Abstract
Background:
Tolvaptan is the only approved treatment in Canada for slowing the progression of kidney enlargement and kidney function decline in people with autosomal dominant polycystic kidney disease (ADPKD). The Canadian Medical Assessment of Jinarc® Outcome Registry (C-MAJOR) registry evaluating long-term clinical outcomes of tolvaptan use was initiated in 2015, as per Health Canada requirements.
Objective:
To describe the study design and methodology of the C-MAJOR registry, and present baseline patient and disease characteristics, time to and reasons for treatment and study discontinuation, time to kidney replacement therapy (KRT), and the long-term safety with tolvaptan.
Design:
C-MAJOR is an ongoing, observational, non-interventional, multicenter registry study.
Setting:
Multicenter study at 24 polycystic kidney disease (PKD) clinics and centers across Canada.
Participants:
People with ADPKD treated with tolvaptan.
Measurements:
The primary study objective was to assess the impact of tolvaptan on health-related quality of life (HRQoL) using patient-reported outcome measures. Secondary study outcomes included time to KRT; mortality rate, and time to and cause of death; markers of kidney function; treatment adherence; time to and reasons for treatment/study discontinuation; and incidence of adverse events. This analysis focused on summarizing baseline demographic, clinical, treatment, and safety data collected between 2015 and December 2023.
Methods:
Outcomes were summarized descriptively or using the Kaplan-Meier method for time-to-event data.
Results:
Among 470 people with ADPKD enrolled and analyzed in C-MAJOR, the mean age at diagnosis and at tolvaptan initiation was 29.8 years and 44.4 years, respectively. Patients had a mean baseline total kidney volume of 2010.8 mL. Most patients (67%) had mild to moderate kidney dysfunction (chronic kidney disease [CKD] category G1-3a) and were at high risk of disease progression (Mayo class 1C-E) at baseline, with hypertension (85%) and hepatic cysts (72%) being common manifestations. Mean baseline patient-reported outcome scores were indicative of minimally impacted HRQoL, with mean ADPKD-Impact Scale scores of 1.6, 2.0, and 2.0 for the physical, fatigue, and emotional scales, respectively, and ADPKD Pain and Discomfort Scale scores of 1.8, 2.0, 1.4, and 2.1 for the Overall Pain and Discomfort, Dull Pain, Sharp Pain, and Discomfort severity scales, among others. Treatment and study discontinuation occurred in 29 and 27% of patients, respectively, with a mean time to discontinuation of 70.5 and 62.1 months. Adverse events were the primary reason for treatment (46%) and study (15%) discontinuations. During follow-up, 8% of patients progressed to KRT with a mean time to KRT of 82.5 months. Adverse events were reported in 93% of patients; most common adverse events (~25% each) were polyuria, nocturia and fatigue.
Limitations:
Recruitment from specialized PKD centers may limit generalizability, and patient-reported outcome analyses were limited to the subgroup enrolled prospectively (~40%) who completed assessments (<25%).
Conclusions:
This first report of 8-year follow-up data from the C-MAJOR registry confirms that most people with ADPKD treated with tolvaptan in Canada are in early CKD category of disease and at risk of rapid progression, with minimally impacted HRQoL at baseline.
Registration number:
NCT02925221.
Introduction
Autosomal dominant polycystic kidney disease (ADPKD) is the most common hereditary kidney disorder 1 and the fourth leading cause of end-stage kidney disease (ESKD). 2 Registry studies have estimated the prevalence of polycystic kidney disease (PKD) among those with kidney failure in 2020 ranged from 115 per million population (pmp) in the United States 3 to 137 pmp across Europe, 4 representing ~5% of kidney transplant cases. Autosomal dominant polycystic kidney disease is characterized by the progressive formation of multiple kidney cysts, which increase in size and quantity with age, leading to kidney enlargement, abnormal kidney structure, loss of function, and ultimately kidney failure. 1 Amongst the manifestations of ADPKD are hypertension, kidney pain, urinary tract infections, and nephrolithiasis. 1 Approximately 50% of people with ADPKD require kidney replacement therapy (KRT) in the fourth to sixth decade of life. 5 Pain, impaired mobility, discomfort, and fatigue significantly impact health-related quality of life (HRQoL) in ADPKD across chronic kidney disease (CKD) categories, with increased burden of disease in category G3b or higher.6-8
Tolvaptan, the selective vasopressin V2-receptor antagonist, reduced kidney growth and decline in estimated glomerular filtration (eGFR) relative to placebo in the phase 3 Tolvaptan (Phase 3 if TEMPO 3:4) Efficacy and Safety Study in Autosomal Dominant Polycystic Kidney Disease (ADPKD) (TEMPO 3:4; NCT00428948), 9 TEMPO 4:4 10 and Replicating Evidence of Preserved Renal Function: an Investigation of Tolvaptan Safety and Efficacy in ADPKD (REPRISE; NCT02160145) 11 randomized controlled trials of early-stage and later-stage CKD. Although ADPKD-specific instruments assessing HRQoL have been recently developed and validated,12,13 reports of the impact of disease on HRQoL among people with ADPKD, including those being treated with tolvaptan, remain scarce. 14
