Abstract
Case summary
A 1-year-old intact female domestic shorthair cat presented for evaluation of worsening respiratory distress and tachypnoea. The cat had been diagnosed with spontaneous pneumothorax 4 days earlier by the primary clinician and treated with therapeutic thoracocentesis. On physical examination, the patient exhibited decreased lung sounds, tachypnoea and increased breathing effort. Recurrent spontaneous pneumothorax was confirmed via thoracic radiography, and therapeutic thoracocentesis was repeated. Lung ultrasound performed after thoracocentesis revealed lung consolidation, pulmonary nodules and multiple cyst-like lesions with irregularly thickened echogenic walls and anechoic centres. Repeat thoracic radiography showed a diffuse moderate bronchointerstitial pattern with multifocal soft tissue nodules. A thorough faecal examination revealed ova resembling those of Paragonimus species, and subsequent molecular analysis confirmed Paragonimus westermani. The cat was initially treated with fenbendazole alone, which resulted in limited improvement and recurrent spontaneous pneumothorax. A second course of fenbendazole combined with praziquantel led to clinical improvement. The cat remained free of clinical signs and was doing well, with no identifiable lung nodules on thoracic radiography 2 years after diagnosis.
Relevance and novel information
This report describes a novel cyst-like lung ultrasound finding characterised by an irregularly thickened echogenic wall and anechoic centre, associated with P westermani infection in a cat presenting with recurrent spontaneous pneumothorax. The report also highlights a successful treatment approach resulting in long-term resolution. The identification of this novel lung ultrasound finding can facilitate early diagnosis and treatment of Paragonimus species infection in cats, especially for those presenting with respiratory distress and pneumothorax.
Introduction
Paragonimiasis (paragonimosis) is a zoonotic, foodborne disease affecting humans and various animals, including dogs and cats.1 –4 Infection typically occurs via ingestion of uncooked or undercooked second intermediate hosts, mainly freshwater crustaceans, such as crabs or crayfish.1,4,5 Once ingested, Paragonimus species metacercariae migrate through peritoneal and pleural cavities to the lungs, where they mature and lay eggs.3,4,6 Patients presenting with lung lesions exhibit clinical signs ranging from mild coughing to life-threatening recurrent spontaneous pneumothorax.1,4,6,7
In veterinary medicine, paragonimiasis is most commonly confirmed by detecting eggs in faecal examinations.4,8 Serological testing can also be helpful when available; alternatively, if the animal is stable enough for anaesthesia and invasive procedures, direct identification of worms in lung histopathological or bronchoalveolar lavage specimens provides definitive evidence.1,4,8 Thoracic radiography is commonly used to monitor pulmonary lesions and evaluate treatment response. 5 However, radiographic findings often resemble other pulmonary diseases, making a definitive diagnosis challenging.1,4,7 For instance, when nodular, cavitary or mass lesions appear on radiography, differential diagnoses include infectious or immune-mediated granulomatous diseases (eg, fungal infections, tuberculosis or other granuloma-forming aetiologies) and malignancies. In addition, consolidation may obscure lesions, further broadening differential diagnoses to include pneumonia, pulmonary oedema or pulmonary haemorrhage. Lung ultrasound, extensively used in human medicine to diagnose lung parenchymal and pleural abnormalities, has gained recognition as a valuable diagnostic tool in veterinary medicine. 9 Despite its potential, reports on the ultrasonographic features of various lung diseases in veterinary medicine remain limited. This report highlights novel lung ultrasound findings in a case of paragonimiasis and documents the therapeutic outcome of a cat with recurrent spontaneous pneumothorax caused by Paragonimus westermani.
Case description
A 1-year-old intact female domestic shorthair cat, weighing 2.64 kg, was referred to the authors’ clinical service. The cat had been rescued from a mountain area in east Asia and adopted 1 month previously, since when progressively worsening coughing had been observed. The cat had been kept exclusively indoors since adoption and had been dewormed with topical selamectin and sarolaner (Revolution Plus; Zoetis) and emodepside and praziquantel (Profender; Bayer). Four days before presentation, the cat developed haemoptysis after playing, followed by acute tachypnoea and respiratory distress. Thoracic radiography performed by the primary clinician revealed a pneumothorax. Therapeutic thoracocentesis removed 80 ml of air from the pleural space, stabilising the cat. Following discharge, the owner noted mild tachypnoea and coughing over subsequent days.
