Abstract
Background
Mobile health (mHealth) offers potential benefits to both patients and healthcare systems. Existing remote technologies to measure respiratory rates have limitations such as cost, accessibility and reliability. Using smartphone sensors to measure respiratory rates may offer a potential solution to these issues.
Objective
The aim of this study was to conduct a comprehensive assessment of a novel mHealth smartphone application designed to measure respiratory rates using movement sensors.
Methods
In Study 1, 15 participants simultaneously measured their respiratory rates with the app and a Food and Drug Administration-cleared reference device. A novel reference analysis method to allow the app to be evaluated ‘in the wild’ was also developed. In Study 2, 165 participants measured their respiratory rates using the app, and these measures were compared to the novel reference. The usability of the app was also assessed in both studies.
Results
The app, when compared to the Food and Drug Administration-cleared and novel references, respectively, showed a mean absolute error of 1.65 (SD = 1.49) and 1.14 (1.44), relative mean absolute error of 12.2 (9.23) and 9.5 (18.70) and bias of 0.81 (limits of agreement = –3.27 to 4.89) and 0.08 (–3.68 to 3.51). Pearson correlation coefficients were 0.700 and 0.885. Ninety-three percent of participants successfully operated the app on their first use.
Conclusions
The accuracy and usability of the app demonstrated here in individuals with a normal respiratory rate range show promise for the use of mHealth solutions employing smartphone sensors to remotely monitor respiratory rates. Further research should validate the benefits that this technology may offer patients and healthcare systems.
Keywords
Introduction
Extensive growth in the development and adoption of remote healthcare tools has been seen in recent years in response to the increasing demand for traditional offerings. 1 Notably, the COVID-19 pandemic has made salient how these mobile health (mHealth) tools may support healthcare systems to manage their patients when resources are pushed to breaking point.2–4 As more widely accessible tools can be used by more people – and therefore offer greater impact – many mHealth smartphone applications (apps) have been developed due to the high global penetration of smartphones. These systems offer a wide variety of services from telemedicine to remote monitoring and self-care, and evidence suggests they may produce improved economic 5 and health outcomes. 6
The respiratory rate (RR) is a fundamental indicator of health status for many health conditions, both general and specific to the respiratory system.7–12 As such, mHealth solutions for monitoring of RR may offer significant value to patients and healthcare professionals (HCPs) alike. Although several hardware and software-based solutions exist to measure RR, currently they often have disadvantages. Hardware-based solutions, including piezoelectric sensors, 13 pulse oximeters 14 and multi-sensor devices,15,16 are typically expensive, vulnerable to limited means of manufacture and distribution 17 and may lack interoperability with other health records, which is cited as a critical risk to the decentralisation of national healthcare systems. 18 Software-based solutions address limitations of cost, manufacture and distribution; however, they typically employ less-stable mechanisms of action. These mHealth apps often use smartphone cameras or microphones,19–21 the latter of which have been evidenced to be vulnerable to environmental noise at the cost of accuracy and usability.20,22
Movement sensors may present a promising alternative software-based solution for mHealth RR monitoring. Research indicates that multi-axial accelerometers and gyroscopes – as found ubiquitously in modern smartphones – can accurately capture RR based on chest movements.23–30 Additionally, due to their mechanism of action, these sensors are significantly less affected by environmental noise. Overall, smartphone-based measurement of RR provides a potential low cost, and a widely available method for RR measurement, both in a remote monitoring environment and in locations where specialised hardware and software are not available.
This article presents the technical validation of a novel user-centric mHealth smartphone app that measures RR using the tri-axial gyroscope. We first conducted a preliminary evaluation of the device and study methods via a small lab-based study and then jointly assessed accuracy and usability on a greater scale and ecological valid environment via a remote study. Ethical approval was provided by the University of Exeter’s Research Ethics Board (application ID: eUEBS004088), and all research was conducted in compliance with the Declaration of Helsinki.
Study 1
Methods
The preliminary evaluation pursued three aims: (a) to establish the accuracy of the novel mHealth smartphone app relative to a reference device cleared by the US Food and Drug Administration (FDA), (b) to understand the usability of the mHealth app and (c) to evaluate the suitability of a novel reference method that would permit accuracy assessments to be conducted via remote and real-world studies. Through a prospective, non-interventional, non-randomised study conducted on healthy volunteers, RR estimates provided by the FDA-cleared reference device were compared to those from the novel mHealth smartphone app and the novel reference. RR measurements were recorded simultaneously with both devices at the same time.
Measurements
Novel mHealth smartphone app
The mHealth app contained a purpose-built user interface (Figure 1) and was designed to monitor RR within the normal range. The user is instructed to hold their smartphone to their upper-middle chest with the screen facing outwards while sitting still and breathing normally for the duration of the 30-s sensor recording. Data is captured from the smartphone’s tri-axial gyroscope and interpolated to achieve an even 100-Hz sample frequency. A low-pass Butterworth filter with 0.4 Hz cut-off is applied to remove high-frequency noise while retaining activity associated with breathing rates within the normal range typically in the 0.16–0.33-Hz range (10–20 breaths per minute (BPM)). RR is calculated by performing an autocorrelation before normalising the resulting signal. A peak-finding routine then identifies prominent peaks corresponding to the cyclical property of breathing movements. The mean inter-peak interval (IPI) is then calculated and converted to a ‘per minute’ RR estimation by division by 60 (seconds) (Figure 2).
