Abstract
Background
While the prevalence of antiphospholipid antibodies (aPL) in venous and arterial thrombotic events had already been estimated by previous studies, the prevalence of aPL in subjects with Thrombotic Microangiopathy (TMA) is still not fully elucidated. Thus, we conducted a systematic review to estimate the frequency of aPL in subjects with biopsy-proven renal TMA.
Methods
We conducted in the PubMed database a search for English-language studies investigating the presence of aPL in subjects with biopsy-proven renal TMA from January 1985 to December 2022. Keywords used in the search included: ‘antiphospholipid syndrome’, ‘antiphospholipid antibodies’ and ‘thrombotic microangiopathy’. Cohorts of HUS patients were excluded due to the risk of over-estimating the prevalence of aPL in these populations. The median frequency for positive aPL including anticardiolipin antibodies (aCL), antibodies against β2-glycoprotein-I (anti-β2GPI) and lupus anticoagulant (LA) was then calculated.
Results
522 articles were identified through the literature search. Six studies, assessing the prevalence of aPL in 211 subjects with renal TMA, were retrieved. The overall aPL prevalence was estimated as 24.4% (range 22–56). The estimated prevalence of aCL (IgG/IgM), anti-β2GPI, (IgG/IgM) and LA was 4.0% (range 3–27), 4.0% (range 3–16) and 18.9% (range 13–25), respectively. APS was diagnosed in 16.3% (range 11–29) of the patients. Of note, a high level of heterogeneity was observed when comparing the reported aPL profiles for each study.
Conclusions
This comprehensive systematic analysis of studies investigating the prevalence of aPL in renal TMA showed that, despite the high heterogeneity of the included studies, aPL are present in about one case out of four renal-TMA cases.
Keywords
Introduction
Thrombotic Microangiopathy (TMA) encompasses a spectrum of complex pathological processes which result in thrombosis of capillaries and arterioles. Clinically, TMA typically manifests as a concurrence of Microangiopathic Haemolytic Anaemia (MAHA), Thrombocytopaenia and organ dysfunction.1–4 TMA can occur in patients with antiphospholipid antibodies (aPL), with or without the full-blown antiphospholipid syndrome (APS).5–9 The prevalence of aPL in various clinical scenarios had already been estimated by previous studies and was found to be strongly associated with cardiovascular events (CVEs), thrombotic events, peripheral artery disease (PAD) and pregnancy morbidity.10–14
While the available data strongly suggest an association between aPL and thrombosis outside of the setting of APS, the frequency of aPL positivity in microangiopathies, such as TMA, is largely unknown. Therefore, the primary objective of this short review was to estimate the frequency of aPL in subjects with biopsy-proven TMA.
Methods
Literature search
Firstly, a detailed literature research strategy has been developed. This strategy was designed to retrieve studies in which cohorts of non-infectious, biopsy-proven renal TMA patients were concurrently assayed for aPL and/or diagnosed with APS. Keywords and subject terms used in the search included MeSH terms such as ‘antiphospholipid syndrome’, ‘antiphospholipid antibodies’ and ‘thrombotic microangiopathy’. The search strategy was applied to PubMed and Ovid MEDLINE (R) In-Process & Other Non-Indexed Citations and Ovid MEDLINE (R) from 1985 to present to December 2022. The present study was performed according to PRISMA guidelines, and research strings are detailed in the supplementary material.
Study selection
Potential studies identified with the abovementioned search strategy were considered and screened for inclusion. Duplicate studies were identified and removed using the filter functions ‘exact duplicates’ and ‘close duplicates’. Two independent reviewers reviewed all potential studies (OA and NT). Eligibility was first determined by review of the title and abstract and then by full article review. Disagreements were resolved by consensus: if consensus was not achieved, a third party (SS) provided an assessment of eligibility. As the data on eligibility were dichotomous (eligible: yes/no), inter-rater agreement about the title, the abstract review and the full article review stages were determined by calculation of Cohen’s κ coefficient (strength of agreement = good).
Inclusion and exclusion criteria
A study was included in the review if (1) it reported on the laboratory investigation of any aPL; (2) it included patients with biopsy-proven renal TMA; (3) it included at least 15 subjects. A study was excluded if no information about the biopsy findings of TMA was given. Review articles, case reports and case series with a sample size of 15 or less were excluded from the analysis.
