Abstract
Background
Currently bubble continuous positive airway pressure (bCPAP) is commonly used in low resource settings to treat respiratory distress. However, the accumulation of condensate in the patient's exhalation limb during operation could significantly increase pressure delivered to the body, which can lead to severe respiratory failure in the infant. The objective of this research was to develop a novel low-cost bCPAP device that can monitor and control the pressure delivered to infants.
Methods
When the neonate expires, the pressure sensor inside the expiratory limb measures the instant positive end-expiratory pressure. The microcontroller decides whether to turn the relay to switch the path of expiration between the two expiratory tubes connected to the valve outlets. This depends on the pressure reading and the cutoff pressure value inserted by the physician.
Results
The system was tested for accuracy, safety, cost, ease of use, and durability. The prototype was accurate in eight iterations at eight different depths of water that were made to monitor and control the pressure. It was safe and provided suitable pressure for the neonate, and the prototype was built in less than 193 USD.
Conclusions
The performance testing of the device demonstrated accurate and safe control and monitoring of continuous positive air pressure (CPAP) and oxygen levels with humidity levels safe for infants. The device provides humidified, blended, and pressurized gas for the patient. It allows physicians to easily monitor and control the accumulation of condensate in the exhalation limb of the CPAP machine accurately and safely.
Keywords
Introduction
Due to the various diseases that neonates face, the period from birth to the first 28 days is the most hazardous period of life.1,2 Globally, in 2017 alone, an estimated 6.3 million children died and nearly half (47%) of the under-five deaths occurred in the first month of life. Africa contributed to one-third of the world's neonatal mortality burden.3,4 In the sub-Saharan region, about 75% of deaths occur during the first week of life, and almost half occur within the first 24 h.3,5 Preterm birth, birth asphyxia, and infections are the major causes of death. 6 Among the mortality reasons, preterm birth complications, which contribute to more than one-third of the global neonatal mortality burden, are the leading cause for which a neonate has a higher risk of developing respiratory distress syndrome (RDS).7–9
RDS is a cause of death for preterm newborns immediately following birth.7,10–12 It causes babies to need extra oxygen and help with breathing.3,10,13 RDS is often seen during the transition from fetal to neonatal life. The successful transition from fetal to neonatal life requires a series of rapid physiologic changes in the cardiorespiratory systems. These changes result in a redirection of gas exchange from the placenta to the lungs and require the replacement of alveolar fluid with air and the onset of regular breathing. 14 Respiratory support is needed to treat RDS that begins immediately after birth to support immature lungs and to establish physiologic stability. 15 These supportive methods are divided into invasive (mechanical ventilation and endotracheal intubation) 12 and non-invasive (various continuous positive air pressure (CPAP) devices, non-invasive mandatory ventilation, and non-invasive pressure ventilation). 15
In low-income countries, invasive mechanical ventilation is often not available for children at risk of death from respiratory failure.13,16 However, CPAP, which is one of the non-invasive methods, and also recommended by the World Health Organization (WHO),12,17 can improve survival in premature neonates with RDS, especially in tertiary centers.9,16,18 In developing nations, early use of CPAP along with early rescue surfactant (InSurE) is the best approach to managing the RDS in preterm and extremely preterm infants. 14 A delivery room with CPAP is feasible and minimizes the use of surfactants and mechanical ventilation by almost 50%. 15
Nevertheless, in higher-income countries, in addition to using CPAP, advanced use of technology such as antenatal corticosteroids, exogenous surfactant therapy, and mechanical ventilation have been used, and have significantly improved the survival of neonates with RDS. 19 Different research studies18,20,21 confirmed that CPAP could improve survival beyond the neonatal period in children with primary pulmonary disease. However, conventional CPAP devices (which is the gold standard method) and mechanical ventilation are expensive options. In addition to this, mechanical ventilation requires high-level expertise and trained personnel, which is not currently affordable in many resource-limited countries.8,10 For instance, even if there are several conventional CPAP devices available, they cost up to US$6000 to US$10,000, 22 while a low-cost bubble nasal CPAP (bNCPAP) system may cost as low as US$350 to US$2000.8,23 This means, comparatively, bNCPAP may cost approximately 15% of the cost of the cheapest mechanical ventilator. 22
