Abstract
We report a case of biopsy-proven renal cortical necrosis in a 42-year-old man after bioprosthetic aortic valve replacement despite prophylactic anticoagulation. He had antiphospholipid syndrome with chronic thrombotic microangiopathy and triple-positive antibodies, leading to renal cortical necrosis manifested as severe non-reversible dialysis-dependent acute kidney injury. We identified 13 previously published reports of renal cortical necrosis due to antiphospholipid syndrome: 3 occurred postpartum, 2 after kidney transplantation, and 1 following salpingectomy for ectopic pregnancy.
Introduction
Renal cortical necrosis (RCN) is a rare etiology of severe acute kidney injury (AKI). 1 The leading cause of renal cortical necrosis in developing countries is obstetric complications. 2 We report a case of severe renal cortical necrosis in a patient with antiphospholipid syndrome (APS) following bioprosthetic aortic valve replacement. We also reviewed reported cases of diffuse renal cortical necrosis secondary to antiphospholipid syndrome in the literature.
Case Report
Presenting Concerns
A 42-year-old man with a history of antiphospholipid syndrome (APS), hypertension, epilepsy, thrombocytopenia, and severe aortic valve stenosis underwent a bioprosthetic aortic valve replacement. He also had chronic kidney disease (CKD) stage G3bA2 secondary to biopsy-proven thrombotic microangiopathy (TMA). The patient had been on aspirin, ramipril, amlodipine, lacosamide, and clobazam prior to admission. Immediately postoperatively, he experienced transient hypotension, which resolved with intravenous fluids. On postoperative day (POD) 1, he was noted to have developed acute kidney injury (AKI).
With regard to his chronic kidney disease course, his serum creatinine had slowly progressed from 140 to 220 µmol/L over the preceding decade, with proteinuria ranging between 1 and 3 g/day based on 24-hour urine collections. At age 40, he underwent a diagnostic kidney biopsy to evaluate the cause of progressive proteinuric chronic kidney disease (CKD). The biopsy demonstrated features of thrombotic microangiopathy (TMA) with both acute and chronic changes (Figure 1A and B). On light microscopy, the renal cortex contained 15 glomeruli, five of which were globally sclerosed. The remaining glomeruli showed moderate to marked mesangial matrix expansion, segmental mesangial hypercellularity, mesangiolysis, and perihilar segmental scarring. Glomerular capillaries remained patent without endocapillary hypercellularity; however, there was diffuse glomerular endothelial cell swelling with segmental double contouring, and glomerular capillary wall thickening. Some glomeruli appeared retracted with periglomerular fibrosis. There were moderate interstitial fibrosis and tubular atrophy, mild interstitial inflammation, severe hyaline arteriosclerosis, and moderate-to-severe arteriosclerosis with mucoid intimal changes. A possible fibrin thrombus was also noted. Immunofluorescence revealed global sclerosis in 34 out of 40 glomeruli, with nonsignificant staining for IgG, IgA, C3, C1q, kappa, and lambda. On electron microscopy, three of nine glomeruli were globally sclerosed. The mesangium was expanded by frequent hyaline electron densities, without immune-type deposits or tubuloreticular inclusions. Subendothelial widening, cellular interposition, and segmental endothelial cell swelling and hypertrophy were observed, consistent with ongoing acute and chronic endothelial injury. The glomerular basement membranes appeared thickened and segmentally duplicated, with segmental podocyte foot process effacement.
