Abstract
Objective
Continuous monitoring and targeted behavioral interventions have been shown to improve health status and quality of life for heart failure patients. Digital therapeutics offer the possibility to make more frequent monitoring and targeted behavioral interventions available for more people.
Methods
We conduct a pilot study with 71 patients who were given a smartphone app and wearables for a 3-month period. Clinical indicators as well as patient-reported outcomes were collected at entry and exit examinations.
Results
The New York Heart Association class remained stable or improved. Most quantitative outcome measures improved (6-minute walk test distance + 21 m, Kansas City Cardiomyopathy Questionnaire summary score + 6.0 points, European Heard Failure Self-care Behavior Scale summary score + 6.6 points, correct answers in the Atlanta Heart Failure Knowledge Test + 2.1), although the changes were mainly not significantly different from zero. There was no change in EQ-5D weight and 9-item Shared Decision-Making Questionnaire summary score.
Conclusions
This before–after comparison shows that an app-based intervention can work as a digital therapeutic for heart failure patients.
Introduction
Worldwide, heart failure (HF) is a widespread disease in the population, with an increasing prevalence, also due to an ageing population.1–3 Poor self-management of the condition often leads to hospitalizations and generally increases costs to health systems. 4 Modern technology can support patients with tools to support lifestyle changes, improve patient knowledge about medical conditions, enhance self-care behavior and guide individual therapy decisions and may hence improve health and avoid costs.5,6 In the following study, we present results from the first empirical assessment of a new application to support patients suffering from HF—the ProHerz app. Key elements of the app—installed on patients’ smartphones or tablets—are easy-to-use monitoring of medical parameters for early detection of disease progression as well as self-help features to guide patients. The research on digital health apps and telemonitoring designed for patients with HF has grown in the past decade. However, these studies are heterogenous, both in terms of what functionality the digital interventions provide as well as outcome measures used to assess their effectiveness. 7
App-based digital therapeutics mainly aim to improve patients’ general health status or the HF condition in particular. Previous research shows mixed results. A popular outcome measure, the New York Heart Association (NYHA) classification for patients with HF, improves for some interventions, does not deteriorate for others but sometimes remains unchanged compared to a control group.8,9 Oxygen uptake, another objective health indicator, can improve with digital interventions. 9 For other objective health status measures like body mass index (BMI) and blood pressure, most studies find an improvement in the treatment group compared to the control group, in some cases, however, small in magnitude.10–13
Effectiveness of app-based digital therapeutics has also been analyzed with more subjective outcome measures like quality of life (QOL). Some randomized controlled trial (RCT) studies find that using targeted applications significantly increases QOL12,14 for patients with HF while others reject a significant improvement. 10 Before–after comparisons also suggest that app-based interventions achieve increases in QOL, 13 whereas studies tracking measurements via web-based tools do not show such effects.15,16
Next to objective health measures and QOL indicators, self-care and self-management behavior are increasingly considered as important outcomes. For patients with HF, self-care and self-management can reduce rapid deterioration and hospitalizations. Results from previous studies targeting these outcomes with app-based interventions are however ambiguous. While some report an overall positive impact of targeted interventions on self-management, 10 others find only minor changes in the overall patient self-care behavior. 11
In summary, previous research shows that improvements in QOL are possible but the specific design of the intervention and the design of the application is crucial. This shows again that the comparison between different studies is difficult since the base of the analyses, the specific design of an app, is varying.
The evidence on the effectiveness of apps for patients with HF is not conclusive yet, but previous work shows that improvements in health status, QOL, and other patient relevant outcome measures are possible. In this article, we provide results from a pilot study to assess the effectiveness of a new app-based intervention for patients with HF, the ProHerz app. The pilot study is based on a small sample of 71 patients who used the app for 3 months with our goal not only to identify first signs of the interventions effectiveness but also to gain information on administrative workflows and patient compliance in an evaluation of a digital therapeutic for patients with HF. The analysis we present here is based on before–after comparisons for several target outcomes which cover health status, QOL, and patient relevant other factors. This study proceeds as follows: in the next sections, we first introduce the app as a digital therapeutic and then the study design and our evaluation approach. After the presentation of the results, we conclude and summarize the most important implications from the ProHerz pilot study.