Risk of bias assessment
Two reviewers (OA and EV) independently assessed the risk of bias of individual studies using the Newcastle–Ottawa Scale (NOS) for cohort studies and for case–control studies. Figure 1 shows the literature search strategy on the association between antiphospholipid antibodies and biopsy-proven TMA. Literature search strategy.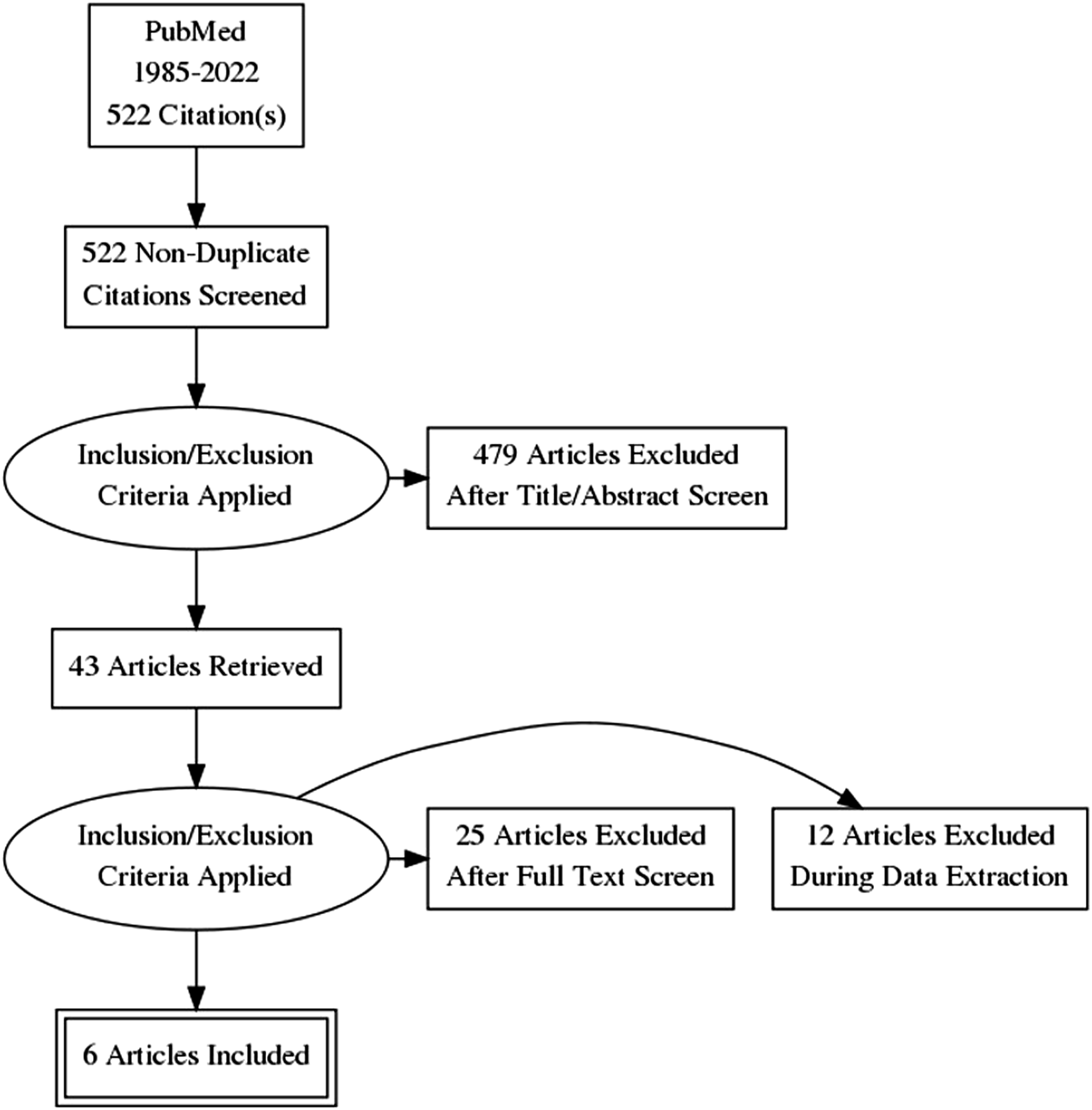
The NOS is a scoring tool used to assess quality of evidence and risk of bias for non-randomised studies included in meta-analyses. This tool is chosen for its content validity as well as its inter-rater reliability has been well established from scientific literature. 15
The criterion validity and intra-rater reliability of this tool were actively determined. The overall quality of evidence was determined using GRADE Criterion and summarised using GRADE profiler.
