Abstract
Case summary
A 9-year-old male castrated domestic shorthair cat from northern California was evaluated for a 12-month history of dermal and subcutaneous dermatitis in the inguinal region. Histopathologic examination of a biopsy revealed severe, chronic, multifocal to coalescing pyogranulomatous dermatitis and panniculitis, accompanied by ulceration and central necrosis. Aerobic bacterial culture of lesions yielded mycobacterial growth. Empiric antimicrobial therapy was initiated with doxycycline and marbofloxacin pending culture and susceptibility. Culture of a biopsy followed by rpoB gene sequencing at a mycobacterial reference laboratory yielded Mycobacterium porcinum after 6 weeks. Ten months after initial antimicrobial administration, the lesions resolved.
Relevance and novel information
To date, in cats, M porcinum panniculitis has been reported from Ohio, Massachusetts and British Columbia in North America; two additional cases were reported from southeastern Australia. In humans, M porcinum infections have been reported from several states in the USA, predominantly in the Midwest and coastal south, but not from the west. This report extends the known spatial distribution of M porcinum to the western USA and strengthens its association with panniculitis in cats. It also demonstrates the need for prolonged incubation for diagnosis of some rapidly growing mycobacteria infections using culture.
Keywords
Introduction
Mycobacterium species are aerobic, acid-fast bacteria with a lipid-rich cell wall of complex mycolic acids.1,2 Mycobacterium species can be broadly divided into two groups: Mycobacterium tuberculosis complex (MTBC) organisms, including M tuberculosis, M bovis and M microti; and non-tuberculosis mycobacteria (NTM). The NTM, in turn, consists of slow-growing NTM (including M avium complex), rapidly growing NTM (RGM) and fastidious mycobacteria, such as M lepraemurium.1,3 The RGM are so-named because growth in culture is generally detectable within 7 days. 4 The RGM are responsible for most cutaneous mycobacterioses in cats. Transmission is thought to occur through cutaneous inoculation, such as after fighting injuries or other wounds, with a tropism for adipose tissue.1,5 The RGM species of clinical significance in cats include those that belong to the M fortuitum complex and the M smegmatis complex, which are ubiquitously found in soil or water.6,7 These were the most frequent isolates in a 2021 study from northern California. 8 The most commonly affected sites are the inguinal skin and subcutaneous tissues. Lesions consist of subcutaneous abscesses and non-healing ulcers with fistulous tracts, collectively referred to as mycobacterial panniculitis.5,7 Treatment requires prolonged courses of antimicrobial therapy. 9
M porcinum is an RGM originally isolated from pigs. 10 To the authors’ knowledge, only two reports describe successful treatment of M porcinum infection in cats: one from British Columbia, Canada, and one from Ohio, USA.11,12 Two additional infections were reported from southeastern Australia and one from Massachusetts, USA.13,14 In humans, M porcinum infection has been associated with wound infections, healthcare-associated infections, catheter-related bacteremia and osteomyelitis.4,10,15 –17 While human cases have been reported from several states in the USA, to the authors’ knowledge, none have been reported from the western USA. Here, we describe the clinical characteristics, diagnosis and successful treatment of a cat from northern California, USA, with M porcinum panniculitis.
Case description
A 9-year-old male castrated domestic shorthair indoor/outdoor cat was evaluated for a 12-month history of subcutaneous nodular lesions. When first evaluated by the referring veterinarian, there were multiple, semi-firm subcutaneous nodules that measured 1–2 cm in the inguinal region. The cat was treated with cefovecin (8 mg/kg SC once). The nodules remained unchanged. Five months later, histopathologic examination of a skin biopsy revealed severe, chronic, multifocal to coalescing pyogranulomatous dermatitis and panniculitis, accompanied by ulceration and central necrosis (Figure 1). No microorganisms were detected with hematoxylin and eosin, Fite-Faraco acid-fast stain, Grocott-Gomori’s methenamine silver stain or periodic acid-Schiff stain. Three months later, aerobic bacterial skin culture of a subcutaneous nodule in the inguinal region yielded a Mycobacterium species on a sheep blood agar media after 8 days of incubation. A referral was then made to the University of California-Davis Veterinary Medical Teaching Hospital (VMTH).

