Abstract
Case series summary
An 8.5-month-old female British Shorthair (case 1) and a 2-month-old female domestic shorthair (case 2) were referred for dyspnoea and lethargy. Echocardiography was compatible with unrestrictive patent ductus arteriosus (PDA) with continuous left-to-right shunting and concomitant iso-systemic pulmonary hypertension (PH) (case 1) and restrictive PDA with mild PH (case 2). Radiography showed cardiomegaly, increased pulmonary vascular markings and diffuse interstitial-alveolar pattern consistent with congestive heart failure (CHF) in both cats. After testing for pulmonary vascular reactivity (case 1) and treatment for acute CHF (case 2), surgical ligation of the PDA was attempted. In case 1, PDA ligation led to severe hypotension and asystole, prompting loosening of the ligature because of suspected intolerance to PDA closure. Cardiac massage was initiated and successfully restored the sinus rhythm. The procedure was aborted after partial PDA attenuation. Postoperative echocardiography raised suspicion of pulmonary artery ligation and supra-systemic right ventricle. No surgical revision was performed as the cat remained clinically stable; however, she died 10 days after surgery. In case 2, severe bradycardia developed 20 mins after duct ligation, followed by cardiorespiratory arrest unresponsive to resuscitation. Anatomopathological examination revealed main pulmonary artery and PDA ligation (case 1), and complete left pulmonary artery (LPA) ligation (case 2).
Relevance and novel information
Inadvertent ligation of the pulmonary artery has been reported in human medicine, and to the best of our knowledge, this is the first report in cats. Prompt recognition and correction of such errors are critical for a favourable immediate perioperative outcome and improved long-term prognosis.
Case series description
Case 1
An 8.5-month-old intact female British Shorthair cat weighing 2.7 kg with a body condition score (BCS) of 3/9 was referred for investigation of chronic lethargy, dyspnoea and coughing. One month before presentation, an echocardiogram performed at another practice raised suspicion of congenital heart disease, for which treatment with furosemide, enalapril, atenolol and aspirin was initiated. On presentation, the cat was quiet but fully responsive with a respiratory rate of 40 breaths/min and mild increased effort. Cardiac auscultation revealed gallop sound but regular rhythm, with a heart rate of 180 beats/min. Physical examination was otherwise unremarkable. An echocardiogram showed increased left ventricular diameters, indicating volume overload and a degree of systolic dysfunction. Right parasternal short-axis views of the heart base at the level of the aorta and pulmonary artery, besides the enlarged left atrium, revealed subjective right ventricular concentric hypertrophy and dilation of the main pulmonary artery and its branches. A left cranial parasternal long-axis view of the pulmonary artery detected a tubular structure compatible with patent ductus arteriosus (PDA) with minimal left-to-right shunt, raising suspicion of pulmonary hypertension (PH) with equalisation of pulmonary and systemic vascular pressures. To test the pulmonary vascular reactivity and the feasibility of PDA closure, therapy with sildenafil (1.16 mg/kg PO q12h) was initiated while continuing with furosemide (1.85 mg/kg PO q12h). The other medications were discontinued. An echocardiogram 1 week later confirmed a large PDA with a tubular shape without restriction in diameter at the pulmonary insertion and showed further left ventricular dilation, suggesting a positive result to vasoreactivity testing and indication for surgical closure of the PDA.
One month later, before surgery, paradoxical breathing, increased bronchovesicular sounds and a systolic grade 2/6 heart murmur over the left parasternal area were detected. Haematology and serum biochemistry were unremarkable. Echocardiography revealed severe left ventricular dilation (Table 1) 1 and a continuous left-to-right shunt with a Doppler flow pattern indicative of a haemodynamically significant PDA, with maximal end-diastolic velocity less than 1 m/s, suggesting unrestrictive pulsatile transductal flow (Figure 1a) (see Video 1 in the supplementary material; please see also the supplementary table for full descriptions of the videos). 2 Thoracic radiography showed severe cardiomegaly, increased pulmonary vascular markings and diffuse interstitial-alveolar pattern; leftward bulge of the aorta, prominent main pulmonary artery and left auricular enlargement were visible in the dorsoventral view. Thoracic CT demonstrated a large tubular PDA, approximately 6.5 mm in diameter, with both the ampulla and the minimal ductal diameter measuring the same (Figure 1b).
