Abstract
Case summary
A 2-year-old, indoor domestic shorthair cat presented with a 3-week history of a progressively growing subconjunctival nodule on the right upper eyelid. Fine-needle aspiration was performed and the sample was submitted for cytological examination. The nodule became an ulceration near the lateral canthus within 1 week after application of ofloxacin-prednisolone eye drops. Yeasts of Sporothrix species were detected and treatment was initiated with itraconazole. As the ulcerated nodule continued to progress, the regimen was modified to terbinafine combined with high-dose itraconazole. Sporothrix schenckii complex was diagnosed from fungal culture, and the antifungal testing reported itraconazole resistance. However, the ulceration showed responsiveness to the combined treatment by day 43. Finally, the ulcerated eyelid was healed after 5 months of antifungal therapy. No recurrence of ocular sporotrichosis was detected at the termination of antifungal medications after 2 years.
Relevance and novel information
Feline sporotrichosis is commonly reported in tropical countries; however, localised nodules and eyelid ulcerations due to S schenckii infection are generally uncommon. To the authors’ knowledge, this is the first report of localised ocular sporotrichosis in a cat in Thailand. Furthermore, although itraconazole resistance was detected in the fungal culture, the combination of high-dose itraconazole and terbinafine proved effective in treating a cat infected with a non-wild-type S schenckii complex in the current study.
Introduction
Sporotrichosis is an emerging mycosis caused by the Sporothrix schenckii complex. These saprophytic, thermodimorphic fungi thrive in soil and organic materials, existing in a mycelial phase at temperatures below 37°C and a yeast phase at body temperature.1 –7 Feline sporotrichosis has been reported in tropical and subtropical regions.1,3,4,8,9 Infections occur in humans and various mammals, such as cats, dogs, cattle, pigs, horses and camels, with cats being the only animal with zoonotic transmission potential.3,5,6 S schenckii is distributed worldwide, including in Southeast Asia.1,10,11 Infection typically occurs through traumatic inoculation of the fungus from soil or via bites and scratches from infected cats.1 –5,7,12 In Thailand, the aetiology of both human and animal sporotrichosis is S schenckii, frequently linked to zoonotic transmission from infected cats.13 –18 In contrast, Sporothrix brasiliensis is the primary zoonotic fungus in South America, transmitted predominantly via an infected cat.4,5,19 S schenckii was first described from a human patient presenting with subcutaneous abscesses on the hand and arm. 20
Feline sporotrichosis comprises three clinical presentations: cutaneous, lymphocutaneous and disseminated systemic forms.1 –3,13 The cutaneous form is the most common, presenting as single or multiple skin ulcers, granulomatous nodules and draining tracts.1 –3,8,14,15 Typically, lesions occur on the head, nose, distal limbs and tail base. 2 In Thailand, feline sporotrichosis was first described in a stray cat with multifocal ulcers; S schenckii was confirmed, showing 100% internal transcribed spacer (ITS)-region sequence identity. 1 Subsequently, human sporotrichosis cases have been described.13,16,17 Evidence suggests that Thailand is an endemic area, but likely underreported. 14 Itraconazole is considered the first-line antifungal therapy.3,10,12,13,17 –22
We report a case of feline sporotrichosis initially presenting as a localised subconjunctival nodule, which progressed to eyelid ulceration without dissemination. Successful treatment was achieved with combined high-dose itraconazole and terbinafine.
Case description
A 2-year-old, neutered male, indoor domestic shorthair cat presented with a nodule on the right upper eyelid. After 3 weeks of treatment with topical antibiotics and non-steroidal anti-inflammatory injections at a local clinic, the nodule failed to resolve and continued to grow progressively. The cat was subsequently referred to the university veterinary hospital.
On day 0, the eyelid nodule measured approximately 2 × 2 cm, with a smooth surface and firm consistency. Ophthalmic examination revealed a subconjunctival nodule and conjunctivitis in the right eye (oculus dexter [OD]) (Figure 1). Fluorescein staining, along with neuro-ophthalmic and fundus examinations were normal. Physical examination and blood profiles were within the reference interval (RI), except for hyperglobulinaemia. Fine-needle aspiration (FNA) was performed to collect a subconjunctival sample for cytology. Amoxicillin-clavulanate (20 mg/kg q12h), tolfenamic acid (3.5 mg/kg q24h) and ofloxacin-prednisolone eye drops were prescribed.

