Abstract
Case summary
A 2-year-old spayed female domestic longhair cat was presented for evaluation of chronic ocular discharge and occasional vomiting. While physical examination findings were consistent with an upper respiratory infection (URI), serum chemistry results revealed increased liver enzyme activities. Histopathologic examination of a liver biopsy identified substantial centrilobular accumulation of copper in hepatocytes – strongly suggestive of primary copper hepatopathy (PCH). Retrospective cytologic examination of a liver aspirate also identified copper aggregates in hepatocytes. After transitioning to a low-copper diet, 1 year of chelation therapy with D-penicillamine achieved normalization of liver enzyme activities and resolution of persistent ocular signs. Subsequently, a long-term regimen of zinc gluconate has been successfully managing the cat’s PCH for almost 3 years. Sanger sequencing of the cat’s ATP7B gene, which encodes a copper-transporting protein, revealed a novel, ‘likely pathogenic’, single nucleotide variation (c.3670t/a [p.Trp1224Arg]), for which the cat is heterozygous.
Relevance and novel information
Recommendations are described for the long-term clinical management of feline PCH – a previously attainable but unreported outcome – with considerations for mitigating the speculated oxidation-exacerbated ocular risks of concurrent URI. This report is the first to include identification of copper aggregates in a liver aspirate from a cat – evidence that liver aspirates from cats could be routinely examined for copper as is standard practice for those from dogs. The cat is also the first reported with PCH and a ‘likely pathogenic’ heterozygous ATP7B genotype, which suggests that normal ATP7B alleles could be recessive to or incompletely/co- dominant with deleterious ATP7B alleles in cats, as has been reported in other species.
Case description
A 2-year-old spayed female domestic longhair cat was presented for chronic ocular discharge and occasional vomiting. Since adoption at approximately 8 weeks old, the cat had lived indoors and eaten wet and dry commercially available feline-formulated food. Physical examination identified bilateral watery ocular discharge, nasal planum ulceration, submandibular lymphadenopathy, mildly depressed mentation and pyrexia (103.0°F) – findings consistent with an upper respiratory infection (URI). A complete blood count was unremarkable and feline pancreas-specific lipase was within normal limits; however, serum chemistry revealed increased alanine aminotransferase (ALT; 996 U/l, 28–109 U/l) and alkaline phosphatase (ALP; 111 U/l, 11–49 U/l) activities; both were within their respective reference interval before the cat’s ovariohysterectomy 18 months prior. Ultrasonographic examination of the abdomen identified diffusely coarse echotexture of the liver and hyperechoic gallbladder walls. The following tests were all negative: fluorescein for corneal ulceration; IgG and IgM titers for Toxoplasmosis gondii; PCR for feline infectious peritonitis (sample: liver tissue); and serologic testing for feline leukemia virus (FeLV) antigens, feline immunodeficiency virus (FIV) antibodies and heartworm antigens.
After administration of buprenorphine (0.06 mg/kg, SC) and vitamin K (2 mg/kg, SC, preoperatively; PO, q24h, 30 days postoperatively), anesthesia was induced with alfaxalone (2 mg/kg, titrated to effect over 30 mins) and maintained with inhalant isoflurane throughout an ultrasound-guided Tru-Cut biopsy of the left medial liver lobe. Histopathologic examination of hematoxylin and eosin-stained biopsy sections identified cytoplasmic copper aggregates in hepatocytes with greatest accumulation in centrilobular regions (Figure 1). Subsequent copper-specific rhodanine staining (Figure 2) permitted digital quantification of the copper 1 (dry weight copper = 3051 µg/g, normal <400 µg/g, secondary copper hepatopathy <700 µg/g, primary copper hepatopathy >700 µg/g) and the results were strongly suggestive of primary copper hepatopathy (PCH). 2 A cytologic re-examination of a modified Wright’s-stained smear of a liver aspirate identified cytoplasmic blue-green refractile and non-refractile aggregates in hepatocytes – the former consistent with copper and the latter possibly lipofuscin or a copper variant (Figure 3).