Echocardiographic parameters at presentation, 1 month after initiation of therapy with sildenafil in addition to furosemide and 1 day postoperatively
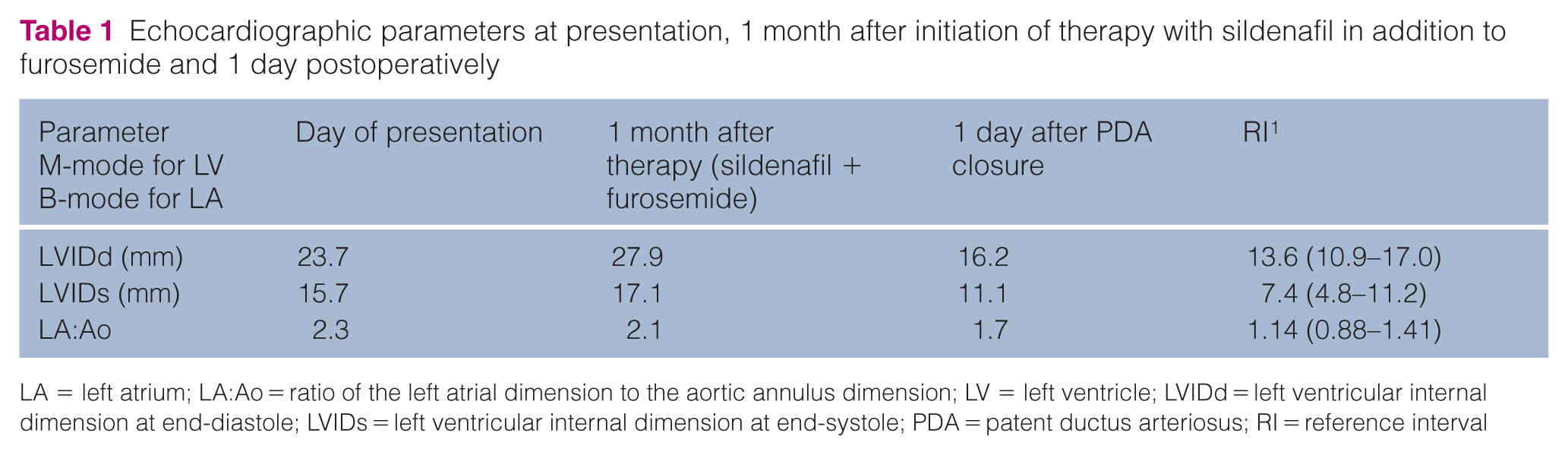
LA = left atrium; LA:Ao = ratio of the left atrial dimension to the aortic annulus dimension; LV = left ventricle; LVIDd = left ventricular internal dimension at end-diastole; LVIDs = left ventricular internal dimension at end-systole; PDA = patent ductus arteriosus; RI = reference interval

(a) Right parasternal short-axis view of the heart base at the level of pulmonary arteries optimised for spectral Doppler interrogation of ductal flow, demonstrating continuous left-to-right shunting with a reduced velocity through the patent ductus arteriosus (PDA) (unrestrictive pulsatile flow) before surgery in case 1. (b) Thoracic radiography (right lateral and dorsoventral view) and CT showing severe cardiomegaly, increased pulmonary vascular markings, diffuse interstitial-alveolar pattern and a large tubular PDA without any constrictions at its ends, respectively. Notice the prominent main pulmonary artery and leftward bulge of the aorta in thoracic dorsoventral view (case 1). (c) Right parasternal short-axis view of the heart base at the level of pulmonary arteries optimised for spectral Doppler interrogation of pulmonary artery/ductal flow after surgical closure of the PDA in case 1, demonstrating predominantly continuous flow below the baseline throughout the diastole, resembling a ‘sawtooth’ flow pattern suspicious of pulmonary artery branch stenosis and reversed PDA
During surgery, when ligation of the ductus was attempted, the cat developed severe hypotension followed by asystole, prompting partial release of the ligature. Cardiac massage was initiated and successfully restored sinus rhythm. The procedure was aborted with partial attenuation of the PDA. Postoperative analgesia with methadone (0.3 mg/kg IV q6h) was administered during hospitalisation. An echocardiogram performed the following day showed a significant reduction in the dimensions of the left heart chambers, dilation of the right heart chambers and bowing of the interventricular septum into the left ventricle, suggestive of supra-systemic right ventricle. Tricuspid regurgitation with a peak systolic velocity of 5.79 m/s and pulmonary regurgitation with peak diastolic velocity of 4.02 m/s further supported the suspected supra-systemic right ventricle. In addition, turbulent flow was observed in the right pulmonary artery, not present before the surgery (see Video 2 in the supplementary material). Continuous-wave Doppler in the pulmonary artery/PDA was different from the preoperative examination, showing predominantly continuous flow below the baseline throughout the diastole with a ‘sawtooth’ pattern suspicious of pulmonary artery branch stenosis and reversed PDA (Figure 1c). 3 Comparing the same views of the pulmonary artery before and after surgery, a significant reduction of the main pulmonary artery and its right branch was evident and raised suspicion of inadvertent pulmonary ligation (see Videos 3 and 4 in the supplementary material). However, given an apparently stable condition and the risk related to general anaesthesia, revision of the surgery was not considered. The cat was discharged 3 days postoperatively and was not monitored at our institution the following week. According to the owners, gastrointestinal signs, lethargy and anorexia developed before death. The exact cause of death could not be determined, although it was likely related to the surgical complication.