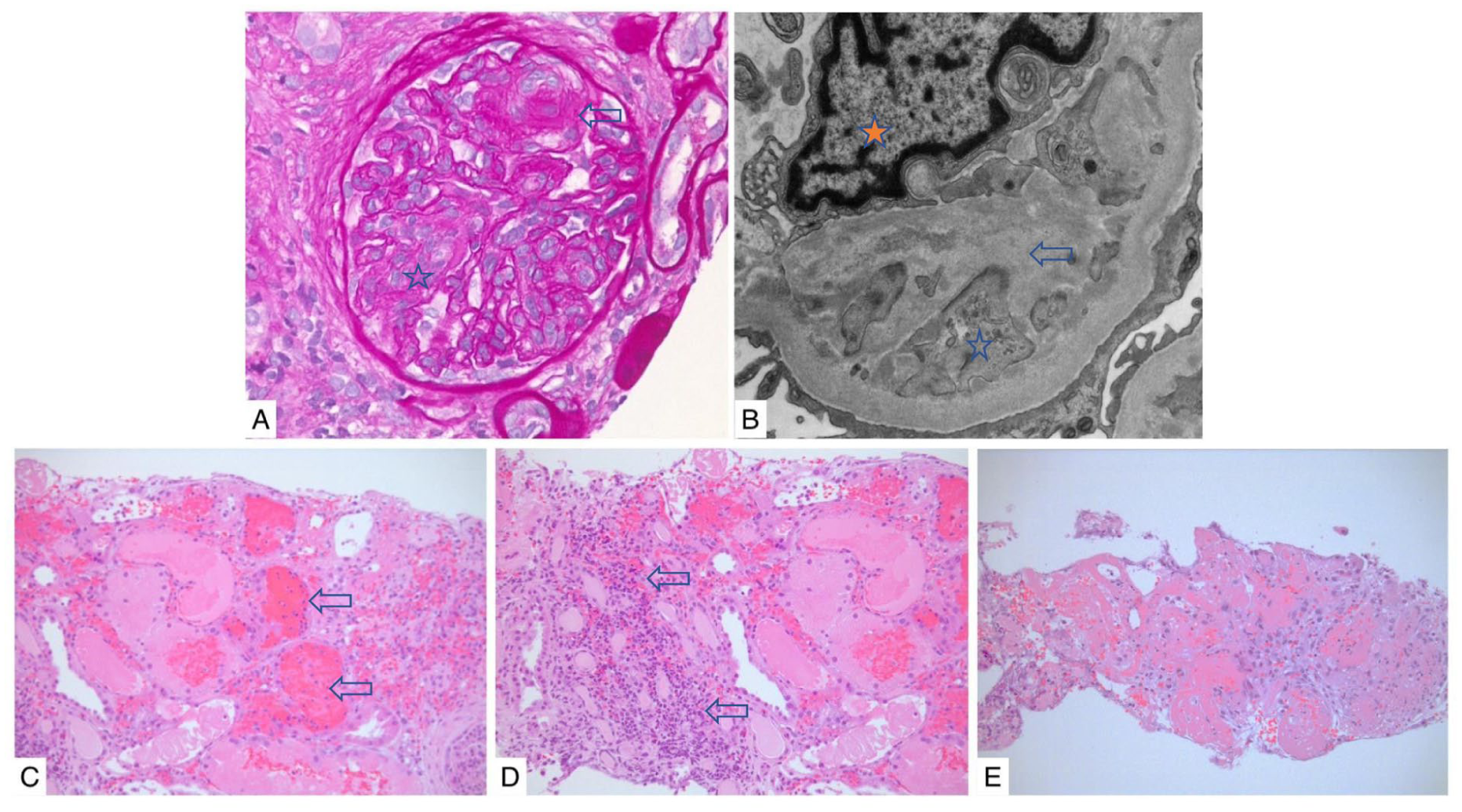
Kidney biopsy findings at initial diagnosis of thrombotic microangiopathy (A, B) and postoperative renal cortical necrosis (C, D, E). Initial kidney biopsy reflecting thrombotic microangiopathy: (A) Glomerulus showing features of acute thrombotic microangiopathy: dilated capillary loops, mesangiolysis (white star) and fibrin thrombus (white arrow). In addition, periglomerular fibrosis due to chronic hypoperfusion. PAS 40×. (B) Ultrastructural features of chronic microangiopathy: subendothelial widening by fluffy proteinaceous material separating the endothelial cell from the GBM (white star) and forming double contours (white arrow). Endothelial cell hypertrophy (orange star) (TEM 20000×). Postoperative kidney biopsy: (C) Extensive necrosis (coagulative) with interstitial hemorrhage (white arrows). (H&E 20×). (D) Coagulative necrosis, interstitial inflammation (white arrows) in areas of reperfusion (H&E, 20×). (E) Extensive cortical necrosis (H&E 4×).
