Abstract
Case summary
A 4-year-old female spayed domestic shorthair cat with chronic anemia was evaluated for acute-onset lethargy, vomiting, abdominal distension, and a palpably enlarged and firm spleen. Abdominal ultrasound confirmed marked splenomegaly and concern for a splenic infarct, prompting exploratory abdominal surgery, where splenic torsion was diagnosed. A splenectomy was performed, and the cat recovered uneventfully. Splenitis was diagnosed on histopathology. Anemia improved postoperatively. The role of chronic anemia and other concurrent findings in the development of splenic torsion in this case remains unknown.
Relevance and novel information
Splenic torsion has not been previously reported in cats, making this the first case of its kind. In cases of splenomegaly and abnormal splenic blood flow, splenic torsion should be considered a differential diagnosis in cats.
Introduction
Splenic torsion is an uncommon, but important, surgical disease, typically affecting large-breed male dogs.1,2 The exact mechanism for its development is incompletely understood, although isolated torsion of the splenic pedicle, called primary splenic torsion (PST), is described most frequently. One study evaluating 102 dogs identified no underlying splenic disease. 2 Acute splenic torsion can cause abdominal signs (severe pain) and hypovolemic shock (weakness, cardiovascular collapse). Splenectomy is recommended for all cases of PST. Human splenic torsion is associated with ‘wandering spleen’, a congenital disorder, with weakness or absence of splenic ligaments, allowing excess splenic movement.3,4 The first case of wandering spleen without torsion in a non-human mammal was reported in a cat with gastrointestinal (GI) lymphoma. 5
In cats, splenic disease is not commonly recognized. Reports indicate that neoplasia accounts for 35% of feline splenic disease, with splenitis rare at 2%. 6 One study evaluating outcomes after splenectomy in 19 cats 7 found mast cell tumor to be the most common reason for surgery, then hemangiosarcoma and lymphoma. The study reported one case each of extramedullary hematopoiesis (EMH), myelolipoma and lymphoid hyperplasia.
Splenic torsion has not been described in cats. This case describes the first report of PST in a cat with chronic anemia.
Case description
A 4-year-old female spayed domestic shorthair cat (4.3 kg) presented to an emergency and referral hospital for acute-onset lethargy, episodic vomiting over 36–48 h and abdominal distension. The cat had pre-existing non-regenerative anemia (diagnosed at 6 months of age) worked up elsewhere with a presumptive diagnosis of ‘myelodysplasia’ (without further detail of affected cell line(s) provided). A summary of diagnostics performed >1 year before presentation is provided in Table 1. No definitive etiology for the anemia was identified; the cat was treated with long-term immunosuppressive therapy (prednisolone) and a course of enrofloxacin after mycoplasma was identified in the liver. The cat had maintained a packed cell volume (PCV) in the range of 10–12% for 2 years with clinical compensation for anemia. Upon presentation, the patient was quiet, alert and responsive, with strong/synchronous femoral pulses and pale pink mucous membranes. The cat’s rectal temperature was 101.2°F (38.4°C) and the heart rate was 184 bpm. A grade II/VI parasternal heart murmur was noted. Its lungs ausculted clearly with eupneic breathing. Abdominal palpation revealed marked distension with fluid ballottement present. A firm mid-abdominal structure with discrete margins was felt on either side of the body wall, with mild discomfort elicited.
Diagnostic results before referral

Records were summarized from a different specialty hospital where the anemia workup was performed previously
CBC = complete blood count; FeLV = feline leukemia virus; FIV = feline immunodeficiency virus; FNA = fine-needle aspiration
Blood was collected for complete blood count (CBC), PCV, total solids (TS), biochemistry and blood typing, and urine was collected via cystocentesis for analysis and culture. Additional infectious disease testing was submitted concurrently (Table 2).