Photomicrograph of a hematoxylin and eosin-stained section of a liver biopsy from a cat with primary copper hepatopathy. Moderate amounts of cytoplasmic brown-orange pigment (arrow) were evident in hepatocytes, extending from the central vein to the portal region (bar = 50 µm) with most of the pigment found in the centrilobular region. The inset is a higher magnification image of the region closest to the central vein to better demonstrate the cytoplasmic pigment (bar = 25 µm)

Photomicrograph of a rhodanine-stained section of a liver biopsy from a cat with primary copper hepatopathy. Copper was appreciable as orange-red aggregates around the hepatic veins (HVs) (bar = 1 mm). The inset is a higher magnification image to better demonstrate cytoplasmic copper aggregates in individual hepatocytes (bar = 100 µm)

Photomicrograph of a modified Wright’s-stained smear of a liver aspirate from a cat with primary copper hepatopathy. The cytoplasm of several hepatocytes contained small amounts of blue-green refractile pigment (arrows), consistent with copper (bar = 10 µm). The more numerous blue-green non-refractile aggregates could be lipofuscin or a copper variant
To treat the liver disease, metronidazole (20 mg/kg, PO, q12h, 7 days; 10 mg/kg, PO, q12h, 14 days), ursodiol (10 mg/kg, PO, q24h) and Denamarin (18 mg/kg S-adenosyl-l-methionine and 1.8 mg/kg silybin A+B, PO, q24h) were prescribed. To reduce nausea and hepatic inflammation, maropitant (1 mg/kg, PO, q24h) and prednisolone (1 mg/kg, PO, q12h) were also prescribed. The cat refused a copper-free diet, so commercially available low-copper wet (Salmon and Rice Entrée in Sauce; Purina Pro Plan) and dry (Urinary Care c/d; Hills) foods were fed instead. Despite the administration of zinc gluconate (10 mg/kg, PO, q24h) to reduce enteric copper absorption, liver enzyme activities continued to increase (maximum: ALT >2000 U/l, ALP = 255 U/l), so the zinc gluconate was replaced with D-penicillamine (15 mg/kg, PO, q12h), which chelates copper systemically.
To treat the URI, famciclovir (50 mg/kg, PO, q12h, 14 days) and Clavamox (10 mg/kg amoxicillin trihydrate and 2.5 mg/kg clavulanate potassium, PO, q12h, 5 days) were prescribed. The ocular discharge persisted with intermittent conjunctivitis progressing to corneal opacity and ulceration of the right eye, which was appreciable grossly and highlighted by fluorescein, although corneal cytology was unremarkable. Erythromycin (1 cm of 5 mg/g ointment, OD, q6h, 5 days) was prescribed for its dual antibiotic and anti-inflammatory properties. 3 In addition, intermittent nausea/vomiting and inappetence were treated using fluid therapy (20–40 ml/kg lactated Ringer’s solution (LRS) with 37.5 mg/cat B Complex 150 and 0.25 mg/cat B12, SC), mirtazapine (0.4 mg/kg, TD, q48h) and ondansetron (0.2 mg/kg, PO, q12h).
Serial serum and urine chemistries were performed after 1, 3, 11 and 12 months of chelation therapy; minor abnormalities (hyperalbuminemia, hyperbilirubinemia, azotemia and proteinuria) were all characteristic of PCH pathogenesis 2 or treatment. 4 After 12 months of chelation therapy, liver enzyme activities had normalized, and all ocular signs had resolved. Client concerns precluded repeating aspiration or biopsy of the liver, so serum copper was measured and found to be within reference limits (Cu = 0.77 ppm, 0.6–1.4 ppm), suggesting copper homeostasis. However, the lack of a pre-treatment serum copper level for comparison and minimal evidence of correlation between serum and liver copper levels 5 were limitations of this approach. Considering these indicators of copper homeostasis, the cat was transitioned from D-penicillamine back to zinc gluconate as previously prescribed; however, the cat continued to vomit. Decreasing the zinc gluconate dose (6 mg/kg, PO, q24h) substantially reduced vomiting, and the cat’s liver enzyme activities have remained within reference limits for almost 3 years, which suggests that this regimen – in combination with the low-copper diet – will effectively manage the PCH in the long term. The cat continues to receive Denamarin, maropitant and ondansetron, as previously prescribed, to support the liver and manage occasional vomiting. The cat has remained chronically pyrexic (103.0–105.0°F) in hospital; however, the cat’s temperature cannot be taken at home to rule out stress.
