Abstract
Case summary
A 1-year-old spayed female mixed-breed cat presented with chronic large bowel diarrhoea. The cat was treated with various antibiotics, prednisolone and dietary supplements without significant improvement. Endoscopic examination revealed an irregular colonic mucosa with multifocal erosion and haemorrhage. Impression smears of the colonic mucosa contained large numbers of unicellular round-to-oval organisms and macrophages. Histologically, granulomatous inflammation with numerous organisms was observed in the lamina propria of the colonic mucosa. The organisms had a cell wall and internal segmentation, which were positive on periodic acid–Schiff and Grocott’s methenamine silver staining. Immunohistochemically, CD204-positive macrophages had accumulated in the lesions. Ultrastructural examination revealed dense bodies and starch granules, and the absence of chloroplasts in the cytoplasm of the organisms. PCR and a sequence analysis detected the Prototheca bovis 18S rRNA gene in formalin-fixed, paraffin-embedded colonic mucosa tissue. Based on these findings, the cat was diagnosed with granulomatous colitis associated with P bovis infection.
Relevance and novel information
This first report of feline intestinal protothecosis suggests that Prototheca species infection should be considered in the differential diagnosis of cats with treatment-resistant chronic large bowel diarrhoea.
Introduction
Prototheca species are saprophytic, achromatic algae widely found in the environment, including in plants, water, soil, and on the skin and in the faeces of humans and animals.1 –3 They exhibit a yeast-like, round-to-oval morphology measuring 10–30 µm in diameter and have a thick cell wall, ultrastructurally characterised by a double-layered structure.1 –4 Reproduction in Prototheca species is by endosporulation, in which the mother cell (sporangium) produces daughter cells (endospores) within the cytoplasm by internal division.1 –3
Protothecosis has been reported sporadically in humans and other animals.1,2,5 In humans and dogs, protothecosis can cause lesions at various sites, including the skin, eyes and internal organs, such as the intestine.1 –3,5 In cats, cases of cutaneous protothecosis have been reported;1,2,4 –6 however, there is no information concerning the intestinal involvement of the pathogen.5,7 This paper describes the pathological findings in a young cat diagnosed with granulomatous colitis associated with Prototheca bovis infection.
Case description
A 1-year-old spayed female mixed-breed cat was initially presented to a primary care veterinary clinic with a 4-month history of large bowel diarrhoea. Rescued as a kitten from the outdoors in Kanagawa Prefecture, Japan, the cat had been kept indoors since, with no history of travel. Retroviral testing for feline immunodeficiency virus and feline leukaemia virus was negative. Faecal PCR examination (Feline Diarrhea RealPCR Panel; IDEXX Laboratories) returned negative results for all pathogens, including Tritrichomonas foetus, Giardia species, Cryptosporidium species, Toxoplasma gondii, Salmonella species, Clostridium perfringens enterotoxin A, feline coronavirus and feline panleukopenia virus. The cat was treated with various antibiotics, including amoxicillin (17.2 mg/kg PO q12h), cefovecin (8 mg/kg SC two doses), chloramphenicol (17.2 mg/kg PO q12h) and metronidazole (13.4 mg/kg PO q12h), along with prednisolone (0.18–0.5 mg/kg PO q24–48h) and was fed an easily digestible gastrointestinal diet (i/d; Hill’s Prescription Diet, gastrointestinal; Royal Canin) without significant improvement.
Five months after the initial presentation, the cat was referred by the primary care veterinary clinic to the gastroenterology department of a referral hospital for further examination because of worsening diarrhoea. On physical examination, the cat weighed 3.45 kg and exhibited mild muscle loss based on the muscle condition score. Its respiratory status and vision were normal. Neurological examination revealed no abnormalities. Blood analysis, including complete blood count (CBC) and chemistry panel, showed elevated ammonia (152 μg/dl, reference interval [RI] 23–78), glucose (196 mg/dl, RI 71–148) and alkaline phosphatase (65 U/l, RI 0–58). All other chemistry and CBC parameters were within the RIs. Faecal smears showed a large number of bacterial organisms, although no specific pathogens were identified. Abdominal ultrasonography revealed diffuse colonic thickening, measuring up to 2.4 mm.