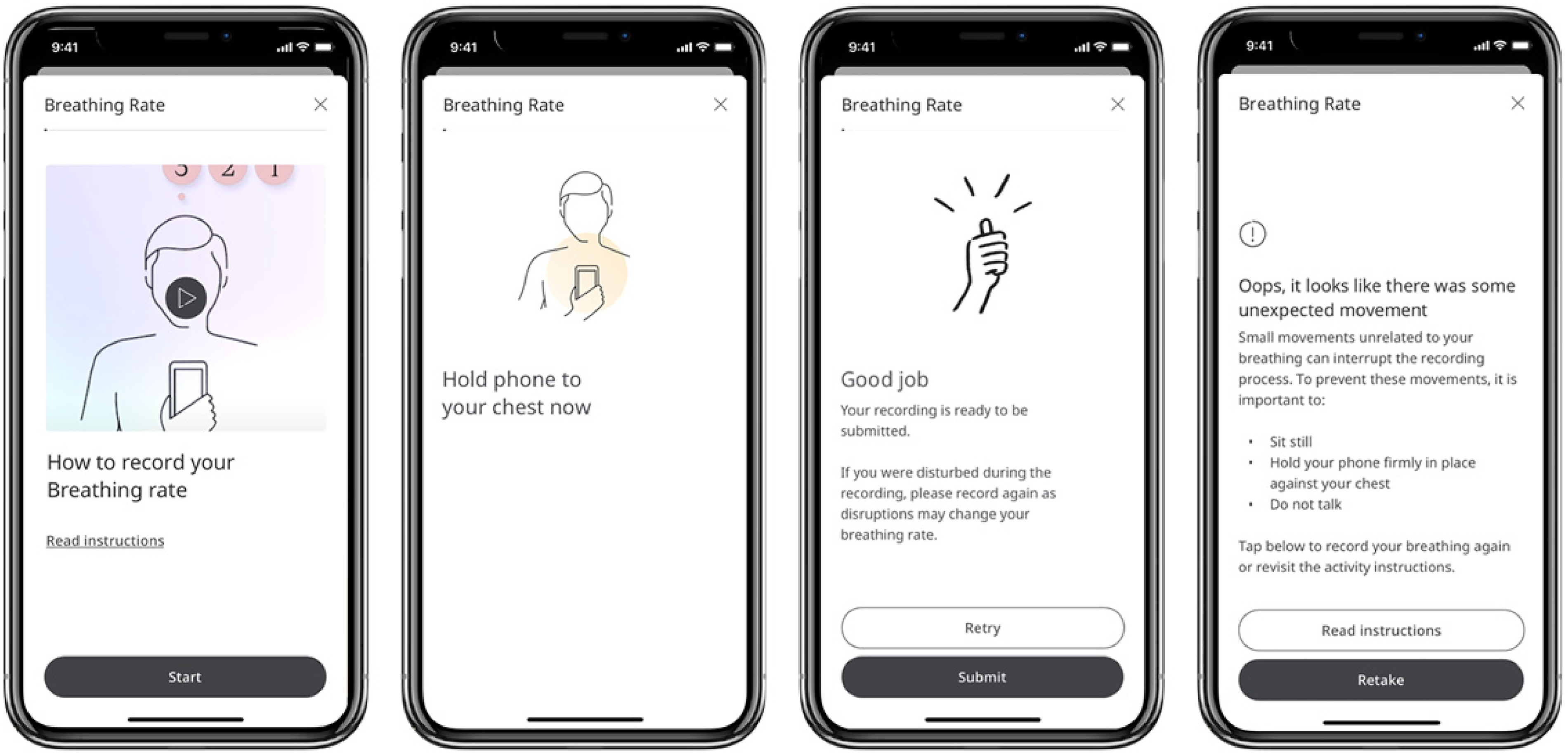
Selected wireframes from the user interface of the mHealth app, depicting (from left to right) an option given to the user to view operation instructions in video or written format, an instruction for the user to hold their smartphone to their chest, a clinical safety feature allowing the user to retake a recording if they were disturbed while taking the original recording, and feedback given to the user if their recording fails the signal check.

Graphical depiction of peak finding following the application of an autocorrelation method. The grey line shows an example of correlation coefficients for a movement sensor (gyroscope) signal correlated with itself at progressive temporal shifts. Black crosses indicate prominent peaks corresponding to the cyclical property of breathing movements. IPIs are depicted by di.
An additional ‘signal check’ routine assesses whether the signal quality is sufficient to accurately derive RR, based on whether the number of autocorrelation peaks or standard deviation (SD) of the individual IPIs meets predetermined thresholds identified via preliminary bench-testing. If a recording fails the signal check, the user is informed via the app’s UI, redirected to the operation instructions and prompted to try again. Passing the signal check within three recording attempts constitutes a successful use of the system, and three consecutive signal check failures constitute an unsuccessful use of the system, after which the user is instructed to seek support or try again later.
