Abstract
Case summary
An 8-year-old male castrated Persian cat was presented to a university teaching hospital for pericardial effusion, fever and hyporexia. Pericardial effusion cytology evaluated previously by the referring veterinarian revealed possible intracellular filamentous bacteria. The cat underwent a CT scan, which revealed severe pericarditis and fibrinous material within the pericardial sac that was adhering to the epicardium, as well as diffuse consolidation of the right middle lung lobe. Subsequently, a median sternotomy, subtotal pericardiectomy and right middle lung lobectomy were performed. Histopathology of pericardial tissue identified a marked chronic, neutrophil-predominant fibrinous pericarditis but no bacteria were visualized. No evidence of infection or neoplasia was identified. The cat’s pyrexia and hyporexia persisted postoperatively and were refractory to several weeks of antibiotic therapy and several days of antiviral administration but rapidly responded to anti-inflammatory steroid administration. The cat was discharged after 12 days in hospital and did not have a recurrence of clinical signs at follow-up 6 months later.
Relevance and novel information
This is the first report of successful management of presumed idiopathic and fibrinous pericarditis and pleural effusion in a cat.
Introduction
Pericardial effusion (PE) in any species may be caused by a variety of etiologies including neoplasia, congestive heart failure, trauma, infection and foreign material. 1 Unlike dogs, who commonly have neoplasia-induced PE, PE in cats is rare and often secondary to cardiac disease.1 –3 Non-cardiac causes of PE in cats are uncommon but have been linked to feline infectious peritonitis (FIP),1 –4 neoplasia,1 –3 bacteremia5 –7 and foreign bodies.8,9 Depending on the character and volume of effusion, life-threatening sequelae, including cardiac tamponade and constrictive pericarditis, may occur.
Due to the rarity of feline PE, treatment recommendations are sparse and non-specific. Survival rates beyond 1 year are below 50%, with 60% euthanized at diagnosis. 2 Recent case studies have reported good outcomes with combined medical and surgical management for feline PE not related to heart failure.5,10 Large-scale studies comparing treatment and outcomes have yet to be conducted.
The aim of this case report was to describe the clinical presentation, diagnosis and successful treatment of a cat with presumed idiopathic fibrinous pericarditis, which has not been previously reported.
Case description
An 8-year-old male castrated Persian cat was referred to a university teaching hospital for further work-up of pericardial effusion, tachypnea and chronic hyporexia. The cat was indoor only, lived with one other cat, was up to date on vaccines and received topical selamectin/sarolaner monthly. The cat had historical right middle lung lobe consolidation noted on radiographs, which was attributed to allergic lower airway disease and treated with oclacitinib (1 mg/kg PO q24h, Apoquel; Zoetis) and intermittent prednisolone tapers (the dose ranged from 2 mg/kg PO q24h to 0.5 mg/kg PO q48h, last administered 18 months prior). Radiographs to check for resolution were not taken. Annual heartworm antigen testing was negative, but antibody testing was not performed. The cat had been seen repeatedly by its primary veterinarian for lethargy, hyporexia and intermittent pyrexia (102.8 °F [39.3 °C]) over the course of a month. At 16 days before presentation to the university hospital, complete blood cell count (CBC) showed an inflammatory leukogram (Table 1); biochemistry and total thyroxine-4 were normal, heartworm antigen was negative and urinalysis was largely unremarkable. Thoracic radiographs revealed historical right middle lung lobe consolidation and a diffuse mild bronchial pattern. The radiologist’s review concluded that these changes were consistent with feline lower airway disease. Due to concern for a secondary bacterial infection, the cat was treated with a 7-day course of both amoxicillin/clavulanic acid (20 mg/kg PO q12h, Clavamox; Zoetis) and enrofloxacin (4 mg/kg PO q24h; Elanco), with buprenorphine (8.6 µg/kg transmucosal q12h; Par) for analgesia, all without improvement (Table 2).