In 2015, Canada became the second country after Japan to approve tolvaptan to slow progression of kidney enlargement in people with ADPKD. Tolvaptan has also been approved in Canada to slow kidney function decline in this same patient population. Tolvaptan has further received approval for the treatment of patients with ADPKD in the European Union, United States and more than 40 other countries. The 2017 and updated 2018 Canadian Expert Consensus recommendations for the optimal management of ADPKD advocate for the use of tolvaptan for adult patients who fulfill enrollment criteria of the TEMPO or REPRISE studies, as well as those identified as having risk factors for rapid progression.15,16 The newly published 2025 Clinical Practice Guideline for the Evaluation, Management, and Treatment in ADPKD issued by Kidney Disease: Improving Global Outcomes (KDIGO), further recommends initiation of tolvaptan should be offered to adults with ADPKD with an eGFR of ≥25mL/min/1.73m2 who are at risk of rapid disease progression, as indicated by either Mayo imaging classification (MIC) class 1C-E or historical rate of eGFR decline of ≥3 ml/min/1.73m2 per year. 17
As per Health Canada mandates, the C-MAJOR (NCT02925221) patient registry evaluating the long-term clinical outcomes of tolvaptan use and the Hepatic Safety Monitoring and Distribution Program established to mitigate the risk of liver injury among patients treated with tolvaptan were both implemented in 2015 and have been running in parallel for 10 years. The present study aimed to describe the design and methodology of the C-MAJOR registry, as well as to present the baseline demographic and disease characteristics, reasons for treatment and study discontinuation, time to KRT, and long-term safety profile among adults with ADPKD initiating tolvaptan in Canada.
Methods
Study Design
C-MAJOR, originally mandated by Health Canada, is an ongoing, observational study of adults with ADPKD treated with tolvaptan in Canada, which was launched in 2015 and initially carried out across 24 study centers. All aspects of treatment and clinical management of participants followed routine clinical practice and all treatment decisions were at the sole discretion of the treating physician prior to, and independently of, participation in the study. In accordance with the non-interventional nature of the study, no assessment visits and no procedures were imposed for the exclusive purpose of the study, and missing a recommended assessment visit did not represent a protocol deviation. All study data were collected from regular patient assessments that are part of routine medical care. Assessments were performed at baseline, and follow-ups were conducted at six months (±three months) after the baseline visit and approximately every six months thereafter. Each study site received approval by an Institutional Review Board or Ethics Committee according to regional requirements.
Patients in C-MAJOR were enrolled under two scenarios. People with ADPKD newly initiated on tolvaptan by their treating physician at the enrollment visit were considered as Pure Prospective Follow-up patients. Patients already on-treatment with tolvaptan at the time of study enrolment were considered as Hybrid Follow-up patients. Study data from routine patient medical care assessments were collected retrospectively (chart review) from the date of patient consent to tolvaptan treatment initiation, and prospectively from the date of patient consent to the end of study/early termination. Patient-reported outcome (PRO) questionnaires were completed by Pure Prospective Follow-Up patients only, either through a secure internet site developed for the purposes of the study, or by use of a paper questionnaire. All patients in the registry provided written informed consent.
Study Population
Patients newly prescribed tolvaptan for the treatment of ADPKD (Pure Prospective), and patients who were on-treatment with tolvaptan at the time of enrolment (Hybrid Follow-up) were included in the C-MAJOR registry. Inclusion criteria were (1) ADPKD diagnosis; (2) aged ≥18 years at the time of tolvaptan initiation; (3) treating physician must have reached the decision to treat the patient with tolvaptan prior to and independently of soliciting the patient to participate in the study; and (4) patient or legal guardian consent. Exclusion criteria were (1) lack of consent; (2) any contraindications to the use of tolvaptan as specified in the Canadian Product Monograph; and (3) patient had any condition which, as per the judgment of the treating physician, prohibited them from participating in the study.
Overall C-MAJOR Study Objectives
The primary objective of the C-MAJOR registry was to describe the impact of tolvaptan on ADPKD-related burden of illness. The secondary objectives were to describe among people with ADPKD treated with tolvaptan: (1) time to KRT; (2) long-term mortality rate and causes; (3) demographics, ADPKD-related comorbidities, disease history, and baseline disease characteristics (including Canadian regional variations), (4) markers of kidney function; (5) patient adherence to treatment; (6) retention rate; and (7) the long-term safety profile of tolvaptan.
Assessments
Baseline assessments in C-MAJOR included demographics (age, sex, race, and Canadian region/province), tolvaptan treatment initiation characteristics (year of tolvaptan initiation, daily prescribed dose), general and ADPKD-related medical history (date of diagnosis, genetic testing, symptoms/signs that led to diagnosis, diagnosis confirmation, imaging modality, family history of ADPKD and early ESKD, hepatic cysts), and vital signs (height, weight, blood pressure). Disease characteristics included total kidney volume (TKV) and height-adjusted total kidney volume (ht-TKV), ultrasound, computed tomography (CT), or magnetic resonance imaging (MRI), kidney length, eGFR, CKD category, and MIC class. Autosomal dominant polycystic kidney disease–related manifestations, including hypertension, cysts, and pain were also assessed.