FDA-cleared reference
The MightySat Rx, 14 developed by Masimo Corporation, was selected as a reference due to its FDA-cleared status, continuous measurement and ease of use. The fingertip pulse oximeter derives RR using photoplethysmography (PPG) (an optical measure of volumetric changes in peripheral blood flow). Continuous estimates of RR produced by this reference were converted to single-weighted averages to facilitate comparison with data derived from the mHealth app.
Novel reference
The novel reference method involves the identification of repeated cyclical peak-trough complexes within smartphone movement sensor signals (Figure 3). Signals of insufficient quality to derive RR are considered to fail the reference method. This method is conceptually similar to reference methods described in peer-reviewed literature reporting the accuracy assessments of multiple RR devices, including successful FDA market clearance applications.14,31–33 This method would permit accuracy assessments to be conducted via remote and real-world studies without a need for additional hardware, offering a significant value in terms of research scale, cost and ecological validity via the avoidance of observation bias.

Graphical depiction of the novel reference method involving the visual inspection of smartphone movement sensor signals by trained clinical and scientific researchers. The grey solid line shows an example of a smartphone movement sensor (gyroscope) signal, with black solid arrows depicting nine full repeated cyclical peak-trough complexes. The grey dotted line indicates the projected continuation of the movement sensor signal past the end of the recording period, with the grey dashed arrow indicating where a tenth full peak-trough complex would end. Hence, the movement sensor signal depicts a total of approximately 9.75 peak-trough complexes, with the final .75 peak-trough complex indicated by the black dashed arrow.
Participants and recruitment
Participants were recruited via convenience sampling. All were employees of the mHealth app manufacturer. Inclusion criteria included were aged 18 or over and willing and able to follow the study protocol and complete an informed consent form.
Procedure
The study took place at the offices of the mHealth app manufacturer. Participants were provided with complete information concerning the study procedures and gave written informed consent to participate. The FDA-cleared reference device was applied to the forefinger of the participant’s left hand. Participants were provided with an iPhone XR, model number MRY42B/A with the mHealth app installed and received verbal instructions on operating the device: namely, to hold the smartphone to their upper middle chest with the screen facing outwards while sitting still and breathing normally during the 30-s recording. Participants were instructed to capture six recordings, disregarding whether each recording passed or failed the signal check. Audiovisual footage was captured during the study and used for offline synchronisation of data captured via the mHealth app and FDA-cleared reference. Specifically, this included sounds produced by the mHealth app indicating the start and end of the app’s recording period and depicting RR estimates displayed on the FDA-cleared reference’s monitor. Participation took around 10 min per participant.
Statistics
The error of the mHealth app and novel reference relative to the FDA-cleared reference was assessed through measures of mean absolute error (MAE), relative MAE and using the Bland–Altman method. 34 Due to the non-normal distribution of absolute error data, confidence intervals (CIs) for MAE and relative MAE were derived via bootstrapping with replacement employing 1000 iterations and a sample size of 100%. The proportion of clinically significant errors, defined as an absolute error greater than three BPM,35,36 was also calculated. Direct relationships between RR estimates generated through the mHealth app, novel reference and FDA-cleared reference were assessed via Pearson Product Moment Correlation (PPMC). The usability of the mHealth app was assessed using the proportion and position of recordings that failed the signal check.
Results
Participants and data
Fifteen participants took part in Study 1 (nine females), for whom six recordings each were collected for a total of 90. Twenty-six (28%) mHealth app recordings failed the signal check and were excluded from analyses, resulting in a dataset of 64 paired samples. Twenty-nine (32%) of recordings failed the novel reference method, so were excluded from analyses, resulting in a dataset of 61 paired samples.
Accuracy
Mhealth app versus FDA-cleared reference
Error results indicated an MAE of 1.65 BPM (SD = 1.49) with a 95% (CI) of 1.32–2.06. Relative MAE was 12.2% (SD = 9.23) with 95% CI of 10.06–14.57. Bias (FDA-cleared reference–mHealth app) was 0.81 (SD = 2.08) with limits of agreement (LoA) of −3.27 to 4.89, indicating RR underestimation by the mHealth app. Eight comparisons (12.5%) had an absolute error greater than 3 BPM. A Bland–Altman plot indicated error values as a function of RR averaged between the reference and mHealth app (Figure 4). PPMC produced a coefficient of r(63) = 0.700, p < .000, indicating a high or strong association between the reference RR estimates and mHealth app RR estimate 37 (Figure 5).