Post-mortem examination of the heart fixed in formalin showed an increase in volume of the left ventricle and severe dilation of the left atrium and auricle. Around the last portion of the pulmonary artery and the origin of the right and left branches, there was a tight ligation completely overhanging the PDA area (Figure 2). The cross-section of both ventricles showed severe concentric hypertrophy of the right ventricle.
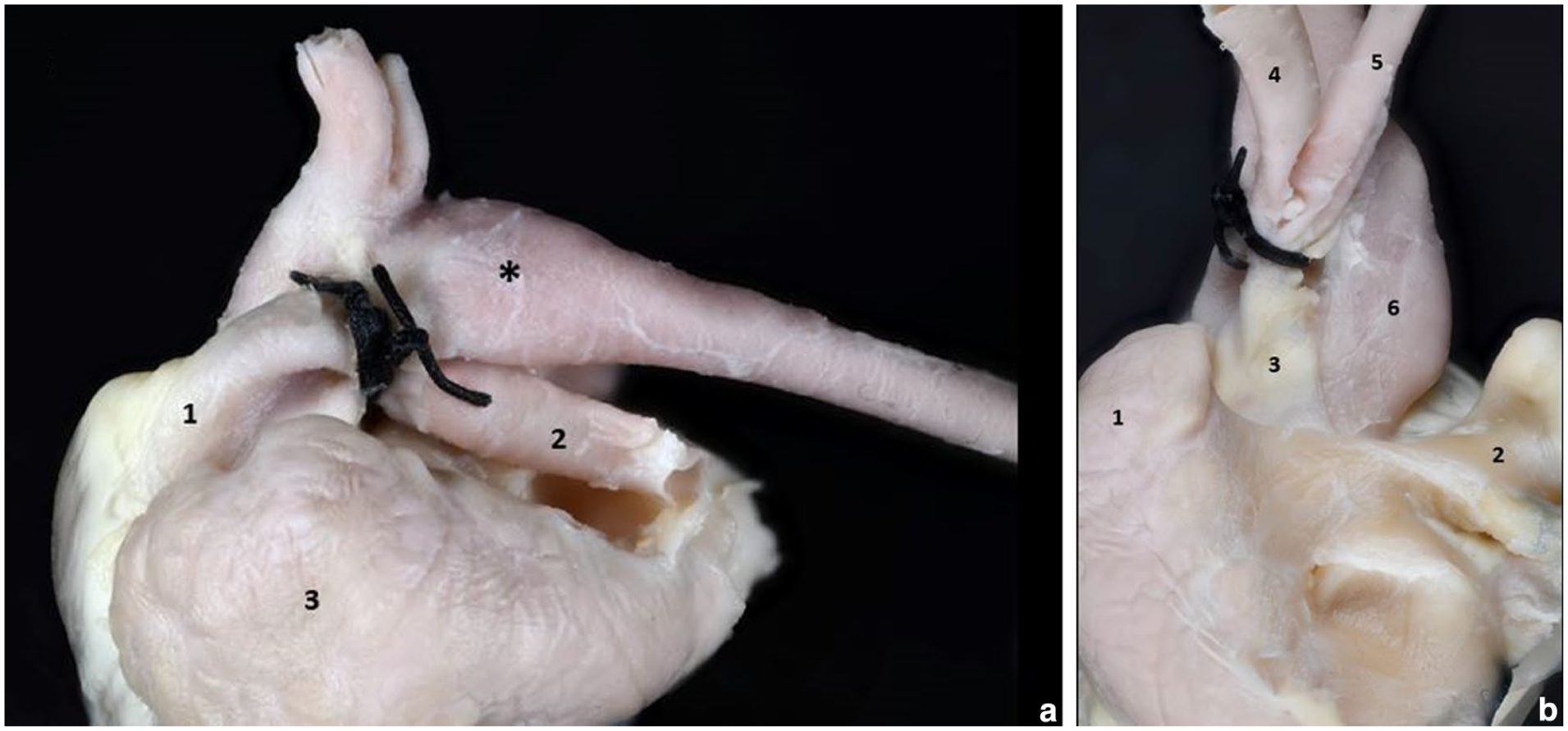
(a) Case 1, left view. Surgical ligation around the last portion of the pulmonary artery and left branch. The patent ductus arteriosus is completely overlaid by the ligature. Note moderate dilation of the aortic isthmus and the first section of the thoracic aorta (*). 1 = pulmonary artery; 2 = left pulmonary branch; 3 = left appendage (severely dilated). Folding of the pulmonary artery wall could indicate dilation. (b) Case 1, caudal view after dislocating the pulmonary artery and aorta dorsally and the right auricle folded to the right. Strong surgical ligation around the last section of the pulmonary artery and at the origin of the right and left branches. 1 = left auricle; 2 = right auricle; 3 = pulmonary artery; 4 = left pulmonary artery; 5 = right pulmonary artery; 6 = aorta
Case 2
A 2-month-old intact female domestic shorthair cat weighing 1.9 kg with a BCS of 2/9 and an unknown medical history, as she had been found as a stray, was referred for a 2-day history of lethargy, dyspnoea and anorexia. Physical examination on admission revealed tachypnoea/dyspnoea, a systolic grade III/VI heart murmur over the left parasternal area and mild hypothermia. Point-of-care thoracic ultrasound showed severe left atrial dilation and coalescent B-lines artefacts, raising suspicion of congestive heart failure (CHF). After stabilisation with repeated boluses of intravenous furosemide and oxygen supplementation, echocardiogram and thoracic radiography carried out 24 h later diagnosed a moderate left-to-right shunting PDA (Figure 3) and cardiogenic pulmonary oedema, respectively. Treatment with furosemide was continued, and surgical closure of PDA was planned in 2 weeks. The patient was premedicated with methadone (0.2 mg/kg IM) and alfaxalone (1.5 mg/kg IM) followed by induction with alfaxalone (0.7 mg/kg IV) and maintenance with isoflurane. At 20 mins after PDA closure, during the placement of intradermal sutures, sudden bradycardia developed, which was followed by cardiac arrest unresponsive to resuscitation.

Right parasternal short-axis view of the heart base at the level of pulmonary arteries optimised for evaluation of ductal anatomy (case 2). AD = ampulla diameter; MDD = minimal ductal diameter; PV = pulmonary valve; RPA = right pulmonary artery; RPV = right parasternal view; TV = tricuspid valve
The necropsy showed poor general condition and a large, shaved area on the right side of the chest, on the skin of which there was a surgical suture. Examination of the chest cavity showed severe congestion and pulmonary oedema. External examination of the heart showed an increased volume of the left ventricle and marked dilation of the left atrium and auricle. Examination of the left side of the origin of the great vessels at the base of the heart showed the PDA and a ligation around the left pulmonary artery (LPA) (Figure 4). Histological examination, performed with routine techniques and with haematoxylin and eosin staining, revealed severe congestion and alveolar oedema in both lungs.

