Abstract
Case series summary
Two castrated male domestic shorthair cats (aged 8 months [case 1] and 13 years [case 2]) were presented at the Small Animal Clinic of the Veterinary Medicine University of Vienna, Austria, both with acute vomiting and distended abdomen, as well as a history of chronic apathy, recurrent vomiting and diarrhoea. Both cats underwent invasive diagnostic procedures approximately 1 month before the diagnosis of sclerosing encapsulating peritonitis (SEP), namely an exploratory laparotomy and a bronchoscopy, respectively. Abdominal ultrasound revealed severely corrugated intestinal loops and, in case 2, the presence of peritoneal effusion. A thick and diffuse fibrous capsule around the intestine was detected and removed surgically, and biopsies were taken from the affected organs confirming the SEP. Case 1 recovered well, was discharged some days after surgery and was clinically unremarkable for the next 2 years. Case 2 showed unsatisfactory improvement directly after surgery and was euthanased a few days later, as the owner declined any further therapy.
Relevance and novel information
SEP is a very rare condition of unclear origins in cats. Here we describe the clinical and diagnostic imaging features, surgical treatment, and outcome of SEP in two cats. The results indicate that prompt diagnosis and appropriate interventions may improve the outcome.
Introduction
Sclerosing encapsulating peritonitis (SEP) is a rare clinical syndrome defined as an acquired inflammatory fibrocollagenous thickening of the peritoneal membranes, eventually resulting in intestinal obstruction.1,2 SEP can be differentiated into primary (idiopathic) or secondary. In human medicine, it is most commonly seen in people who undergo peritoneal dialysis.1,3–5 Abdominal surgery, foreign material or substances in the abdominal cavity are other common causes of SEP in people.5,6 The suspected underlying pathomechanism suggests the activation of a proinflammatory and proangiogenic cytokine cascade, promoting the detachment of mesothelial cells and their differentiation into mesenchymal cells, thus resulting in a fibrocollagenous cocoon that alters the peritoneum’s function.7–11 In dogs, SEP remains extremely rare, although various cases have been reported.12–23 SEP must also be histologically differentiated from sclerosing peritoneal mesothelioma. 24 In cats, very few cases of SEP have been reported18,25–27 and the condition is believed to be even more infrequent.
Case series description
Case 1
An 8-month-old castrated male shorthair outdoor cat was referred to our clinic with a 3-month history of diarrhoea, vomiting and progressive lethargy. The cat had previously been treated with pre- and probiotics, deworming, diet trials and vitamin B12 administrations by the referring veterinarian, who also performed an exploratory celiotomy, without clinical improvement. The operative findings were reported as unremarkable. Thirty days later, clinical signs worsened, and the cat was referred to our clinic. On physical examination, only mild dehydration and mild abdominal distension were present. The patient was normothermic. Feline leukaemia virus (FeLV) antigen test and feline immunodeficiency virus (FIV) antibody test (SNAP FIV/FeLV Combo Test; Idexx Laboratories), as well as parvovirus antigen test, were negative. Complete blood count (CBC) and serum biochemistry showed mild lymphocytosis (6453/µl, reference interval [RI] 099–5100/µl) and increased serum amyloid A (SAA) (149.6 mg/l, RI <5 mg/l) (see Table 1 in the supplementary material). Abdominal ultrasound and radiographs showed multiple corrugated small intestinal loops, a mildly dilated stomach filled with fluid and ingesta, as well as prominent abdominal lymph nodes. However, neither a foreign body nor the presence of peritoneal effusion were visualised. Based on the persistent clinical and ultrasonographic findings, an exploratory celiotomy was performed. Minimal ascites was present, and a greyish white 1-mm thick, fibrous capsule was covering the small intestine. The intestinal loops were tightly compressed in this mantle of tissue, completely impairing motility (Figure 1a). The fibrous coating started at the descending duodenum, extending down to the ileocaecocolic junction. Moreover, the ventral spleen extremity was partially coated in the same fibrous tissue. Incision of the capsule revealed mild adhesions to the underlying intestinal serosa. As the fibrotic layer could be elevated, all adhesions were ablated and an immediate onset of motility was noticed.