Although the existing CPAP machine helps prevent the collapse of alveoli in the lungs and increases survival rates with the CPAP machine, some babies still develop Bronchi pulmonary dysplasia, which is a serious lung condition that affects newborns that need oxygen therapy.22–24 In addition to this, the main engineering design gap with the existing CPAP machine was that the condensate in the exhalation limb of the patient circuit during bubble CPAP (bCPAP) could significantly increase the pressure delivered to the neonate. The back and forth movement of this condensate fluid cause oscillations in airway pressure that are much greater than the oscillations created by gas bubbling out of the exhalation tube into the water bath 25 and allows delivered pressures to reach pressures significantly higher than those intended. This can result in serious physical consequences such as air leaks, over-distention, and gastric distention. Moreover, the accumulation of condensate in the expiratory limb of the bCPAP circuit increases the delivered pressure to the infant, which will cause additional resistive loading of the respiratory system, causing serious physiological complications (such as increased PaCO2, reduction in venous return, and compromised cardiac output) and potentially leading to respiratory failure by imposing large airway pressure oscillations that are out of phase with the neonate's intrinsic respiratory efforts.15,26 Since neonates are very sensitive, disconnecting the expiratory tube for cleaning will leave them at a higher risk. In addition to this, there is still a lack of knowledge regarding factors influencing the implementation of the existing CPAP machine in resource-limited settings. 5 Due to this, developing countries need to address these preventable deaths by scaling up efforts to implement innovative and yet effective low-tech interventions to achieve sustainable development goals. The objective of this project was, therefore, to develop a bCPAP machine that has features to control the delivered CPAP to keep it in a safe range at minimum cost. The developed bCPAP machine delivers safe therapy and monitors the CPAP to protect the infants from the excessive pressure caused by the condensate accumulation in the expiratory limb of the circuit.
Material and methods
The proposed design
In this study, a low-cost bCPAP machine that has a dual function of monitoring and controlling the excess pressure caused by condensate accumulations in the expiratory tube by using a pressure sensor and two-way solenoid valve that help for alternating breathing was proposed. The device includes components such as the Arduino Mega microcontroller (which controls and processes the entire system based on the uploaded script), a pressure sensor that continuously measures the airway pressure (for this project, the XFPM-050KPGP1 pressure sensor was incorporated to measure the pressure inside the expiratory tube). In addition to this, a solenoid valve that alternatively provides positive end-expiratory pressure (PEEP) depending on the condensate accumulation on either side of the expiratory tube was integrated. Moreover, a plastic jar or water reservoir was required to submerge the two expiratory breathing tubes directly from the two-way solenoid valve to the depth necessary (which was adjusted manually by the user) to establish PEEP (see Figure 1). A flow meter that measures the mixed oxygen and atmospheric pressure flowing through the tube, and a pressure gauge used to regulate the pressure of these gases to the desired value were also integral parts of the device (see Figure 1). Furthermore, a keypad was used for the physician to fill the amount of pressure range based on the oxygen-blending value (state in Supplemental information, Table SI1). Figure 1 shows the functional and general block diagram of the proposed design. First, the neonate is given blended and regulated oxygen (from any oxygen source, we had used an oxygen concentrator machine for testing purposes) with atmospheric air (from a compressor). The compressor has been integrated inside the pack of the final developed prototype (see supplemental information Figure SI1). As shown in Figure 1, the tube which contains the blended and regulated gas has been connected to one side of the Y-tube, and the pressure sensor has been connected to the other side of the Y-tube. The tube in which the pressure sensor is attached is connected to the inlet of the two-way solenoid valve. Then the two expiratory tubes were connected to the two outlets of the solenoid valve and finally immersed into the water jar (see Figure 1). In the existing CPAP machine, the air travels through the expiratory limb and forms a bubble inside the water jar. This bubble helps to create the expected amount of air inside the neonate's lung. This is what we call PEEP. The depth of the tube inside the jar determined the PEEP value and has a direct proportionality relationship. The depth of immersion of these two expiratory tubes is manually adjusted using a rotating pin in the jar and has been done based on the required PEEP value from the oxygen blended table (see supplemental information Table SI1).

