded within future text messages. See Figure 2 for an example of the web-based intervention.

Examples from web-based iENDURE intervention.
Text message-based intervention
The second intervention phase consisted of daily text messages, delivered in partnership with Marigold Health, over a span of 8 weeks (56 days receiving messages), which focused on (a) promoting motivation for remaining in treatment and maintaining recovery and (b) providing adaptive skills for tolerating emotional and physical discomfort. Each day during the text-message intervention period, participants were provided with a morning message emphasizing their personal reasons, derived from the decisional balance exercise, to persist with their goals. This was followed by a midday “check-in” assessing for participants’ mood and presence of cravings. Participants were queried (a) “How are you feeling in general today?” and prompted to respond on a Likert scale, ranging from 1 to 5 (1 = really bad, 5 = great) and (b) “Any cravings or urges to use in the past 24 h?” and prompted to respond with “yes” or “no.” Immediately following the response, participants received a DT skill recommendation tailored to their mood/cravings assessment, which was selected from the participant-generated pool, The automated message schedule provided a reminder after two hours if no engagement with the intervention was detected and delivered recommendations for low mood/yes cravings if no response was captured within a four-hour response window. Additionally, the intervention consisted of a participant-driven text feature to provide tailored support. Participants were educated about this component at the time of enrollment. For example, if a participant texted “USE” to the automated program, indicating the need for extra support following an episode of returning to opioid use, they would receive a relevant skills-based message. On-demand keywords to obtain additional support available to participants also included “CRAVING,” “MOOD,” “GUILT,” and “PAIN.”
Measures
Appropriate copyrights and necessary permissions for all applicable tools/questionnaires in the study had been obtained prior to data collection.
Substance use measures. The online Timeline Follow-Back 43 was implemented to capture self-reported daily opioid use, use of other illicit substances (e.g., cocaine), and buprenorphine adherence. Results of urine toxicology screens were collected during routine clinical care, made available through the electronic medical record (EMR), and were used to (a) screen for the presence of opioids and other illicit substances and (b) confirm adherence to buprenorphine. Retention in treatment services was captured via a retroactive EMR review.
Readiness ruler (RR). The RR is a single question assessing the current level of motivation to change opioid use, indicated by selecting a value between 0 and 10 (Please indicate to what extent you feel ready to change your opioid use.). The RR in the present study was adapted from the LaBrie et al. 44 RR, which was validated to briefly assessing motivation to change drinking behavior.
Confidence scale. This item assessed participant's confidence in meeting their recovery goals. Participants were prompted “Please indicate to what extent you feel confident in reaching your goals for treatment” and were expected to respond on a 11-point Likert scale, ranging from “0” = “I do not think I will reach my goals” and “10” = “I think I will definitely reach my goals.” Higher score indicated greater levels of confidence in meeting goals.
Opiate Craving Scale (OCS). The OCS is a 3-item measure of craving that was delivered to capture a current desire to use opioids. The first item asks participants directly about their current opiate cravings. The second and third items prompt participants to rate their opiate cravings after imagining themselves in an environment that may trigger a desire to use. Participants are expected to respond on an 11-point Likert scale, ranging from “0” = “Not at all” and “10” = “Extremely.” Higher scores indicate greater presence of cravings. The Craving Scale was validated for use in people with substance use disorders, and with opioid use. 45
DT Scale (DTS). The DTS is a 15-item self-report measure of one's ability to tolerate psychological distress. 20 Items are rated on a 5-point scale ranging from “1” = “Strongly agree” to “5” = “Strongly disagree. Low scores on the DTS indicate that an individual can tolerate higher levels of distress. Higher scores reflect low DT (i.e., distress intolerance). The scale has been found to demonstrate good internal consistency, good test—retest reliability, and discriminant validity with measures of negative affect. 46
Positive and Negative Affect Scale (PANAS). The PANAS is a 20-item mood scale which evaluates the extent to which participants’ experience positive and negative affectivity over the past week. The scales are highly internally consistent, largely uncorrelated, and stable over a 2-month period. 47 Items are divided to calculate a “Positive Affect Score” and “Negative Affect Score.” Adding items 1, 3, 5, 9, 10, 12, 14, 16, 17, and 19, scores can range from 10 to 50; higher scores representing higher levels of positive affect. Adding items 2, 4, 6, 7, 8, 11, 13, 15, 18, and 20, scores can range from 10 to 50; higher scores representing higher levels of negative affect.