An endoscopic examination (GIF-XP290N, 5.8 mm scope; Olympus) revealed a markedly shortened colon, with a total length of 14 cm from the anus to the ileocecal valve and a colonic stricture located 11 cm from the anus. The colonic mucosa exhibited irregularities with multifocal erosion and haemorrhage (Figure 1). In addition, the colonic mucosa was fragile, inelastic and tore easily during insufflation. The gastric, duodenal and ileal mucosa appeared unremarkable on endoscopic examination. Mucosal samples from the stomach, duodenum, ileum and colon were endoscopically collected using forceps (VH-142-B52; Olympus) for a 2.2 mm channel.
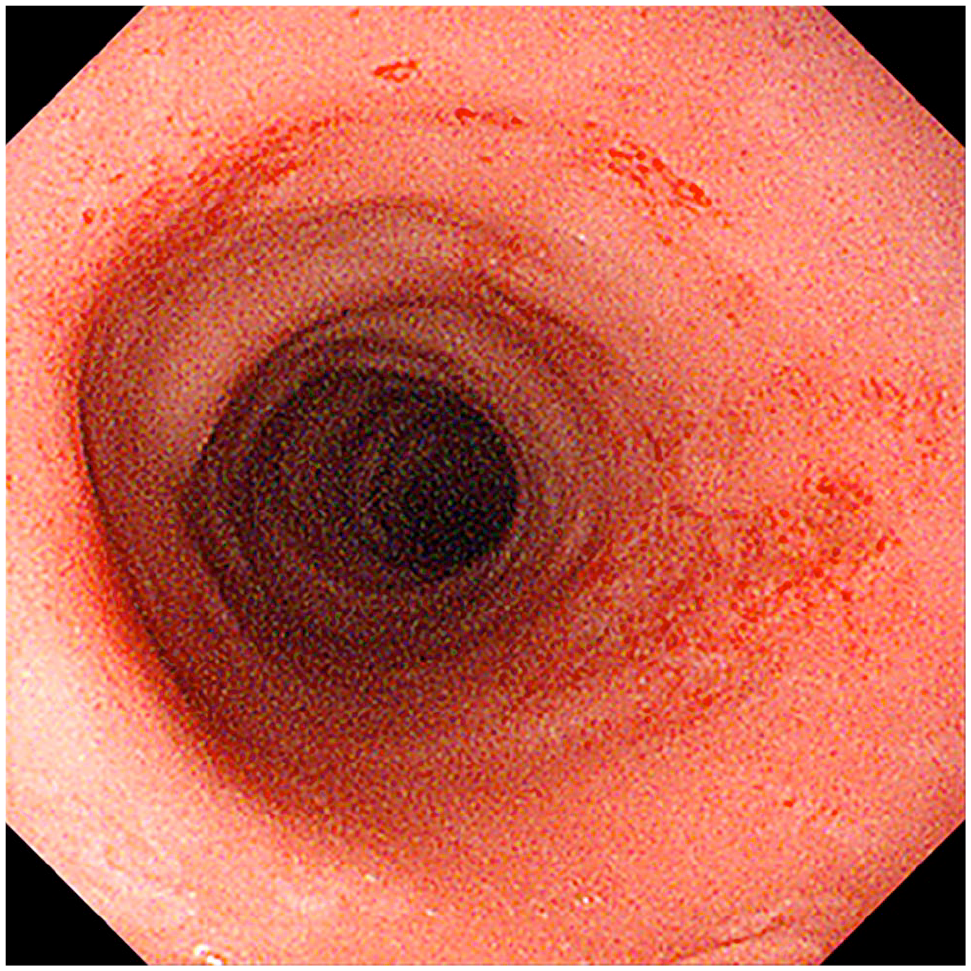
Endoscopic image of the colon. Irregular colonic mucosa with multifocal erosion and haemorrhage
Impression smears of Wright–Giemsa-stained colonic mucosa specimens revealed numerous unicellular round-to-oval organisms and macrophages (Figure 2). The organisms were approximately 10 µm in diameter, featured internal endospores and had unstained cell walls. Several organisms were observed within phagocytic macrophages.