Risk of publication bias assessment
To assess publication bias, a visual review of the symmetry of the funnel plot was performed. The limitations of using the funnel plot for the assessment of publication bias (particularly in a topic area with relatively few and relatively small publications) were also considered.
Data extraction
All the papers were scrutinised for the following: (1) study design (retrospective, prospective, case–control, cross-sectional and case series); (2) number of patients, sex and age (mean, range); (3) type of outcome; (4) number and type of aPL tests used (criteria tests vs non-criteria); (5) definition of ‘positive criteria aPL’ (low, medium or high titre, or other) as per the study’s definition; (6) confirmation of criteria aPL, at least 6 weeks or 12 weeks apart; and (7) frequency of positive aPL in the study population (defined by sex and age range).
Data were explored to determine if sources of heterogeneity could be explained by the following hypotheses: study design (cohort vs case–control); reported medical comorbidities in the patient population; and the aPL type and methods used for laboratory aPL testing (including the number of aPL tests performed). Indeed, we can hypothesise that each of these sub-group analyses could account for potential heterogeneity in the pooled estimate. The median frequency for positive aPL including anticardiolipin antibodies (aCL), antibodies against β2-glycoprotein-I (anti-β2GPI) and lupus anticoagulant (LA) was then calculated.
Given the non-parametric distribution of our data, we expressed aPL frequency as median (range). First, we calculated the frequency of aPL positivity of any aPL test. When possible, the frequency was estimated for each of the aPL criteria tests (aCL, anti-β2GPI and LA).
Results
Main characteristics of the retrieved studies.
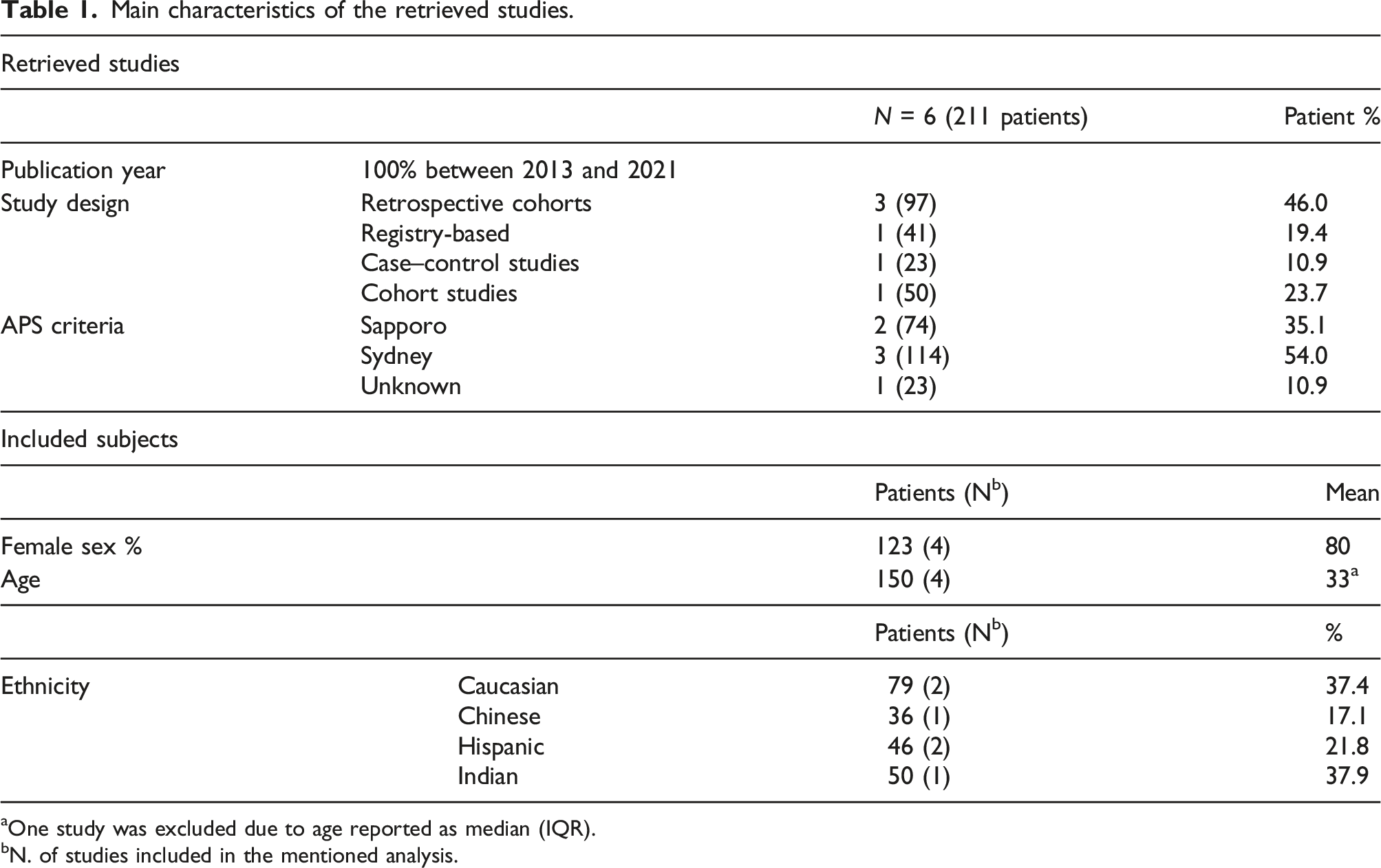
aOne study was excluded due to age reported as median (IQR).
bN. of studies included in the mentioned analysis.
Frequency of any aPL, aCL, anti-β2GPI, LA and APS in patients with renal TMA.
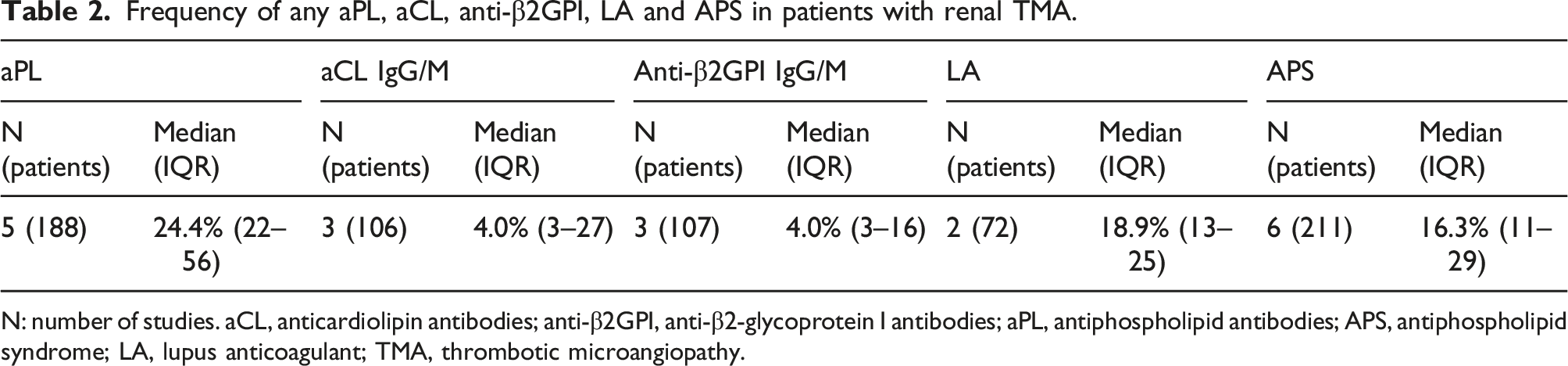
N: number of studies. aCL, anticardiolipin antibodies; anti-β2GPI, anti-β2-glycoprotein I antibodies; aPL, antiphospholipid antibodies; APS, antiphospholipid syndrome; LA, lupus anticoagulant; TMA, thrombotic microangiopathy.