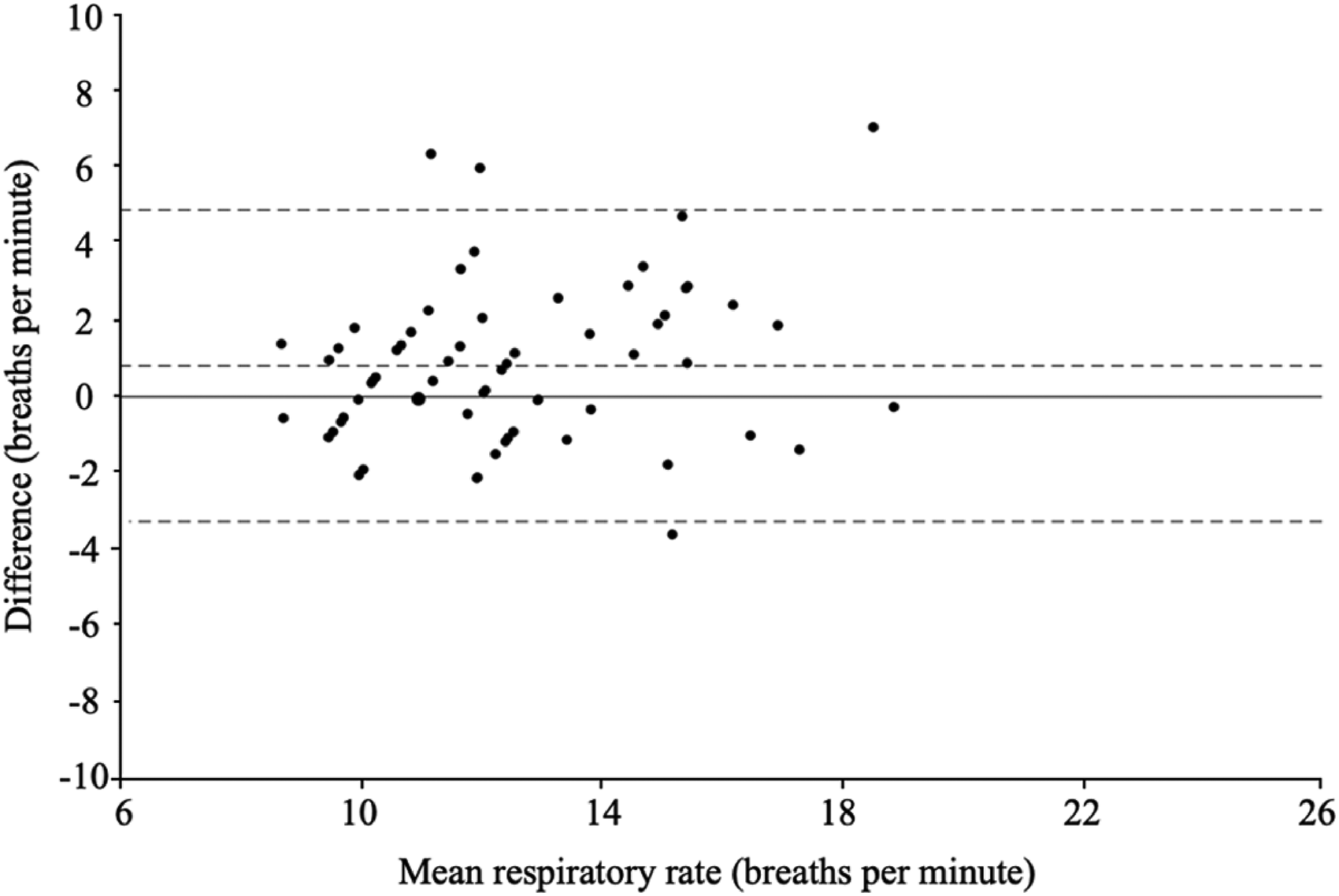
Bland−Altman plot for RR estimates provided by the mHealth app and FDA-cleared reference. The x-axis indicates RR estimates averaged between the mHealth app and FDA-cleared reference, and the y-axis indicates the difference between RR estimates from each source (FDA-cleared reference−mHealth app). The solid horizontal line depicts a mean difference (bias) of 0, and dashed lines from top to bottom represent the upper limit of agreement (4.89), the observed mean difference (bias: 0.81), and the lower limit of agreement (−3.27). Marker size is proportional to the number of observations for each combination of values.

Scatterplot for simultaneous RR estimates provided by the mHealth app (x-axis) and FDA-cleared reference (y-axis). The solid line indicates the gradient y = x. Marker size is proportional to the number of observations for each combination of values.
Novel reference versus FDA-cleared reference
Error results indicated that MAE was 1.69 BPM (SD = 1.61) with a 95% CI of 1.23–2.22. Relative MAE was 12.8% (SD = 11.60) with 95% CI of 9.96–15.64. Bias (FDA-cleared reference–novel reference) was 0.22 (SD = 2.34) with LoA of −4.36 to 4.79, indicating slight RR underestimation by the mHealth app. Nine comparisons (15%) had an absolute error greater than 3 BPM. A Bland–Altman plot indicated error values as a function of RR averaged between the FDA-cleared and novel references (Figure 6). PPMC produced a coefficient of r(59) = 0.701, p < .000, indicating a high or strong association 37 (Figure 7).

Bland−Altman plot for RR estimates provided by the novel reference and FDA-cleared reference. The x-axis indicates RR estimates averaged between the novel and FDA-cleared references, and the y-axis indicates the difference between RR estimates from each source (FDA-cleared reference−novel reference). The solid horizontal line depicts a mean difference (bias) of 0, and dashed lines from top to bottom represent the upper limit of agreement (4.79), the observed mean difference (bias: 0.22), and the lower limit of agreement (−4.36). Marker size is proportional to the number of observations for each combination of values.