Further work-up to determine the underlying cause of thrombotic microangiopathy revealed triple-positive antiphospholipid antibodies: anticardiolipin IgG >160 GPL-U/mL (reference <20), anticardiolipin IgM 32 MPL-U/mL (reference <20), β2-glycoprotein 1 (β2GP1) IgG >160 U/mL (reference <20), β2GP1 IgM 98 U/mL (reference <20), positive Dilute Russell’s Viper Venom Time (DRVVT) test, and positive lupus sensitive PTT assay. Hemoglobin was 138 g/L, and platelet count was mildly reduced at 139 ×109/L. Lactate dehydrogenase (LDH) was elevated at 5.5 ukat/L (reference 2-3.6), while INR and PTT were within normal limits. Total bilirubin was 6 µmol/L (reference < 22), and haptoglobin was 0.4 g/L (reference 0.3-2). Complement levels were suppressed, with C3 at 0.67 g/L (reference 0.90-1.80) and C4 at 0.08 g/L (reference 0.10-0.40). Autoimmune serologies, including antinuclear antibodies (ANA), anti-double-stranded DNA, and extractable nuclear antigen (ENA) antibodies, were negative. Genetic testing for atypical hemolytic uremic syndrome (aHUS) did not reveal any known pathogenic mutations in C3, CD46, CFB, CFH, CFHR5, CFI, or THBD.
Given the absence of clear evidence supporting anticoagulation in antiphospholipid syndrome with chronic thrombotic microangiopathy without arterial or venous thrombotic event, he was treated with aspirin only. With better control of his blood pressure, his proteinuria decreased to below 0.5 g per day and his serum creatinine stabilized around 195 µmol/L.
Clinical Findings
His serum creatinine began rising on postoperative day one despite hemodynamic stability. His serum creatinine immediately prior to operation was stable around baseline of 195 µmol/L. By day seven, serum creatinine had increased to 485 µmol/L, accompanied by bilateral flank pain, gross hematuria, and livedo reticularis. Blood tests showed low platelets 50 × 109/L (142 × 109/L preoperative), low hemoglobin 80 g/L (145 g/L preoperative), elevated LDH 8.3 ukat/L, and high-normal total serum bilirubin 16 µmol/L, with no schistocytes on blood smear. By day 10, his serum creatinine reached 850 µmol/L.
Diagnostic Focus and Assessment
Transthoracic echocardiogram demonstrated normal left ventricular ejection fraction (55%-60%) with a well-seated bioprosthetic valve and no intracardiac thrombus. Kidney ultrasound showed echogenic kidneys without hydronephrosis. Renal magnetic resonance angiography revealed normal arteries and veins.
A repeat kidney biopsy showed extensive coagulative cortical necrosis, severe interstitial fibrosis and tubular atrophy, severe arteriolar hyalinosis, and arterial sclerosis (Figure 1C and E).
Therapeutic Focus and Assessment
Postoperatively, he received prophylactic low molecular weight heparin. In light of the extensive cortical necrosis, escalation to full-dose anticoagulation was not pursued.
Follow-up and Outcomes
Platelet counts gradually improved and normalized 1 month postoperatively, reaching 163 × 109/L. Hemoglobin levels improved more slowly, eventually reaching 110 g/L at three months post-op. Despite supportive measures, the patient remained dialysis-dependent six months postoperatively.