Serial leukogram results

Timeline of medication administration

After 12 days, the cat was presented to an emergency clinic for continued inappetence and labored breathing. Physical examination revealed increased respiratory effort. A fever of unknown origin panel was negative for all agents tested (Table 3) and N-terminal pro B-type natriuretic peptide was within normal limits. A CBC showed mild normocytic, normochromic anemia and lymphocytosis (Table 1). Point-of-care ultrasound (POCUS) revealed moderate PE and scant pleural and peritoneal effusion. The cat was hospitalized at a specialty facility in an oxygen cage, where furosemide (2.19 mg/kg IV q12h; Merck Animal Health), maropitant (1 mg/kg IV q24h; Zoetis) and mirtazapine (2 mg/cat q24h; Dechra) were administered. An echocardiogram identified a progressive, large volume of echogenic PE, fibrin attachments to the pericardium and normal cardiac structure. Pericardiocentesis produced 25 ml of brown, serosanguinous fluid (analgesia not listed in record). Cytology of the effusion showed increased cellularity (>5000/μl) and protein (>3.0 g/dl) levels. The pathologist’s readout reported activated macrophages and degenerate neutrophils occasionally containing fine, filamentous rod-shaped structures suggestive of degenerate bacteria. The cat was administered ampicillin/sulbactam (30 mg/kg IV q8h, Amoxicillin/Sulbactam; Hikma Pharmaceuticals) and enrofloxacin (5 mg/kg IV q24h). Pericardial effusion was submitted for aerobic, anaerobic and mycobacterial cultures but no growth was detected. The cat’s PE persisted despite repeated pericardiocentesis; therefore, the patient was referred to a university hospital.
Fever of unknown origin PCR panel submitted to IDEXX Laboratory on 31 July 2024

On presentation, the cat had a mildly increased respiratory effort, muffled heart sounds and a temperature of 104.6 °F (40.3 °C). POCUS revealed marked PE. Biochemistry revealed mildly increased creatinine kinase and hypoproteinemia (Table 4). Given the concern for septic pericarditis, it was elected to continue broad spectrum antibiotic therapy. The cat was continued on ampicillin/sulbactam (30 mg/kg IV q8h) to provide Gram-positive and anaerobic coverage (including Actinomyces species) and switched to pradofloxacin (7.5 mg/kg PO q24h, Veraflox; Elanco) for Gram-negative coverage. The patient was also started on intravenous crystalloids (45 ml/kg IV q24h, Normosol M with D5W; ICU Medical) and placed in an oxygen cage with FiO2 at 0.4.
Relevant chemistry panel results from 3 August 2024
The following morning, the patient was placed under general anesthesia and thoracic CT revealed pericardial thickening, large volume PE, moderate pleural thickening, diffuse bronchial thickening with multifocal mucus plugging, right middle lung lobe consolidation, diffuse pulmonary atelectasis and sternal lymphadenopathy (Figure 1).

Dorsal (a) and transverse (b) contrast-enhanced thoracic CT scan in soft tissue window. There is moderate pleural thickening, a small volume of bilateral pleural effusion and a large volume of pericardial effusion. Right ventricular free wall flattening is also appreciated, which may be secondary to cardiac tamponade or could be an artifact from the phase of the cardiac cycle. Not shown: a scant right-sided pneumothorax, diffuse bronchial thickening with multifocal mucus plugging, right middle lung lobe collapse, diffuse pulmonary atelectasis and sternal lymphadenopathy. No masses or pulmonary nodules were detected
The patient was prepared for surgery, and an intercostal block was performed bilaterally with 12.5 mg of bupivacaine (Hikma Pharmaceuticals) and 2.5 mg of dexmedetomidine (Zoetis). Exploratory median sternotomy revealed serosanguinous pleural effusion, thickened pericardium and collapse of the right middle lung lobe (Figure 2). A subtotal pericardiectomy was performed; there was a large amount of fibrinous material within the pericardial sac, which was adhering to the epicardium (Figure 2). The material was gently debrided from the epicardium using blunt finger dissection and cotton-tipped applicators. A right middle lung lobectomy was performed, and a chest tube was placed. The thoracic cavity was copiously lavaged with sterile saline and closure was routine. The patient was maintained on a fentanyl (Hospira) constant rate infusion (CRI; 3 µg/kg/min IV) intraoperatively and for the first 24 h postoperatively.