Histopathology image showing a pyogranuloma centered around an adipocyte with intralesional filamentous bacteria (arrow) (hematoxylin and eosin stain, × 20 magnification)
On examination at the VMTH, the cat was bright, alert and responsive with normal vital parameters and a body condition score of 9/9. Five skin lesions were noted in the ventral abdominal and inguinal region, each measuring 1–3 cm in diameter. Three lesions were subcutaneous, firm and moveable with an irregular texture, and two were flat, cutaneous, freely moveable plaque-like lesions with multifocal crusting (Figure 2). A complete blood count (CBC) revealed mild leukocytosis (14,090 cells/µl; reference interval [RI] 4500–14,000) characterized by neutrophilia (10,750/µl; RI 2000–9000). A serum biochemistry panel revealed hyperglobulinemia (5.8 g/dl; RI 2.8–5.4). A urinalysis revealed a urine specific gravity of 1.048 with inactive sediment. Abdominal ultrasound examination and thoracic radiographs were unremarkable. Punch biopsies of affected skin and subcutaneous tissue were sent to a mycobacterial reference laboratory (National Jewish Health Advanced Diagnostic Laboratories, Denver, CO, USA) for culture and susceptibility testing using broth microdilution. Antimicrobial therapy with doxycycline (6.6 mg/kg PO q12h) and marbofloxacin (2.6 mg/kg PO q24h) were started, pending culture and susceptibility. On the sixth week of incubation, culture yielded M porcinum as identified using PCR sequencing of the rpoB gene. Susceptibility results are shown in Table 1.

Inguinal region of a cat infected with Mycobacterium porcinum on initial examination at the referring hospital. Multifocal ulcerative cutaneous lesions are present
Results of antimicrobial susceptibility tests for Mycobacterium porcinum determined by broth microdilution

I = intermediate; R = resistant; NI = no Clinical and Laboratory Standards Institute interpretative guidelines for humans and this antibiotic/organism combination; S = susceptible
Based on clinical response, doxycycline and marbofloxacin were continued, and lesions resolved over 9 months (Figure 3). Doxycycline was then discontinued and marbofloxacin continued for 2 months. Serial monitoring of hematology and biochemistry results during treatment revealed resolution of neutrophilia and hyperglobulinemia. Three months after marbofloxacin was discontinued (14 months after starting treatment), the owner noted a new skin lesion on the cat’s ventral abdomen, where previous lesions had been present. Physical examination revealed a 0.5 cm focal area of mild erythema and superficial thickening of the skin. Skin biopsies and cultures were not performed. Marbofloxacin was recommenced (2.5 mg/kg PO q24h); 4 weeks later, the lesion had resolved. Marbofloxacin was continued for an additioanl 4 weeks. An examination at the VMTH 32 months after initial presentation revealed no new lesions, after which time the cat was lost to follow-up.