Feasibility and Acceptability. We collected acceptability of the current intervention through the Client Satisfaction Questionnaire (CSQ-8). The CSQ-8 was validated within residential substance use treatment settings. 48 Feasibility was evaluated through (a) retention across the study period, including percent of participants who completed assessments, (b) engagement with the text messages, including participant reports of interacting with/reading messages and average response rates, and (c) perceived usability of the intervention via the System Usability Scale (SUS). The SUS has a valid measure to assess the usability of internet-based interventions when used by professionals in mental healthcare. 49
Qualitative interviews. Research staff conducted interviews over the phone or in-person (when possible) at the end of the 8-week intervention period. The purpose of these interviews was to elicit feedback on perceived acceptability of the intervention as well as ways to improve/refine intervention content and delivery for the subsequent phase of the study involving a randomized clinical trial.
Data analysis
This pilot study sought to evaluate the feasibility and acceptability of the two-part iENDURE intervention. Preliminary clinical outcomes included: opioid use, adherence to buprenorphine, opioid-related craving, treatment attendance and retention, readiness to change opioid use, confidence in the ability to achieve treatment goals, and tolerance of distress. Additionally, changes to mood (degree of positive and negative affect) were assessed as secondary outcomes. Qualitative data analysis was conducted using the Framework Matrix approach, 50 a structured process to categorize qualitative data by thematic (deductive) and data driven (inductive) codes, such that highly relevant and emergent concepts can be easily captured and compared across participants. All interviews were coded separately by two research assistants, who met regularly to establish code concordance. Any discrepancy was resolved by consultation with the Principal Investigator.
Results
Feasibility
Participants (n = 10) were administered in-person or remote follow-up assessments at 1-, 4-, and 8-weeks following enrollment in the text message component of the intervention. Seven participants completed the 1-week assessment, while 9 individuals were retained for the 4- and 8-week assessments and closing interviews. As indexed by self-report in the qualitative exit interviews, 100% of retained participants (n = 10) engaged with the text messages throughout the 8-week period. While participants received compensation for completing follow-up assessments, no incentives were administered for text message responsiveness; indicating that engagement with the iENDURE program is consistent with natural response behaviors. The level of engagement, however, varied across individuals. Analysis of text data indicated that the intervention daily response rate averaged 66.6% (SD = 34.6). While six participants responded to more than 75% of messages and two participants responded to more than 55%, two participants engaged with less than 10% of messages. During the exit interviews, the participants with the lowest response rates were prompted about barriers to engaging with the intervention. Participant 3 endorsed reading all messages, despite not responding to the check-in for current mood and cravings. Additionally, the participant admitted to not being interested in supportive resources outside medication, providers, friends, and family. This participant suggested that the iENDURE program would “benefit someone without a good support system.” Participant 7 attributed their lack of responsiveness to not having access to their cellphone throughout the workday: “Just not having my phone next to me at work. I don’t really keep my phone on me at work because it will get destroyed.” The average rating on the SUS at the end of the 8-week program was 65.3 (possible range = 0–100), suggesting that iENDURE was relatively easy to use.
Acceptability
Table 2 provides a summary of the questions and responses regarding satisfaction with various components of iENDURE. Global ratings for the quality of services and satisfaction with the program were high in participants relating to both the web-based intervention and following the 8-weeks of text messages. One participant negatively reported on iENDURE. However, this participant, nonetheless, indicated they would recommend the program to a friend. Otherwise, participants uniformly responded that iENDURE helped manage substance use more effectively and that they would come back to iENDURE if they decided to seek help again.
Participant satisfaction with enhancing distress tolerance to uplift motivation in recovery (iENDURE).
