Abstract
Case summaryA 4-year-old male castrated domestic shorthair cat presented with a 2 cm painful cutaneous mass on its right lateral abdominal wall. The cat inflicted self-trauma to the lesion site causing secondary ulceration and mild haemorrhage. Fine-needle aspiration or incisional biopsy was advised, along with diagnostic imaging; however, excisional biopsy was preferred by the owners. The mass was surgically removed with 1 cm lateral margins and a deep fascial plane. The histopathological features were most consistent with a diagnosis of a glomus tumour; the diagnosis was supported by subsequent immunohistochemistry. The tumour was completely removed and there were no signs of recurrence at the 6-month follow-up. In this case, surgery is expected to be curative.
Relevance and novel information To the authors’ knowledge, this is the first report of a glomus tumour of the body wall in a cat and its association with pain and self-trauma. This report aims to add more data to the diagnosis and presentation of glomus tumours in animals.
Introduction
Glomus tumours (GTs) are rare, usually benign, tumours arising from the glomus cells and are typically found in specialised arteriovenous shunts or anastomoses mostly within the dermis and subcutis. 1 Reports of GTs in the veterinary literature are scarce, described in primates, 2 cows,3,4 horses,5 –7 a hyena, 8 dogs9 –13 and two cats.14,15 In the human literature, these tumours are uncommon, accounting for approximately 1–5% of soft tissue tumours of the hand (main location), 1 with other anatomical locations reported.16 –18 The diagnosis of GT in humans is challenging because of the often-small size of the tumour, and it is achieved with a thorough examination and the aid of multiple diagnostic imaging modalities (radiography, focused ultrasound, CT or MRI). 19 In people, pain is usually the main symptom aiding the diagnosis through multiple pain-focused tests. 20
This report describes the presentation and an unusual location of a GT in a cat.
Case description
A 4-year-old male castrated domestic shorthair cat presented with an acute and painful swelling on the right lateral abdominal wall. Days before the mass appeared, discomfort at gentle palpation of the area was reported. Subsequently, lethargy and self-trauma were noticed, prompting veterinary consultation.
At presentation, the physical examination was unremarkable, except for a solitary and ulcerated mass on the right lateral abdominal wall measuring 1.5 × 2 cm (Figure 1). The mass was mobile and seemed to arise from the cutaneous or subcutaneous layer. At palpation, the patient was very painful; therefore, further examination was preferred under sedation or general anaesthesia.

Glomus tumour on the right lateral abdominal wall. (a) The haemorrhage secondary to self-inflicted trauma and (b) the well-circumscribed nodular tumour
Although fine-needle aspiration and/or incisional biopsy were discussed to further characterise the mass, along with diagnostic imaging modalities (focused ultrasound or CT), the owners preferred excisional biopsy and histopathology. They were made aware of the risk of incomplete excision of the mass, recurrence and possible unknown metastatic disease.
The cat was pre-medicated with methadone (0.3 mg/kg IM) and medetomidine (0.01 mg/kg IM), and an intravenous catheter was placed on the left cephalic vein. General anaesthesia was induced with a slow dose of propofol (2 mg/kg IV) and maintained with isoflurane in oxygen. The patient was surgically prepared and positioned in left lateral recumbency.
The mass was surgically resected via a pre-marked circular incision with measured lateral margins measuring 1 cm and one deep fascial plane. Primary closure was easily achieved.
The cat received a postoperative subcutaneous (SC) injection of meloxicam (0.2 mg/kg), followed by a 5-day course of meloxicam (0.05 mg/kg PO q24h). The mass was fixed in 10% formalin and sent for histopathology.
Histopathological examination identified the presence of an ulcerated, circumscribed dermal to subcutaneous tissue mass that was composed of neoplastic round or occasionally plump spindle-shaped cells, with an impression of packeting. Scattered mitotic figures were present within the neoplastic cells (Figure 2a). Numerous dilated vessels were present within and at the margins of the mass. Periodic acid–Schiff (PAS) staining (Figure 2b) revealed a basement membrane around individual and small clusters of neoplastic cells. These neoplastic cells stained positively for α smooth muscle actin (Figure 3) and vimentin and failed to stain for desmin; this staining pattern supports the diagnosis of a GT, since glomus cells are modified smooth muscle cells and contain intermediate filaments and therefore stain for vimentin, and myofilaments that express α smooth muscle actin and other muscle actin isoforms. 21 The histopathological features combined with the immunohistochemical staining pattern were therefore considered to be consistent with a diagnosis of a GT, which was completely excised.