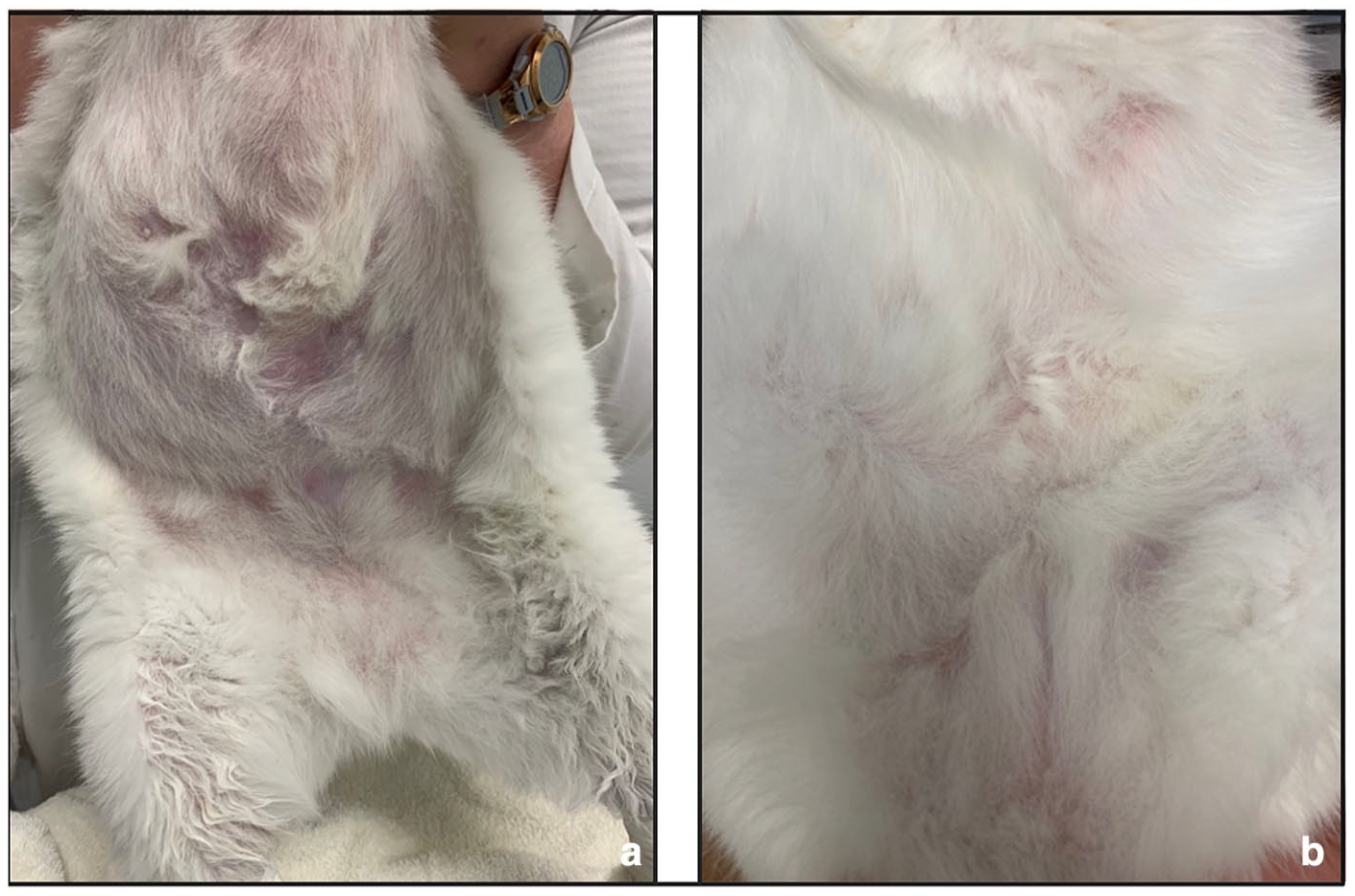
(a) Ventral abdomen and inguinal region after 6 weeks of antimicrobial therapy. (b) Ventral abdomen and inguinal region after 16 weeks of antimicrobial therapy
Discussion
Mycobacterial infections in cats occur sporadically and can be challenging to diagnose and treat.1,9 This is the first report of M porcinum panniculitis in a cat from California, USA. The clinical signs were consistent with previous reports of mycobacterial panniculitis in cats. 18 Initially isolated from submandibular lymphadenitis in pigs, M porcinum belongs to the M fortuitum complex, a group of closely related RGM species, including M fortuitum, M peregrinum, M porcinum and M septicum.1,10,19 In humans, M porcinum infections have been recognized in association with wound infections and healthcare-associated infections in multiple states in the USA,4,10,15 –17 including a 2018 outbreak of 101 vaccine-site infections associated with workplace vaccination of humans in Indiana, Kentucky and Ohio, 16 but not from the western USA.