The ProHerz app
The ProHerz app is a software application for patients suffering from HF. It aims to provide positive care effects to the patients, in particular improved health status and QOL as well as increases in health literacy, self-care behavior, and better involvement of patients in medical decisions. The digital diagnostic component of the app consists of regular measurement and documentation of relevant vital signs, visualization of the vital parameters, and personalized recommendations and educational lessons. Additionally, the app allows for archiving and easy access to medical documents and daily medication reminders. Vital signs collected by the app are blood pressure, heart rate, blood oxygen saturation, body weight, and temperature. These measurements are collected either by medical devices via Bluetooth or by manual patient input. Regular screening of vital signs offers a more detailed surveillance than the current standard of care in many countries, including Germany. The measurements—graphically visualized and presented to the patient with additional information in an easily comprehensible way. As a behavioral intervention, the app provides instant and regular feedback on changes to the health status (in traffic-light color coding for easy understanding) and targeted instructions for potential improvements as well as background information on the different parameters. This information enables patients to realize the effects of lifestyle changes and incentivizes them to adjust their behavior accordingly. Following detection of extremely deviating patterns according to medical guidelines (e.g. for signs of decompensations), the digital therapeutic component of the app advises patients to seek medical advice in early stages of disease progression and incentivizes behavioral changes. The app also regularly tracks patient well-being and keeps track of possible comorbidities. In addition, the app offers a platform where all medical documents are saved and where information can easily be shared with health providers. Finally, the app sends reminders for medications and patients can track their medication plan. Two screenshots from the patient view of the ProHerz app are shown in Figure 1.

User view of the ProHerz app.
Evaluation
This study was registered at the German Clinical Trials Register under ID DRKS00024798. Patients were included in the study in April and May 2021. The follow-up examinations took place in August 2021. Inclusion criteria were a diagnosed HF (ICD-10 Code I50), age over 18 years, the ability to speak and read German, the ability to use a smartphone or another portable device and the consent of the study participant. Exclusion criteria were pregnancy and terminal kidney failure (ICD-10 Code N18.5). Staff of the Cardiovascular Center at Klinikum Nuremberg performed a screening of suitable inpatient and outpatient participants. Suitable patients were contacted and informed about the study. Additionally, participants were recruited on site when a suitable patient visited. After a conversation with a physician, patients gave their written consent to participate in the study. In total 71 patients were included in the study. In the entry examination, a physician collected patient characteristics (sex, age, BMI, comorbidities from the Elixhauser index, smoking status and whether the patient lives on their own) into the study's electronic clinical research files. Next, patients filled in a set of questionnaires on a dedicated tablet and conducted a 6-minute walk test. After these initial data collections, the patient was introduced to the ProHerz app and received Bluetooth devices for the regular measurements (blood pressure device, pulse oximeter, scale, body thermometer). After 3 months, the follow-up appointment was scheduled and a second examination took place, again collecting data in the form of questionnaires, and the 6-minute walk test. Two physicians performed the entry and exit examinations in this study but there was no rule that the same physician performed both examinations for one patient.
The focus of this study is to evaluate how measures that indicate the effectiveness of the digital therapeutic in terms of improved health status, QOL, self-care behavior, health literacy, and general involvement of patients in medical decisions change after 3 months of using the app. The detailed measures for the outcomes are the following:
Change in the NYHA classification. This is a scheme to classify the severity of HF from class I, the least severe condition to class IV, the most severe condition.
17
Differences in health status are evaluated as the change in distance walked in meters in the 6-minute walk test. The 6-minute walk test is a widely used measure of physical performance, where larger distances indicate better health status.
18
Change in EQ-5D-L index compared before and after the intervention. The EQ-5D-L questionnaire addresses health-related QOL and has been validated for Germany.
19
Its values can range from 0 to 1 where larger values indicate higher QOL. Change in Kansas City Cardiomyopathy Questionnaire (KCCQ) index. The KCCQ is a measurement tool validated in German to assess QOL for patients with chronic HF.
20
A summary scale from the 12 different questions is transformed to range between 0 and 100, where higher values indicate higher QOL. Change in the 9-item European HF Self-care Behavior Scale (G9-EHFScBS). The G9-EHFScBS is a health behavior questionnaire validated in German for patients with HF.