Patient-reported outcome measures assessed among Pure Prospective Follow-up patients in C-MAJOR included the Short-Form 12 Health Survey version 2 (SF-12v2), 18 ADPKD-Impact Scale (ADPKD-IS), 12 ADPKD-Pain and Discomfort Scale (ADPKD-PDS), 13 healthcare resource utilization, and Work Productivity and Activity Impairment (WPAI) questionnaire. 19
The SF-12v2 is a 12-item questionnaire that assesses HRQoL in the past month. It comprises two summary scores, the Physical Component Summary (PCS) and Mental Component Summary (MCS) score transformed to standardized
The ADPKD-IS is a validated tool for the measurement of the impact of ADPKD on patient’s HRQoL over the past two weeks. The ADPKD-IS comprises 18 questions capturing three domains: physical, emotional, and fatigue, and also contains 4 items outside these domains: guilt, sleep, size/shape of abdomen, and urinary urgency/frequency. 12 The ADPKD-PDS is a validated instrument assessing pain and its effect on patient’s life, based on a seven-day recall period. 13 It uses a 22-item questionnaire to measure the magnitude of three types of pain: acute pain, dull pain, and fullness and discomfort. The ADPKD-PDS scores are: Overall Pain and Discomfort Severity, Dull Pain Severity, Sharp Pain Severity, and Discomfort Severity, as well as Dull Pain Interference, Discomfort Interference, and Sharp Pain Interference. Both ADPKD-IS and ADPKD-PDS scores are calculated on a Likert scale with responses ranging from one (not at all or not difficult at all or not bothered at all) to five (completely or extremely difficult or extremely bothered). Based on prior studies,6,20 for both tools, a score <3 on all three subscales was considered as good HRQoL, whereas a score ≥3 on at least 2 subscales was equated with poor HRQoL. A score of ≥3 on only one subscale was considered as neutral. Healthcare resource utilization is assessed with a questionnaire evaluating institutional use (ambulance services, hospitalizations, emergency room, and intensive care unit visits), and healthcare consultations (general practitioners, kidney specialist physicians, and other specialists) between follow-up visits. The WPAI questionnaire evaluates the impact of ADPKD on work and productivity loss and impairment of daily activities. 19 Patient responses are scored from 0%-100%, with 0% indicating no impairment, and 100% indicating complete impairment.
Follow-up assessments in C-MAJOR reported the prescribed tolvaptan dose, vital signs, disease characteristics, ADPKD-related manifestations, adverse events, and reasons for study/treatment discontinuation. Pure Prospective Follow-Up patients were asked to provide consent to continue with PRO questionnaire completion.
Endpoints
Primary objective endpoints in C-MAJOR were assessments of SF-12v2, ADPKD-IS, ADPKD-PDS, WPAI, and healthcare resource utilization. Secondary observational outcomes were time to KRT, including dialysis and transplantation; mortality rate, and time to and cause of death; markers of kidney function; adherence to treatment; time to and reasons for treatment/study discontinuation; and incidence of adverse events. Treatment discontinuation in C-MAJOR did not necessitate study discontinuation, and patients may have continued to be followed-up as per routine medical care with data collected after discontinuing tolvaptan. Study discontinuation may have occurred either when a patient withdrew consent for study participation or when a patient who had previously withdrawn treatment consent (ie, treatment discontinuation), withdrew study participation consent at a later time. Patients discontinuing study participation may have continued tolvaptan treatment under standard medical care, with no further data collected after study discontinuation.
Statistical Analysis
The present analyses were conducted using the registry study patient population, comprising all patients enrolled in the study from 2015 until December 31, 2023, who received more than one dose of tolvaptan.
Baseline demographics and clinical characteristics were summarized using descriptive statistics. Statistical comparisons across Western (ie, Alberta and Saskatchewan), Ontario, Quebec, and Eastern (ie, New Brunswick and Nova Scotia) regions of Canada and across Canadian provinces (Supplemental Table S2) were performed based on analysis of variance for continuous variables and Chi-square test or Fisher exact test for categorical variables. Due to small enrollment numbers in certain provinces, only results stratified by Canadian region are retained in the main body of the report. The same statistical comparisons were conducted across MIC classes.
To account for the observational nature of the study, baseline PRO questionnaires were defined as the assessment completed nearest to the in-person baseline visit, within a three month window.
The number of patients discontinuing treatment or study or those progressing to KRT (dialysis and transplantation) during the course of the study were summarized with descriptive statistics. Reasons for patients discontinuing treatment and the study were summarized among those that discontinued treatment and the study, respectively. Kaplan-Meier estimates of the survival function were used to estimate the mean and median time to discontinuation, and KRT.