Intraoperative findings in the two cases of sclerosing encapsulating peritonitis. (a) Case 1: intraoperative presentation of the small intestines. Thick, opaque fibrous tissue coated the descending duodenum, the jejunum and ileum, terminating at the ileocaecocolic junction. There were no signs of intestinal motility visible. The remaining abdominal organs were of normal appearance. (b) Case 2: intraoperative presentation immediately after incision of the abdominal wall, cranial to the left. All abdominal organs, including the liver and stomach, are coated in a layer of thick, fibrous material, with the small intestines being wrapped in a further outer layer of fibrous tissue, forming a dense, opalescent capsule. (c) Case 2: intraoperative presentation after incision of the outer fibrous membrane covering the intestines (cranial to the left). The layer of fibrous tissue coating the small bowel and stomach was carefully incised and could be easily removed with minimal irritation to the underlying intestines. In the background, the stomach is visible, being coated in the same, opaque layer of fibrous tissue. (d) Case 2: intraoperative presentation after partial removal of the fibrous tissue from the jejunum (cranial to the left). The intestinal loops expand and have an otherwise unremarkable appearance
Full-thickness jejunum sections of approximately 2–3 mm, a mesenteric lymph node and multiple beige-greyish membrane-like structures ablated from the external intestinal wall were examined histologically. The specimens were fixed in 4% neutral buffered formaldehyde for 24 h; thereafter the samples were dehydrated in rising concentrations of ethanol and embedded in paraffin wax. Sections of 4-µm thickness were stained with haematoxylin and eosin (H&E), Masson trichrome staining (MTS) or Prussian blue staining (PBS) to highlight ferrous salt deposits. Histopathological analysis confirmed the severe thickening of the denudated visceral peritoneum covering the intestine, composed of a broad fibrous encapsulating membrane approximately 400–500 µm in size (Figure 2a). Most of the tissue samples comprised a deep layer of densely packed collagen fibres with numerous active fibroblasts and a superficial layer with a loose collagen matrix (Figure 2b) admixed with multifocally interspersed light basophilic, mucinous deposits. The partially degenerated collagen fibres were interspersed with markedly activated slightly pleomorphic fibroblasts (Figure 2c). The fibrous tissue was oligofocally spread especially on the serosal surface, accompanied by exudated fibrin deposits (Figure 2d). Furthermore, minor oligofocal haemorrhages (Figure 2e) and multifocal aggregates of lymphocytes and plasma cells (Figure 2f) were identified. Ferrous salt deposits were detected in solitary locations (Figure 2g). The jejunum layers and the mesenteric lymph node were histologically unremarkable.