The vast majority of the patients included in our analysis suffered for SLE,16–20 and 5 out of 6 of the retrieved studies included only patients with concomitant lupus.16–20 The study by Marties and co-workers 5 included different connective tissue diseases: patients with systemic lupus erythematosus (n = 18), primary Sjögren's syndrome (n = 7), systemic sclerosis (n = 11), mixed CTD (n = 2) and 2 cases of vasculitides, including 7 overlapping forms and 8 cases of primary APS.
Due to high heterogeneity across studies, a random-effect model for meta-analysis was chosen.
21
Studies show a range of prevalence variation between 10% and 84%. The pooled estimate of the prevalence, based on five included studies, is 39% with a 95%CI ranging from 0.12 to 0.66. The pooled estimate is statistically significant such as the heterogeneity test; in fact, the I2 is 95.68%. The I2 statistic describes the variation across studies due to heterogeneity, rather than due to chance.
22
The prevalence and pooled estimate of aPL in the studies included in our meta-analysis are represented in Figure 2. Prevalence and pooled estimate of aPL in the studies included in our meta-analysis.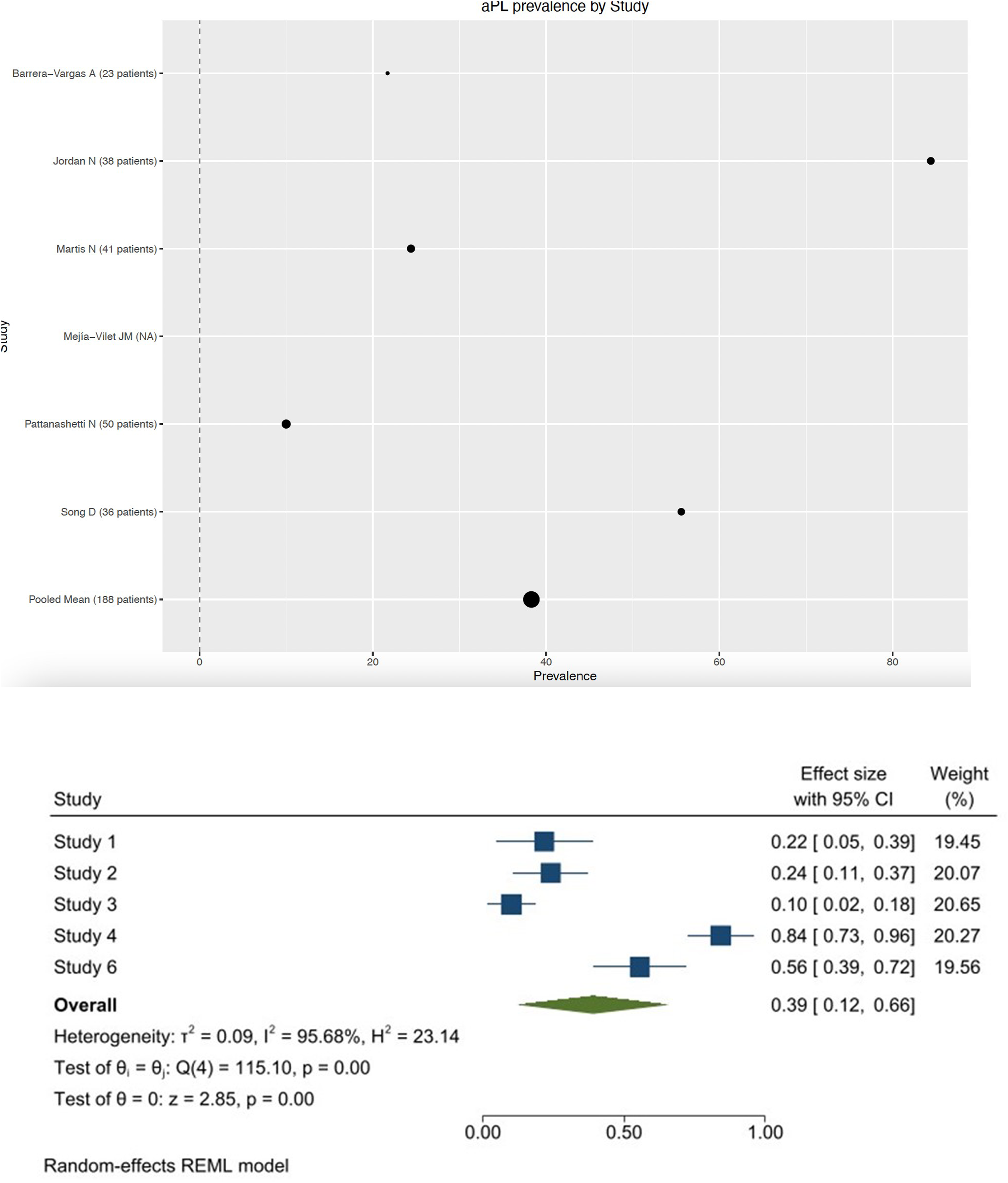
Finally, due to the low number of studies and low sample size, we cannot conclude that no publication bias exists but only a variation due to chance.