Other cases of feline PCH have been linked to variations in copper-transporting ATPase 2,5,6 a transmembrane protein encoded by the ATP7B gene that maintains copper homeostasis by exporting excess cellular copper. 7 Using previously published methods, 5 genomic DNA extracted from the cat’s whole blood was Sanger sequenced, and the coding regions of the cat’s ATP7B gene were aligned with a feline consensus sequence (NCBI: XM_023251165.1) using CLC Sequence Viewer. Alignment identified 23 single nucleotide variations (SNVs), which included six missense variations, two of which had not been previously reported in healthy cats (c.1268c/t and c.3670t/a) (Table 1). Using the 99 Lives Consortium data set (n = 340 cats), c.1268c/t was called with an allele frequency of 0.134, whereas c.3670t/a was not called.
Missense single nucleotide variations (SNVs) identified in the ATP7B gene of a cat with primary copper hepatopathy*

ATP7B genome position: chromosome A1: 19,541,514-19,614,548 forward strand
Bold text indicates a previously unreported SNV
SNV = single nucleotide variation
To determine whether these novel SNVs were pathogenic, the consensus amino acid sequence of the feline ATP7B gene was aligned with seven other species using previously described methods (Figure 4). 5 Substitutions in functionally important amino acids are more likely to be pathogenic and therefore purged by natural selection (ie, functionally important amino acids are highly conserved across species). While the amino acid affected by the c.1268c/t SNV (p.Thr423Ile) was only conserved across three of the eight species (Figure 4a), the amino acid affected by the c.3670t/a SNV (p.Trp1224Arg) was conserved across all eight species (Figure 4b), which suggests that the c.3670t/a SNV and resulting p.Trp1224Arg substitution could be pathogenic.
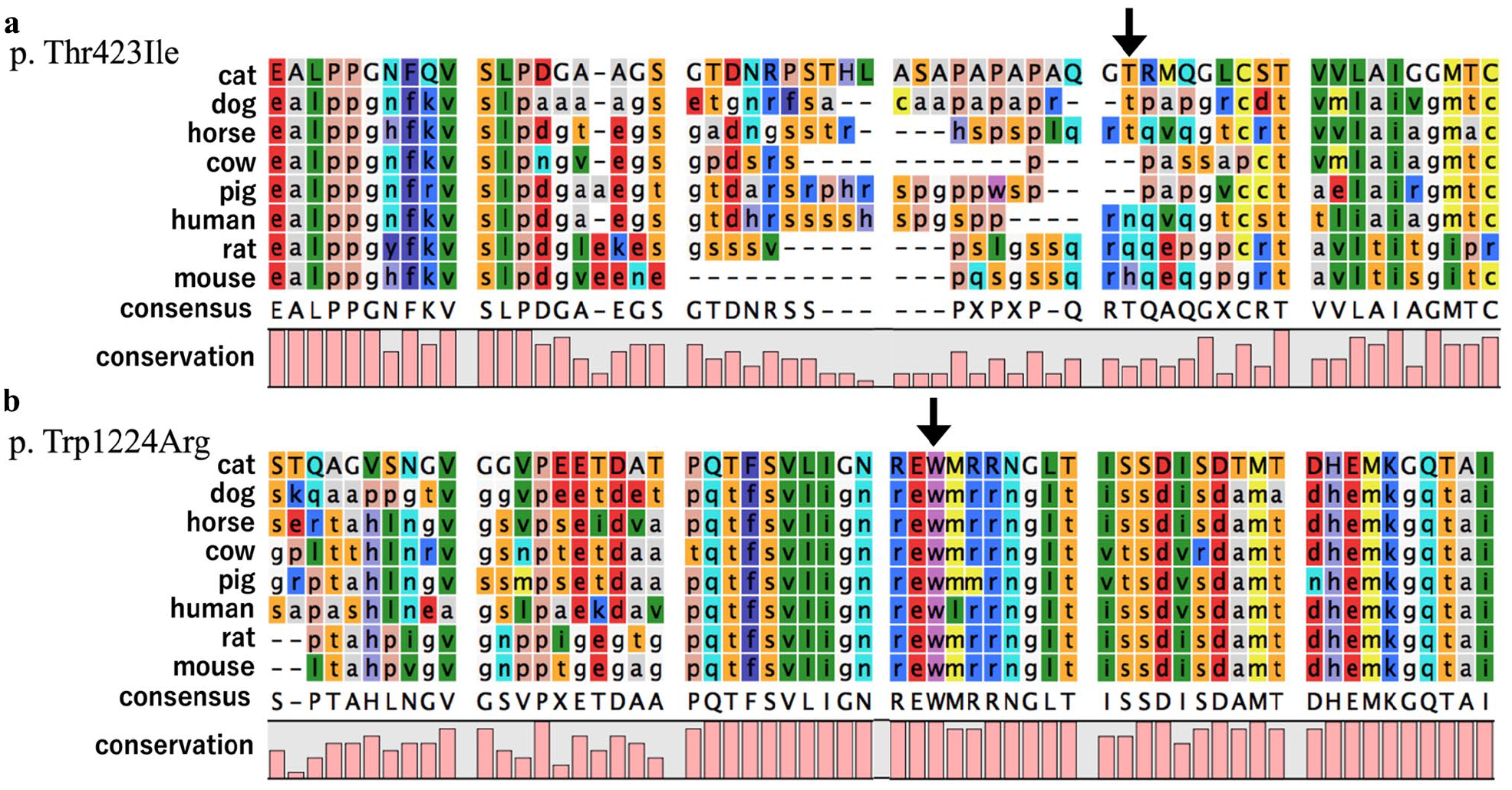
Amino acid sequence alignment of ATP7B across eight species in the regions of both previously unknown single nucleotide variations (SNVs) found in a cat with primary copper hepatopathy. The black arrows indicate the locations of amino acid substitutions owing to the novel SNVs. (a) The amino acid affected by the c.1268c/t SNV (p.Thr423Ile) was only conserved across 3/8 species, (b) whereas the amino acid affected by the c.3670t/a SNV (p.Trp1224Arg) was conserved across 8/8 species. NCBI Reference Sequences: dog NP_001020438.1, horse XP_005601246.1, cow XP_002691840.1, pig XP_020920945.1, human NP_000044.2, rat NP_036643.2 and mouse NP_031537.2