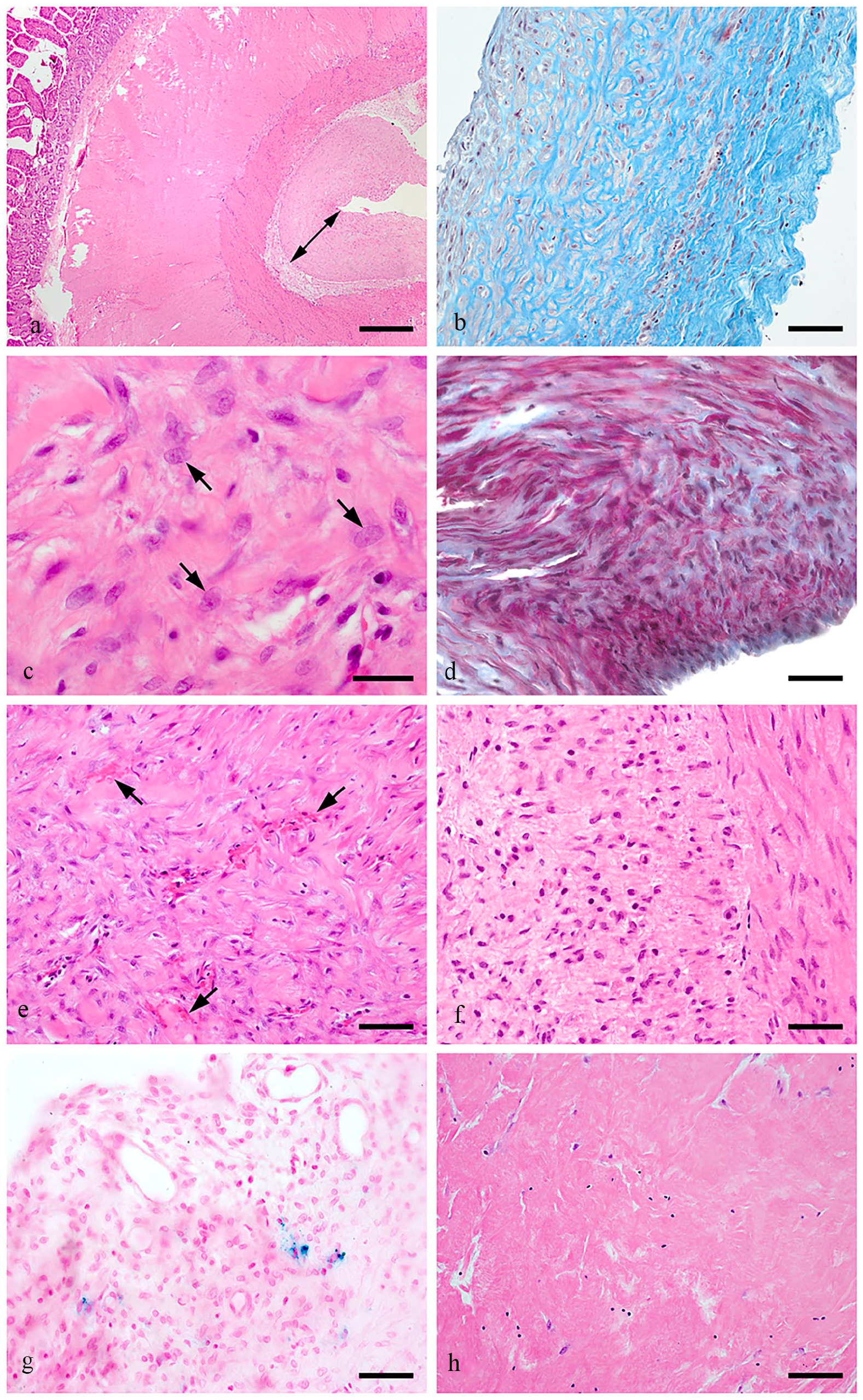
Histopathological findings of the examined biopsy samples. (a) Case 1: severely thickened visceral peritoneum (double arrow) mostly composed of sparsely cellular, fibrous tissue; haematoxylin and eosin (H&E), bar represents 400 µm. Figure 2 (Continued) (b) Case 1: photomicrography of ablated fibrous membranes showing local variation in distribution of interlacing bundles of collagen fibres (blue); Masson trichrome staining (MTS), bar represents 80 µm. (c) Case 1: enlarged, activated fibroblasts (black arrows) interspersed throughout the fibrous membrane containing large nuclei with 1–2 evident nucleoli; H&E, bar represents 30 µm. (d) Case 1: locally abundant fibrin deposits (red) with interspersed activated fibroblasts and tender collagen fibre production (blue) reflecting initiated organisation of the exudative mass; MTS, bar represents 40 µm. (e) Case 1: neovascularisation and perivascular bleeding (black arrows) accompanying the fibrous proliferation; H&E, bar represents 80 µm; and (f) mild mononuclear cell infiltration predominantly composed of lymphocytes and plasma cells; H&E, bar represents 40 µm. (g) Case 1: areas of iron deposition (blue) in encapsulating membrane; Prussian blue staining, bar represents 40 µm. (h) Case 2: regional low cellularity of fibrous membrane covering the intestine; H&E, bar represents 80 µm
Aerobic and anaerobic bacterial cultures of dissected specimens were negative. Postoperative treatment included intravenous fluids, buprenorphine 15 µg/kg IV q6h, meloxicam 0.05 mg/kg IV q24h and maropitant 1 mg/kg PO q24h. The cat was discharged soon after surgery and was unremarkable until the last follow-up at 30 months.