Adverse events were classified according to the medical dictionary for regulatory activities (MedDRA) dictionary of terms (Version 27.0) and summarized using the total number of adverse events, the total number and percentage of patients who experienced an adverse event overall, and the number and percent of patients who experienced an adverse event within each system organ class (SOC) and preferred terms within individual SOCs. Patients experiencing the same adverse event multiple times were counted once for the corresponding preferred term; if a patient experienced multiple adverse events within the same SOC, that patient was counted only once for that SOC.
All analyses were performed using the SAS Version 9.4 software package.
Results
Between 2015 and December 31, 2023, a total of 487 patients from 24 sites were enrolled in C-MAJOR of whom 470 patients were included in the registry study patient population. A total of 185 patients were enrolled as part of the Pure Prospective Follow-up population and 285 were part of the Hybrid Follow-up population.
Baseline Characteristics
Demographics
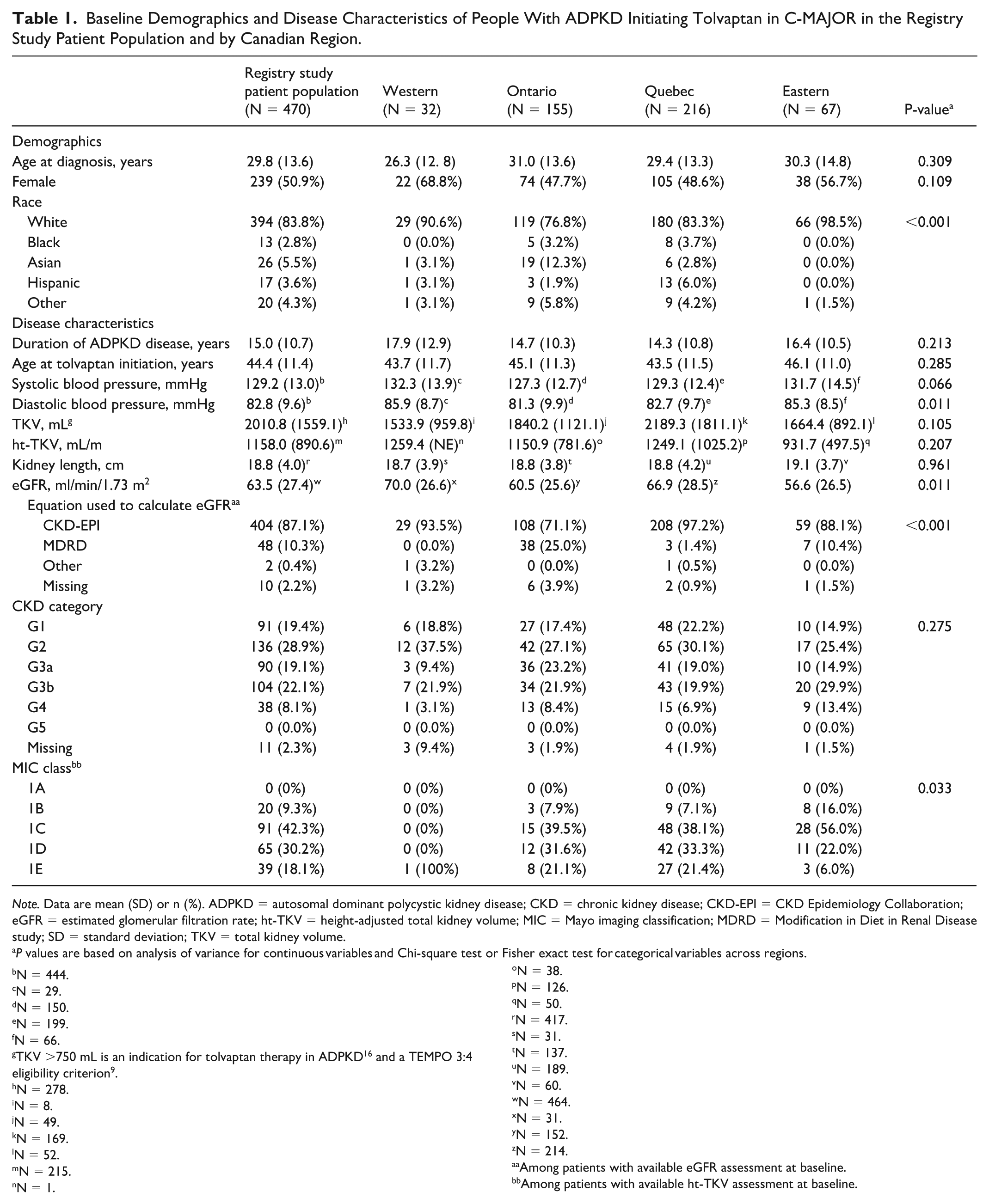
In the registry study patient population, mean (SD) patient age at ADPKD diagnosis was 29.8 (13.6) years, 51% of patients were female, and majority were White (84%; Table 1). Most patients were from Quebec (46%) and Ontario (33%), with 14% and 7% of patients from Eastern and Western Canada, respectively. Baseline demographics across Canadian regions were balanced (Table 1).