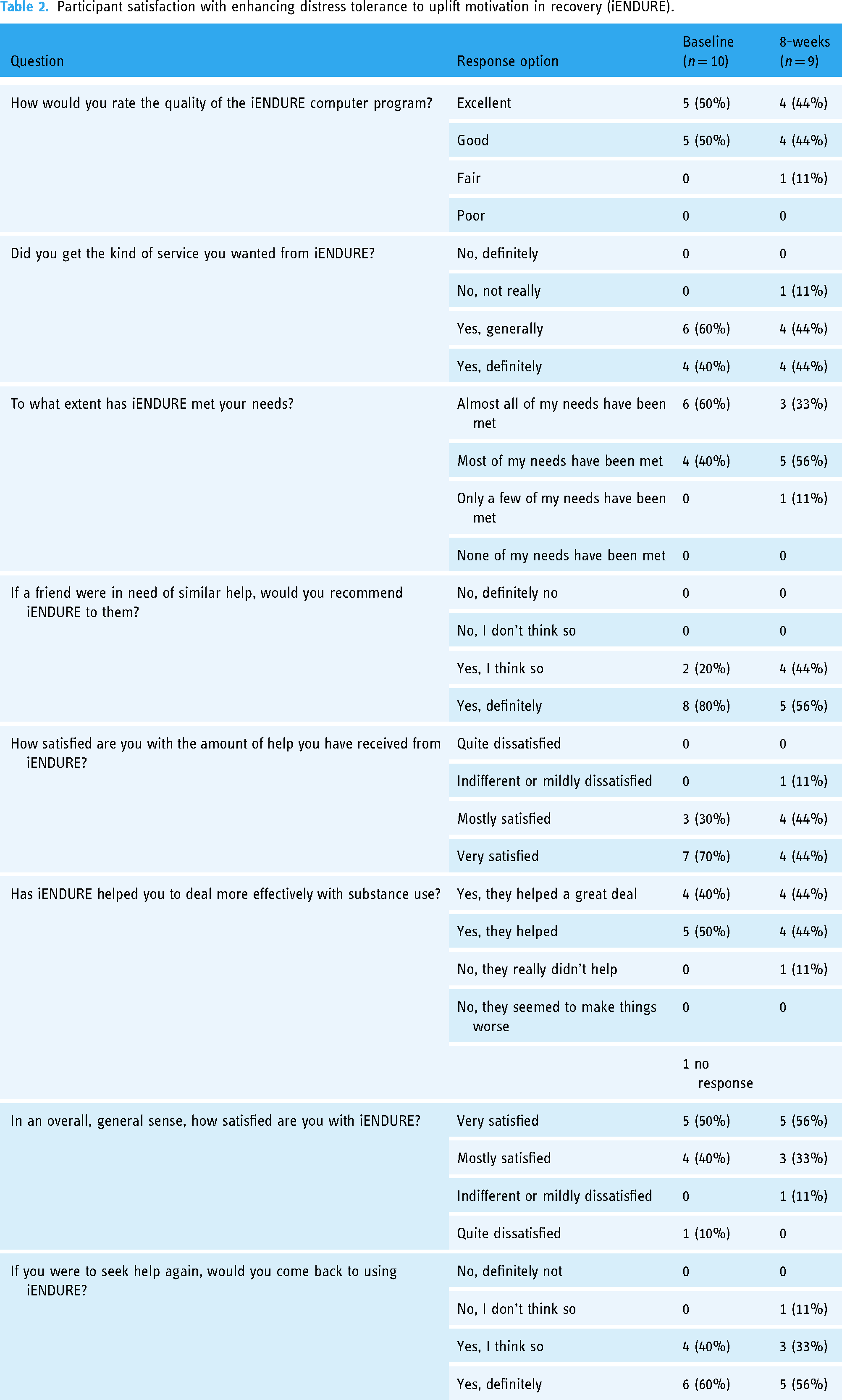
The mean score on the CSQ-8 was 28.6 (SD = 2.55) following the web-based intervention and 27 (SD = 5.05) at the end of the text message program. Scores on the CSQ-8 range from 8 to 32, indicating a high degree of satisfaction with the intervention. Overall, participants endorsed positive experiences with the intervention content. Specifically, following the delivery of each text message, participants were asked to rate their satisfaction with the content on a 1–5 scale (1= “very dissatisfied,” 3= “neutral,” 5= “very satisfied”). Across the 8-week period, morning messages were, on average for each participant (n = 10), rated as 4.10 (SD = 0.76) and afternoon messages were rated as 4.22 (SD = 0.84), suggesting that participants were highly satisfied with the content delivered. This sentiment was supported by qualitative interviews with participants noting that the intervention was “…a positive experience. It kept me focused and motivated on the task at hand,” and “it's helped with my overall, … Supporting me … It was like some of my armor when I'm going into battle.”