The effects of both amino acid substitutions on the function of ATPase 2 were predicted using PolyPhen2, SIFT, PROVEAN and PANTHER, using previously described methods (Table 2). 5 Substantial effects were not predicted for p.Thr423Ile but were predicted for p.Trp1224Arg. The p.Trp1224Arg substitution changes the primary structure of copper-transporting ATPase 2’s ATP-binding cytoplasmic domain, which would likely prevent active transport of copper across membranes.
Protein function analysis of two novel amino acid substitutions found in a cat with primary copper hepatopathy*

PolyPhen2 results closer to 1 and SIFT results closer to 0 suggest more pathogenic effects. The c.3670t/a SNV is just slightly above SIFT’s 0.05 cutoff for significance. For both PROVEAN and PANTHER, more negative results suggest more pathogenic effects
Collectively, these results support conservative characterization of the c.3670t/a SNV as ‘likely pathogenic’ 8 (Table 3) and a likely cause of this cat’s PCH despite occurring on only one chromosome (ie, the cat is heterozygous for this SNV).
While there could be sufficient evidence to characterize the c.3670t/a SNV as ‘pathogenic’ (1 strong: PS1; 2 moderate: PM1 and PM2; <2 supporting: PP2, PP3 and PP4), some of the evidence requires extrapolation from other species, so the variation was conservatively characterized as ‘likely pathogenic’ instead. This approach allows for omission of either one moderate and all supporting criteria, both moderate criteria, or the sole strong and one supporting criteria
PCH = primary copper hepatopathy
Discussion
Feline PCH is rarely reported2,5,6,14–16 and has not been as comprehensively studied as in other species. For example, Wilson’s disease (WD) is a type of human PCH associated with 1400 known ATP7B variations, 10 many more than described from only two sibling 5 and three other cats 6 with PCH. These preceding cats were all homozygous for their respective SNVs, unlike the cat reported here. If this cat is truly heterozygous for this ‘likely pathogenic’ variation (ie, not compound heterozygous with undetected pathogenic variations, such as in non-coding DNA), then normal feline ATP7B alleles could either be recessive to or incompletely/co- dominant with deleterious ones. There are also reports of ATP7B heterozygous WD patients, 17 yet WD is misleadingly described as a recessive disease. Similarly, ATP7B heterozygotes with clinical signs have also been described in laboratory mice, 11 and ATP7B heterozygous Dobermans 12 and Labrador Retrievers 13 express an intermediate phenotype. Further research is needed to fully implicate ATP7B variations in PCH for non-laboratory species, but the potential for ATP7B heterozygotes to be both carriers and clinically ill is evident.