21
The summary scale from nine different questions is transformed to range between 0 and 100, with higher values indicating better self-care behavior.
22
The 9-item Shared Decision-Making Questionnaire (SDM-Q-9) score, questionnaire for evaluating patient participation during treatment.
23
A summary scale from the nine different questions is transformed to range between 0 and 100, where higher values indicate better participation in the decision making process. Atlanta Heart Failure Knowledge Test (AHFKT), validated knowledge test on HF.
24
A larger number of correct answers at the 30 questions indicates better health competency.
Sample description
In total, 71 patients took part in the entry examination and 64 completed both examinations of the trial (7 patients did not show up for the exit examination). Mean patient age at baseline was 61.81 years and 67.2% of the patients were male. Patients had an average BMI of 30.44 kg/m2 and the average Elixhauser score based on the van Walraven algorithm 25 was 10.62 where possible values range from −19 to 89 and higher values indicate more severe comorbidities. Approximately 29.7% of the patients lived in a single household and 21.9% were smokers. Most recruited patients fall in the NYHA classes II and III, the medium severity classes for HF. An overview of the descriptive statistics is presented in Table 1.
Descriptive statistics.

Descriptive statistics for the 64 subjects who completed both examinations out of the 71 who were included in the trial.
BMI: body mass index; NYHA: New York Heart Association; SD: standard deviation.
Evaluation results
As a first step, we analyze how health status changed from entry to exit examination. Figure 2 shows how the NYHA class of our sample changed. Patients who were categorized in the NYHA class I in the entry examination (the least severe HF) all kept their classification after the intervention. Among all patients in the NYHA class II at the entry, 42% improved their status and were categorized in class I in the exit examination. In total, 58% remained in the NYHA class II. Patients with the NYHA class III HF in the beginning either remained in class III (43%) or improved their health condition (6% to class I and 51% to class II). Among the patients with the most severe HF at the entry examination (NYHA class IV), 50% improved to class III, 50% remained in class IV. A chi-square test for equal distribution of the NYHA class supports the change from entry to exit examination (χ2 = 19.5, df = 3, p < .001). In summary, no patient was grouped worse after the intervention than before—all patients either maintained their NYHA class or experienced an improvement.

Changes in New York Heart Association class from entry to exit examination.
In Table 2, we present the outcome measures and how they changed between entry and exit examinations. The average distance walked in the 6-minute walk test increased by 21.2 m from a baseline of 377 m. This difference is however not significantly different from zero. There was almost no change to the average EQ-5D utility weights, our first QOL indicator. Average EQ-5D utility weights stayed at the baseline value of 0.84. Our second QOL indicator, the KCCQ summary score increased on average from 53 to 59, although the increase was again not significantly different from zero. The EHFScBS summary score for self-care behavior increased significantly from 63 to 70 over the study period. Overall patient participation in medical decision making measured by the SDM-Q-9 summary score did not change significantly and remained on average at 64 points. The average number of correct answers in the 30 question AHFKT increased statistically significantly from 16 to 18. In Figure 3, we provide an overview of the distributions of all outcomes in the entry and the exit examination. These distributions indicate that in particular for the KCCQ and EHFScBS scores and the correct answers in the AHFKT a lot of probability mass is shifted upwards from the very low scores while the increase in the 6-minute walk test distance is distributed over the full outcome range.

Distribution of outcomes at entry and exit examination. AHFKT: Atlanta Heart Failure Knowledge Test; EHFScBS: European Heard Failure Self-care Behavior Scale; KCCQ: Kansas City Cardiomyopathy Questionnaire; SDM-Q-9: 9-item Shared Decision-Making Questionnaire.
Change in outcome measures over time.

p-values from two-sided t-tests.
AHFKT: Atlanta Heart Failure Knowledge Test; EHFScBS: European Heard Failure Self-care Behavior Scale; KCCQ: Kansas City Cardiomyopathy Questionnaire; SDM-Q-9: 9-item Shared Decision-Making Questionnaire.