Preliminary outcomes
To assess the impact of the intervention on treatment engagement and adherence, self-report (Timeline Follow-Back) and medical record data were utilized to examine compliance with buprenorphine, rates of opioid use, and treatment attendance. Of the 9 people who were retained at the 4-weeks follow-up period, 100% reported buprenorphine adherence and 8 individuals reported abstinence from opioids. At the end of the intervention, at the 8-week follow-up assessment, 7 participants reported buprenorphine adherence and 8 participants reported abstinence from opioids. Regarding treatment retention, 8 out of 10 participants had scheduled appointments at the 4-week follow-up, and 7 out of 10 participants had active appointments scheduled at the 8-week follow-up.
Table 3 provides the means and standard deviations of self-reported outcome variables. Participants reported high motivation for change and high confidence in their ability to reach their goals at the start of the intervention (RR: M = 8.40, SD =1.96; Confidence Scale: M = 8.20, SD = 1.99). One week following enrollment, participants reported, on average, slightly lower motivation for change and lower confidence in the ability to reach goals (RR: M = 7.86, SD = 2.27; Confidence Scale: M = 6.67, SD = 2.42). However, by the end of the 8-week intervention, participants overall motivation and confidence increased (RR: M = 8.88, SD = 1.46; Confidence Scale: M = 8.22, SD = 1.99). Participants’ cravings, as indexed by the OCS, decreased from the start of the intervention (M = 5.33, SD = 2.88) to week 1 (M = 3.67, SD = 1.91), and decreased further by week 4 (M = 3.24, SD = 2.23). By the end of the 8-week intervention, participants overall cravings remained lowered relative to the start of the program (M = 4.33, SD = 3.01).
Assessments.
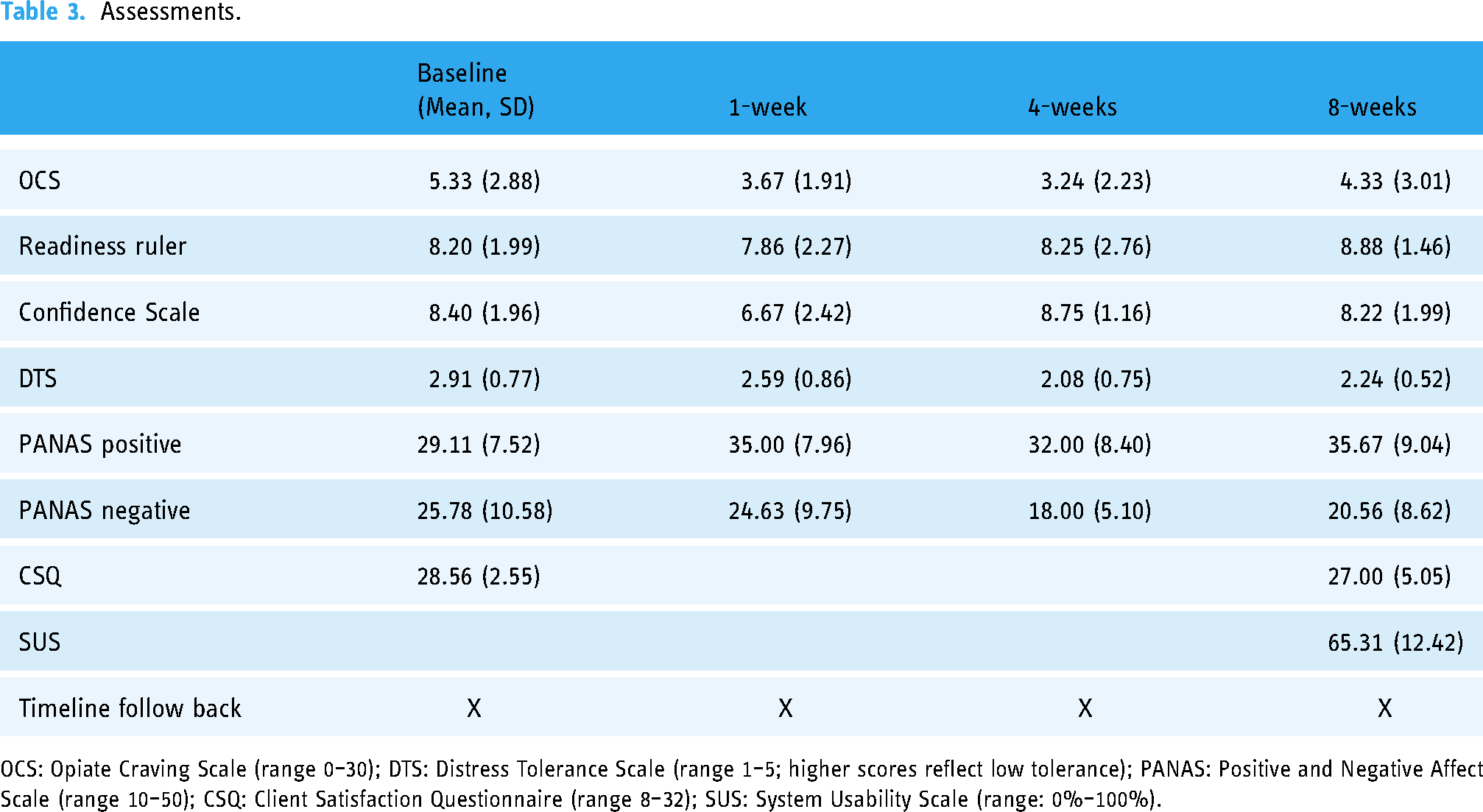
OCS: Opiate Craving Scale (range 0–30); DTS: Distress Tolerance Scale (range 1–5; higher scores reflect low tolerance); PANAS: Positive and Negative Affect Scale (range 10–50); CSQ: Client Satisfaction Questionnaire (range 8–32); SUS: System Usability Scale (range: 0%–100%).
