Abstract
Case summary
An 8-year-old castrated male cat presented with a 7-year history of urinary incontinence (UI) and severe urinary scalding (US) caused by continuous urine leakage after prepubic urethrostomy (PPU) performed at 5 months of age for urinary trauma. The patient experienced significant pain and discomfort associated with US. Despite attempts at medical management, the patient’s condition did not improve. Consequently, a hepatic occluder (HO) device was implanted around the shortened urethra after the PPU. Gradual occluder inflation was conducted over approximately 13 weeks to facilitate sphincter function recovery. The HO device effectively improved and controlled the UI, significantly enhancing the patient’s quality of life and satisfying the owners.
Relevance and novel information
This case report describes the successful use of an HO to manage UI in a cat with a structurally irregular and shortened urethra resulting from traumatic injury and multiple prior surgeries. Urethral sphincter occluders, including HO, may represent a viable salvage option for managing UI in cats with complex urethral anatomy.
Introduction
Urinary incontinence (UI), defined as involuntary urine leakage during the storage phase, 1 is uncommon in cats, comprising up to 4% of lower urinary tract diseases. 2 Severe UI can significantly affect quality of life (QoL) and may lead to euthanasia. Urethral sphincter occluders, including artificial urethral sphincters (AUSs) and hydraulic occluders, are adjustable implants placed around the urethra to treat refractory UI. 3 Several feline case reports suggest that such devices can improve continence and QoL;4 –8 however, data on their feasibility in cats with a short urethra after prepubic urethrostomy (PPU) remain limited.
This report describes successful UI management using a hepatic occluder (HO) device in a cat that underwent PPU after traumatic urethral injury.
Case description
An 8-year-old castrated male cat (weight 3.48 kg, body condition score 3/5) was referred for evaluation of persistent urinary scalding (US) secondary to UI. At 5 months of age, the cat sustained a pelvic urethral rupture and tail injury from a traffic accident, and was treated with PPU and tail amputation; however, 2 months later, urethral stenosis developed at the abdominal wall. Despite multiple surgeries to enlarge the urethral orifice and reconstruct the urethra, UI and US progressively worsened, accompanied by recurrent bacterial cystitis and struvite urolithiasis. Antibiotic therapy failed to resolve the UI, and frequent hospitalisation for catheter management was required because of declining QoL.
On initial evaluation, the cat had a slightly palpable bladder, severe US and a continence score (CS) 9 of 1/10 (1 = constant leakage, 10 = complete continence). Haematology and serum biochemistry were unremarkable. Abdominal radiography confirmed a small, normally positioned bladder, and ultrasonography revealed a thickened, irregular urethra. An 8 Fr Foley catheter was placed to manage US, and the patient remained hospitalised with closed-system catheter care for 46 days at the owner’s request. After US resolved, phenylpropanolamine (PPA) (1.8 mg/kg q12h, Proin; PRN Pharmaceutical) was administered for 1 week after catheter removal, but no improvement in UI was observed (CS remained 1/10) and US recurred. Increased aggression led to discontinuation of PPA. Surgical management was elected, and CT followed by HO implantation was performed on day 76.
Preoperatively, atropine (0.04 mg/kg SC; Mitsubishi Tanabe Pharma) and fentanyl citrate (2.5 µg/kg IV; Terumo) were administered. Anaesthesia was induced with propofol (6 mg/kg IV; Mylan Seiyaku) and maintained with 1.4–1.8% isoflurane (IsoFlo; Zoetis) in oxygen via a 4.5 mm endotracheal tube. CT revealed no urinary tract uroliths or anatomical abnormalities, but the intraperitoneal urethra was short (approximately 11 mm) and irregular because of previous surgeries, including PPU and reconstruction (Figure 1). Aseptic surgical procedures were subsequently performed. Intra- and postoperative management included continuous rate infusion of dopamine (2.5–5.0 µg/kg/min; Teva Takeda Pharma) and fentanyl (1.25–10 µg/kg/h), and intravenous ampicillin (20 mg/kg; Meiji Seika Pharma).