The cat presented to the referral clinic with worsening respiratory distress and tachypnoea. Thoracic auscultation revealed decreased lung sounds bilaterally. Oxygen supplementation, sedation and intravenous catheter placement were provided for stabilisation and stress reduction. Recurrent pneumothorax was confirmed by thoracic radiography (Figure 1). Point-of-care lung ultrasound was initially performed in lateral recumbency to rapidly identify areas lacking lung sliding and locate the lung point, facilitating thoracocentesis. After hair clipping and aseptic preparation, a 23 G butterfly needle with a three-way stopcock and 20 ml syringe was inserted cranial to the rib into the thoracic cavity, slowly removing 75 ml of air. After further stabilisation, comprehensive lung ultrasound scanning covered the maximal explorable area. Upon identifying the curtain sign at the costophrenic recess, scanning began from the caudal thorax. The probe was positioned transversely within intercostal spaces, tilted for optimal visualisation of the pleural line and subpleural space, and gradually advanced cranially to systematically assess each successive intercostal space in each hemithorax. Lung ultrasound revealed partial lung consolidation with irregular margins, air bronchograms, comet-tail artefacts, peripheral pulmonary nodules and multiple cyst-like lesions with irregularly thickened echogenic walls and anechoic centres (Figure 2). The size of the cyst-like lesions was in the range of 3.5 × 3.5 mm to 6 × 8.5 mm. Thoracic radiography repeated after thoracocentesis revealed a diffuse bronchointerstitial pattern with multifocal soft tissue nodules approximately 8 mm in size (Figure 3).

Initial right lateral thoracic radiography revealed a large-volume pneumothorax, predominantly affecting the caudal lung fields. A moderate bronchointerstitial pattern was observed in lung regions not significantly affected by the pneumothorax

Lung ultrasound performed after thoracocentesis revealed multiple cyst-like lesions with irregularly thickened echogenic walls and anechoic centres. These lesions varied in size, had irregular, round margins and were partially surrounded by consolidated lung tissue