23
An assessment of publication bias is represented by a funnel plot in Figure 3. Funnel plot for the five studies included in the meta-analysis.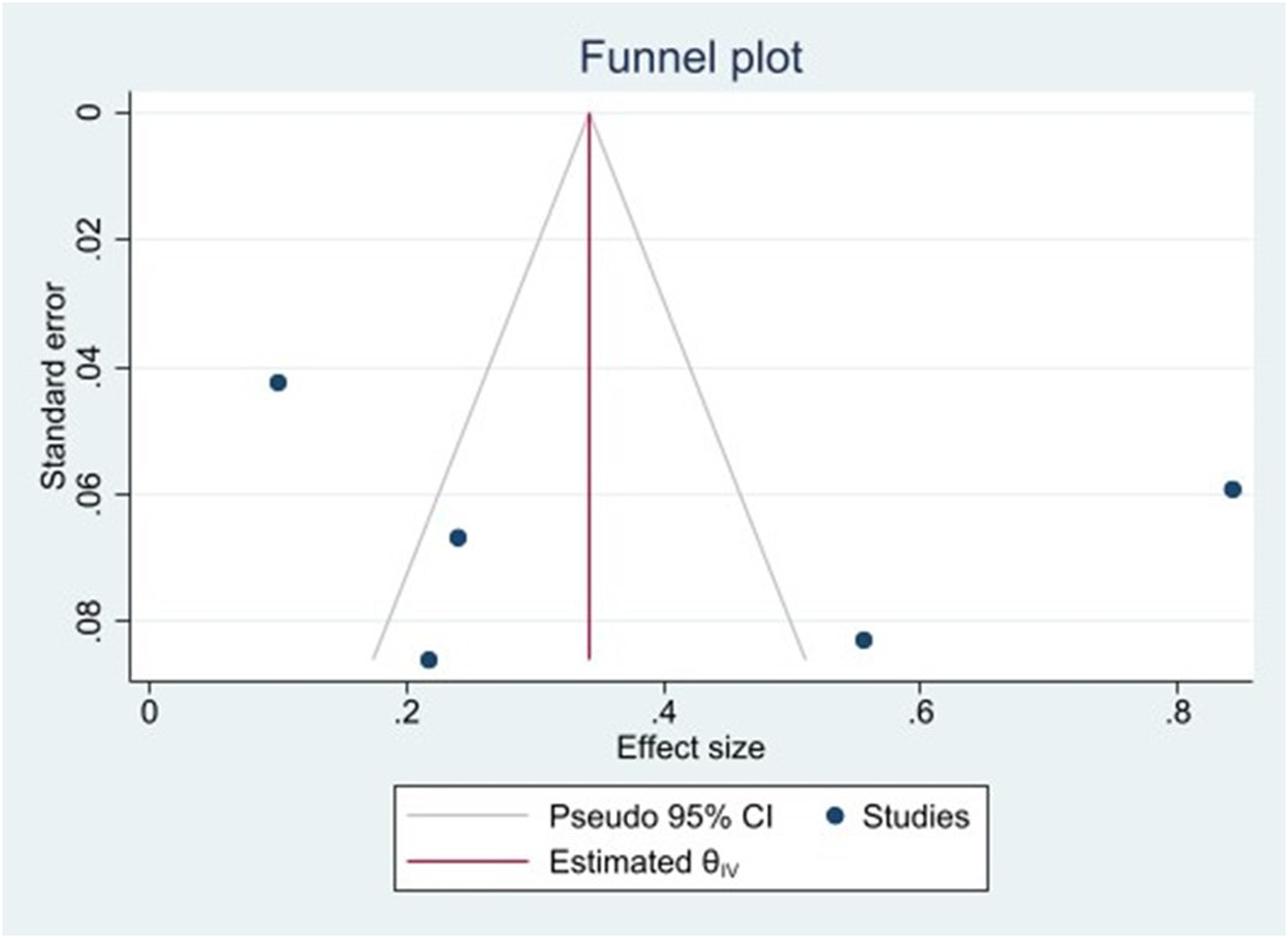
Discussion
TMA can develop in a variety of clinical scenarios and is associated with an increased risk of major cardiovascular events, neurologic events, dialysis and death.24–26 To our knowledge, this is the first comprehensive systematic analysis of studies investigating the association between aPL and TMA. Our results demonstrate that aPL positivity can be observed in approximately one out of four non-infectious, biopsy-confirmed TMA cases.