Baseline Demographics and Disease Characteristics of People With ADPKD Initiating Tolvaptan in C-MAJOR in the Registry Study Patient Population and by Canadian Region.
N = 444.
N = 29.
N = 150.
N = 199.
N = 66.
TKV >750 mL is an indication for tolvaptan therapy in ADPKD 16 and a TEMPO 3:4 eligibility criterion 9 .
N = 278.
N = 8.
N = 49.
N = 169.
N = 52.
N = 215.
N = 1.
N = 38.
N = 126.
N = 50.
N = 417.
N = 31.
N = 137.
N = 189.
N = 60.
N = 464.
N = 31.
N = 152.
N = 214.
Among patients with available eGFR assessment at baseline.
Among patients with available ht-TKV assessment at baseline.
Clinical characteristics
In the registry study patient population, the mean (SD) duration of ADPKD was 15 (10.7) years and the mean (SD) age at tolvaptan initiation was 44.4 (11.4) years (Table 1 and Supplemental Tables S2 and S3). The majority of patients (72%) initiated treatment with tolvaptan between 2016-2019 (Supplemental Table S2). Nearly all patients (99%) were initially prescribed a tolvaptan daily dose of 60 mg, as per product monograph, 21 with the exception of three patients whose initial dose was 90 mg, and one patient each with an initial dose of 45 mg and 30 mg (Supplemental Table S2). The mean (SD) TKV was 2010.8 (1559.1) mL (Table 1), indicative of considerably enlarged kidney size, in comparison with the 750 mL TKV eligibility criterion in the TEMPO 3:4 study. 9 Mean (SD) ht-TKV was 1158.0 (890.6) mL/m and the mean (SD) kidney length was 18.8 (4.0) cm. The mean (SD) eGFR was 63.5 (27.4) ml/min/1.73 m2, and the most commonly used equation to calculate the eGFR was the CKD Epidemiology Collaboration (CKD-EPI; 87%).
During the study period, among patients with available TKV measurement (N = 278), MRI (72%) was the predominant method for assessing TKV, both prior to (before 2017; 71%) and following (2018 onwards; 73%) the evolution of practice, marked by the 2017 and the updated 2018 Canadian Consensus recommendations (Figure 1A).15,16 When comparing the period before 2017 with that of 2018 onwards, the use of ultrasound declined from 15 to 7%, while the use of CT increased from 14 to 17% and other methods of assessing TKV increased from 0 to 3%.

Method used to measure TKV over time in the registry study patient population for the overall study period (A), and across Canadian regions for the overall study period (B), prior to 2017 (C), and 2018 onwards (D).
Most patients (67%) had early-stage CKD (CKD G1-3a) at tolvaptan initiation, and approximately one-third had later-stage disease (CKD G3b-5; Table 1). For patients with available ht-TKV (N = 215), 90% were classified as at high risk of disease progression (class 1C-E) using the MIC prognostication tool (Table 1 and Figure 2). Baseline patient and disease characteristics by MIC classes are detailed in Supplemental Table S4. Overall, the most common ADPKD clinical manifestations were hypertension (85%), hepatic cysts (72%), kidney pain (24%), gross hematuria (14%), nephrolithiasis (14%), and other cysts (14%; Figure 3).

Baseline MIC class by CKD category at tolvaptan initiation in the registry study patient population with available ht-TKV.

Most common (>10%) ADPKD clinical manifestations at tolvaptan initiation in the registry study patient population.
Mean TKV and ht-TKV levels were similar across Canadian regions (Table 1), although imaging modalities used to determine TKV differed significantly (Figure 1B). Magnetic resonance imaging was the predominant method of TKV measurement in Quebec (83%), Ontario (63%), and Eastern Canada (52%), whereas ultrasound was most common in Western Canada (88%), however the sample size was small; use of ultrasound in Eastern Canada reached 38% (Figure 1B). Similar patterns of imaging modality use were observed prior to (before 2017; Figure 1C) and following (2018 onwards; Figure 1D) the release of the Canadian Consensus recommendations.15,16 The distribution of patients with CKD G1-5 across Canadian regions was consistent, and similar to that in the registry study patient population (Table 1). Significant differences were seen in the mean (SD) eGFR, ranging from 56.6 (26.5) ml/min/1.73 m2 in the Eastern region to 70.0 (26.6) ml/min/1.73 m2 in the Western region (
PRO measures
Among Pure Prospective Follow-up patients who completed the SF-12v2 questionnaire, mean (SD) PCS and MCS baseline scores were 51.4 (8.5) and 48.6 (9.3), respectively (Table 2). Mean (SD) ADPKD-IS baseline scores were 1.6 (0.8), 2.0 (1.0), and 2.0 (1.0) for the physical, fatigue, and emotional scales, respectively. The mean (SD) Overall Pain and Discomfort Severity score assessed with the ADPKD-PDS, was 1.8 (0.8), with mean (SD) scores for Dull Pain Severity, Sharp Pain Severity, and Discomfort Severity of 2.0 (1.0), 1.3 (0.8), and 2.1 (1.1), respectively. Mean (SD) scores of 1.5 (0.9) for Dull Pain Interference, 1.6 (0.9) for Discomfort Interference, and 1.4 (0.8) for Sharp Pain Interference scores were reported. Among patients who completed the WPAI questionnaire at baseline, mean (SD) scores were 1.4 (4.8) for absenteeism, 15.3 (22.2) for presenteeism, 15.1 (20.9) for work productivity loss, and 16.9 (24.1) for activity impairment.