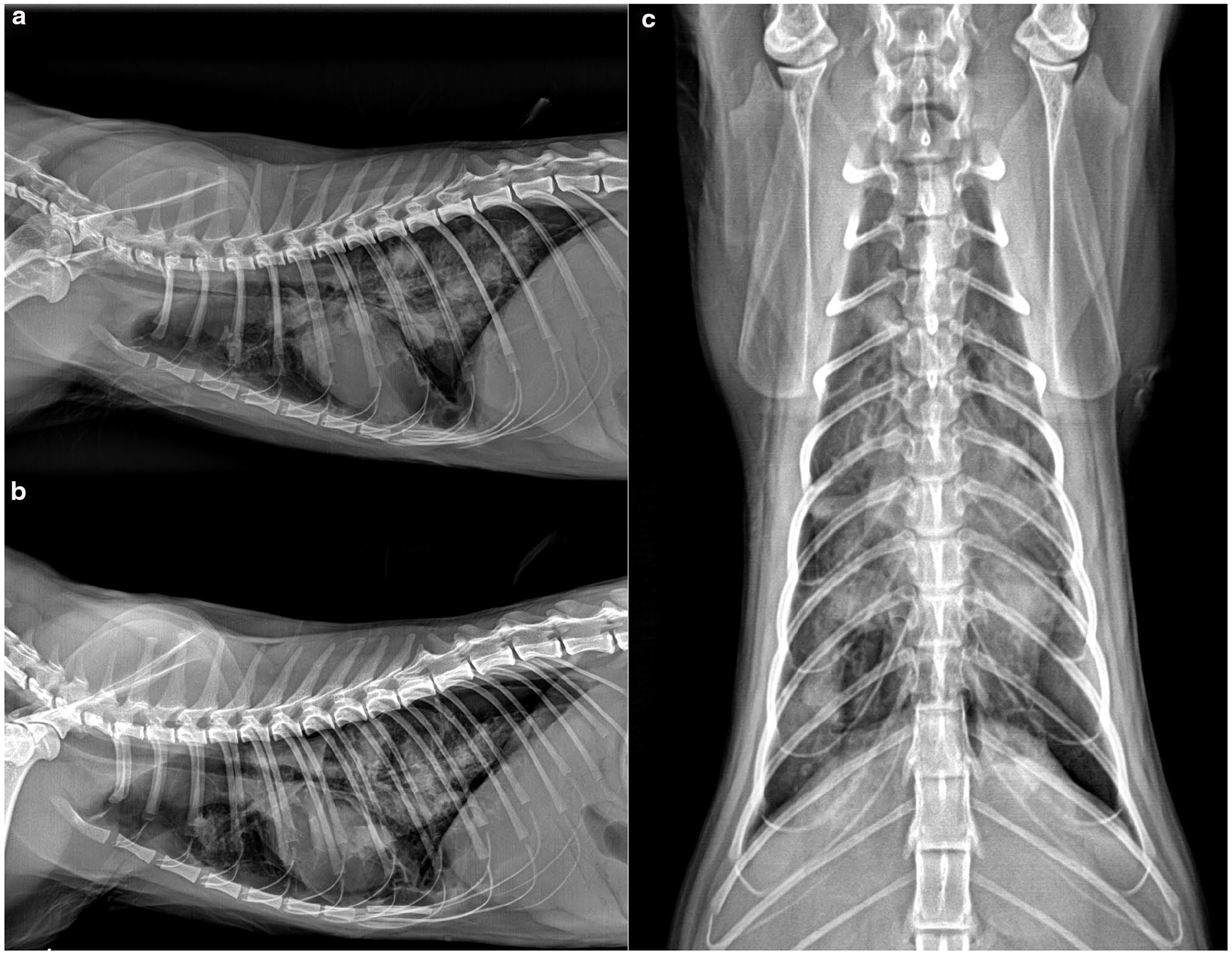
Post-thoracocentesis thoracic radiographs, including (a) right lateral, (b) left lateral and (c) ventrodorsal views, revealed a small volume of pneumothorax. Diffuse, moderate bronchointerstitial patterns were observed, along with multiple nodular lesions and areas of patchy interstitial-to-alveolar infiltration
A minimum database was collected. Haematology and serum biochemistry were unremarkable except for increased globulin (5.1 g/dl) and a low albumin: globulin ratio (0.63) (see File 1 in the supplementary material). Serological tests for feline heartworm antibody, Toxoplasma gondii IgG antibody, cryptococcal antigen, feline immunodeficiency virus antibody and feline leukaemia virus antigen were negative.
Although the owner reported previous deworming, given the young age of the cat, the presence of pneumothorax, previous mountainous living environment and uncommon lung ultrasound findings, a thorough faecal examination was conducted to investigate a parasitic aetiology. Faecal smear, flotation and Baermann tests were negative. However, faecal sedimentation testing discovered ova (size 70 μm) suspected of being Paragonimus kellicotti but smaller (Figure 4). The faecal samples underwent molecular analysis via PCR, confirming P westermani (see File 2 in the supplementary material). 10

Paragonimus westermani ova identified in the cat’s faecal sample using the sedimentation technique. The ova measured approximately 70 μm in length, displayed a yellow-brown coloration and had an ovoid to elongated shape. They featured a thick shell with distinct asymmetric thickening at one pole (arrows)
On the day of diagnosis (day 0), fenbendazole (Panacur; Intervet) was prescribed at 50 mg/kg PO q12h for 2 days, followed by 28 mg/kg PO q12h for 19 days. Despite treatment, the cat continued coughing daily, and pneumothorax recurred 10 days later. A third thoracocentesis was performed, removing 85 ml of air. A second faecal sedimentation test on day 22 remained positive for P westermani ova. Praziquantel (Droncit; Bayer) was added (25 mg/kg PO q8h for 3 days), followed by a second 21-day course of fenbendazole (Axilur vet; Intervet; 28 mg/kg PO q12h).