A domestic shorthair cat presented with a subconjunctival nodule on the right upper eyelid, featuring a smooth surface, on day 0
On day 7, the eyelid nodule ruptured, resulting in ulceration on the temporal side. Cytology revealed neutrophils and phagocytic macrophages containing intracellular pleomorphic yeasts, characterised by small, round-to-oval or cigar-shaped forms with thin, unstained capsules compatible with Sporothrix species (Figure 2). Conjunctivitis and marked chemosis OD, protruding from the palpebral fissure, were detected (Figure 3a). Discharge was collected for fungal culture. Topical therapy was discontinued and oral itraconazole was prescribed at 11 mg/kg q24h. The owner was advised on zoonotic precautions when handling the cat.

Fine-needle aspiration of the subconjunctiva shows neutrophils and phagocytic macrophages containing both free and intracellular pleomorphic yeasts. These yeasts exhibit characteristic small, round-to-oval and cigar-shaped forms, encapsulated with thin, unstained capsules (arrows). The morphology is compatible with fungi of the genus Sporothrix. (Diff-Quik stain 100 × magnification)

Subconjunctival sporotrichosis of the right upper eyelid following treatment with itraconazole on (a) day 7, and after combination therapy with itraconazole and terbinafine on (b) day 14, (c) day 28, (d) day 35, (e) day 43, (f) day 53, (g) day 88, (h) day 123, (i) day 151 and (j) day 217
On day 14, the eyelid nodule enlarged and became severely ulcerated, accompanied by notable swelling of the lower eyelid. The nodule compressed the cornea, and mucoid-serosanguineous discharge was present; however, fluorescein staining remained negative (Figure 3b). Sodium hyaluronate (0.1%) artificial tear was prescribed q4h for lubrication. Fungal culture confirmed Sporothrix species, prompting the addition of terbinafine (37 mg/kg q24h) to the existing itraconazole regimen.
On day 28, the ulceration had worsened, resembling a conjunctival mass that extended to the lower eyelid (Figure 3c). Consequently, the itraconazole dosage was increased to 15.5 mg/kg q24h. Because of a lack of clinical improvement by day 35, ocular discharge was submitted for species identification and susceptibility testing (Figure 3d). Fungal isolation was performed by inoculating the discharge onto Sabouraud Dextrose Agar with Brain Heart Infusion Agar supplemented with amoxicillin/chloramphenicol. Cultures were incubated at 30°C and 35°C, and fungal growth was monitored for 5–14 days. Yeast morphologies were evaluated and measured using an ocular micrometre using imaging software (cellSens v3.2; Olympus). The minimum inhibitory concentration (MIC) was determined via the broth microdilution. While awaiting results, the itraconazole was increased to 23 mg/kg q24h and terbinafine was continued. In addition, dermatitis was observed beneath the right lower eyelid and nasal area.