Case 2
A 13-year-old castrated male shorthair indoor cat was referred to our clinic with a 4-week history of recurrent vomiting, diarrhoea, progressive lethargy and abdominal distension. Four days before presentation, the referring veterinarian had suspected nasopharyngitis after performing a rhinoscopy and bronchoscopy and treated the cat with maropitant and dexamethasone.
Upon presentation, abdominal distension and moderate dehydration were detected. The rest of the clinical parameters were normal. CBC showed mild non-regenerative normochromic normocytic anaemia (haematocrit 28.8%, RI 30.3–52.3%) and mild neutrophilia (14380/µl, RI 2300–10290/µl) (see Table 1 in the supplementary material). FeLV and FIV tests were performed as in case 1 and were negative. Marked ascites and a thin capsule surrounding the intestine, liver and part of the spleen were found on abdominal ultrasonography (Figure 3) and confirmed by CT (Figure 4). These imaging features were consistent with SEP and a celiotomy was performed.
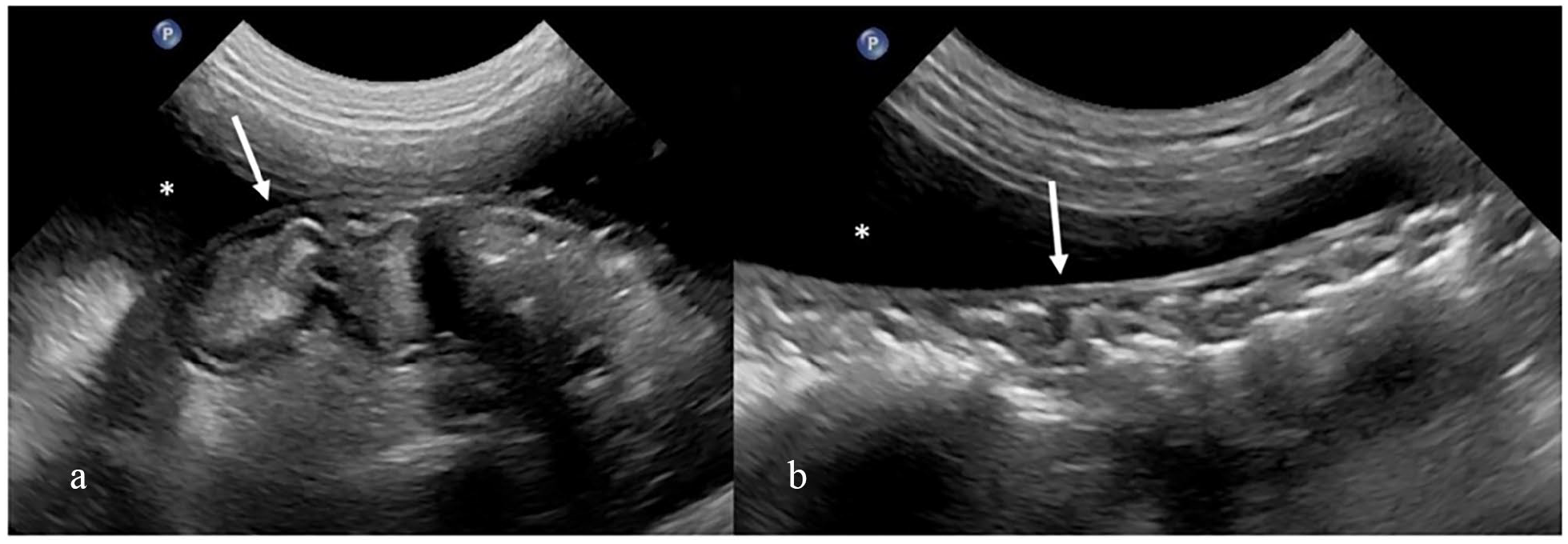
Ultrasound images of (a) the small intestine and (b) large intestine of case 2. Note the plication of the small intestine and the marked corrugation of the descending colon encased by a smooth capsule (arrow) and surrounded by peritoneal effusion (*)

Transverse CT images of case 2 in a soft tissue window at the level of (a) the second, (b) third and (c) fourth lumbar vertebra. Abdominal organs together with mesenteric fat (**) are encapsulated in the centre (‘cocooning’) and surrounded by peritoneal effusion (*) in the periphery. (b, c) The plication of the small intestine can be appreciated. C = colon; L = liver; LK = left kidney; RK = right kidney; SI = small intestine; Sp = spleen; St = stomach
A greyish-white fibrous capsule up to 1-mm thick was covering the liver, omentum, stomach and the entire small intestine (Figure 1b). The intestinal adhesions were ablated, and the fibrous capsule was removed from the gastrointestinal tract and the liver (Figure 1c,d). Bacterial cultures along with real-time PCR for feline coronavirus from the abdominal fluid were negative. Postoperative treatment was similar to case 1 with the addition of ondansetron 0.3 mg/kg IV q8h. Capsule-like structures ablated from the intestine and liver, as well as adhesive fibrous tissue spanning between the organs, were submitted for histopathological examination and were processed as described previously. As in case 1, the samples mainly depicted cell-poor fibrous tissue accompanied by large amounts of collagen fibres. Some areas were nearly acellular (Figure 2h) and were sometimes associated with reasonable amounts of fibrin. Activated fibroblast showed no signs of malignancy.