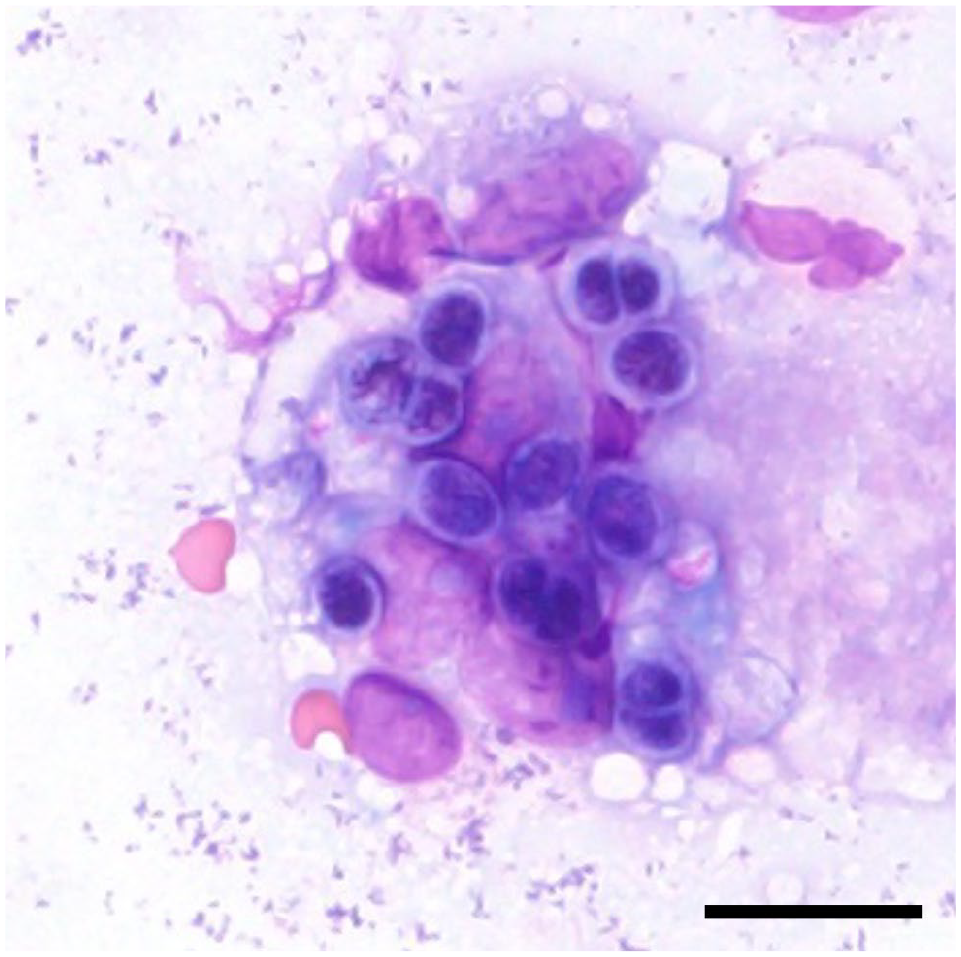
Impression smear of the colonic mucosa. Unicellular round-to-oval organisms and macrophages. The organisms are approximately 10 µm in diameter, feature internal endospores and have an unstained cell wall. Some organisms are phagocytosed by macrophages. Wright–Giemsa staining. Bar = 20 μm
For histopathological analyses, biopsy samples were fixed in 10% neutral-buffered formalin and embedded in paraffin wax. Formalin-fixed, paraffin-embedded (FFPE) tissues were sectioned at a thickness of 3 μm and stained with haematoxylin and eosin. Serial sections were subjected to periodic acid-Schiff (PAS), Grocott’s methenamine silver (GMS) and immunohistochemical staining. For macrophage identification, immunohistochemical staining was performed using mouse anti-CD204 antibodies (1:50, clone DCS-6; TransGenic). After reacting with the primary antibody, the sections were incubated with Envision polymer (Dako), visualised with diaminobenzidine tetrahydrochloride and counterstained with PAS. Histologically, granulomatous inflammation with numerous organisms was observed in the lamina propria of the colonic mucosa (Figure 3a). The organisms had cell walls and often exhibited random or radial internal segmentation (Figure 3b). Each segment contained wedge-shaped endospores. Some organisms lacked internal content. The cell walls and internal segments were positive for PAS and GMS (Figure 3c). Double staining with immunohistochemistry and PAS revealed the infiltration of CD204-positive macrophages around the organisms (Figure 3d). Some organisms were observed in the cytoplasm of the CD204-positive macrophages. Mild infiltration of lymphocytes, plasma cells and neutrophils was observed within the lesions. A small number of organisms were observed in the mucosal epithelium and on the surface of the mucosa. No significant lesions were observed in the mucosa of the stomach, duodenum or ileum.