On day 43, the culture was confirmed as S schenckii complex, identified as a non-wild-type strain based on colony morphology, microscopic appearance and characteristic thermal dimorphism. Antifungal susceptibility testing revealed resistance to itraconazole and posaconazole (Table 1). Despite these findings, partial improvement was observed after the increase in itraconazole dosage, evidenced by a reduction in eyelid lesion size (Figure 3e). Repeat complete blood count (CBC) and serum biochemistry were within the RI, except for persistent hyperglobulinaemia, which was consistent with inflammation. Consequently, itraconazole and terbinafine were continued at their current dosages.
Antifungal susceptibility testing of the Sporothrix schenckii complex collected from ocular discharge on day 35
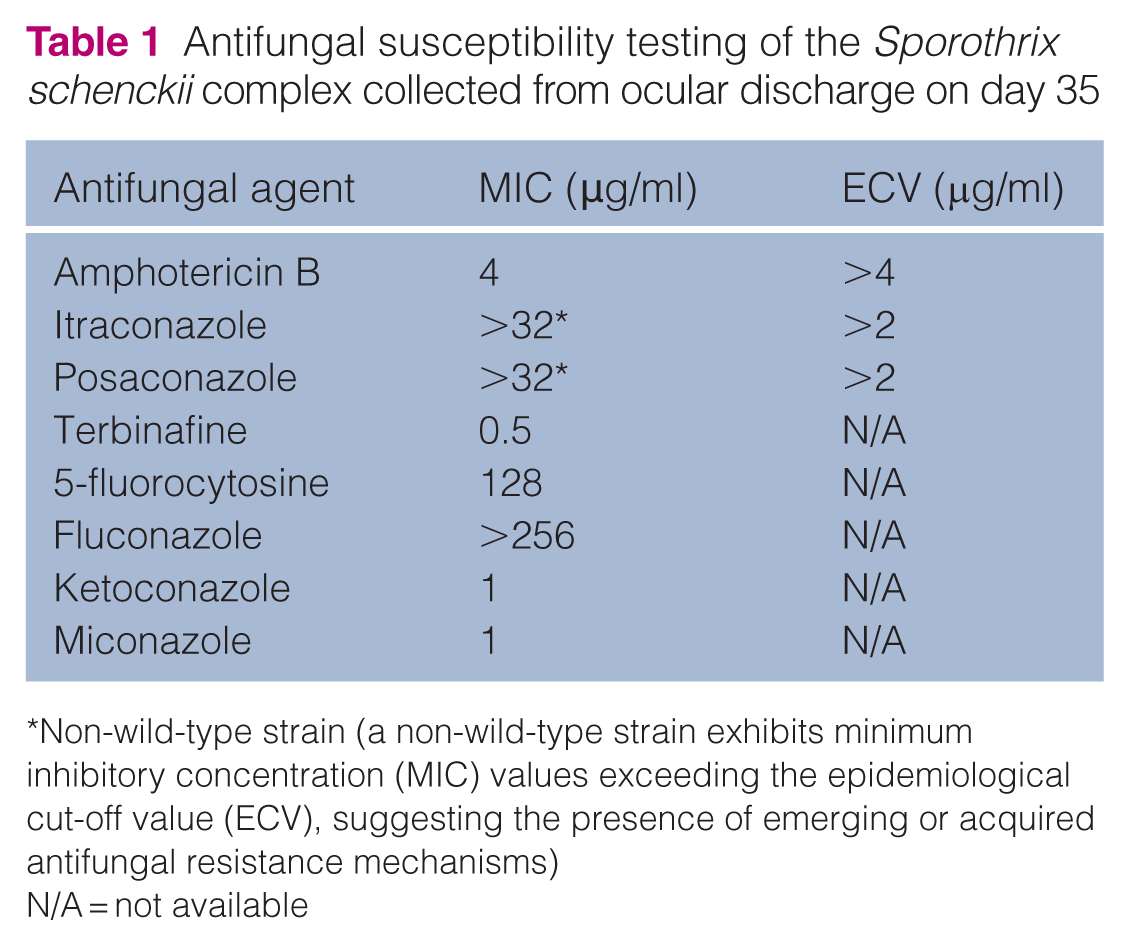
Non-wild-type strain (a non-wild-type strain exhibits minimum inhibitory concentration (MIC) values exceeding the epidemiological cut-off value (ECV), suggesting the presence of emerging or acquired antifungal resistance mechanisms)
N/A = not available
On day 53, the regression of the lesion allowed for visualisation of the cornea, which showed mild keratitis. In addition, dermatitis of the lower eyelid and nasal area had improved (Figure 3f). By day 88, the lesion had markedly subsided. The palpebral reflex was present, but blinking was incomplete. Conjunctivitis with mucoid discharge persisted. Consequently, the artificial tears were replaced with an ophthalmic gel containing carbomer/cetrimide (q8h). On day 123, the ulceration and conjunctivitis further improved, dermatitis near the nasal and medial canthus persisted (Figure 3g), and incomplete blinking remained unchanged (Figure 3h).
On day 151, the ulceration had completely resolved, leaving a permanent partial defect of the upper eyelid. Approximately 50–60% of the eyelid margin was absent and mild blepharitis was noted. The facial dermatitis had also resolved (Figure 3i). Mild keratitis with inactive superficial vascularisation was observed. Terbinafine was discontinued on day 175 (total treatment duration: 168 days), while itraconazole was continued for an additional 2 months.
By day 217, the eyelid ulceration was completely healed. An eyelid agenesis-like lesion was observed on the upper eyelid, while other ophthalmic findings were unremarkable (Figure 3j). Conjunctival cytology revealed only neutrophils, with no evidence of S schenckii. Itraconazole was discontinued on day 245, at which point blood profiles remained normal. The owner was advised to monitor for secondary ocular signs, such as trichiasis. No recurrence was detected during the 2-year follow-up period.