Diagnostic results from samples collected at the time of emergency room presentation and before splenectomy

Reference laboratory report
ALP = alkaline phosphatase; ALT = alanine aminotransferase; CBC = complete blood count; FeLV = feline leukemia virus; FIV = feline immunodeficiency virus; HCT = hematocrit; HGB = hemoglobin; MCV = mean cell volume; MIC = minimum inhibitory concentration; NRBC = nucleated red blood cell; PCV = packed cell volume; PLT = platelet count; RBC = red blood cell; TP = total protein; USG = urine-specific gravity; WBC = white blood cell
Abdominal ultrasound with a 13 MHz transducer revealed a large volume of anechoic peritoneal effusion with hyperechoic strands consistent with fibrin. Other findings included marked splenomegaly, hepatomegaly with multifocal, heterogeneous hepatic nodules and an enlarged mesenteric lymph node (LN). The spleen was characterized with mixed echogenicity and a heterogeneous ‘honeycomb’ appearance (images unavailable). Abnormal splenic blood flow was identified with color-flow Doppler, with concern for complete splenic infarct. No splenic or portal vein thrombus was visualized. Ultrasound-guided splenic fine-needle aspiration (FNA) was performed and cytology submitted. Abdominocentesis yielded 250 ml dark yellow cloudy fluid, which was also submitted for analysis and cytology. The TP was 3.2 g/dl, many non-degenerate neutrophils were seen and no etiologic agents were identified. Findings were consistent with a high-protein transudate and suppurative inflammation (Table 2).
Thoracic radiographs revealed mild cardiomegaly but were otherwise unremarkable, without evidence of congestive heart failure. Echocardiography revealed myocardial hypertrophy with biatrial enlargement.
The very firm enlarged spleen and ultrasonographic findings of a heterogeneous ‘honeycomb’ matrix suggested splenic infarction. Exploratory abdominal surgery was scheduled for the next day. Overnight hospitalization included intravenous fluids (Plasmalyte-A 40 ml/kg/day IV; Abbott), maropitant citrate (Cerenia 1 mg/kg IV; Zoetis), buprenorphine (buprenorphine hydrochloride 0.01 mg/kg IV q24h; Par Pharmaceutical), methadone (methadone hydrochloride 0.2 mg/kg IV every 4–6 h for analgesia; Mylan). Antimicrobial therapy was initiated with ampicillin sulbactam (UNASYN 30 mg/kg IV q8h; Pfizer) for neutropenia.
A midline celiotomy was performed, revealing the markedly enlarged spleen torsed around the splenic pedicle (Figure 1). Multiple raised hepatic lesions were noted; a 4 mm punch biopsy was obtained. An FNA was obtained from an enlarged mesenteric LN and submitted for cytology. Splenectomy was performed by ligating the torsed pedicle with two encircling ligatures using 2–0 polydioxanone (PDSII; Ethicon), with care taken not to untwist the spleen during removal. Gastrosplenic ligament laxity was not commented upon in the surgery report. No other gross abnormalities were identified.