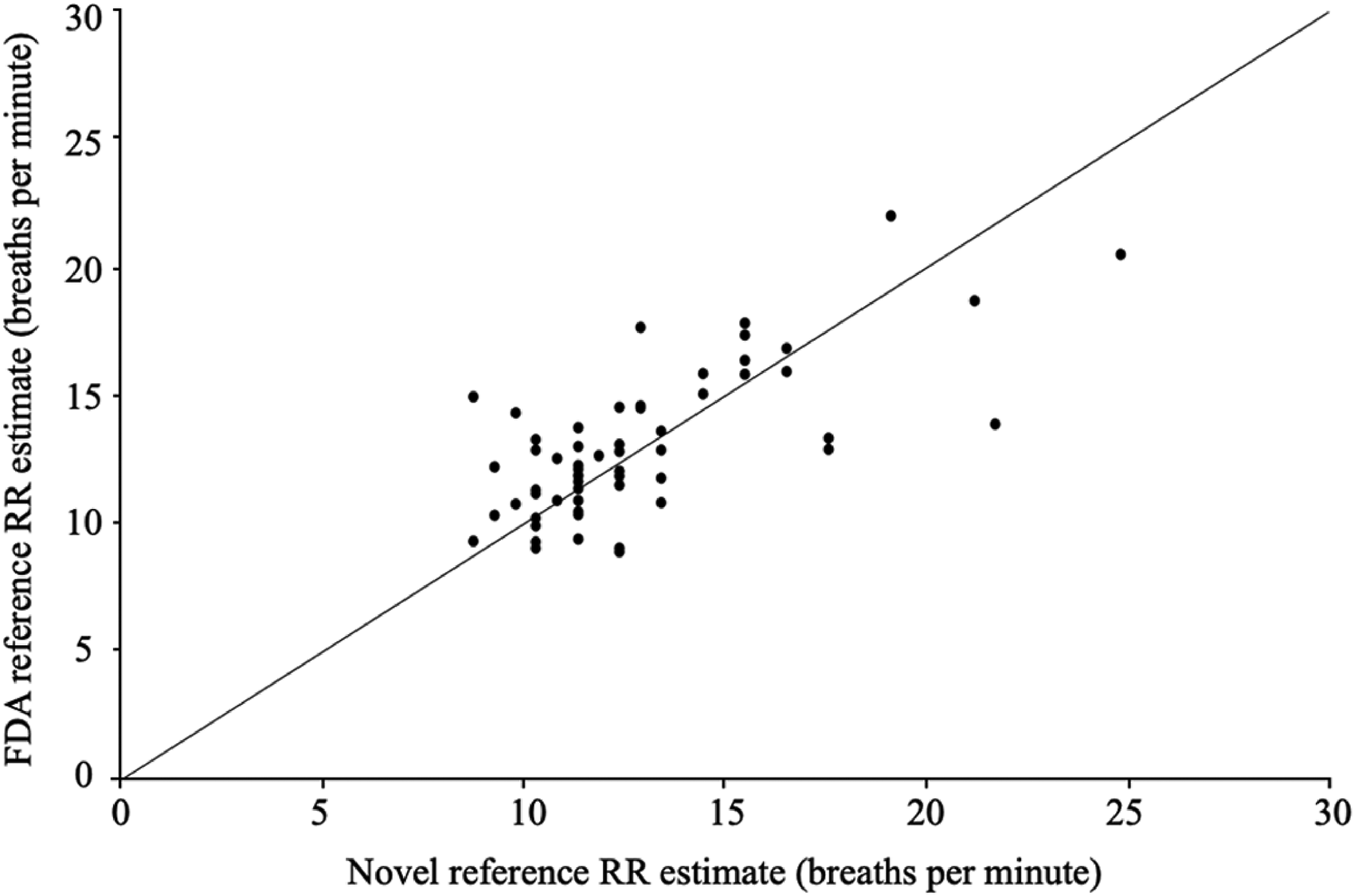
Scatterplot for simultaneous RR estimates provided by the novel reference (x-axis) and FDA-cleared reference (y-axis). The solid line indicates the gradient y = x. Marker size is proportional to the number of observations for each combination of values.
Usability
Fourteen of 15 participants (93.3%) were able to use the system successfully on their first try (Table 1; Figure 8). Specifically, this indicates that they could capture one or more recordings that passed the signal check within the first three attempts. All participants were able to use the system successfully by the end of their second try.

Line graph indicating the proportion of Study 1 participants who were able to generate a recording that passed the signal check on each of six consecutive recording attempts. The black dashed line indicates the proportion of individuals who were able to generate a recording that passed the signal check by the end of their first use of the system (three consecutive recordings), and the grey dashed line indicates the proportion of individuals who were able to do so by the end of their second use of the system.
Number and proportion of participants able to use the system successfully on consecutive attempts in Study 1 (n = 15).