In addition to the data collected in the entry and exit examinations, we also recorded how often patients used the app to store measurements in the app during the study period. Hence, in contrast to other medical studies, a direct measure of take-up is available. Therefore, we can compare the effects of the app on outcome measures between patients with high and low usage. Previous research shows that higher usage intensity is correlated with larger improvement. 10 Our usage measure is the average recorded measurements per day of a patient (heart rate, temperature, oxygen saturation, weight). On average, the patients document 3.86 measurements per day. Figure 4 shows the distribution of the usage index in more detail. We observe that most of the study participants recorded between 3 and 6 measurements on average a day and that the app was used actively among all patients with a minimum of 1.75 recorded entries per day.

Distribution of app usage—average number of measures (heart rate, temperature, oxygen saturation, weight) taken per day.
Next, we analyze whether usage intensity affects the change in patient outcomes. We perform t-tests on the differences in the changes in the outcomes from entry to exit examination between patients that used the app more or less compared to the median. The results in Table 3 show that the distance walked in the 6-minute walk test increased significantly more in the high usage group. In contrast, the EHFScBS summary score mainly improved in the low usage group. All other changes in outcome measures do not differ between the usage groups.
Difference in changes in outcome measures between entry and exit examination for above and below median use intensity.
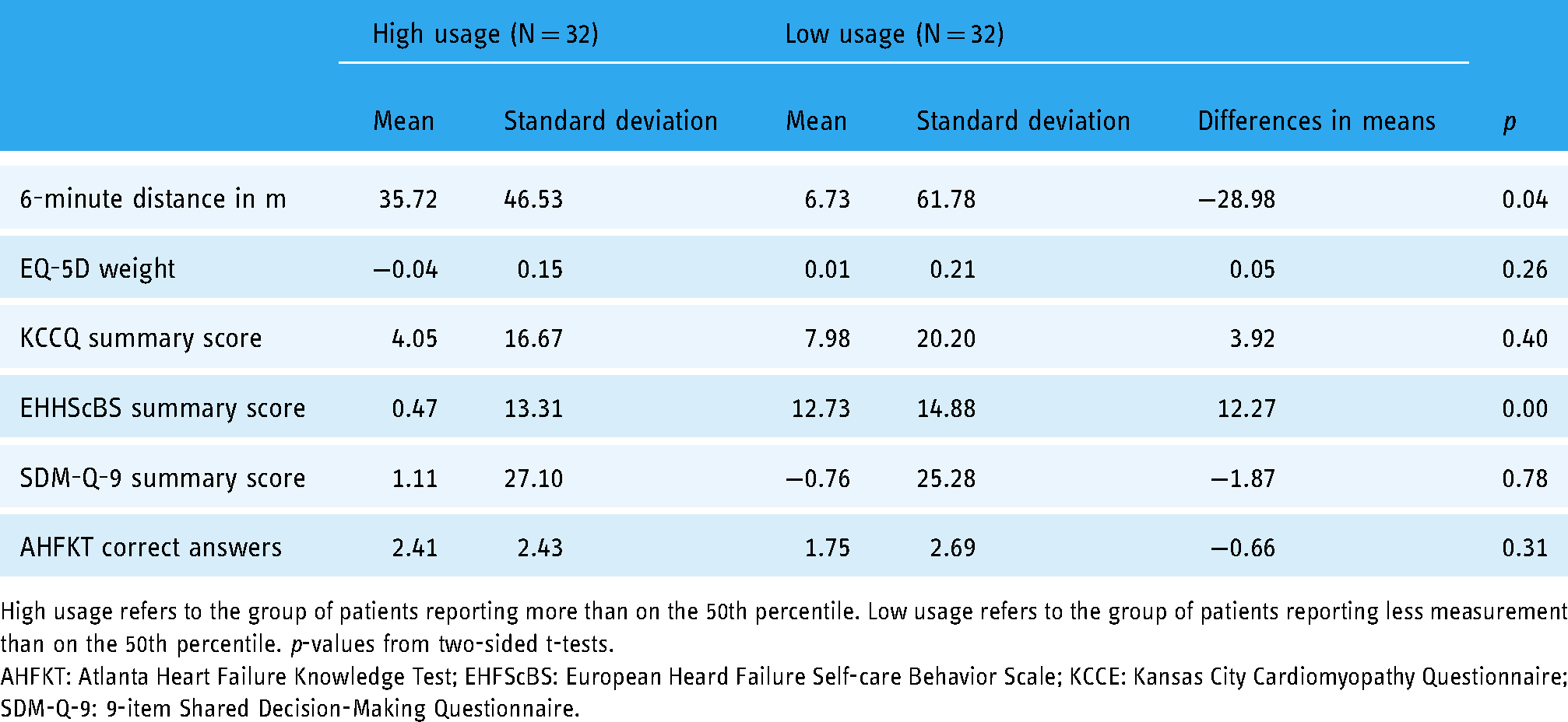
High usage refers to the group of patients reporting more than on the 50th percentile. Low usage refers to the group of patients reporting less measurement than on the 50th percentile. p-values from two-sided t-tests.