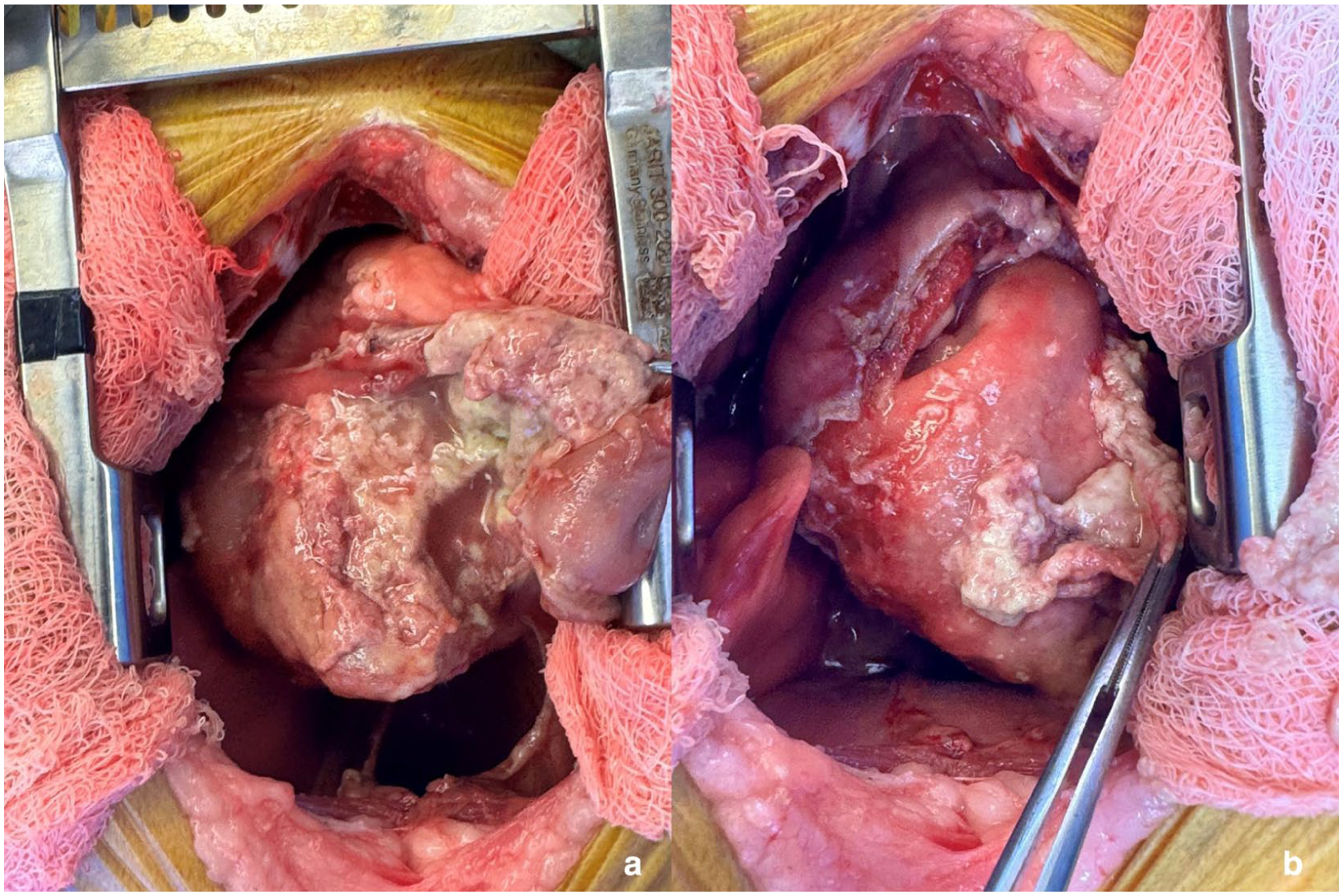
In both intraoperative images, the patient’s head is toward the top of the image. (a) The pericardium has been opened partially and is severely thickened, with fibrinous material adhering to it. A mild amount of fluid is seen in the pericardial sac. (b) The pericardium is retracted out of the chest. It is severely thickened, with a large amount of caseous to fibrinous material lining its inner surface. More fibrinous material is seen covering the epicardial surface
The cat recovered from surgery without complication; pain was monitored using the Glasgow Feline Composite Measure Pain Scale. Pradofloxacin was discontinued. Trimethoprim/sulfamethoxazole (16 mg/kg IV q12h; Somerset Pharmaceuticals) was added out of concern for potential filamentous bacteria, and metronidazole (10 mg/kg IV q12h; Sun Pharmaceutical Industries) was added for increased anaerobic coverage. Ampicillin/sulbactam was continued for Gram-positive and Actinomyces species coverage. By 24 h postoperatively, fentanyl CRI was discontinued and replaced with buprenorphine (20 µg/kg IV q8h as needed) and pregabalin (1.5 mg/kg PO q12h; Ascend). The chest tube was removed 2 days postoperatively as fluid production was minimal. Over the next week, the cat remained eupneic with minimal pleural effusion but persistent pyrexia and inappetence. The cat was started on mirtazapine (1.5 inches transdermal q24h) on day 4 postoperatively, with no improvement in appetite.
Histopathology reported fibrin aggregates admixed with numerous degenerate neutrophils, consistent with a fibrinosuppurative exudate, leading to the diagnosis of fibrinous pericarditis (Figure 3). Although the neutrophilic component of the inflammation was suggestive of a bacterial infection, perivascular inflammation along the pleura in conjunction with the history of tri-cavitary effusion also raised a concern for FIP. Following Gram, Grocott’s methenamine silver and acid-fast stains, no intralesional microorganisms, embedded foreign material or neoplastic cells were observed. Feline coronavirus PCR of the pleural effusion and immunohistochemistry of the pericardium were performed. GS-441524 (17 mg/kg PO q24h; Stoke’s Pharmacy) was started 6 days postoperatively while awaiting results. All test results returned negative including the FIP tests mentioned above, Aspergillus species, Coccidioides species and Toxoplasma gondii serology, pericardium cultures (aerobic, anaerobic, mycobacterial and fungal) and serum antinuclear antibody testing.