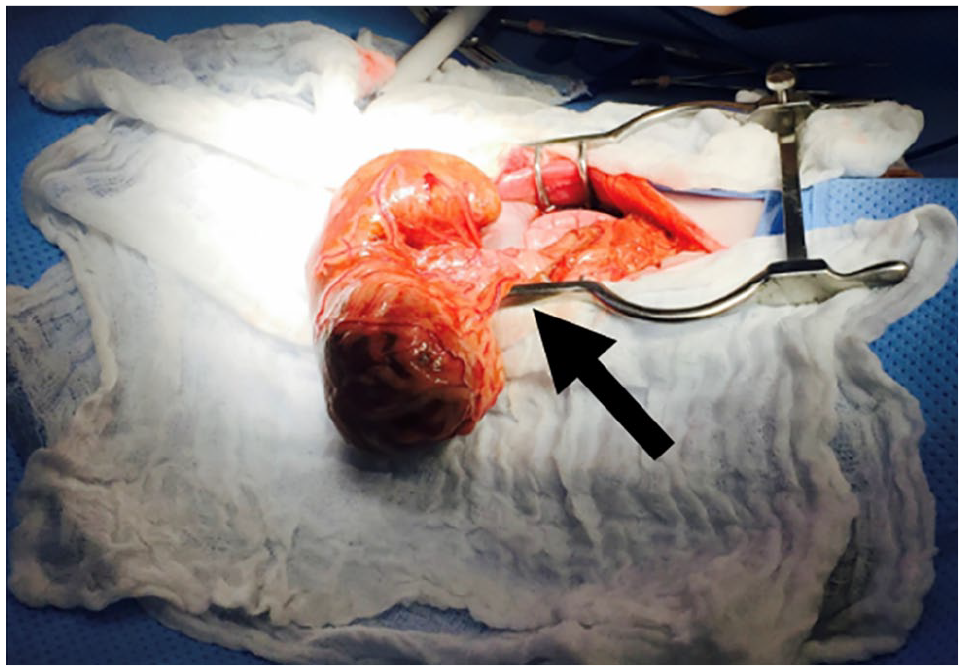
Intraoperative finding of splenic torsion. Note the marked splenomegaly and torsion around the splenic pedicle (arrow)
Histopathology and cytology results are summarized in Table 3. The cytology identified a mixed lymphoid population with small and medium-sized lymphocytes as well as lymphoblasts, and this population was mixed with fewer plasma cells, and increased neutrophils and histiocytic cells. These changes were consistent with mixed-cell splenitis with mild lymphoid hyperplasia. Erythrophagocytosis by histiocytic cells was noted. Normal clumps of splenic stromal cells were seen. No infectious agents or neoplastic cells were identified. Hepatic lesions demonstrated extensive EMH and hemangioma. The LN demonstrated reactive lymphoid hyperplasia.
Diagnostic results after surgery

FNA = fine-needle aspiration; LN = lymph node; PCV = packed cell volume
Postoperative recovery was uneventful. The cat was maintained on IV methadone (methadone hydrochloride; Mylan) for analgesia. The cat’s postoperative weight was 4.08 kg, a decrease of 220 g from its weight before splenectomy. PCV improved before discharge (Table 4). The cat was alive and well 8 months after splenectomy with a stable PCV (24%), and was doing well 2 years later.
Packed cell volume (%) before and after splenectomy

Discussion
This is the first reported case of feline PST in the veterinary literature. The abdominal presentation (pain, distension) is indicative of acute torsion as described in dogs; weakness and cardiovascular collapse are common due to tissue necrosis and altered splenic circulation.1,2 Ultrasonographic findings in dogs and humans include free abdominal fluid, splenomegaly, heterogeneous parenchyma, coarse ‘lacy’ hypoechogenicity and lack of splenic vessel flow on Doppler.2,3,8 –11 Hyperechoic abdominal fat and pancreatitis have been seen in dogs.9,10 Ultrasonographic changes described here compare with findings in dogs; however, the description in this case was limited without archived imaging available for review. The authors acknowledge this missed opportunity.
Causes of PST in other species include inflammatory disease states, splenic ligamental laxity as a congenital abnormality (wandering spleen) or acquired due to gastric dilation (large-breed dogs), neoplasia or peritonitis, accessory splenic tissue and trauma.3,4,12,13 As PST has not been described clinically in cats, conclusions regarding the specific etiology in this case are unavailable. Chronic use of corticosteroids could be a risk factor for gastrosplenic laxity,14,15 but use is common in cats while splenic torsion appears exceedingly rare.
The relationship between PST and chronic anemia in this case remains undetermined. The torsion is possibly incidental and unrelated; however, the anemia was persistent and stable for years then dramatically improved postoperatively, suggesting the circulatory changes and decreased blood pooling were involved with the rapid erythron recovery.