Sagittal image from preoperative abdominal contrast-enhanced CT. The intraperitoneal urethra, affected by prior surgical procedures such as prepubic urethrostomy and urethral reconstruction, was shortened to approximately 11 mm in length and measured about 11 mm in diameter. In addition, its shape was irregular, displaying significant anatomical changes
The patient was positioned in dorsal recumbency and a midline celiotomy was performed from 1 cm cranial to the PPU stoma to the cranial edge of the pubis. Adhesions between the bladder and abdominal wall were dissected, and a stay suture was placed on the cranial bladder. A 10 mm HO device (LePort; cuff width 7 mm, thickness 2 mm; Norfolk Vet Products) was placed around the urethra caudal to the bladder and secured with 3-0 polypropylene (Prolene; Johnson & Johnson) through the cuff eyelets (Figure 2). The actuation tube was connected to an access port, which was fixed to the femoral fascia with 4-0 polypropylene (Prolene) (Figure 3). The system was primed with sterile saline per manufacturer instructions. After confirming urethral occlusion at 0.5 ml inflation, saline was withdrawn. The abdomen was routinely closed and a urethral catheter was placed for US management.

Intraoperative image showing the placement of an inflatable silicon vascular occluder around the dissected portion of the urethra, caudal to the bladder. The cuff was positioned around the urethra at the dissected area and prepared for fixation with a polypropylene suture passed through the eyelets

Intraoperative image showing the placement of the actuating tube and access port. The access port was positioned medially on the thigh, as recommended by Norfolk Vet Products. The tube was carefully advanced from the hepatic occluder cuff placement site on the urethra to the access port on the medial thigh without tension or twisting. The tube was not constrained to ensure proper positioning. The blue boot at the end of the catheter was held to facilitate passage through the subcutaneous tunnel to the access port
Surgical duration was 80 mins, and the patient recovered well postoperatively. Blood tests on postoperative day (POD) 1 showed no abnormalities. The patient received a subcutaneous injection of 20 mg/kg cefovecin sodium (Convenia; Zoetis). Postoperative radiographs confirmed appropriate placement of the occluder and access port (Figure 4). The patient was hospitalised until POD 14, and the urinary catheter was removed before discharge.
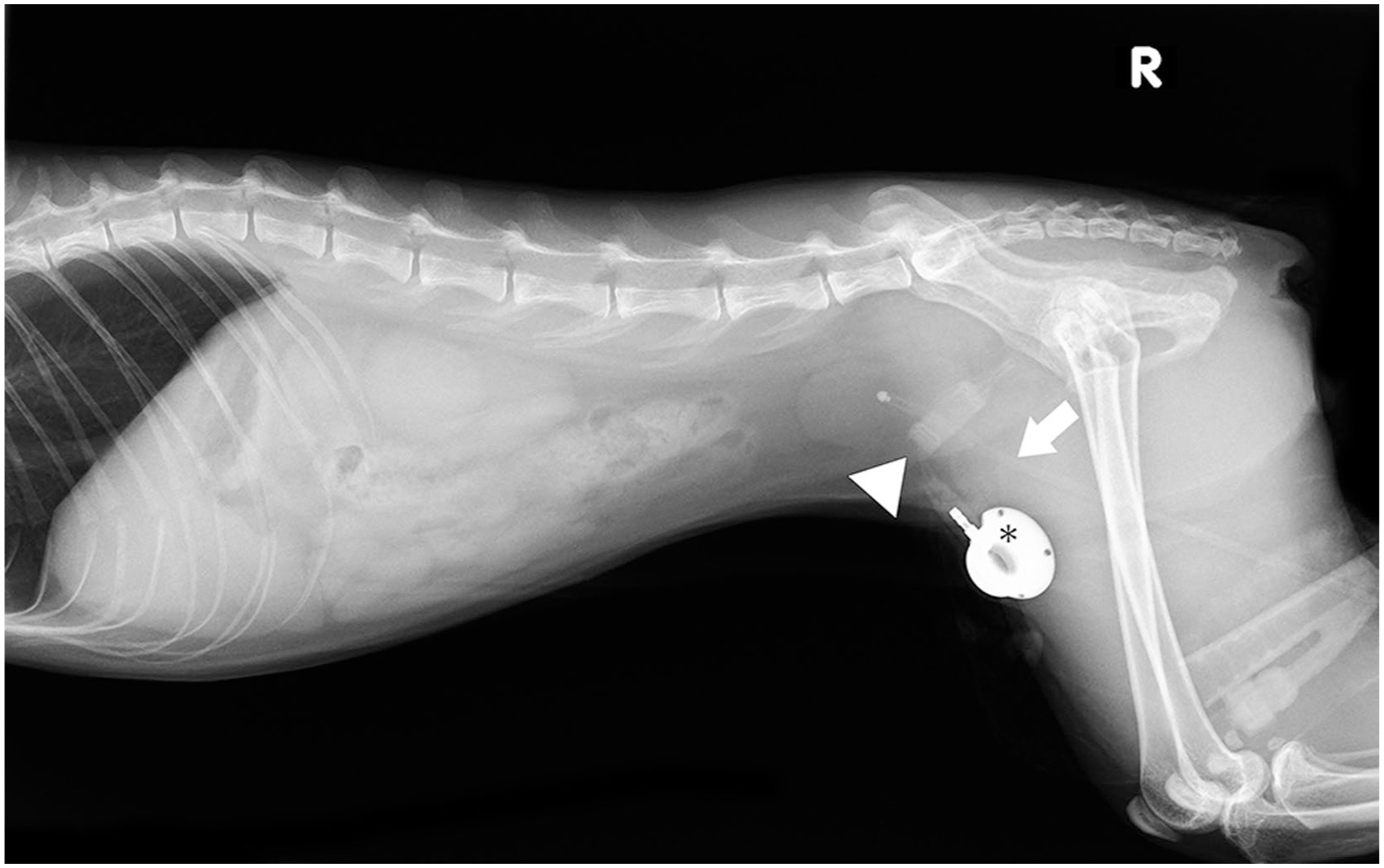