In 50% of WD patients with hepatopathy, copper can also accumulate in the cornea as pathognomonic Kayser–Fleisher rings. 18 While copper was not grossly visible in this cat’s corneas, copper-related oxidation was speculated to have contributed to the cat’s unusually persistent ocular signs. Oxidative environments are favorable for viruses, especially feline herpesvirus-1 (FHV-1), 19 a common cause of chronic URIs with ocular signs. 20 This cat’s coincident return to copper homeostasis and resolution of persistent ocular signs suggests a shared mechanism needing further research. Meanwhile, the potential increased risk of corneal damage from FHV-1 in cats with PCH could be reduced using antioxidant supplements, FHV-1 vaccination and antiviral therapies.
Identifying copper aggregates on cytologic evaluation of liver aspirates from dogs is standard practice, yet evidence of copper aggregates in a liver aspirate from a cat has not been previously published. In modified Wright’s-stained smears of liver aspirates, copper aggregates are identifiable as blue-green refractile aggregations within the cytoplasm of hepatocytes. 21 Since copper can be misidentified as lipofuscin, a slightly darker cytoplasmic pigment associated with oxidative damage and aging, additional copper-specific staining, such as rhodanine, is recommended. Identification of copper aggregates in liver aspirates could support recommendations for more invasive and expensive diagnostics (eg, biopsies, genetic testing) in pursuit of a definitive PCH (or other) diagnosis. This case suggests that evaluation for copper could become part of the standard cytologic assessment of liver aspirates from cats.
PCH manifests in young adult cats.2,5,6,14,16 Before this cat’s ovariohysterectomy at 6 months old, all serum chemistry results were within reference limits; therefore, the PCH was undetectable with routine testing. If this cat had not presented 18 months later for chronic ocular discharge, the occasional vomiting might have been overlooked (likely attributed to longhair cat hairballs) until the cat presented with more significant clinical signs, which have been associated with hepatocellular carcinomas 2 and grave outcomes.5,14,16 In contrast, this cat’s PCH has been well managed for nearly 3 years and is the first reported case with this outcome. Since PCH (and other slowly developing metabolic diseases) may not be detectable on pre-neuter blood panels, routine serum chemistry testing in young adult cats could be used for early detection of these life-threatening diseases.
Conclusions
Serum chemistry testing could be used as an early screening tool for feline PCH, especially in young adult cats. Copper aggregates can be identified on cytologic evaluation of liver aspirates from cats and used to recommend further testing. D-penicillamine chelation therapy can effectively stabilize feline PCH to enable long-term management with zinc gluconate and a low-copper diet. For cats with PCH, ocular signs associated with chronic URIs are speculated to be unusually persistent due to copper-related oxidation of the cornea. Lastly, the heterozygous ’likely pathogenic’ ATP7B genotype identified in this case of feline PCH suggests that normal feline ATP7B alleles could be recessive to or incompletely/co- dominant with deleterious ones.
Footnotes
Acknowledgements
The authors would like to acknowledge Kathleen Kelly DVM PhD of the Center of Immunology at the Cornell University College of Veterinary Medicine (histopathological examination), Denise Kessler DVM and Elizabeth Berger DVM of Affectionately Cats and Sharon Center DVM of the Department of Clinical Sciences at the Cornell University College of Veterinary Medicine (clinical and editorial consultations) and Hajime Asada DVM PhD of the Department of Veterinary Internal Medicine at the University of Tokyo Graduate School of Agricultural and Life Sciences (genetic consultation). DNA extraction was performed at the Vermont Integrative Genomics Resource DNA Facility supported by University of Vermont Cancer Center, Lake Champlain Cancer Research Organization and the UVM Larner College of Medicine. The authors would also like to acknowledge the use of 99 Lives Genome Sequencing Consortium data and the data mining and analysis performed by Jess Hayward PhD of the Cornell Veterinary Biobank at the Cornell University College of Veterinary Medicine.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS Open Reports. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (verbal or written) for their use in the publication was obtained from the people involved.