An elevation in alanine aminotransferase (ALT) of uncertain cause was observed on blood examination after the first course of fenbendazole (ALT increased from 48 to 1769 U/l; reference interval [RI] 12–130). Since drug- or adjuvant-related hepatic toxicity was among the potential differential diagnoses for the elevated liver enzyme levels, a different fenbendazole formulation was selected for the second course. The cat’s ALT subsequently returned to the normal RI in follow-up blood examinations.
The cat had intermittent coughing but no further respiratory distress after thoracocentesis on day 10. By day 77, thoracic radiography showed resolution of the soft tissue nodules, and faecal sedimentation testing was negative for P westermani ova. The cat’s breathing normalised and its coughing gradually resolved over the following months. By day 427, the cat remained free of clinical signs, without significant nodular or infiltrative lesions on radiography (Figure 5). At the time of writing, 2 years after diagnosis, the cat remains clinically well.

Follow-up thoracic radiographs taken on day 427, including (a) right lateral, (b) left lateral and (c) ventrodorsal views, show resolution of radiographic changes associated with Paragonimus westermani infection. Previously noted nodular lesions were no longer detectable
Discussion
Specific lung ultrasound findings of paragonimiasis have not been reported in the small animal literature. Ultrasonographic features in this case corresponded to the known pathogenesis of paragonimiasis. Paragonimus species migrates from the intestines, penetrates the pleura and feeds on the host’s lung (Figure 6), creating cyst-like lesions surrounded by granulomatous tissue, 4 consistent with the observed lung ultrasound findings.

The typical life cycle of Paragonimus species begins with the excretion of eggs in the faeces or sputum of a definitive host. These eggs hatch in freshwater, releasing miracidia that infect various species of Melania and Tricula snails, which serve as the first intermediate hosts. Within the snail, asexual reproduction produces cercariae, which are then released and infect aquatic arthropods, primarily freshwater crabs or crayfish (crustacea), serving as second intermediate hosts. Within these hosts, the cercariae develop into metacercariae, which can infect definitive hosts when they consume raw or undercooked crustaceans or other second intermediate hosts. After ingestion, the metacercariae migrate to the lungs of the definitive host, where they mature and begin producing eggs. Paratenic hosts, such as wild boar or deer, can also harbour metacercariae after consuming infected second intermediate hosts. These metacercariae remain infective to definitive hosts, further contributing to the transmission cycle
Radiographic findings evolve with disease progression and treatment.2,3,7,11,12 Thoracic radiographs typically show structural interstitial patterns, soft tissue opacities, lung nodules or cysts, sometimes with pleural effusion and pneumothorax.3,11,13,14 Nodules develop when parasites induce granuloma formation. 4 Fluid-filled lesions may appear as focal consolidations or nodules; if connected to bronchi, they present as bubble-like, thin-walled rings or cysts. 3 Pneumothorax can result from lesion rupture and air leakage.3,11,14,15 During recovery, fibrotic or calcified densities may replace nodules.11,14 Nevertheless, these radiographic changes are non-specific and overlap with other pulmonary diseases, including pneumonia, oedema, haemorrhage, granulomatous diseases and malignancies. 16 Lung nodules, cystic lesions and pleural effusion cannot reliably distinguish paragonimiasis from malignancies or other granulomatous infections. Though pneumothorax is common in paragonimiasis, radiographs taken during these episodes may further compromise a cat’s condition. Lung ultrasound is increasingly utilised as a first-line imaging modality for cats with respiratory distress. 17 Identifying cystic lesions with anechoic centres and thickened echogenic walls, along with relevant history, can raise suspicion of paragonimiasis during initial assessment. These ultrasonographic findings possibly reflect underlying pathogenesis, as previous studies described worm cysts with eccentric wall thickening producing an eclipse-like appearance due to adherent live worms, accompanied by pericystic consolidation, cavitary lesions, thick- or thin-walled pneumatocysts and fluid-filled granulomas.5,7
Cats with paragonimiasis typically present with mild coughing but may develop respiratory distress due to pneumothorax, a debilitating yet poorly understood complication. The precise mechanism of pneumothorax in paragonimiasis remains unclear, 14 but it is suspected to result from lung penetration by migrating larvae or rupture of pleural-adjacent cysts, leading to air seepage.3,6,7,12,18,19