Interim conclusion
Study 1 results indicated strong relationships between the FDA-cleared reference and both the mHealth app and the novel reference. Notably, these relationships were highly comparable to functional outcomes for alternative FDA-cleared RR monitoring devices.13,15,36,38 Accordingly, these results supported both the continued assessment of the mHealth app and the application of the novel reference to accuracy analyses in Study 2, as described below.
Study 2
Methods
Study 2 aimed to establish the accuracy of the mHealth app ‘in the wild’ via remote data capture, compared to the novel reference validated in the Study 1. The usability of the mHealth app was additionally assessed in a larger sample. Measures and statistics were as described for Study 1.
Participants and recruitment
Participants were recruited via an online research platform, with study enrolment controlled to ensure a proportionate distribution of age, gender and smartphone ownership (iOS vs Android). Inclusion criteria included were being aged 18 or over, having access to a smartphone of minimum requirements to download the mHealth app and being willing and able to follow the study protocol and complete an informed consent form. As researchers would not monitor participants during their participation, additional safety criteria excluded individuals who were pregnant and breastfeeding, had a pacemaker or self-reported a condition that might affect their breathing, such as asthma, or might affect their movement such as tremor.
Procedure
Participants were directed to online documentation containing full information about the study procedures before completing an online eConsent procedure. They then completed a baseline questionnaire concerning their demographics, including age, sex, ethnicity, height and weight, before receiving instructions to download and activate the mHealth app. Participants were requested to follow instructions provided within the mHealth app to capture 10 RR recordings, including recordings that both passed and failed the signal check, before completing a System Usability Scale (SUS) 39 and providing separate qualitative feedback on their experience using the mHealth app. Study-specific procedures took approximately 20 min, for which participants were reimbursed £2.50 through the research platform.
Results
Participants and data
One Hundred and sixty-five participants enrolled in the study, of whom 152 completed the baseline questionnaire concerning their demographics (Table 2). Medical conditions reported included asthma (respiratory), arthritis and Parkinson’s disease (movement). Five participants were excluded due to significant deviation from the study protocol, resulting in a participant cohort of 160, for whom a mode of 11 mHealth app recordings each was captured. Nine hundred and eighty-seven recordings passed the signal check and were included in accuracy analyses. Recordings were submitted from 64 unique smartphone models, 46 (71.9%) of which were Android and the rest were iPhone models.
Demographic characteristics for Study 2 participants (n = 152).

Accuracy
Error results indicated an MAE of 1.14 BPM (SD = 1.44) with a 95% CI of 1.02–1.26. Relative MAE was 9.5% (SD = 18.70) with 95% CI of 8.38–10.72. Bias (novel reference–mHealth app) was 0.08 (SD = 1.84) with LoA of −3.68 to 3.51, indicating slight RR underestimation by the mHealth app. Sixty-one comparisons (6.2%) had an absolute error greater than 3 BPM. No difference in MAE was found between Android and Apple devices. A Bland–Altman plot indicated error values as a function of RR averaged between the reference and mHealth app (Figure 9).
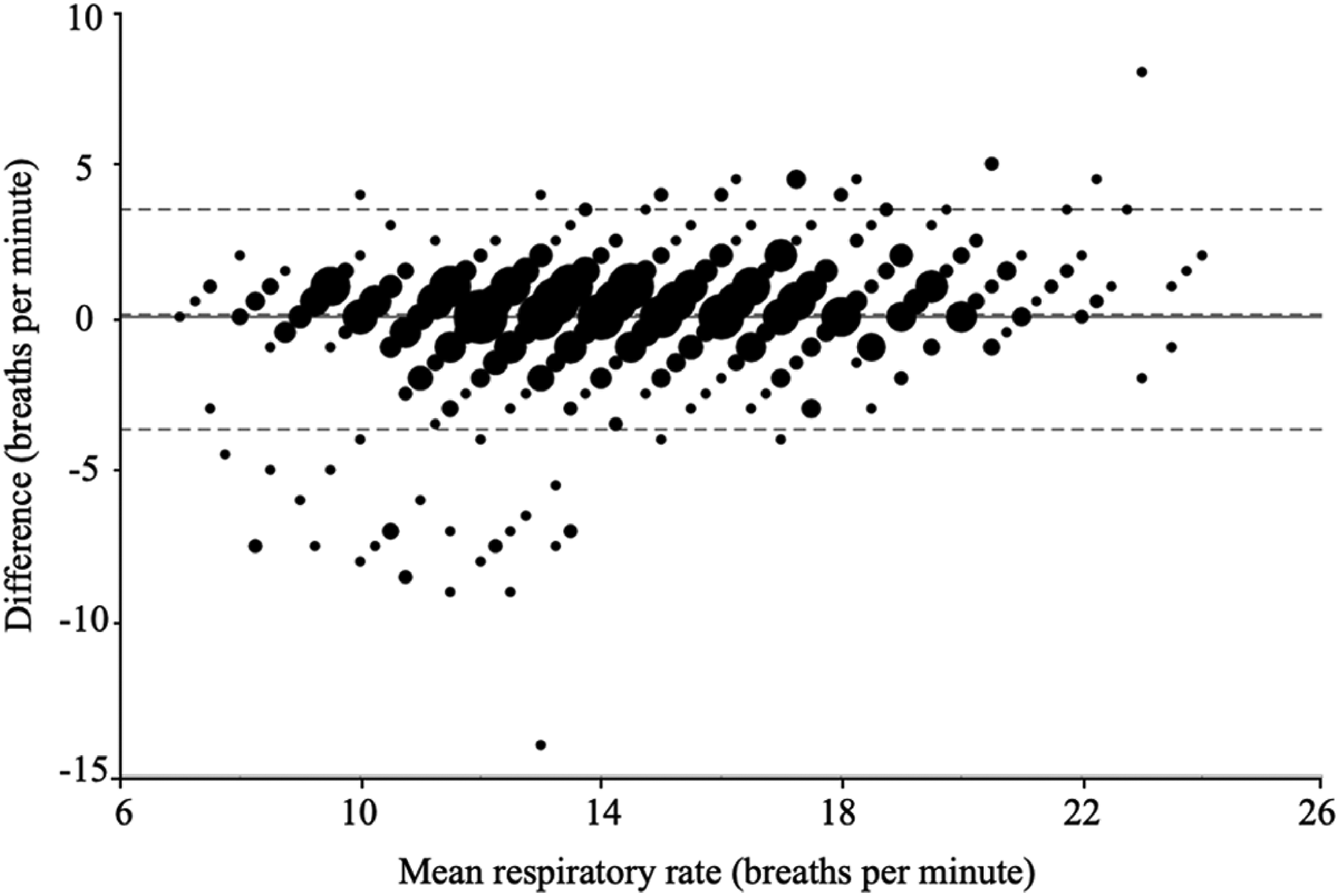
Bland−Altman plot for RR estimates provided by the mHealth app and novel reference. The x-axis indicates RR estimates averaged between the mHealth app and novel reference, and the y-axis indicates the difference between RR estimates from each source (novel reference−mHealth app). The solid horizontal line depicts a mean difference (bias) of 0, and dashed lines from top to bottom represent the upper limit of agreement (3.51), the observed mean difference (bias: 0.08), and the lower limit of agreement (−3.68). Marker size is proportional to the number of observations for each combination of values.
PPMC produced a coefficient of r(986) = 0.855, p < .000, indicating a high or strong association 37 (Figure 10).