Functional and general block diagram of the system.
When the device is operated, the physician inserted a safe range (the maximum and minimum safe range) pressure from the oxygen blending table provided by WHO. When the neonate exhales, the pressure sensor placed above the inlet of the two-way solenoid valve measures the instant PEEP in a centimeter of water. The reading value is dispatched to the microcontroller and displayed in liquid crystal display (LCD). If the condensate is accumulated in the expiratory limb, and the reading of the PEEP value is above the manually inserted blended oxygen range (safe range) by the physician early using the keypad, the microcontroller makes a verdict to switch the pathway of blended air to the alternate expiratory tube and block the other expiratory tube that contained the condensate accumulation in order to save the neonate. This alternating airway path to either side of the expiratory tube helps to decrease excessive pressure that would have occurred due to the accumulation of condensate which later on leads to the damage of the neonate's lung. Nevertheless, when the pressure is low beyond the safe range and high enough beyond the required range and if the solenoid valve does not switch to the alternate side of the expiratory tube to decrease the pressure, the alarm would be automatically triggered to remind the caregivers.
Materials used
Table 1 lists the materials used in this work.
Demonstrates the materials and their specifications used to construct the prototype.

Results
Simulation result
The design was simulated using Proetus Simulation software and Arduino IDE before prototype construction and real testing. The inputs used for simulating the system were the value from the pressure sensor (XFPM-050KPGP1) and the keypad to insert the safe range PEEP value from the oxygen blended table. Since it is not possible to use the real neonate in the simulation, we have assumed that when the neonate expires there would be a condensate accumulation in the expiratory tube and the pressure would increase. Therefore, based on this assumption, we manually increased and decrease the pressure from the sensor for concept proofing of the simulation test. As shown in the Figure, to simulate the system, we manually inserted the safe range pressure using the keypad based on the oxygen blending table provided by WHO. When the PEEP pressure reading is in the range of the manually set safe range by the physician, our device continues to work and no alarming and red light-emitting diode (LED) blinks (Figure 2). But if the pressure reading is above the safe range due to the condensate accumulation, the relay would not switch the expiratory path to the alternate tube, both the alarm buzzer and red LED were triggered to notify the physician (see Figure 3).

Both alarm and light-emitting diode (LED) are off when the pressure is within the range.

Both the alarm and LED are ON when the pressure is greater than the safe PEEP range manually set.
Prototype iteration
Different prototype iterations have been escorted to come up with the final design. The final design was completed to obtain a safe and reasonably priced bCPAP device with a pressure monitoring and control system. Figure 4 shows the component integration of the final design. The design includes a delivering mechanism of continuous positive air (from oxygen source and compressed air) section, pressure monitoring (pressure sensor, LCD, and alarm) part, and pressure controlling (pressure sensor, Arduino mega, and two-way solenoid valve) compartment. In addition to this, the flow meter, expiratory tube, relay, and water jar were components used in the final developed prototype. During the demonstration, a test lung was used to represent the infant. The system draws 5 V from an external battery to power the Arduino microcontroller. Figure 5a and b shows the final design of the AutoCAD and the final prototype of a low-cost bCPAP device with a pressure monitoring, and controlling system, respectively. You can find a video of the prototype at http://doi.org/10.17605/OSF.IO/JE9DB divided into three parts.