Histological images of the colon mucosa. (a) Granulomatous inflammation with numerous organisms in the lamina propria. H&E. Bar = 100 μm. (b) The organisms have a cell wall and often exhibit random or radial internal segmentation. Each segment contains wedge-shaped endospores. Some organisms lack internal contents. H&E. Bar = 20 μm. (c) The cell walls and internal segmentation were subjected to Grocott’s methenamine silver staining. Bar = 20 μm. (d) CD204-positive cells surrounding organisms. Some organisms are observed in the cytoplasm of CD204-positive cells. Double staining with immunohistochemistry and periodic acid–Schiff. Bar = 20 μm. H&E = haematoxylin and eosin
For electron microscopy, FFPE tissue from the colonic mucosa was deparaffinised, treated with phosphate-buffered saline (pH 7.4) and then refixed in 1% osmium tetroxide. The samples were dehydrated and embedded in epoxy resin. Ultrathin sections were stained with uranyl acetate and lead citrate and examined using a transmission electron microscope (JEM-1400Plus; JEOL). Ultrastructural examination revealed dense bodies, starch granules and the absence of chloroplasts in the cytoplasm of the organisms (Figure 4a). The organism and internal endospores had thick, double-layered cell walls (Figure 4b).
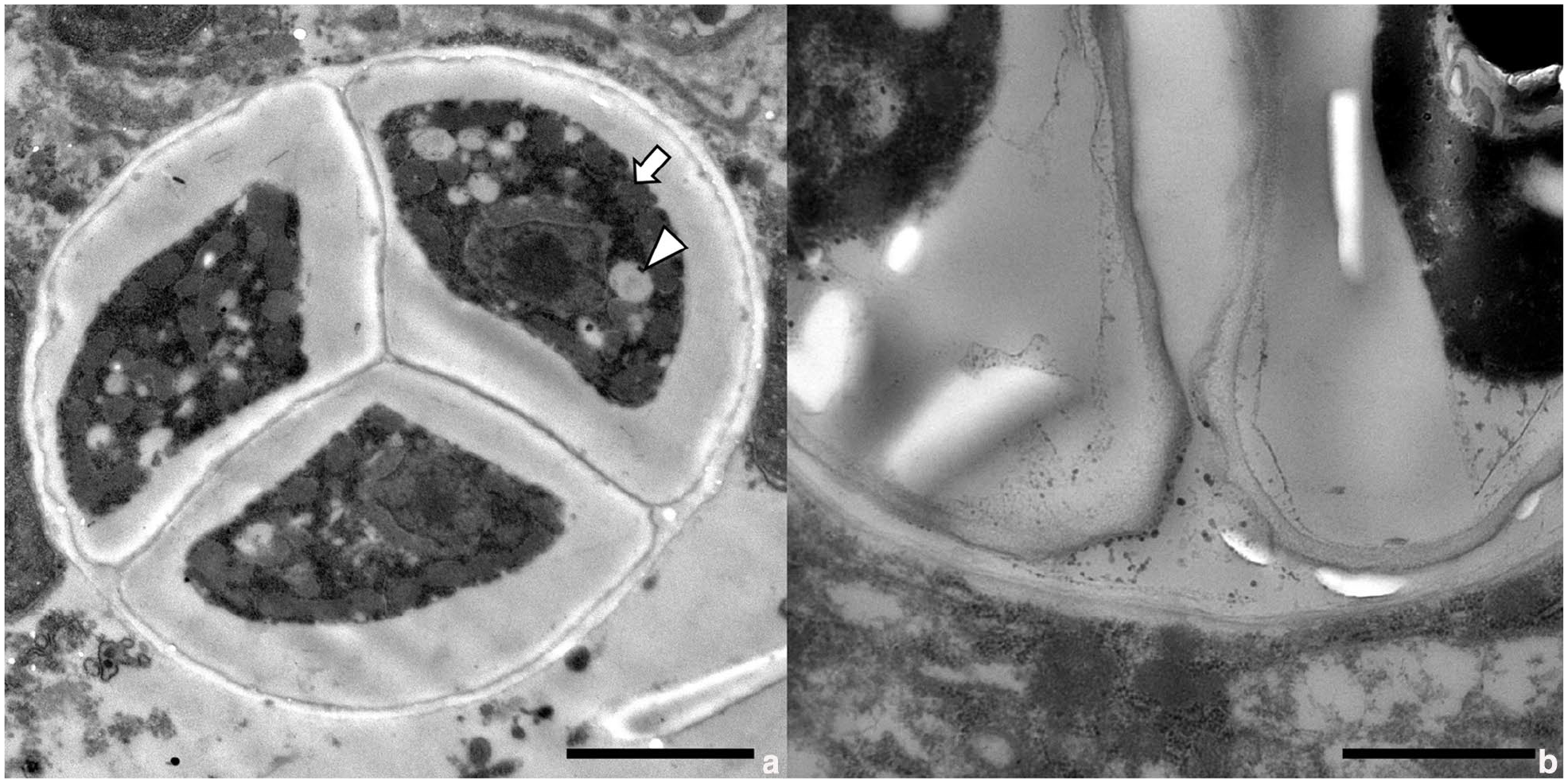
Ultrastructural images of the organism. Transmission electron microscopy: (a) dense bodies (arrow) and starch granules (arrowhead) and the absence of chloroplasts in the cytoplasm of the organisms (bar = 2 μm); and (b) the organism and the internal endospores have double-layered thick cell walls (bar = 1 μm)