Discussion
In this report, we describe a case of postoperative severe bilateral renal cortical necrosis secondary to antiphospholipid syndrome. Antiphospholipid syndrome is an autoimmune thrombophilic disease that is diagnosed based on the development of venous, arterial, or microvascular thrombosis and/or obstetrical morbidity in the presence of persistent circulating antiphospholipid antibodies (lupus anticoagulant, anticardiolipin antibody, and/or B2 glycoprotein I antibody). 3 The presence of antiphospholipid antibodies is not sufficient to provoke vascular thrombosis. Another precipitating factor like surgery, pregnancy, or infection usually represents the second hit at the time of thrombosis. 4
Antiphospholipid syndrome can affect the kidneys targeting renal vessels of different sizes. This usually manifests as renal vein thrombosis, renal arterial thrombosis or stenosis, renal infarction, and renal small vessel disease also known as antiphospholipid syndrome nephropathy. 3 Renal cortical necrosis is a rare manifestation of antiphospholipid syndrome. We conducted a comprehensive review of the literature by searching PubMed and Google Scholar using the terms “antiphospholipid syndrome” and “renal cortical necrosis.” Only English-language, peer-reviewed publications were included in the review. In addition to our report, there are 13 other reports in the literature of diffuse renal cortical necrosis secondary to antiphospholipid syndrome.
Table 1 summarizes the 14 reports. While cortical necrosis could be attributed to perioperative hypotension, the hypotensive episode was mild, transient, and rapidly responsive to intravenous fluid resuscitation, without requiring vasopressor support. Furthermore, the patient’s hematologic profile showed features suggestive of active thrombotic microangiopathy (TMA), including worsening anemia, thrombocytopenia, and elevated levels of lactate dehydrogenase (LDH) and total bilirubin. Although schistocytes were not observed on peripheral smear, which limits a definitive diagnosis of active hematologic TMA, the overall clinical and laboratory picture remains concerning for ongoing microvascular injury.