Despite the minimal bleeding during surgery, anaemia progressed along with a marked increase of alanine aminotransferase, aspartate aminotransferase, creatine kinase, potassium and phosphate immediately after surgery. Upon the owner’s request, the cat was discharged on day 4 after surgery, and was euthanased on day 6 after surgery due to unsatisfactory improvement.
Discussion
SEP is rarely diagnosed in veterinary medicine, and is mainly reported in experimental animals used for peritoneal dialysis. 28 The paucity of published feline cases makes feline SEP even rarer than in dogs and the condition seems to have a poor prognosis. To our knowledge, this is the first case report of two cats that describes the clinical findings together with the imaging characteristics, surgical therapy and outcome.
To date, the pathophysiology of SEP is thought to start either from a predisposing or triggering condition or to be idiopathic. In dogs, SEP is usually secondary, as it is frequently related to ongoing abdominal processes,17,24 systemic infections 22 or most commonly to intestinal foreign bodies.13,18,19,23,27 While the pathomechanism in cats appears to be similar to dogs, very few cases have been described to hypothesise what promotes the development of SEP. Case 1 underwent an exploratory laparotomy by the referring veterinarian, which could have served as a trigger. However, congenital encapsulating peritoneal sclerosis cannot be fully excluded. In case 2, a possible trigger is even more difficult to hypothesise.
Peritoneal effusion is a common clinical finding of SEP in both dogs and cats. We hypothesise that the chronicity and severity of encapsulation play a role in the severity of ascites, although this remains to be proven.
A rapid and accurate diagnosis of this rare and clinically non-specific disease can be challenging, as specific biomarkers are not available. In contrast to case 2, case 1 presented with increased SAA, which decreased after surgery. Hence, although non-specific, SAA can perhaps be associated with an acute manifestation of SEP when other comorbidities are ruled out.