Finally, participants demonstrated an increase in tolerance of distress throughout the first month of the intervention which was evident by a decrease in their DTS scores from baseline (M = 2.93, SD = 1.25) through week 1 (M = 2.51, SD = 1.39) and week 4 (M = 2.20, SD = 1.24) post-baseline. Gains made in the first month of the intervention were maintained throughout the 8-week follow-up period (M = 2.207, SD = 1.14). Participants also evidenced a change in affect across the study period. Specifically, levels of positive affect increased [baseline (M = 29.11, SD = 7.52); week 8 (M = 35.67, SD = 9.04)] and levels of negative affect decreased [baseline (M = 25.78, SD = 10.58); week 8 (M = 20.56, SD = 8.62)] throughout the 8-week intervention.
Discussion
Findings from this pilot development trial suggest that iENDURE, a two-part intervention designed to support buprenorphine treatment for OUD, is, in general, feasible to administer and acceptable to patients engaged in outpatient care. iENDURE differs from other existing treatments for OUD in that it (a) combines motivational enhancement with DT skill building and (b) is delivered through a highly accessible digital health platform. This unique combination of intervention content and delivery method offers patients with OUD continuous access to evidence-based and personalized support.
The iENDURE intervention was not only feasible to administer, but also highly engaging and perceived as easy to use. Nine out of ten (90%) participants were retained in the study throughout the entire 8-week period. Additionally, participants responded to daily text messages at a rate of 66.6%, with more than half of the sample responding to messages at least 75% of the time. Interestingly, participants who did not respond to the daily queries, nonetheless, indicated that they read most of the motivational and coping skill messages. This is noteworthy as meta-analyses suggest that one-way communications are still effective in producing behavior change. 39
Although this study did not involve a comparison condition, nor was it powered to detect effect sizes, we observed trends in the predicted direction for levels of motivation, confidence, cravings for opioids, DT, and positive/negative affect from baseline to the 8-week follow-up assessment. At the conclusion of the study, 7 participants endorsed adherence to buprenorphine and 8 participants reported abstinence from opioids. Further, 7 participants were still actively engaged in treatment at the 8-week follow-up as evidenced by the EMR. Given that recent estimates suggest that nearly 50%–75% of patients who initiate buprenorphine will discontinue treatment within the first three months, 50 our findings provide preliminary support for improved engagement and retention in care.