Components of the low-cost bubble CPAP (bCPAP) machine with a pressure controlling system.

(A) Final AutoCAD design, (b) final developed prototype.
Prototype test results
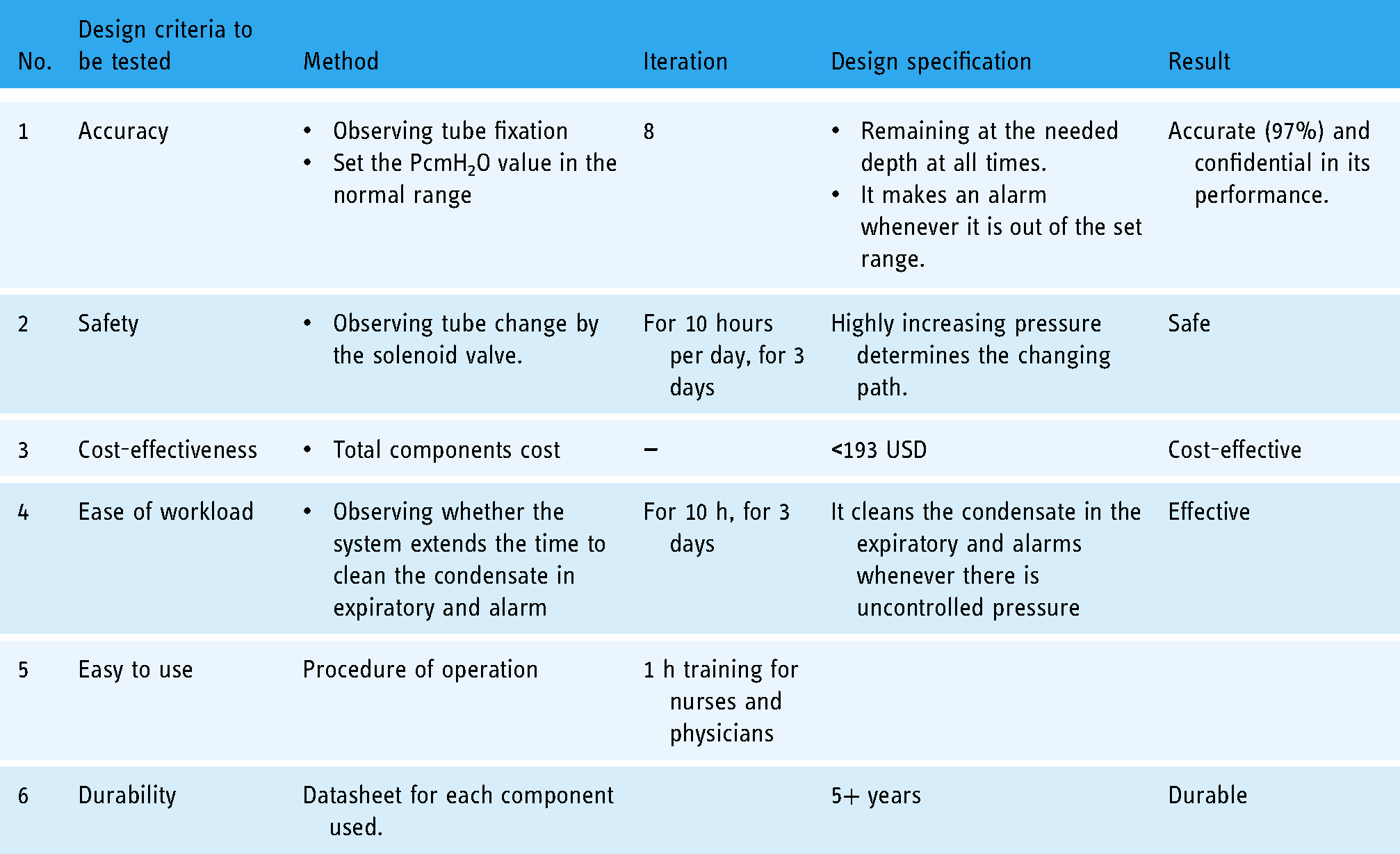
Accuracy, safety, cost-effectiveness, portability, ease of use (ease of workload), durability, a mechanism to control the pressure delivery, and adjustable flow rate ability were the criteria used to design and construct the prototype. We acknowledge that we were unable to test the device using models or in the real world due to a lack of realistic animal or other models. But we have tested our device by squeezing the artificial lung/ballon manually and seeing the effect for eight different iterations. We acknowledge that tests on the animal model should be done before clinical testing and evaluation on humans. The test results were conducted against the design criteria. Accordingly, the accuracy of the prototype units was checked by performing different tests. Table 2 demonstrates the testing methods and test results obtained. The accuracy test was done for condensate accumulation testing and staying of the expiratory tube at its fixed depth inside the water jar. Observations were performed sequentially at eight different depths underwater (2.0–9 cm). Observing each depth for 24 h solely did not slip from its fixed depth. But since we have not been able to get a chance to test our system on real-world the condensation accumulation test was done using balloon as a lung model and see the effect for eight different iterations. For the first four iterations, we kept the machine at room temperature (25.5°C) and the balloon started to breathe so that the machine functioned in the normal range. On the other hand, in order to build up the moisture in the tubing and create condensate accumulation, we place the water jar in a high-temperature area and heated the humidifier, and squeeze the balloon with an increased speed of breathing rate than the normal one. Now, when condensate accumulates inside the expiratory tube and the pressure rises above the safe level, the microcontroller switches the air pathway to the alternate expiratory tube and makes an alarm until it changes its pathway to the other side of the expiratory limb. The team, along with clinical collaborators, tried this step for the last four different iterations and ensure the system is accurate in its function. In the same fashion, we did an experiment on the Pumani bCPAP machine, made in the USA to compare our device performance with this gold standard device and we got good almost 97% accuracy on its performance.