Baseline PRO Measures Among Pure Prospective Follow-up Patients.
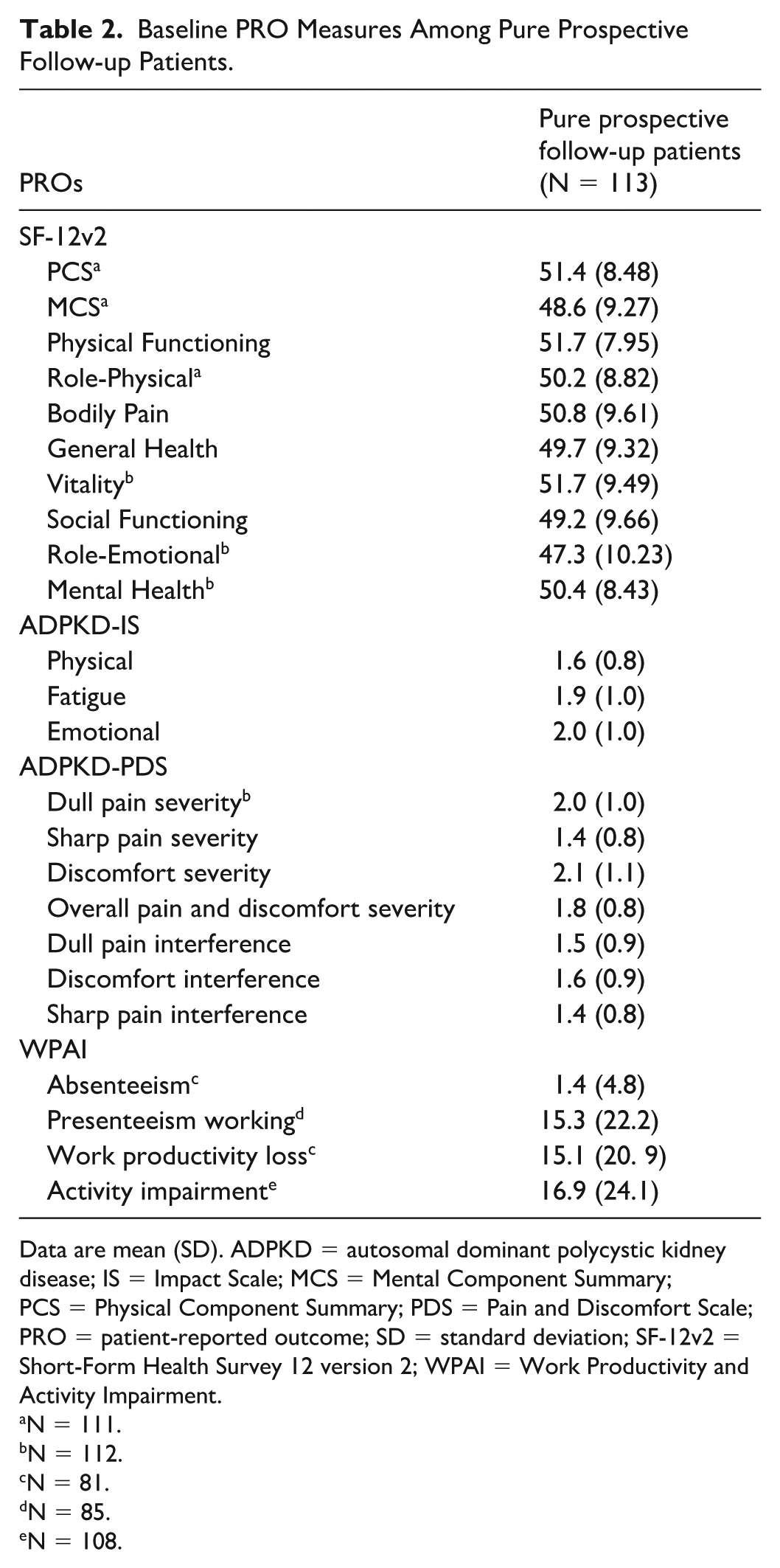
Data are mean (SD). ADPKD = autosomal dominant polycystic kidney disease; IS = Impact Scale; MCS = Mental Component Summary; PCS = Physical Component Summary; PDS = Pain and Discomfort Scale; PRO = patient-reported outcome; SD = standard deviation; SF-12v2 = Short-Form Health Survey 12 version 2; WPAI = Work Productivity and Activity Impairment.
N = 111.
N = 112.
N = 81.
N = 85.
N = 108.