Scatterplot for simultaneous RR estimates provided by the mHealth app (x-axis) and novel reference (y-axis). The solid line indicates the gradient y = x. Marker size is proportional to the number of observations for each combination of values.
Usability
One Hundred and forty-nine (93.1%) of a total of 160 participants who captured mHealth app recordings were able to use the system successfully on their first try (Table 3; Figure 11). One Hundred and fifty-five (96.9%) did so by their second try.

Line graph indicating the proportion of Study 2 participants who were able to generate a recording that passed the signal check on each of ten consecutive recording attempts. The black dashed line indicates the proportion of individuals who were able to generate a recording that passed the signal check by the end of their first use of the system (three consecutive recordings), and the grey dashed line indicates the proportion of individuals who were able to do so by the end of their second use of the system.
Number and proportion of participants able to use the system successfully on consecutive attempts in Study 2 (n = 160).

The mean SUS score was 73.2 (SD = 5.39). Of the sub-scales, each scored between 0 and 4, those most agreed with by participants were: I would imagine that most people would learn to use this system very quickly (3.2), I thought the system was easy to use (3.1) and I felt very confident using the system (3.0). The lowest scoring, indicating participant disagreement, was: I thought that I would need the support of a technical person to be able to use this system (0.5) and I needed to learn a lot of things before I could get going with this system (0.8).
Discussion
Principal findings
To the authors’ knowledge, this is the first technical validation to assess at scale a user-operated novel mHealth smartphone application designed to capture a user’s RR using smartphone movement sensors, considering both accuracy and usability in an ecologically valid study environment. Outcomes for the mHealth app were highly comparable to results published for medical devices available on the market today (Table 4). In addition, as changes in breathing rate greater than 3 BPM may indicate clinical deterioration,35,36 observations that error values for the mHealth app were typically less than this threshold suggest the device may carry low clinical risk. Study 2 revealed a small cluster of substantial overestimation errors (5–10 BPM) for lower RRs (8–14 BPM). Although this observation was not found in Study 1, this may be due to the smaller sample size in that analysis. The nature of these overestimations is unclear based on the present analyses. The overestimation of RR carries clinical risk with regard to both the underdiagnosis of bradypnea (low RR) and the overdiagnosis of tachypnea (elevated RR) that may lead to clinical decision-making based on misinformation, although it should be noted that RR is rarely used in isolation to inform clinical decision-making. Future research should seek to identify and mitigate the cause of these errors.
Comparison of mHealth app results to alternative devices available on the market today.

Concerning usability, most participants could successfully operate the mHealth app on their first or second use of the system. Although no industry standards for successful operation exist, results observed here appeared to be broadly similar to values that could be estimated from the available literature regarding other physiological measurement mHealth apps, which were typically in the range of 95% or higher.40–42 Subjective usability outcomes were also promising, with an overall SUS score well above the industry average of 68. 43 Study 2 revealed a general trend of high signal check pass rates for later sequential recording attempts, suggesting that participants found it easier to capture RR recordings the more they used the mHealth app. Although this learning effect was not observed in the Study 1 results, this may be due to observer bias and a small sample size within that study setting. This observation holds promise for improved usability with the long-term use of the mHealth app, although it may indicate greater clinical risk during the early use of the system. Future research may seek to steepen the learning curve to minimise clinical risk.