While this study highlights the feasibility and acceptability of text-based interventions, such research needs to be augmented by findings on ways to improve accessibility to interventions. Specifically, the COVID-19 pandemic and the subsequent adoption of digital technologies in the provision of health services revealed stark inequalities in access to resources required for telehealth such as the internet, reliable cellphone data, or cell phones.51,53 Research that develops digital intervention without addressing these systemic barriers will continue to exclude a significant portion, and often the most vulnerable, portions of populations from accessing care. Two aspects of the current study augmented its feasibility. Firstly, text messaging-based interventions, as opposed to cellphone application-based interventions (or apps), are accessible across various types of cellphones (e.g., smartphones, burner phones, older models) and digital literacy. Thus, research delivering intervention via text messages may have a wider reach. Secondly, incorporating this research program into standard care received by participants provided opportunities for human touch points, which then bolstered retention. For example, if a participant obtained a new cellphone number, research staff were able to collect this information during the participant's treatment visit.
There are several limitations to the current study. First, our sample was limited regarding gender, racial, and ethnic diversity as well as overall sample size. Future trials, with larger sample sizes and adequate representations of gender, racial and ethnic minorities, are needed to determine whether iENDURE is feasible and acceptable to the general population, especially those from underrepresented backgrounds. Second, given that this was a pilot development trial, the present study did not employ a control group. Thus, it is difficult to determine whether the clinical improvements observed throughout the study period, were indeed uniquely related to the iENDURE intervention or related to initial engagement in treatment for OUD, given that all participants had recently enrolled in MOUD program. Relatedly, given the study was focused on assessing feasibility and acceptability, sample size was not calculated. To fully evaluate the effectiveness of iENDURE, future work should utilize a randomized controlled trial design that incorporates an appropriate control condition and involves apriori calculation of sample size. Thirdly, we were unable to assess demographic differences among those who agreed to participate in the intervention at baseline and those who did not. Such differences would be important to investigate in future studies in order to make improvements in the intervention. And, finally, many of the studied variables were assessed via self-report. While an attempt was made to increase scientific rigor through objective verification of certain outcomes via the EMR, there is still the possibility of recall bias.
Conclusion
In conclusion, the results obtained from this pilot study demonstrate promising trends in the feasibility and acceptability of a combined web- and text message-delivered intervention in improving adherence to and engagement in buprenorphine treatment for OUD. Our sample was comprised of patients actively engaged in buprenorphine treatment in an outpatient setting. While preliminary, our data suggests that when combined with treatment as usual, incorporating motivational enhancement and distress tolerance skills training via a digital health platform, may hold promise in improving treatment outcomes for individuals with OUD.
Footnotes
Contributors
Prachi H. Bhuptani contributed to the conceptualization of study, data analysis, and writing (original and revised draft) of the manuscript. Amanda Block and Paola Jiménez Muñoz contributed to the data analysis, writing (original and revised draft) of the manuscript. Mariel S Bello contributed to the writing (review and editing) of the manuscript. Susan Ramsey, Megan Ranney, Kate Carey, and Josiah Rich contributed to the conceptualization of study, writing (review and editing). Kirsten Langdon contributed to the conceptualization of study and writing (original and revised draft).
Conflict of interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study received approval from Rhode Island Hospital's Institutional Review Board (1777545-7).
Funding
The authors received the following financial support for the research, authorship, and/or publication of this article: This work was supported through grants from the National Institutes of Health (NIH, PI: Langdon, K23DA046482) and the National Science Foundation Graduate Research Fellowship (NSF, PI: Bello, DGE-1418060). The content is solely the responsibility of the authors and does not necessarily represent the official views of the CDC, NIH, or NSF.
Guarantor
K.L.