The prevalence of aPL in venous and arterial thrombotic events had already been estimated by previous studies. A 2013 review of 120 studies conducted by Andreoli et al on behalf of the APS ACTION estimated that criteria-aPL frequency was 6% in patients with pregnancy morbidity, 13.5% in stroke, 11% in myocardial infarction and 9.5% in deep vein thrombosis. 27
Additionally, a 2015 systematic review of 43 studies assessing the prevalence of criteria-aPL in young patients below the age of 50 who presented with cardiovascular events (CVE), conducted by Sciascia et al, estimated the prevalence of aPL to be 17.2% in any CVE, 17.2% in stroke and 11.7% in transient ischaemic attack, concluding that aPL positivity confers a fivefold risk for stroke when compared to controls. 28
Our study potentiates the conclusions yielded by the aforementioned studies and together with those studies, suggests that aPL positivity may be strongly associated with the development of thrombotic and thromboembolic events.
Our study is not devoid of limitations. First, repeat aPL testing was not performed in all of the included studies. In 5 studies aPL testing was repeated and confirmed at least 12 weeks apart, while in 1 study it was not specified if aPL positive testing was confirmed, with one 1 of the 6 included studies only testing for aPL at the time renal biopsy was performed. 17 In addition, selection biases in the analysed cohorts cannot be ruled out.
Second, reporting of aPL was not consistent across all studies, for example, some studies not specifying the aPL profile of aPL-positive patients. Information that could enhance the accuracy of risk estimation, such as adjustment for clinical and historical factors, information obtained via physical examination and apposite diagnostic tests, was not always available, and presentation of available information was heterogeneous across the studies assayed in our analysis.
Similarly, it is worth mentioning that to address the impact of aPL positivity in TMA, patients without SLE should be investigated. However, the number of available cohorts of patients with biopsy-proven TMA and not SLE systematically tested for aPL is scarce, and, while our research strategy was not designed to include only patients with SLE, we failed to retrieve any studies including patients without a connective tissue disease meeting the a priori defined inclusion criteria. Further studies comparing the prevalence of aPL in TMA in patients with and, more critically, without SLE are warranted.
In addition to these limitations, heterogeneity in patient populations, lack of standardisation in aPL testing methods, diagnostic criteria used and language bias also represent non-negligible limitations to our analysis. Furthermore, while our research strategy was a priori designed, we cannot exclude that some pertinent studies might have been missed (e.g. due to heterogeneity in indexing keywords). Besides, we acknowledge that testing for funnel plot asymmetry in analysis including less than 10 studies reduces the power of the tests to distinguish chance from real asymmetry.
In conclusion, our study demonstrates that aPL can be detected in a non-negligible rate of patients with biopsy-proven TMA and suggests that the presence of aPL may confer an increased risk for the development of TMA. However, it is important to mention that investigating the pathogenic role of aPL in the development of TMA was out of the scope of this analysis and we cannot exclude that in some cases the presence of aPL might represent an epiphenomenon. Once validated by prospective studies, aPL testing in this high-risk population may enhance the diagnostic workup, risk stratification, prognostic evaluation and therapeutic approach in the context of TMA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