(a) Case 2, left side of the chest. Severe dilation of the left auricle (1) and enlargement of the left ventricle (2) are evident. Note the patent ductus arteriosus (PDA) (white arrow) and the surgical ligation (black arrow) around the left pulmonary artery (LPA). (b) Case 2, left side of the chest. Opening of the pulmonary artery (1), PDA (arrow) and the first tract of the thoracic aorta (2). Note the presence of the surgical ligation around the LPA
Discussion
In cats, PDA is reported less frequently than in dogs, with a prevalence of 5%. It also appears to be more commonly associated with PH, likely related to ductal morphology that shows minimal tapering at the pulmonary artery ostium, unlike what is typically seen in dogs.4,5 Radiographic signs of pulmonary over-circulation were evident in this case, which are typical findings of left-to-right PDA; however, they have also been reported in some cats with right-to-left PDA. 5 After birth, in patients with a large PDA (case 1), the drop in pulmonary vascular resistance creates a degree of left-to-right shunting; however, pressures in the aorta, ductus arteriosus, pulmonary artery and both ventricles remain equal. Managing PDA in the presence of PH remains an important dilemma, even in humans. 6 Decision-making is frequently based on pathophysiological considerations and expert opinion in referral centres, where a treat-and-repair approach is considered based on a predominantly reversible pulmonary vascular disease vs predominantly irreversible (pre-Eisenmenger). 7 In case 1, therapy with sildenafil revealed a positive response to vasoreactivity testing based on increased left ventricular diameter in diastole, indicating increased pulmonary blood flow (Table 1). Indeed, it is the authors’ opinion – based on several observations – that a positive vasoreactivity test result should not solely rely on velocity measurements or pressure-gradient estimates across the PDA, as these parameters are unlikely to change significantly. This is especially true in cases such as this, where the PDA type was most likely in a transitional stage between type 3b and type 4, with equal pressures in both ventricles and great arteries.6 –8 In case 2, there was a degree of restriction to the flow in the PDA and clinical signs were mostly related to left-sided CHF (type 3a). 8
Although different occlusion devices have been used to close the PDA in cats, including vascular plugs, coils and Canine Duct Occluders,9 –11 surgical closure remains the first choice of treatment for many veterinarians. 5 Attention should be given to both procedures when concurrent PH is present, as closure may not be feasible in all patients because of the onset of systemic hypotension or right heart failure after PDA closure.12 –14 Typically, long-term therapies are recommended to manage PH after PDA closure in both human and small animal patients.7,12,13 Indeed, treatment with sildenafil was continued for 1 month in a cat 12 and for several months in a dog. 13 However, long-term follow-up data are lacking in veterinary medicine.