This cat’s chronic disease included a non-regenerative macrocytic anemia with marrow supporting myelodysplasia. Myelofibrosis may have developed over time, resulting in marked EMH in the liver. Both liver and spleen provide the necessary vasculature and microenvironment to support EMH. 16 The lack of EMH in the spleen may be evidence of altered blood flow to this organ and chronic torsion. Chronic splenic torsion is described in dogs, posing diagnostic challenges with vague clinical signs, including intermittent vomiting and malaise.8,10 Periods of remission of clinical signs may occur, with exacerbations as acute complications, including thrombosis, develop within the compromised splenic parenchyma.
Splenitis in this case was identified both cytologically and on histopathology. Human PST can occur with inflammatory disease states.5,17 The mild preoperative neutropenia makes splenic neutrophilic sequestration another possibility, which may be cytologically difficult to distinguish from inflammation. While the neutropenia may be partially due to sequestration, mixed-cell splenitis supports an inflammatory process rather than splenic sequestration alone.
The spleen is the primary site of EMH, along with the liver in domestic animals and humans.16,18 Hepatic hemangioma was unexpected in this case. This type of vascular tumor is a benign and often incidental finding. In cats, hemangiomas are rare and tend to occur on the eye, with one report of an arteriovenous hemangioma in a cat with a vascular skin lesion. 19 Hepatic hemangioma is poorly described in cats. Its significance here is unknown and no evidence of EMH was noted on microscopic description.
Chronic corticosteroid treatment could contribute to anemia through marrow retention of iron or GI blood loss from ulceration, although functional or absolute deficiency is associated with microcytosis. 20 Macrocytosis can also result from deficiencies in vitamin B12 and folates, which can be seen with chronic GI disease and malabsorption, 21 but with no history of GI disease in this case, biopsies were not collected during surgery. Previous anemia workup did not include GI investigation and the conclusion was a primary marrow disorder (myelodysplasia).
The cardiac abnormalities demonstrated (ventricular hypertrophy, atrial dilation) were consistent with anemic cardiomyopathy, as reported previously. 22 Primary cardiac disease could not be ruled out.
A significantly increased hematocrit was observed postoperatively. This rebound was considered the result of pre-existing EMH, where erythroid precursor cells were seen in the liver (Table 3), coupled with removal of an area of blood pooling and blood flow redistribution (splenectomy). Unfortunately, full CBC was not performed concurrently, and precise timing of the regenerative response was not tracked. The source of the chronic anemia remains speculative and could involve both myelodysplasia and chronic splenic torsion. Financial concerns prohibited re-evaluating the original diagnosis in the acute setting.
There is a report of anemia and thrombocytopenia in a dog with acute PST that resolved after splenectomy. 12 The cause of anemia was presumed to be splenic sequestration. However, sequestration alone does not explain the chronic anemia here. Anemia appears to be a common feature in dogs with chronic splenic torsion.9,13 Splenic pathology may contribute to chronic anemia in some cats, but mechanisms are yet to be fully understood. The theory of anemia-induced hypersplenism is a feature of human medicine 23 and may provide a rationale for anemia in animals with PST. In a case series reporting intermittent severe hemolytic anemia in Abyssinian and Somali cats, splenomegaly was documented in 10/13 cases, 24 further supporting this theory. Increased destruction by splenic reticuloendothelial pathways is associated with extravascular immune-mediated hemolytic anemia (IMHA), although classic signs of IMHA (icterus) were not present in this case, nor did prior anemia investigations support this diagnosis.
Conclusions
No specific underlying cause for the PST was determined. The splenitis and chronic anemia could have predisposed to torsion, or a chronic partial torsion may have contributed to the anemia and splenic inflammatory process. Evidence is lacking regarding splenectomy indications in cats without splenic neoplasia, 8 although PST should be added to the list. Regardless of etiology, the PST resolved with splenectomy, and the cat’s anemia improved and has remained stable for 2 years.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support from the Center for Integrative Mammalian Research, Ross University School of Veterinary Medicine.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.