Discontinuations
Tolvaptan discontinuations
A total of 135 (29%) patients in the registry study patient population discontinued tolvaptan (Table 3). Patients who discontinued tolvaptan could remain enrolled in the study with follow-up data collected as per routine care. The mean (standard error [SE]) and median (95% confidence interval [CI]) time to tolvaptan discontinuation were 70.5 (1.7) months and 93.9 (85.4-not estimable) months, respectively. The primary reasons leading to treatment discontinuation were adverse events (46%) and lack of treatment efficacy (12%).
Treatment and Study Discontinuations in the Registry Study Patient Population.

Among patients in the registry study patient population (N = 470).
Among patients who discontinued treatment (N = 135).
Among patients who discontinued from the study (N = 126).
Study discontinuations
A total of 126 (27%) patients in the registry study patient population discontinued from the study (Table 3). Patients who discontinued study participation could continue tolvaptan treatment under standard medical care, with no further data collected. The mean (SE) time to study discontinuation was 62.1 (1.2) months; median time to study discontinuation was not estimable due to lack of sufficient events. Adverse events were the primary reason leading to study discontinuation (15%).
Kidney replacement therapy
During follow-up, 36 (7.7%) patients required KRT. Mean (SE) time from tolvaptan initiation to KRT was 82.5 (0.8) months (Supplemental Figure S1A). Thirty (30) events of dialysis were reported among 30 (6.4%) patients with a mean (SE) time from tolvaptan initiation to dialysis of 83.2 (0.8) months (Supplemental Figure S1B). There were nine events of kidney transplantation among nine (1.9%) patients, with a mean (SE) time from tolvaptan initiation to transplantation of 78.0 (0.3) months (Supplemental Figure S1C). Three (3; 0.6%) patients required both dialysis and kidney transplantation during the course of the study, with two patients requiring transplantation after dialysis and one patient requiring dialysis subsequent to transplantation.
Adverse events
Adverse events were reported for 438 (93%) patients in the registry study patient population (Figure 4). The most commonly reported adverse events included polyuria, nocturia and fatigue, each affecting approximately a quarter of patients. In the registry study patient population, 30 (6.4%) patients experienced adverse events leading to study discontinuation. These adverse events most frequently involved general disorders and administration site conditions (nine patients; 1.9%), and renal and urinary disorders (eight patients; 1.7%). Adverse events resulting in treatment discontinuation occurred in 91 (19.4%) patients and primarily involved renal and urinary disorders (33 patients; 7.0%) including polyuria (11 patients; 2.3%), and general disorders and administration site conditions (25 patients; 5.3%), such as drug ineffectiveness (11 patients; 2.3%).

Adverse events in >10% of patients in the registry study patient population.
Discussion
To assess the long-term impact of tolvaptan use in ADPKD since its approval in Canada in 2015, patients initiating tolvaptan were offered participation in the Health Canada-mandated, product-specific Canadian tolvaptan patient outcomes registry. This report is the first to describe real-world demographic and clinical characteristics of the C-MAJOR registry participants initiating tolvaptan for the treatment of ADPKD in Canada.
Patients initiating tolvaptan in Canada generally had early-stage CKD (CKD G1-3a) and were identified as ADPKD fast progressors, defined as MIC class 1C-E as per the 2017 and 2018 Canadian Expert Consensus recommendations15,16 and the 2015 KDIGO clinical guidelines. 3 According to the Canadian Expert Consensus recommendations, treatment with tolvaptan should be considered for patients with MIC class 1D and 1E, as well as 1C patients who have other risk factors. 16 The 2025 KDIGO clinical guidelines further recommend treatment initiation with tolvaptan should be offered to adults with an elevated eGFR rate (≥25mL/min/1.73m2), who are at risk for rapidly progressive disease. 3 Use of tolvaptan therapy in patients with early-stage disease adheres to expert opinion advocating for early treatment when kidney parenchyma is relatively preserved. 22 Nearly all patients in C-MAJOR were prescribed an initial dose of 60 mg, in alignment with approved dosing, 21 similar to that reported in the Australian controlled monitoring and distribution program IMADJIN, where over half of patients remained on tolvaptan 60 mg. 23 According to C-MAJOR, Canadian practice employed MRI as the preferred method for TKV measurement since 2015.