Tests conducted.
Discussion
Despite its drawbacks when compared to our developed devices, many scholars have worked hard to solve this problem. For instance, the Rice 360° Institute for Global Health (Houston, Texas, USA) developed the Pumani system for low-income countries 27 with a system that includes a driver unit with a built-in bubble bottle for pressure control and a single inspiratory tube connected to Hudson prongs. The bubble bottle is connected to the inspiratory limb, which works as a pressure release valve upstream of the patient. However, the flow diverted to the bubble bottle will not reach the patient and the bubbling does not represent the flow that the patient receives. The system was later redesigned and presented by Brown et al. 8 in 2013 with a capped expiratory limb and the bubble bottle moved to the inspiratory limb. With this design and a situation with no leakage at the interface or through the mouth, there would be total rebreathing with an accumulation of carbon dioxide and subsequent respiratory failure. The Pumani system has since been revised with a bleed port added to the expiratory limb (previously capped) with sufficient flow through the bleed port. 26 However, with this design and a situation with no leakage at the interface or through the mouth, there would be total rebreathing with an accumulation of carbon dioxide and subsequent respiratory failure. On the other hand, B & B Medical’s bubbler 27 is designed to deliver between 0 and 10 cm H2O CPAP for infants weighing <10 kg. It has a dual-chamber wall for monitoring fluid levels. It does not have to be disconnected to see the water level. Disrupting the therapy is unnecessary. The drainable air overflow chamber does not allow the water to rise above the prescribed level. This limits the fluid level to be within an optimal range. A fill port is provided for adding fluid without disengaging the circuit, and rotating the CPAP dial with a setting lock reduces the risk of inadvertent setting changes. However, the B & B bubbler is a fixed pressure manifold that only opens if the pressure is more than 12.5 cm of H2O and this will make it difficult for healthcare personnel to give the treatment if the neonate requires a pressure above 12.5 cm of H2O and if the pressure required by the neonate for treatment is far below 12.5 cm of H2O. This makes the gadget specific to the pressure and limited to use. In addition to this, the B & B Bubbler is very expensive, making it unaffordable for middle- and low-income countries.