Reported Cases of Renal Cortical Necrosis Secondary to Antiphospholipid Syndrome.
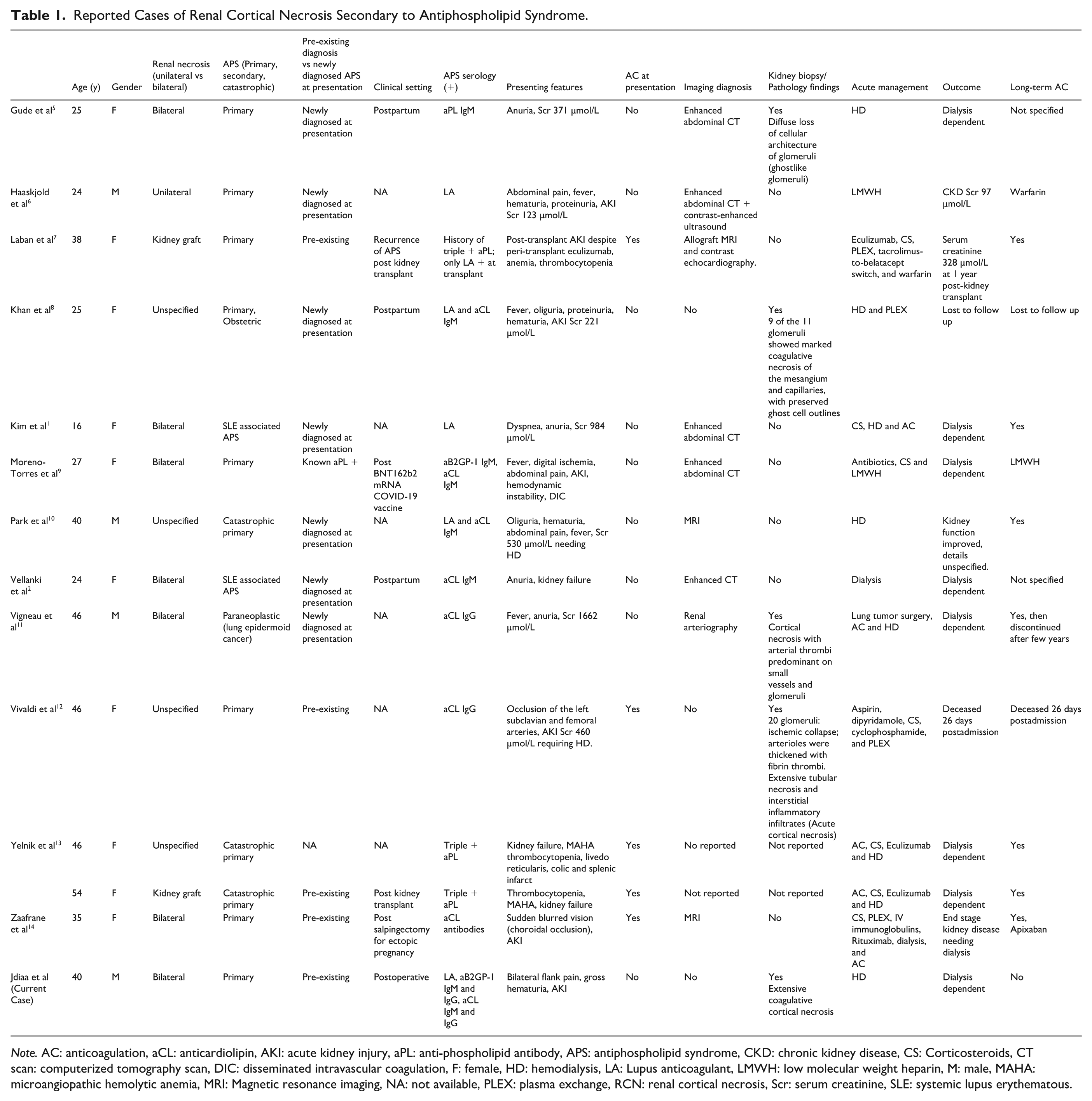
Note. AC: anticoagulation, aCL: anticardiolipin, AKI: acute kidney injury, aPL: anti-phospholipid antibody, APS: antiphospholipid syndrome, CKD: chronic kidney disease, CS: Corticosteroids, CT scan: computerized tomography scan, DIC: disseminated intravascular coagulation, F: female, HD: hemodialysis, LA: Lupus anticoagulant, LMWH: low molecular weight heparin, M: male, MAHA: microangiopathic hemolytic anemia, MRI: Magnetic resonance imaging, NA: not available, PLEX: plasma exchange, RCN: renal cortical necrosis, Scr: serum creatinine, SLE: systemic lupus erythematous.
There is no clear guidance regarding the optimal treatment for antiphospholipid syndrome induced chronic renal limited thrombotic microangiopathy and antiphospholipid syndrome nephropathy. 3 Postoperatively, patients with antiphospholipid syndrome are considered high risk for both thrombosis and bleeding which can occur simultaneously. There is a knowledge gap in the best approach to manage antiphospholipid syndrome perioperatively. Meroni et al described successful eculizumab prophylaxis therapy prior to arterial bypass surgery in a patient with antiphospholipid syndrome and recurrent arterial thrombosis. Meroni et al 15 also demonstrated the immunofluorescence positivity for b2GPI, b2GPI co-localizing IgG, C3, C4, and C1q on the endothelium of occluded femoral artery proposing classical pathway complement activation in the pathophysiology of antiphospholipid syndrome induced thrombotic event. Also, eculizumab has been used to prevent vascular thrombotic events and thrombotic microangiopathy in patients with catastrophic antiphospholipid antibody syndrome undergoing kidney transplantation. 3 Since surgical intervention can be the second hit for thrombotic event in patients with high-risk antiphospholipid antibodies, clear guidance is needed for perioperative management of this population with and without prior large or small vessel thrombotic event.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Informed Consent
Patient provided informed consent to publish this case.