Diagnosis of paragonimiasis typically depends on detecting parasite ova. Identifying worms in the lung confirms infection but is often limited by the invasiveness of sampling. Serological tests, including Paragonimus species antibody or antigen assays, assist in diagnosis but may yield false positives due to antigen cross-reactivity.4,20,21 Although serology might be an initial screening tool, definitive diagnosis requires ova detection. 21 In early infection, ova are undetectable, but detection is achievable in the later phase. 4 Because of high specific gravity, Paragonimus species ova can be missed by faecal flotation tests; sedimentation techniques are more sensitive, particularly for low ova counts. 18 Haematological and biochemical evaluations often provide little diagnostic insight. In this case, bloodwork was unremarkable, consistent with studies showing only transient neutrophilic leukocytosis and eosinophilia, which declined within 2 weeks after inoculation.8,18 By the time clinical signs develop, routine bloodwork is typically normal.
Reported treatments for paragonimosis in dogs and cats include albendazole, fenbendazole and praziquantel.4,5,8,16 In the present case, fenbendazole was initially selected because of its documented success in treating paragonimiasis;4,16 however, regional differences in response among Paragonimus species and individual treatment variability have been noted. 4 In the present case, the first fenbendazole course failed to produce clinical improvement, and faecal examination remained positive for P westermani. Interestingly, a previous case report on P kellicotti infection in a cat described clinical improvement after the first fenbendazole treatment; however, the faecal examination remained positive, and the infection was only considered successfully eliminated after a second course of fenbendazole. 5 Whether clinical improvement in the present case resulted from praziquantel, a second fenbendazole course or their combined effects remains unclear. Clinicians should be aware of potential individual variations in treatment response, as a single course of fenbendazole may not be sufficient to eliminate Paragonimus species infection in some cats.
Paragonimus species infection should be suspected in endemic regions in patients presenting with compatible respiratory signs and a relevant history, such as consuming raw freshwater crabs or crayfish. Given the wide endemic range of Paragonimus species – which mainly includes, but is not limited to, North America, West Africa and South Asia – it is crucial to consider this infection in differential diagnoses.1,4,7,12,19 In addition, human cases of paragonimiasis have been reported worldwide, likely due to increased international travel and the growing availability of diverse foods online. Reports of canine paragonimiasis after ingestion of raw or undercooked meat from paratenic hosts, such as wild boar or deer, have also been increasing, with a higher prevalence observed compared with previous studies.20 –22 As raw food diets and wild game dishes become more popular in companion animals, Paragonimus species also poses a potential health threat that should not be overlooked, even in cats without direct outdoor access or a history of crustacean ingestion. Veterinarians should recognise the potential for a rise in paragonimiasis cases in the future.
Conclusions
This is the first report of the specific ultrasonographic features of paragonimiasis on lung ultrasound in veterinary medicine. It underscores the importance of raising awareness about Paragonimus species infection in endemic regions and highlights the potential application of lung ultrasound for the early diagnosis of paragonimiasis in cats.
Supplemental Material
File 1
Hematology and biochemistry results on the first day of presentation.
Supplemental Material
File 2
Detailed information regarding molecular analysis.
Footnotes
Acknowledgements
The authors express their sincere gratitude to the cat’s owners for rescuing the cat and providing unwavering dedication and attentive care, which greatly contributed to the successful management of the case.
Supplementary material
The following files are available as supplementary material:
File 1: Hematology and biochemistry results on the first day of presentation.
File 2: Detailed information regarding molecular analysis.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The manuscript was funded by National Taiwan University (NTU-CDP-114L7748).
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