Patient-reported outcomes measured at baseline indicated that the MCS score was minimally affected by ADPKD, specifically in the role-emotional domain, though other SF-12v2 domain scores were generally at or above the established norm. Levels of physical and emotional functioning, as well as fatigue and pain indicate these domains were not impacted by ADPKD at baseline in this study. Low levels of work productivity loss and activity impairment were observed. These findings are consistent with previous studies of patients in early-stage ADPKD, showing overall HRQoL is highest in patients with CKD G1-3a.20,24
The tolvaptan discontinuation rate of 29% found in the present analysis was similar to that reported in the TEMPO 3:4 study of 23%, 9 as well as the 24% observed in the IMADJIN program. 23 Rates and types of adverse events reported in this study were comparable to those in TEMPO 3:4, 9 with polyuria and nocturia (both 24%) reported most frequently, as may be expected considering the tolvaptan mechanism of action. 25 Fatigue was reported for 23% of patients, consistent with findings of fatigue/tiredness being one of the most burdensome symptoms of ADPKD among patients with early-stage disease.26,27
The study has several limitations. Patients were recruited from a subset of participating PKD clinics and centers in Canada, which may limit the generalizability of findings to all PKD or kidney medicine clinics in Canada and in other jurisdictions. The participating sites may be biased to those centers with an expertise in PKD management. Furthermore, approximately 40% of the cohort was enrolled prospectively, and fewer than 25% completed PRO assessments, limiting the PRO data analysis to this subgroup. Geographic representation was also limited, with few centers from Western and Eastern Canada participating in the registry, suggesting that practices such as the reliance on ultrasound as the primary imaging modality for kidney size measurement may not fully reflect practices across these regions. Due to the observational design of the study, no placebo-controlled comparator was included, which may limit the ability to attribute observed effects solely to the intervention.
Future studies will evaluate the evolution of ADPKD and its treatment in Canada, persistence of tolvaptan dosage and titration by region, changes in PROs over time with tolvaptan, aquaretic effects of tolvaptan by age and CKD category, as well as eGFR rates with tolvaptan. Although this first report included approximately 18% of all people with ADPKD being treated with tolvaptan nationwide from 2015 to 2023, the C-MAJOR Canadian registry allows for a detailed characterization of this patient population.
Conclusions
The C-MAJOR registry evaluates baseline demographic and clinical characteristics, as well as HRQoL and disease progression among people with ADPKD initiating tolvaptan in the real-world Canadian setting. Present findings confirm that majority of patients in Canada treated with tolvaptan are at risk of rapid ADPKD progression (MIC class 1C-E) and in early stages (CKD G1-3a) of the disease, with minimally impacted HRQoL at the time of treatment initiation.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581251404128 – Supplemental material for Eight Years of Canadian Real-World Assessment of Tolvaptan in Patients With Autosomal Dominant Polycystic Kidney Disease: The C-MAJOR Registry
Supplemental material, sj-docx-1-cjk-10.1177_20543581251404128 for Eight Years of Canadian Real-World Assessment of Tolvaptan in Patients With Autosomal Dominant Polycystic Kidney Disease: The C-MAJOR Registry by Philip McFarlane, Daniel G. Bichet, Ahsan Alam, Suzy Bubolic and Annick Laplante in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors thank the participants, investigators (Supplemental Table S5) and study site personnel across Canada for contributing to this research. The authors would also like to thank Dr Patrick S. Parfrey, Memorial University of Newfoundland, St. John’s, NL, for acting as a scientific advisor (2015-2022), and Dorothee Oberdhan, Director Patient Centric Outcomes Global Value Real World Evidence, Otsuka Pharmaceuticals Development and Commercialization, Inc for substantive review of sections of this manuscript pertaining to patient-reported outcome measures. Data analysis and writing services for this manuscript were provided by JSS Medical Research (Montreal, Quebec, Canada) funded by Otsuka Canada Pharmaceutical Inc.
Ethical Considerations
The study is performed following Good Clinical Practice and conducted in compliance with the protocol and all other applicable local laws and regulatory requirements. The ethical review board Canadian SHIELD evaluated the ethical, scientific, and medical appropriateness of the study (approval number 15-12-002). All patients in the registry provided written informed consent.
Consent to Participate
Patients to be initiated on tolvaptan were offered participation in the manufacturer and product-specific Canadian patient outcomes registry. Patients who expressed a willingness to consider participation were given a consent form to review. If they had any questions related to participation in the registry, these were answered by the principal investigator or designee. Patients were required to sign the voluntary consent form. Patients who consented to participate in the registry received a signed and dated copy of the consent form. Informed consent was obtained before any study data were collected.
Consent for Publication
All results presented are in aggregate form and no personal identifiable information was used for this study.
Author Contributions
Conceptualization/Study design: PM, DGB, AL; Data curation: PM, DGB, AA, AL; Analysis and interpretation of data: all authors; Writing, original draft: all authors; Writing, review and editing: all authors. All authors have read and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Otsuka Canada Pharmaceutical Inc.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PM: advisor, investigator and overall study supervisor, and speaking bureau for Otsuka Pharmaceutical.
DGB: consultant, grants and speaker bureau for Otsuka Pharmaceutical; consultant for Lupin Pharma.
AA: consultant, grants, investigator and speaker bureau for Otsuka Pharmaceutical; grant from Paladin Pharma; consultant for Lupin Pharma; investigator for Vertex Pharmaceuticals.
SB: employee of Otsuka Pharmaceutical receiving salary.
AL: employee of Otsuka Pharmaceutical receiving salary.
Dorothee Oberdhan: employee of Otsuka Pharmaceutical Development & Commercialization receiving salary.
Data Availability Statement
Anonymized data generated and/or analyzed as part of the C-MAJOR registry may be made available upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