Discussion
Feline conjunctivitis is commonly caused by feline herpesvirus-1 and Chlamydophila felis. Calicivirus and Mycoplasma species are also documented aetiologies, although they occur less frequently.23 –26 However, conjunctivitis with subconjunctival granuloma is rarely associated with these aetiologies. Granulomatous conjunctivitis is the most common ocular pathology of sporotrichosis in cats and humans.13,19,27 Consequently, sporotrichosis should be included as a primary differential diagnosis for conjunctivitis or blepharoconjunctivitis presenting with granulomatous lesions, particularly in endemic and tropical countries.1,6,28 In the current case, granulomatous conjunctivitis was observed in the superior palpebral conjunctiva, a finding consistent with other reports in cats and humans.17,19 Clinical reports of feline sporotrichosis remain limited in Southeast Asia, likely due to underreporting because of a lack of accessible diagnostic laboratories.1,10 This issue requires urgent attention, as it represents an emerging zoonotic disease in Thailand.14,16
Cytology is a pivotal initial diagnostic tool, utilising FNA from nodules or impression smears from ulcerations with Romanowsky staining, which reveals pleomorphic yeasts measuring 3–5 × 5–9 μm.1 –5 Conjunctival cytology is a cost-effective and rapid method; 19 however, the risk of misdiagnosis with other fungi, such as Cryptococcus neoformans and Histoplasma capsulatum, must be considered. 14 Therefore, fungal culture remains a key for confirming sporotrichosis. Here, a definitive diagnosis was confirmed by culture, identifying the S schenckii complex within 7 days. Antifungal susceptibility testing was performed to guide appropriate drug selection.2,14 In addition, other definitive diagnoses include ITS-region sequencing and matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry. 13
The cat was considered immunocompetent, with serial CBCs remaining within the RI and negative feline leukaemia virus antigen/feline immunodeficiency virus antibody tests. Serum biochemistry was also unremarkable, with no evidence of concurrent systemic disease. However, progressive worsening of the periocular lesions was observed at days 7–35; clinical improvement was subsequently noted after an increased antifungal dosage and the initiation of combination therapy. These findings suggest the infection was unlikely to be self-limiting. In sporotrichosis, protective immunity is mainly mediated by cell-mediated immune responses, specifically Th1-dependent pathways involving CD4+ cells and activated macrophages. However, long-term immunity may be incomplete and reinfection can occur. 3
Itraconazole is the first-line antifungal; alternative drugs reported in humans and cats include ketoconazole, posaconazole, voriconazole, terbinafine, fluconazole, amphotericin B and potassium iodides.1,3,13,18 In the current case, itraconazole was initiated at 11 mg/kg and increased to 15.5 mg/kg on day 28 because of lesion progression. On day 35, the dosage was increased to 23 mg/kg before obtaining antifungal sensitivity results. Despite a high MIC (>32 μg/ml) indicating itraconazole resistance, partial improvement was observed by day 43. This clinical-laboratory discrepancy emphasises the importance of integrating laboratory findings with clinical responses to guide therapy.
Based on antifungal testing, the isolate was susceptible to amphotericin B; however, this drug was not selected owing to concerns regarding nephrotoxicity after systemic administration. Furthermore, local administration may result in abscess formation, tissue irritation and injection-related pain, all of which were particularly concerning in the periocular tissue. 29
The duration of itraconazole treatment was 245 days, with the final dosage maintained for 210 days. Complete remission of ocular lesions was achieved within 151 days (approximately 5 months) from a non-wild-type S schenckii infection. In Brazil, primary conjunctival sporotrichosis in cats was successfully treated within 4 months, although the specific strain was not identified. 19
Itraconazole-resistant S schenckii and S brasiliensis infections in cats have been reported in Malaysia and Brazil, respectively.10,22 A combination of itraconazole and potassium iodide was reported to be effective against resistant S brasiliensis. 22 Itraconazole-resistant feline sporotrichosis reflects the lack of clinical responses to treatment with standard doses, as seen in the current case. Similarly, in Malaysia, cats infected with S schenckii failed to respond to itraconazole at 10 mg/kg for 2 months but were eventually cured using a high-dose protocol. 10 Future studies should focus on the efficacy and complications of combining high-dose itraconazole with other antifungal drugs for resistant sporotrichosis.
As a result of the risk of hepatotoxicity from prolonged administration of high-dose itraconazole, close monitoring is essential. In this case, physical examinations, haematology and serum biochemistry profiles remained within the RI throughout the treatment period.
Primary conjunctival sporotrichosis has been reported worldwide in cat owners, even without traumatic scratches or bites.17,21 Ocular discharges may facilitate transmission, as positive cultures have been obtained from cats without apparent ocular disorders. 28 Fortunately, the owner in this case remained uninfected, highlighting the necessity of educating owners on zoonotic risks and rigorous self-prevention measures.
Conclusions
We reported a case of cutaneous sporotrichosis, initially presenting as a localised subconjunctival lesion in an indoor cat living with his owner. Successful clinical resolution was achieved after 5 months of combined high-dose itraconazole and terbinafine therapy. Veterinarians and pet owners should remain vigilant regarding zoonotic transmission when handling cats with conjunctival nodules or ulcerated eyelids unresponsive to conventional antimicrobial and/or anti-inflammatory therapies.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore, additional informed consent for publication was not required.