Based on cytological, histological and ultrastructural findings, the organism was suspected to belong to the Prototheca species. PCR was performed to detect Prototheca species genes. Total DNA was extracted from FFPE tissues of the colonic mucosa using a QIAamp DNA FFPE Tissue Kit (Qiagen). PCR was performed using a PCR master mix (KOD One PCR Master Mix-Blue; TOYOBO) and the following primer pairs: primer pair 1, targeting the Prototheca species 23S rRNA gene; primer pair 2, targeting the P bovis 18S rRNA gene; and primer pair 3, targeting the Prototheca wickerhamii 23S rRNA gene (Table 1). 6 Distilled water, instead of template DNA, was used as the negative control. PCR-amplified products were electrophoresed on 1.5% agarose gels and the bands were visualised by UV exposure. The amplified products were purified using the High Pure PCR Product Purification Kit (Roche) and sequenced using the Sanger method (FAS-MAC). Sequence data were queried using (National Center for Biotechnology Information) BLAST (Basic Local Alignment Search Tool; https://blast.ncbi.nlm.nih.gov/Blast.cgi). Gel electrophoresis of the PCR-amplified products revealed bands specific for primer pairs 1 and 2, whereas no bands were observed for primer pair 3 (Figure 5). The sequences of the PCR products from primer pairs 1 and 2 showed >99% homology with P bovis reference sequences (GenBank accession numbers: NC_045059.1 and NC_045058.1, respectively).
The PCR primers used in the present study

bp = base pairs.

Gel electrophoresis of the PCR-amplified products. PCR-positive bands for Prototheca species 23S rRNA gene and Prototheca bovis 18S rRNA gene. bp = base pairs; N = negative control; M = 50 bp ladder
Based on these findings, the cat was diagnosed with granulomatous colitis associated with P bovis infection. After conducting the endoscopic biopsy, the cat was treated with probiotics (Slab 51 [SivoMixx]; Ormendes SA, Streptococcus faecalis 129 BIO 3B-R [Biofermin R]; Biofermin), a high-fibre diet (Gastrointestinal Biome; Hill’s Prescription Diet) and antifungal therapy using itraconazole (4.0 mg/kg PO q24h for 8 weeks); however, diarrhoea persisted throughout the 223-day observation period.