Abdominal radiograph obtained immediately postoperatively to assess the spatial relationship between the hepatic occluder cuff, access port and urethra. The patient was positioned in right lateral recumbency during imaging. Key structures: urethral catheter (white arrow), hepatic occluder cuff (white arrowhead) and access port (asterisk)
All saline injections into the access port were performed without sedation. On PODs 18 and 20, 0.1 ml was injected each time because of worsening US (CS 4/10), resulting in a total cuff volume (TCV) of 0.2 ml. As signs worsened, 0.2 ml was withdrawn on POD 22, and a urethral catheter was placed. US resolved by POD 40 and the catheter was removed. Subsequent injections included 0.3 ml on POD 40 (CS 7/10), 0.05 ml on POD 43 (TCV 0.35 ml) and withdrawal of 0.05 ml on POD 50 because of urinary retention (TCV 0.3 ml). Minor adjustments followed: 0.01 ml on PODs 81 and 94 (TCV 0.32 ml). By POD 197, CS was 9/10, with no recurrence of US, normal blood test results and high owner satisfaction (Figure 5).

Comparison of abdominal photographs. (a) At the initial consultation, the patient had a continence score (CS) of 1/10, with severe urine scalding (US) and significant pain. (b) By postoperative day 197, the CS had improved to 9/10. Although complete resolution of incontinence was not achieved, US did not recur and the patient’s condition significantly improved. The degree of US is visibly reduced in the photographs, illustrating the patient’s progress
Discussion
UI has various causes5,10,11 and may occur as a post-operative complication of PPU, often due to nerve injury or reduced urethral length.6,12 –14 In our case, UI was likely related to urethral shortening or neuropathy after PPU. Neurological deficits, such as hindlimb paralysis, were absent, and normal posture during urination and defecation was observed before HO placement. These findings suggested intact bladder contractility and no storage dysfunction, supporting the suitability of HO placement.
PPA, an alpha-adrenergic agonist, is considered effect-ive for the management of acquired urethral sphincter mechanism incompetence in dogs; 15 however, its efficacy is still controversial in cats.5,8,10,16 –18 It has been suggested that feline urethral smooth muscle may exhibit a lower responsiveness to PPA compared with dogs, possibly due to morphological differences in the lower urinary tract.19,20 In our case, PPA administration resulted in minimal clinical improvement, which aligns with anecdotal reports describing limited efficacy in cats. Further studies are warranted to clarify the therapeutic potential of PPA in feline UI, particularly in cases with structural or neurological involvement.