However, our design called a low-cost bCPAP machine with pressure monitoring as well as a control system has effectively solved problems related to excess pressure resulting from condensate accumulation in the expiration limb. Any design is preferable if it is precise and simple to use. In this sense, the developed design is simple to use and user-friendly. Vividly, the developed prototype has the following traits: (1) adjustable flow rates, (2) ability to mix oxygen into the flow stream, (3) mechanism to control the pressure delivered to the patient, (4) low cost, (5) safe, (6) durable, (7) easy to use and repair, and (8) accurate operation. In our country, there is a limited number of professionals in the Neonatal Intensive Care Unit to the number under bCPAP follow-up. The developed bCPAP extends the time required to clean the condensate in the expiratory tube and alarms nurses to check the neonate whenever there is uncontrolled pressure.
The proposed design allows physicians to easily monitor and control the accumulation of condensate in the exhalation limb of the CPAP machine accurately and safely. The prototype costs <193 USD, which makes it easily affordable in a low-resource setting. The accuracy of the system has been conducted under the supervision of a physician from the Jimma Medical Center. The prototype is accurate in the simulating test. As compared with already existing BCPAP machines (the gold standard one), when the condensate is accumulated inside the expiratory tube and the pressure exceeds the safe range, the microcontroller switches the pathway of air to the alternate expiratory tube. As a result, the device is dependable and the setting is trusted. The developed prototype provides a high level of safety. It is free from electric shock, contamination, and any type of hazardous radiation exposure. Nevertheless, until now the device has not been tested on a human subject, as the validation policy was not done on the system.
Conclusions
The accumulation of condensate in the exhalation limb of the breathing circuit during bCPAP significantly increases pressure delivery to neonates and harms them quietly. To solve this, our design, the low-cost bCPAP device with a pressure monitoring and controlling system, has the function of both monitoring and controlling this accumulation of condensate and taking action whenever necessary. Cost-effectiveness is a very important consideration in resource-poor regions of the world and has to be considered before any intervention is scaled globally. Therefore, our developed device is relatively more cost-effective than the already existing one. Moreover, the prototype was built and underwent different testing and iteration mechanisms, and it is dependable in monitoring and controlling the accumulation of condensate and the life of the neonate. The proposed method will have a significant impact in low-resource settings where expertise is scarce.
Supplemental Material
sj-docx-1-dhj-10.1177_20552076221109060 - Supplemental material for A novel low-cost bubble continuous positive airway pressure device with pressure monitoring and controlling system for low resource settings
Supplemental material, sj-docx-1-dhj-10.1177_20552076221109060 for A novel low-cost bubble continuous positive airway pressure device with pressure monitoring and controlling system for low resource settings by Etagegnehu Dagnachew Feleke, Eyerusalem Gebremeskel Gebremaryam, Feven Tadele Regassa, Hawi Rorissa Kuma, Hayat Solomon Sabir, Ahmed Mohammed Abagaro and Kokeb Dese in Digital Health
Footnotes
Acknowledgements
We would like to thank Dr Abraham (a Pediatrician at Ayder Hospital), Dr Abenezer Hosiso (Medical Intern at Jimma Medical Center), and Dr Handsome (a Pediatrician at Black Lion Specialized Hospital) for their invaluable clinical assistance and advice.
Contributorship
ED, EG, and FT contributed to conceptualization, visualization, software, methodology, and writing of original draft, reviewing, and editing. HR and HS contributed to formal analysis, validation, investigation, data curation, and writing of the original draft. AG and KD contributed to supervision, data curation, visualization, evaluation, reviewing, and editing. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This research did not involve humans, animals, or other subjects. According to Jimma University's Institutional Review Board, no formal ethics approval was required in this particular case.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Guarantor
Jimma University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