AHFKT: Atlanta Heart Failure Knowledge Test; EHFScBS: European Heard Failure Self-care Behavior Scale; KCCE: Kansas City Cardiomyopathy Questionnaire; SDM-Q-9: 9-item Shared Decision-Making Questionnaire.
Discussion and conclusion
In this article, we present empirical results from a pilot trial that evaluates ProHerz app, a smartphone application designed to support patients with HF. All patients in this study received the intervention (access to the application) for 3 months. Our study shows that it is possible to evaluate the effects of a digital therapeutic for patients with HF, both from an administrative point of view as well as in terms of patient compliance. Seven out of the originally 71 patients recruited for the study dropped out (did not show up for the exit examination), an insight which helps to power further studies on the topic. To get an idea of the benefits of the intervention, we estimate the effect as the change in outcomes from entry to exit examination. Our results show improvement of patients’ NYHA class, an indicator for HF severity in the exit compared to the entry examination. Patients’ distance walked within 6 minutes, a measure of physical health, increased, although not significantly. General QOL measured by EQ-5D-L did not change while the disease-related QOL measured by the KCCQ did increase. Furthermore, the self-care of patients as well as their health literacy improved significantly after the 3-month intervention while perceived patient participation in the medical decision-making process remained unchanged. Hence, for most areas targeted with the intervention (health status, QOL, self-care of patients as well as health literacy) the results of the pilot trial are a clear indication for positive effects of ProHerz. Not all of the positive changes are significantly different from zero and all of the differences are quantitatively smaller than what is presented in previous studies. Those previous studies, however, mainly use a longer intervention period and a superior control group design in comparison to our explorative study. Additionally, we find small and often insignificant differences in the moderating effect of app usage intensity on changes in outcomes. This pattern could partly be explained by the overall high usage rates in our sample. Additionally, patients may select into the right intensity of usage themselves to meet their needs. Finally, there is the general concern that motivated patients self-select into the use of digital therapeutics (or studies on their effectiveness) which would lead to overestimations of the true benefits. As a next step, a prespecified RCT with a larger sample and a longer intervention period is needed to credibly identify causal effects. Such an RCT can be designed with the parameters from this pilot in mind and will then allow to separate changes in the outcomes unrelated to the intervention from the true causal effects of using ProHerz.
Footnotes
Acknowledgments
We would like to thank Georg Zweyer for implementing the e-CRF tool and Sophie Terheyden as well as Till Gladow for excellent project management and valuable comments. The publication of this article was funded by the Open Access Fund of the Leibniz Association.
Contributorship
SR, FH, TF, MP, and JK conceived the study. SR, SS, and JS researched the literature. SR and SS conducted the data analysis and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TF received compensation and honoraria from lecturing and advising by LivaNova/Sorin and BioStable. JK works on advisory boards in the field of healthcare technologies and digital health of different associations of medical professionals, industries, and political authorities. In the past 5 years, JK received compensation and honoraria from lecturing and advising by Medical Valley Digital Health Application Center; RoxHealth, UCB, Desitin, Bial Deutschland; Celgene, Lundbeck-Foundation, Novartis, EverPharma; JK holds shares of Portabiles HealthCare Technologies, Portabiles, Alpha-Telemed. JK has a patent related to gait assessments pending
Ethical approval
The ethics committee of the Bavarian medical association (BLAK) approved this study (REC number: 21022).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financed by ProCarement GmbH, the developers of the ProHerz App evaluated in this article.
Guarantor
SR.