Contrast-enhanced CT allowed confirmation of a continuous segment with sufficient diameter for HO placement. Previous studies have demonstrated that CT excretory urography is superior to ultrasonography and fluoroscopy in identifying complex lower urinary tract anomalies in dogs.15,21 In our case, abdominal ultrasonography and radiography did not allow the accurate measurement of urethral diameter; however, postoperative lateral radiography was helpful to confirm proper cuff positioning. Postoperative radiography can be repeated periodically to screen for complications including device migration and rotation. This approach is consistent with the previous studies that reported on serial radiography-guided decisions regarding device inflation and surgical revision.9,22
Preoperative CT demonstrated a luminal urethral diameter of approximately 11 mm and a total intraperi-toneal urethral length of only 11 mm; therefore, an HO with a 10 mm internal diameter and a 7 mm cuff width was selected. The smallest feline-specific artificial urethral sphincter (AUS) device was an 11 mm cuff width, which was deemed unsuitable considering the required space for implantation. Evidence-based guidance on appropriate cuff sizing in cats is currently limited.4 –8 In dogs, one study recommended selecting a cuff diameter equal to approximately half of the urethral circumference. 23 Another series reported favourable continence outcomes when the cuff circumference matched or slightly exceeded the urethral measurement. 24 In the absence of robust feline-specific data, individualised device selection based on urethral anatomy and spatial constraints, followed by staged postoperative adjustment, may represent the most pragmatic approach.
Although open surgery was required in our case, the HO allowed postoperative cuff volume adjustment to modulate urethral pressure non-invasively. 3 Current guidelines recommend delaying inflation for 4–6 weeks to promote urethral tissue revascularisation and reduce ischemic risk;24,25 however, feline studies indicate earlier reperfusion may occur.26 –28 For example, contrast studies showed urethral healing within 5–14 days after catheter realignment 26 and submucosal vascular maturation by POD 12 after perineal urethrostomy. 27 In our case, initial adjustment was performed on POD 18 to allow adequate reperfusion and sphincter recovery. Cuff inflation was guided by prior reports suggesting 0.1 ml increments while maintaining urinary flow,25,29,30 although some animals achieve continence without reinflation because of the semi-rigid cuff design.4,5,9,23-25,31 The infused volume (0.32 ml) was consistent with previous feline cases (0.1–0.4 ml).4 –8 Adjustments were made at short intervals per the owner’s wish to prevent recurrent US.
The current American College of Veterinary Internal Medicine guidelines do not recommend routine postoperative antibiotic use for urinary incontinence. 15 However, in this case, cefovecin sodium was administered because of concern about implant-associated infection. In dogs, a study reported bacterial colonisation based on positive culture from fibrous tissue or the device itself in all six cases with urethral stricture after AUS placement. 22 Although rare, implant-associated infection involving the subcutaneous port or occluder system remains a potential risk. In our case, prophylactic antibiotic administration was considered appropriate for two reasons: the cat was unable to receive oral medications during PPA therapy, and postoperative urethral catheterisation was anticipated. Catheter placement may increase the risk of urinary tract infection with antimicrobial-resistant organisms and could contribute to bacterial seeding of the implant.
Although the urinary catheter was used for a limited time in our case, its presence may still pose risks to ur-ethral healing. In particular, catheterisation has been implicated in the development of urethral strictures at stoma sites, including those created during prepubic or perineal urethrostomy. A retrospective study evaluating complications of perineal urethrostomy in cats identified urethral stricture as a notable postoperative concern, with contributing factors such as local inflammation and mechanical irritation. 32 In our case, the urinary catheter was maintained for 46 days before HO placement. During this period, a closed-collection system and strict aseptic protocols were employed to minimise mechan-ical trauma and reduce the risk of catheter-associated complications at the stoma site.
Assessing treatment outcomes for UI can be difficult because of variability in sign severity. In our case, UI was evaluated using the owner’s subjective CS, a simple 1–10 scale previously reported. 9 Although easy to apply, the CS lacks objective standardisation and may be prone to bias. Urethral pressure profilometry, a component of uro-dynamic testing, could provide more objective data on sphincter function, as recommended in human medicine before procedures that increase urethral resistance. 33 Similar principles may apply to veterinary cases, although urodynamic testing is rarely used in cats because of the need for sedation, technical challenges and inconsistent reliability. 3 As a result, profilometry was not performed in our case. Nonetheless, incorporating such methods in future studies may improve standardisation. Despite the subjective nature of CS, the marked improvement from 1/10 preoperatively to 9/10 postoperatively indicated effective UI control and owner satisfaction.
Conclusions
This case report describes the successful use of an HO to manage UI in a cat with a structurally irregular and shortened urethra resulting from traumatic injury and multiple prior surgeries. Urethral sphincter occluders, including HO, may represent a viable salvage option for managing UI in cats with complex urethral anatomy.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