Histopathology of the pericardium: hematoxylin and eosin stain at the ×10 objective level (corresponding to ×100 magnification). The inner aspect of the pericardium is overlaid with a thick fibrin mat admixed with degenerate neutrophils (stars), and the subjacent pericardial fibrous tissue is infiltrated by moderate numbers of neutrophils and macrophages. The outer pericardial fibrous tissue is thickened by multifocal, perivascular, high-density aggregates of lymphocytes, plasma cells, macrophages and fewer neutrophils (arrows). No intralesional microorganisms, metazoan parasites, embedded foreign material or evidence of neoplasia were observed in the examined pericardial sections
At 8 days postoperatively, with no improvement on antibiotics and antiviral agents, dexamethasone sodium phosphate (0.15 mg/kg IV q24h; MWI) was administered to reduce the patient’s temperature. At 5 h after administration, the patient’s pyrexia resolved and the cat remained normothermic for the remainder of his hospitalization. At the same time, the cat was started on capromorelin (1.5 mg/kg PO q24h, Elura; Elanco) and started eating well on day 9. GS-441524, trimethoprim/sulfamethoxazole and metronidazole were discontinued on day 10.
At time of discharge, after 12 days of hospitalization, the cat was transitioned from dexamethasone sodium phosphate and ampicillin/sulbactam to prednisolone (1 mg/kg PO q24h; Lloyds Pharmacy) and amoxicillin/clavulanic acid (12 mg/kg PO q12h), respectively. The prednisolone dose was not tapered, and the cat was discharged with a 3-week course of amoxicillin/clavulanic acid to cover for an undiagnosed bacterial component because of cultures being performed after antibiotic administration. The cat was also continued on pregabalin, capromorelin and mirtazapine for comfort and appetite support. At 6 months after discharge, the cat is still managed on long-term prednisolone (1 mg/kg PO q24h) and has a persistent scant pleural effusion, but clinically he is doing well, with no return of pyrexia or hyporexia.