Our radiographic findings are consistent with the findings reported in the literature. 27 Some articles reported that intestinal barium studies can more easily identify the conglomerated bowels in the so-called ‘cauliflower sign’.2,25,26 Abdominal ultrasonography is presumably the most commonly available imaging technique that may provide the first evidence of SEP. Common findings are peritoneal effusion and encased intestinal loops with a plicated and/or corrugated appearance.21,27 Abdominal CT with contrast enhancement is the most specific diagnostic examination both in human and veterinary medicine2,27 as it clearly identifies and localises the intestinal encasement, the peritoneum’s thickening and adhesions.
Definitive diagnosis requires surgical exploration and histopathological examination. A white and thick appearance of the peritoneum, encased intestines and/or other organs are typical surgical findings.
Histologically, the covering membrane-like structures resemble fibrotic regions of simple sclerosis. The histological features of SEP that help discriminate these two pathomorphologically similar conditions are as follows: high expression levels of podoplanin; presence of numerous activated fibroblasts; denudation of the mesothelium; calcification areas; acellular regions; fibrin deposits; signs of vasculopathy; acute inflammation; and iron deposition. 29 Podoplanin expression is identified in various feline tumours, in the majority of cancer-associated fibroblasts 30 as well as in several canine tumours 31 and therefore would be an interesting feature to investigate in the future. Decreased cellularity was suggested to be a late stage finding; 29 the latter could explain the poor outcome of case 2. Honda et al 32 categorised the newly formed membranes into three stages. Applied to our cases, we could speculate that case 1 was a stage I because of high amounts of fibrin deposits and high cellularity, while case 2 showed signs of stage II or III.
Medical treatment of SEP is usually ineffective. A non-surgical approach is normally attempted when the SEP grade is mild.5,33 In animals, this approach is not described, probably because of the higher stage of SEP at diagnosis. When conservative management is ineffective, drugs, such as tamoxifen, corticosteroids, colchicine and others, have been suggested in humans.2,3,5,33 A combination between these drugs – especially corticosteroids and tamoxifen – seems to be more effective in people.2,5 In dogs, the addition of tamoxifen has been positively described in some case reports,19,23 but so far its use has not been reported in cats.
The prognosis of SEP in small animals is reported to be guarded to poor.15,18 In the published feline cases, 6/7 cats were euthanised soon after diagnosis and just one cat was still alive 11 months after surgery.18,25–27 Here, case 1 had an excellent outcome. Case 2 showed unsatisfactory improvement and was euthanased upon the owner’s request. It is possible that the clinical and haematological deterioration in case 2 (see Table 1 in the supplementary material) may be related to a reperfusion syndrome that occurred after the removal of the capsule and the chronic adhesions. On the other hand, liver injury during removal of the liver capsule and liver manipulation could have also contributed to the marked increase in liver enzymes. Given the above, we surmise the prognosis could be positively associated with young age or early stage of disease and negatively with the chronicity of the disease.
Conclusions
Herewith we describe two cases of SEP in cats. SEP is a very rare, but potentially underdiagnosed condition in cats that has a guarded to poor prognosis. Given the positive outcome in case 1, early diagnosis and prompt surgical management may improve prognosis. Future prospective controlled studies are needed to better understand the disease in cats and to evaluate prognosis; however, this will be challenging given the condition’s rarity.
Supplemental Material
sj-docx-1-jor-10.1177_20551169231178447 – Supplemental material for Sclerosing encapsulating peritonitis in cats: a two-case report and literature review
Supplemental material, sj-docx-1-jor-10.1177_20551169231178447 for Sclerosing encapsulating peritonitis in cats: a two-case report and literature review by Caterina Kiniger, Janina N Janssen, Kristina A Lederer, Karoline Lipnik and Pavlos G Doulidis in Journal of Feline Medicine and Surgery Open Reports
Footnotes
Accepted: 10 May 2023
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support for the publication of this article was provided by the Division for Small Animal Internal Medicine, University of Veterinary Medicine Vienna. The authors received no financial support for the research and authorship of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
Supplementary material
The following file is available online:
Supplementary Table 1: Haematology and serum biochemistry before and after surgery.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