Discussion
Prototheca species are classified into 18 species, and infections of some species have been reported in humans and animals: P wickerhamii, P bovis, Prototheca ciferrii, Prototheca cutis, Prototheca miyajii and Prototheca blackhead.1 –3,8 P wickerhamii is the most common species in human and feline protothecosis, and mostly causes cutaneous lesions.1 –3,5,7 P wickerhamii has also been associated with cutaneous protothecosis in dogs.1,9 In contrast, P bovis is associated with bovine mastitis.1,2,4,6 P bovis is the most common species of canine protothecosis and often causes systemic infection.1,2,5,8,10 Previous reports have shown that most cases of canine systemic protothecosis initially present with diarrhoea due to colonic infection.1 –3,5,10 In cats, only two cases of P bovis infection have been reported.11,12 In one of these cases, the cat had neurological signs and P bovis was identified in the cerebrospinal fluid. 11 In the present case, the cat presented with chronic large bowel diarrhoea and was diagnosed with granulomatous colitis associated with P bovis infection. This report suggests that the manifestations of protothecosis in cats may differ among Prototheca species: P wickerhamii causes cutaneous lesions, whereas P bovis can cause lesions in various organs, including the intestine.
Intestinal protothecosis is rare, with only a few cases reported in humans and dogs, and a variety of gross lesions described.1,13 –18 In the present case, the shortened colon and colonic stricture were observed. In addition, histological examination revealed the severe infiltration of macrophages and mild infiltration of lymphocytes, plasma cells and neutrophils, which has also been described in intestinal protothecosis in humans and dogs.14,17,18 In cats, short bowel syndrome and colonic stricture have been suggested to result from chronic inflammation; 19 therefore, chronic inflammation associated with P bovis infection may have contributed to the lesion observed in this case.
The diagnosis of intestinal protothecosis in humans and dogs is based on microscopic examination, culture and PCR analysis.5,10,13,15,17,18 In the present case, numerous organisms were observed by microscopic examination, and the P bovis 18S rRNA gene was detected by PCR analysis using endoscopic samples of colonic mucosa. This case suggests that endoscopic biopsy of the intestinal mucosa is useful for diagnosing intestinal protothecosis in cats.
Previous reports have shown that faecal examination did not reveal the presence of Prototheca species in cases of canine intestinal protothecosis.14 –16 Similarly, no Prototheca organisms were observed in the faecal smears in the present case. Histologically, these organisms were observed in the lamina propria of the colonic mucosa, but rarely in the mucosal epithelium or the surface of the mucosa. This histological finding suggests that the organisms were rarely shed into the faeces, which may have contributed to the difficulty in detecting Prototheca species by faecal examination. In contrast, impression smears allowed detection of the organisms within the mucosa, including the lamina propria.
Treatment of protothecosis in humans and animals involves surgical resection and antifungal therapies such as amphotericin B, ketoconazole and itraconazole.2,3,5,7,8,10,13,17,18 However, previous reports have shown that most cases of canine intestinal protothecosis are resistant to antifungal therapies.8,14 Although clinical evidence is limited, some reports of canine intestinal protothecosis suggest that retention enemas with antifungal agents may be effective.8,20 In the present case, the cat was treated with itraconazole, but diarrhoea persisted. Further studies involving additional cases, alternative antifungal agents and approaches such as retention enemas are needed to evaluate the efficacy of antifungal treatment in feline intestinal protothecosis.
Conclusions
This is the first report of granulomatous colitis associated with P bovis infection in a young cat with chronic large-bowel diarrhoea. Therefore, intestinal protothecosis should be included in the differential diagnosis of cats with treatment-resistant chronic diarrhoea.
Footnotes
Acknowledgements
The authors thank Dr Tomoko Nakano and Dr Mika Fukuda of Fujimino Animal Hospital for their valuable contribution in providing part of the clinical data used in this case report. The authors thank Dr Kimura for his excellent technical assistance with the electron microscopy.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved. For any animal or humans individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