Discussion
To the authors’ knowledge, this is the first report describing the management of presumed idiopathic suppurative and fibrinous pericarditis in a cat. A retrospective study reports idiopathic effusion in only 4% of cats with PE; characteristics of the effusion and treatments have not been well described. 2 In contrast, idiopathic PE in dogs is reported in 20% of cases and is commonly hemorrhagic. 11
Historically, feline PE is considered to be a mild and incidental finding, with congestive heart failure secondary to hypertrophic cardiomyopathy.1 –3 Non-cardiac etiologies had larger quantities of effusion but better prognosis, with median survival near 1 year. 2 These included neoplasia, FIP, sepsis, disseminated intravascular coagulation, trauma, peritoneopericardial diaphragmatic hernia and renal failure.1 –3
Previously reported cases of feline septic PE included hepatic abscess, 5 pyometra, 6 recent dental surgery 7 and migrating foreign bodies. 9 Bacterial agents identified in these cases included Escherichia coli,5,6 Peptostreptococcus species, 7 Pasteurella species 8 and Actinomyces species. 9 In a cat with fibrinous PE, a pericardial abscess was identified on necropsy, and colonies were identified as Moraxella oslonesis, Bacteroides pyogenes and Brevundimonas vesicularis. 12 Another cat with eosinophilic PE had Bartonella species implicated as the source. 11 The wide variety of bacterial pathogens implicated in feline septic PE, including Gram-negative, Gram-positive, aerobic, anaerobic and intracellular bacteria, makes empiric antibiotic treatment challenging and in part contributed to the variety of antibiotics trialed in this case. Among the above cases, survival to discharge was more common in those that received pericardiectomy and antibiotic therapy vs medical management alone. Median survival time was also longer in dogs with PE that received a subtotal pericardiectomy. 13 Given the above reports of bacterial causes of PE and the relative success of combined antibiotic therapy and surgical intervention, this course was pursued for this patient.
FIP was suspected in this case due to the presence of tri-cavitary effusions, serosal fibrin deposits and pericardial inflammation in the absence of intralesional microorganisms. However, PCR and immunohistochemistry for feline coronavirus were negative, and the patient did not respond to GS-441524 or trimethoprim/sulfamethoxazole. Furthermore, the cat had no deterioration after stopping the antivirals. Therefore, idiopathic pericarditis was suspected, and the patient was started on anti-inflammatory doses of steroids, after which the cat improved.
In this case, no inciting cause of PE was determined despite thorough diagnostic evaluation. Sterile pyogranulomatous pericarditis has also been reported in one dog; despite pericardiectomy and antibiotics, the dog’s pyrexia only resolved with anti-inflammatory steroids. 14 At the time of the cat’s first pericardiocentesis, possible intracellular filamentous bacteria were seen on cytology; however, all cultures were negative. The initial cultures were collected before the cat was restarted on antibiotics, but because of prior antibiotic use within the previous week, growth may have been inhibited. Given the cat’s lack of clinical response to all antibiotics, and its positive response to anti-inflammatory steroids, a bacterial etiology is considered unlikely although not definitively ruled out.
In humans, idiopathic pericarditis is the most common etiology of PE, at just over 80%, but in many of these cases an unidentified viral cause is suspected. 15 Non-steroidal anti-inflammatory drugs (NSAIDs) are the treatment of choice, while corticosteroids are avoided due to their higher adverse effects profile in people. However, the preference of anti-inflammatories has not been determined in veterinary medicine, and NSAIDs were not effective in the dog with pyogranulomatous pericarditis described above. 14
Conclusions
This is the first report describing the management of presumed idiopathic suppurative and fibrinous pericarditis in a cat. As in a recent canine report, pericardiectomy and anti-inflammatory steroids led to the resolution of effusion and fever, respectively. This contrasts with human treatment recommendations. The low incidence and diagnostic challenges of idiopathic PE have limited our current understanding, and further studies in small animals are needed.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