Several complications have been reported after surgical ligation of the PDA, such as left-sided laryngeal paralysis, voice change, difficulty swallowing, fever, haemorrhage, chylous effusion and iatrogenic lung injury. 15 However, to the best of the authors’ knowledge, inadvertent ligation of the pulmonary artery has not yet been described in cats.
In a human case report, a long-term survivor of more than 30 years without medical treatment was described, in whom investigations for suspected PH revealed an accidental ligation of the LPA. 16 A review of the literature revealed inadvertent LPA ligation in more than a dozen patients, and most of them survived with or without corrective surgery carried out 1 day to 3 years later. Similarly, in one dog investigated for a PDA that had been attempted to be closed surgically 2 weeks before consultation, inadvertent LPA ligation was identified, and after PDA closure, the dog was still alive 3.5 years later. 17 This complication, observed in case 2, was fatal, likely due to acute increased afterload on a non-adapted right ventricle. In addition, it caused flow and pressure shifting to the right lung lobes without closure of the PDA in a patient who had suffered recent pulmonary oedema and therefore had compromised pulmonary function, which further reduced the ability to tolerate this complication. In case 1, concomitant partial closure of the main pulmonary artery and PDA was likely better tolerated, as the right ventricle was well adapted to the coexisting pre-Eisenmenger stage. However, the error resulted in a supra-systemic right ventricle, which likely contributed to the fatal outcome over time. The inability to perform a histopathological examination of the lungs is a limitation of this case.
Conclusions
Inadvertent ligation of the pulmonary artery during surgical closure of a PDA is a serious complication that, although rarely reported in humans, may also occur in cats and has been described in one dog. Consequently, clinical signs such as severe hypoxia and hypotension, along with the acute onset of pulmonary hypertension characterised by pronounced right ventricular dilation, should raise suspicion for this surgical error. Prompt recognition and correction are essential to achieving a favourable immediate perioperative outcome and improving the long-term prognosis.
Supplemental Material
Supplementary table
Full descriptions of the video supplementary material.
Footnotes
Author note
Altin Cala is currently affiliated with Linnaeus Veterinary Limited at North Downs Specialist Referrals, Bletchingley Surrey, RH1 4QP, UK, where he holds the position of cardiology clinician.
Supplementary material
The following files are available as supplementary material:
Video 1. Transthoracic two-dimensional colour-flow Doppler examination of left-to-right shunting through a patent ductus arteriosus. Right parasternal short-axis view of the heart base at the level of pulmonary arteries showing a continuous red laminar flow towards the pulmonary trunk. Notice the blue laminar flow in the right pulmonary artery.
Video 2. Transthoracic two-dimensional colour-flow Doppler examination of left-to-right shunting through a patent ductus arteriosus after surgical closure. Right parasternal short-axis view of the heart base at the level of pulmonary arteries showing turbulent flow in the right pulmonary artery, not present before surgery. The continuous red laminar flow towards the pulmonary trunk is less evident on this occasion.
Video 3. Transthoracic two-dimensional examination of the pulmonary artery and its branches before surgical closure of a patent ductus arteriosus. Right parasternal short-axis view of the heart base at the level of pulmonary arteries showing a dilated main pulmonary artery compared to the aorta. The right and left pulmonary branches are also very prominent.
Video 4. Transthoracic two-dimensional examination of the pulmonary artery and its branches after surgical closure of a patent ductus arteriosus. Right parasternal short-axis view of the heart base at the level of pulmonary arteries showing a significant reduction in the size of both right and left pulmonary branches postoperatively, indicative of inadvertent pulmonary ligation.
Supplementary table. Full descriptions of the video supplementary material.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written for their use in the publication was obtained from the people involved.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