Outdoor access may be a risk factor for mycobacterial infections in cats. In the UK, only 2% of 339 cats with mycobacterial disease had an indoor-only lifestyle. 18 The cat in this report had both indoor and outdoor access. While a fight or a penetrating injury was not directly observed, the mode of transmission was suspected to be cutaneous inoculation. Obesity may have increased the risk of infection due to the proclivity of mycobacteria for adipose tissue.5,7
Treatment of NTM infections typically requires several months of antimicrobial therapy,18,20 with resolution of clinical signs requiring a median of 7 months of therapy in one study. 20 In the cat described in this report, therapy with doxycycline and marbofloxacin was commenced while awaiting culture and susceptibility. Treatment with pradofloxacin and doxycycline was associated with resolution of M porcinum infection in a cat from Canada. 11 Another cat with M porcinum panniculitis from Ohio was successfully treated with marbofloxacin and doxycycline. 12 Combination therapy consisting of doxycycline and fluoroquinolone has been recommended for RGM panniculitis.13,21 Combination therapy may reduce the chance of selection for resistant strains. In the case reported here, doxycycline and marbofloxacin were chosen based on previously published reports of successful treatment and clinical suspicion of RGM. 8 Although the dose of marbofloxacin was lower than label recommendations, it was selected based on recommendations in the ABCD mycobacteriosis guidelines; 9 given the cat’s obesity, dose per lean body weight was likely sufficient, as supported by the response to treatment. Given predicted resistance to doxycycline based on susceptibility testing, doxycycline could have been discontinued earlier to optimize antimicrobial stewardship; however, the attending clinician initially chose to continue both drugs based on the following: (1) the response to and recommendations for combination therapy; (2) the fact that for mycobacteria, breakpoints for companion animals do not exist and susceptibility test results do not always correlate with clinical response; and (3) susceptibility can differ when drugs for which resistance is predicted are used in combination owing to synergistic effects. Although susceptibility testing indicated that other options for combination therapy existed (ie, clarithromycin or trimethoprim/sulfamethoxazole), these options were associated with added expense and logistical challenges, given the COVID-19 lockdown processes in place at the time, as well as the potential for other adverse effects, including the difficulties of administering trimethoprim/sulfamethoxazole to cats. Considering the susceptibility test results, when all lesions had resolved, doxycycline was discontinued to reduce the need for twice daily therapy and possible adverse effects of doxycycline (such as hepatopathy). Ideally, combination therapy should have continued for the duration of treatment. Based on predicted susceptibility to moxifloxacin, pradofloxacin may have been preferable to marbofloxacin due to its dual-site mechanism of action, which reduces the potential for selection of resistant bacteria; 22 however, for financial reasons, marbofloxacin was chosen. This case illustrates some of the challenges that relate to administering treatment to cats with mycobacterial infections for prolonged periods, and the need for novel approaches to treatment and prevention of these conditions.
The absence of acid-fast bacteria on histopathologic examination is consistent with the low sensitivity of acid-fast staining for RGM infections.1,23 –25 This may be due to the removal of tissue lipid during routine processing, strong decolorization of the acid during the staining process, low organism loads and variable mycolic acid expression in RGM.5,23,26 The slow growth of the isolate in this case was notable; culture in other cases of M porcinum panniculitis in cats yielded growth within several days.11,14 We described the slow growth of another RGM, M thermoresistibile, in a cat with mycobacterial panniculitis. 27 In the case reported here, contamination was unlikely given the consistent clinical presentation, previous isolation of a mycobacteria by the referring veterinarian and treatment response. Given that growth was reported to the referring veterinarian after 8 days, slow growth on the second attempt may have reflected low organism numbers. Culture of deeper layers of adipose tissue rather than the dermis may have been more rewarding. Regardless, this case illustrates the possibility of slow growth and the importance of prolonged incubation, potentially in specialized media, for some RGM. PCR testing on a biopsy specimen may have aided in a more rapid diagnosis but was not carried out owing to previous isolation of a mycobacteria by the referring veterinarian and our interests in obtaining susceptibility test results.
Conclusions
This report extends the known spatial distribution of M porcinum to the western USA and strengthens its association with panniculitis in cats. It demonstrates the need for prolonged incubation for the diagnosis of some RGM infections using culture. Because mycobacterial infections are sporadic, case reports such as this contribute to our progressive understanding that risk factors, approach to diagnosis, disease manifestations and antimicrobial susceptibility vary with mycobacterial species. Such knowledge aids clinicians in optimal approaches to diagnosis, treatment and prevention of mycobacterial infections in cats.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental, including cadavers, tissues and samples) for all procedures undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