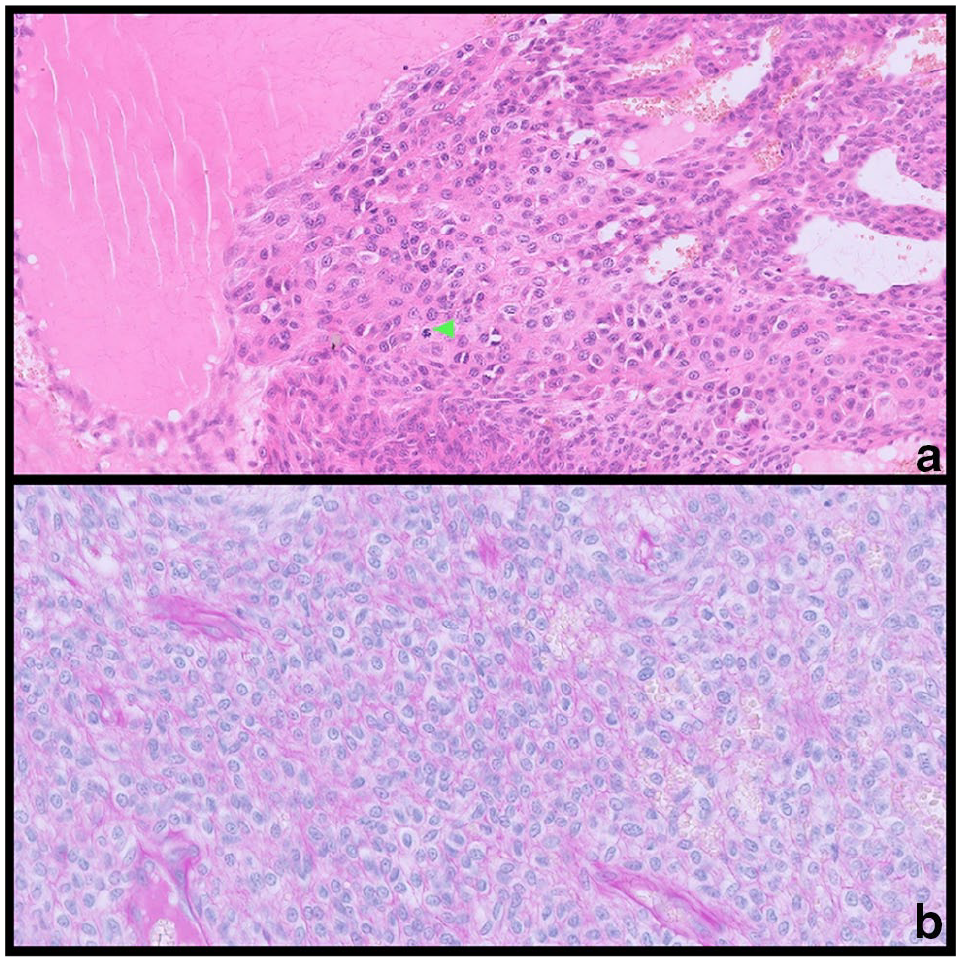
Glomus tumour of the abdominal wall of a cat. (a) Closely packed round and (bottom of photomicrograph) plump spindle-shaped cells with eosinophilic cytoplasm and occasional mitotic figures (green arrowhead) with numerous vascular structures (haematoxylin and eosin, × 40). (b) Individual tumour cells are surrounded by a PAS-positive basement membrane (periodic acid–Schiff, × 40)

Glomus tumour. The neoplastic cells show strong cytoplasmic and membrane staining for α smooth muscle actin (α smooth muscle actin, ×40)
At the 6-month telephone follow-up, the cat was fully recovered; its behaviour normalised immediately postoperatively, with no further pain or self-trauma reported. There was no evidence of recurrence.