Wearable devices equipped with PPG sensors provide another alternative method for low cost and the remote measurement of RR. However, PPG methods have hardware and processing power requirements that are higher than smartphone movement sensor methods. Smartphone devices are also used more widely and therefore have greater availability in remote locations or low income countries. 44
Strengths of the present study include the application of a remote study design that lends ecological validity to the results and selective recruitment to ensure a heterogeneous participant cohort, which suggests good generalisability of the results. In addition, the inclusion of usability assessment allows a holistic perspective on the mHealth app to be generated. In all, these results hold promise for the use of smartphone movement sensors as a viable means of remote RR monitoring. Software-based mHealth may offer cost and scalability benefits compared to hardware-based monitoring. Additionally, movement sensors may better protect RR signal quality than alternative devices that use microphone and camera sensors, as these are vulnerable to noise from environmental light and sound that is difficult to control. These benefits suggest that RR monitoring based on smartphone movement sensors may support healthcare systems to care for their patients when they are outside of the clinic better than currently available alternatives.
Study limitations
As all participants had RR within the normal range, it is unclear how the observed results may extrapolate to healthcare patients who would be likely real-world users of the mHealth app, particularly those with abnormal breathing rates and patterns due to a respiratory condition. For example, individuals receiving ventolin for asthma may have a medication induced tremor, which may affect gyroscope recordings. This is also true for individuals with disorders that are associated with tremor such as Parkinson’s disease. Additionally, measuring RR using the mHealth app in the presence of a chronic, persistent cough, like those associated with severe asthma or COPD may require additional signal processing considerations. Additional signal processing considerations may be required for clinical use cases that require the detection of tachypnea or bradypnea. Participants both from Study 1 (employees of the mHealth app manufacturer) and Study 2 (members of an online research community) were likely to be technologically confident and may have therefore been predisposed to successfully operating the mHealth app. Future research should seek to incorporate individuals of low technical literacy and target end-users with relevant medical conditions to better understand these results’ generalisability.
Concerning methodology, the FDA-cleared reference used in Study 1 has its own measurement error. 14 Hence, error estimates presented here are, in fact, an unknown combination of errors associated with the FDA-cleared reference and mHealth app versus true RR. The Study 2 reference also underwent only limited validation in Study 1 and should be assessed more rigorously. Future research may wish to apply a wider range of reference methods, including gold and industry-standard references, to reduce the vulnerability of the mHealth app to shortcomings of any single reference.
Additionally, the present research design does not directly address potential benefits the mHealth app may offer if applied in a healthcare setting. Although expectations that moving health assessments outside of a clinical setting via mHealth technologies will improve healthcare economics have been somewhat supported by literature, 5 clinical evidence suggests that mHealth technologies are highly heterogeneous in their ability to improve health outcomes.45,46 Suggestions that mHealth may help to overcome social, economic and geographical barriers to healthcare are also yet to be validated.47–49 Future research should seek to understand the clinical, economic and social outcomes associated with real-world use of the mHealth app.
Conclusions
Decentralised healthcare technology holds the potential to offer clinical and economic benefits to patients, HCPs and healthcare systems. Breathing is an important indicator of health, and although solutions for remote RR monitoring exist, many entail significant shortcomings that limit their ability to capitalise on potential benefits of mHealth. Results from this technical validation hold promise for the use of smartphone movement sensors as a robust means for remote RR monitoring. However, future research should address residual questions and risks associated with the technology identified in this article and seek to validate the impact of similar technologies as applied in the real world.
Footnotes
Acknowledgements
This research was sponsored by Huma Therapeutics Ltd. The authors are grateful to Huma’s wonderful developers Michele Colombo, Stanislas Heili, Leonardo Festa, Davide Mascitti, Emanuele Distefano, Matteo Puccinelli and Matteo Vigoni for their diligent efforts in preparing the mHealth app for this research. Additional thanks to Emily Binning for her continued strategic support that helped this research come to fruition.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article. All authors are the current or previous employees of Huma Therapeutics Ltd, which is the developer of the mHealth smartphone app. No additional conflicts of interest relevant to this study are declared.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Huma Therapeutics Ltd (Grant No. N/A).
Author contributions
SV, AC, BK, MD and MB drafted the manuscript, conducted statistical analyses and interpreted the results. DP aided in statistical analysis, interpreted the results and revised the manuscript for intellectual content. MA and DP revised the manuscript for intellectual content. All authors approved the final manuscript for submission.
Guarantor
All authors are willing to take full responsibility for the article, including for the accuracy and appropriateness of the reference list.
Ethical approval
Ethical approval was provided by the University of Exeter’s Research Ethics Board (application ID: eUEBS004088), and all research was conducted in compliance with the Declaration of Helsinki.
Informed consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial registration
Not applicable, because this article does not contain any clinical trials.