Discussion
Located in the stratum reticularis of the dermis, the glomus body is a specialised arteriovenous anastomosis, modulated by the sympathetic nervous system, thought to be involved in thermoregulation. 20 It is composed of the Suquet-Hoyer canal, which connects the afferent arteriole and the efferent venule. The canal wall is composed of modified smooth muscle cells known as glomus cells. 20 GTs are tumours arising from glomus cells and are described as either benign or malignant, typically occurring as a solitary lesion. 17
The first description of GTs in the human literature dates back to 1812, when Wood described multiple cases of painful subcutaneous tubercles. 22 In the human literature, GTs are reported mainly in the subungual region of the hand while body wall GTs are extremely rare, with only five cases described in humans, two of which had characteristics of malignancy. 18 Clinical presentation includes point tenderness, severe paroxysmal pain crises and temperature sensitivity. The presence of nerve fibres in GTs, containing immunoreactive substance P (a neuropeptide acting as a pain transmission mediator), might explain the tenderness and paroxysmal pain. 20 Diagnosis of GT in humans is frequently delayed due to the generally small size of the mass, with one paper reporting an average of 10 years between the onset of clinical signs and diagnosis. 23 Investigations include Love’s test, in which pain is elicited by applying blunt point pressure to the lesion; the Hildreth ischaemia test (limited to extremities), in which a tourniquet is applied around the finger to relieve pain; and the cold sensitivity test.17,23 The presence of pain should always raise suspicion of a GT. 23
From both human and veterinary literature, plain radiography is sometimes helpful, as it may reveal osteolytic lesions.5,16 CT may show abnormalities and tissue erosions. 24 The imaging modalities of choice to delineate the tumour in humans are high-resolution MRI1,16,25 or focused Doppler ultrasonography 26 (investigated mainly on digital GTs). Most GTs show high signal intensity on T2-weighted spin-echo MRI images, 25 while on ultrasound, they appear as small, solid, homogeneously hypo- or isoechoic, well-demarcated nodules characterised by hypervascularity. 26 Nevertheless, the abovementioned diagnostic imaging tools may be negative even when a GT is present, 27 making the correlation of investigations with physical examination and symptoms crucial. Confirmation of GT can only be obtained through histopathological examination and immunohistochemistry. 17
As in this case, the only two previously reported feline GTs14,15 are well-demarcated subcutaneous and/or dermal masses, typically with numerous small nerve fibres and vessels associated with the mass, particularly at the margins. The majority of the mass is generally composed of round cells and resembles GT proper in humans, 21 although variable numbers of plump spindle cells may also be present. Each tumour cell or small group of tumour cells are surrounded by a PAS-positive basement membrane. Entrapped vascular structures are common and may be compressed. 21 Differentiation from other tumours composed of round cells is achieved by immunohistochemistry, as GTs retain cytoplasmic expression of vimentin, α smooth muscle actin and pan-muscle specific actin. 21 The latter stain was not available for use in this case.
In humans, GTs are predominantly benign tumours, although malignant cases (which are locally aggressive) have been reported, along with their distant metastatic potential. 28 In 2013, the World Health Organization (WHO) changed the malignancy criteria for GTs in humans. The presence of atypical mitotic figures and marked nuclear atypia satisfies the malignancy requirements, while tumour size (>2 cm) and deep location are now considered indicative of tumours of uncertain malignant potential, which require close monitored. 29
Surgical excision remains the standard therapy for GTs, with clear margins usually indicating the effectiveness of treatment. 24 Because of the paucity of reports, there are no guidelines regarding GT surgical resection and margins. The recurrence rate, extrapolated from human literature, can vary from low to 30%, depending on papers. 16 In a recent human study involving 31 cases, there was no recurrence in the short-term follow-up (5.8 months), in which three cases did not have clean margins. 16
There are only two previous reports of GTs in cats: one on the digit, 14 reflecting the most common location described in the human literature, and one on the head. 15 Their clinical presentation differed from that in this report, with both appearing to be non-painful.
Conclusions
To the authors’ knowledge, this is the first case describing a GT located on the abdominal wall in a cat and its association with pain (a characteristic symptom for human GTs) and secondary self-trauma. Although GTs are rare in domestic animals, they should be considered a differential diagnosis for any cutaneous/subcutaneous masses, particularly when pain is present. Surgical excision is the treatment of choice and usually results in permanent relief of symptoms. Currently, there are no margin guidelines; however, a 1 cm lateral margin and a deep fascial plane resulted in complete tumour removal in this case, with no recurrence observed at the 6-month follow-up. Definitive diagnosis of GT can only be achieved with histopathology and immunohistochemistry, which also help identify any potential for malignancy in accordance with WHO criteria from the human literature. Further reports of GTs in animals might help in understanding their incidence, biological behaviour, treatment and prognosis in small animals.
Footnotes
Acknowledgements
The authors would like to thank Michał Mól for the support and constructive criticism during the preparation of the manuscript.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
