Abstract
Introduction:
People with hemodialysis-dependent kidney failure experience significantly higher cardiovascular (CV) morbidity, mortality, symptom burden, and cognitive impairment compared to the general population. Hemodialysis-induced ischemia contributes to myocardial stunning, which worsens CV outcomes. Intradialytic cycling has the potential to mitigate cardiac stunning, but its effects have not been assessed in randomized controlled trials (RCTs).
Methods:
This multicenter RCT evaluates the impact of 12 weeks of intradialytic cycling on hemodialysis-induced myocardial stunning. The study will recruit 160 adults (80 intervention and 80 control) receiving maintenance hemodialysis at five Canadian centers and one Australian center. Outcome assessments are conducted at baseline, 12 weeks, and 16 weeks. An intervention implementation process evaluation is part of this study.
Outcomes:
The primary outcome is change in number of regional wall motion abnormalities (RWMAs) at peak hemodialysis stress from baseline to 12 weeks. Secondary outcomes include changes in cognitive function, hemodialysis recovery time, and symptom burden.
Statistical Analysis:
Change in number of RWMAs at peak hemodialysis stress from baseline to 12 weeks are compared between study groups using an independent 2-tailed t test and using a Poisson generalized linear mixed model with study group as a fixed effect, and time and mean ultrafiltration rate as random effects.
Conclusion:
Compared with standard care, we anticipate that intradialytic cycling will decrease myocardial stunning, resulting in improved symptom burden, and functional and cognitive status. This trial provides critical evidence regarding the benefits and feasibility of exercise during hemodialysis, potentially informing clinical practice, and guidelines for hemodialysis care.
Introduction
Globally, most individuals with kidney failure receive facility-based hemodialysis, which is associated with poor quality of life, cognitive impairment, and adverse outcomes.1,2 Up to 80% of people receiving hemodialysis suffer burdensome dialysis-related symptoms, such as fatigue, cramps, pruritis, and pain. 3 Physical activity and physical function levels are up to 60% lower than in the general population and decline over time.4,5 Rates of cardiac death are 10 to 20 times higher than in the general population; however, these high rates are not explained by traditional cardiovascular (CV) risk factors. 6 Hemodialysis itself causes repetitive episodes of transient myocardial ischemia that is associated with worsening functional status, worse hemodialysis-related symptoms, increased cardiac failure rates, sudden cardiac death, and increased mortality. 7
Transient myocardial ischemia leading to persistent left ventricular (LV) dysfunction is known as myocardial stunning. 8 Over 60% of people receiving facility-based hemodialysis experience recurrent myocardial stunning due to reduced myocardial blood flow resulting in cardiac regional wall motion abnormalities (RWMAs) during hemodialysis treatments. 9 Exact mechanisms of hemodialysis-induced myocardial stunning are unclear. Contributing factors include increased susceptibility to demand ischemia due to pre-existing heart disease, myocardial ischemia due to arterial stiffness from arteriosclerosis, LV hypertrophy, impaired microcirculation, and disturbed vasoregulation in individuals receiving hemodialysis. 10
Hemodialysis-related myocardial stunning is theorized to be exacerbated by removal of large volumes of fluid and intradialytic hypotension but can occur during hemodialysis without significant clinical signs or symptoms.11,12 This has been demonstrated by intradialytic magnetic resonance imaging and positron emission tomography measurements of myocardial blood flow. 12 Importantly, even though age and CV disease can exacerbate hemodialysis-induced myocardial stunning, 11 it also occurs in people receiving hemodialysis who have few comorbidities and are perceived to have low CV risk.9,13 During the last 30 minutes of hemodialysis, the volume of fluid removed often peaks and blood pressure nadirs, making this a time of “peak hemodialysis stress” and the ideal time to assess for RWMA. 10
Myocardial stunning is a valid marker of recurrent hemodialysis-related cardiac injury. 14 Although commonly elevated in individuals on hemodialysis, pre-hemodialysis levels of high-sensitivity troponin T (hsTnT) are independently associated with increasing number and severity of hemodialysis-related RWMA. 15 Individuals with no stunning at baseline, but who developed stunning after one year demonstrated longitudinal increases in pre-hemodialysis hsTnT. 16 Similarly, increasing post-hemodialysis hsTnT levels have been associated with decreased LV global longitudinal strain (GLS). 17 Furthermore, presence of myocardial stunning during hemodialysis is associated with a 10% to 12% decline in LV ejection fraction, a 20% reduction in survival, and a 30% increase in CV events over one year. 18
Reduction in organ perfusion related to myocardial stunning leads to worsening hemodialysis-related symptoms, compromised cognitive and physical function, and poor quality of life (Figure 1).15,19,20 Decreased brain perfusion associated with myocardial stunning can lead to cognitive impairment in individuals receiving hemodialysis. 21 Furthermore, individuals on hemodialysis can suffer from post-dialysis fatigue, or the delayed ability to perform one’s usual activities, 22 which significantly impacts functional status, CV events, and mortality.22-24 Worsening myocardial stunning at the end of hemodialysis was independently associated with an almost 2-fold risk of severe post-hemodialysis fatigue. 19 Interventions to reduce hemodialysis-induced ischemic-based tissue injury and improve patient-important outcomes, such as post-dialysis fatigue, are needed.

Conceptual framework of effect of intradialytic exercise on patient-important outcomes.
Intradialytic exercise has been proposed as an intervention strategy that may decrease myocardial stunning given its cardioprotective ischemic preconditioning effect.24-28 The application of small repetitive ischemic insults enabling the heart to cope with more significant ischemic insults, such as acute myocardial infarction is called ischemic preconditioning. In short term, exercise can provide cardioprotective effects similar to those described with ischemic preconditioning. 26 In both animal and human models, this protection has been shown to occur immediately after exercise and last for two to four hours. Then it returns approximately 24 hours after exercise, continuing for up to five days.29,30 Exercise as preconditioning improves cardiac function and survival after myocardial infarction in animal models and has been associated with improved outcomes in human observational studies.29,31 Exercise training (multiple bouts of exercise rather than a single session) confers sustained benefits over and above the protection a single exercise session provides.29,30 Exercise training appears to preserve the ability of preconditioning to prevent endothelial ischemic injury in older adults. It supports the cardioprotective role of exercise training in populations with pre-existing cardiac risk factors, including people receiving hemodialysis.32,33
Cross-sectional and pilot studies have shown the potential effects of exercise on hemodialysis-related myocardial stunning. A cross-sectional study of 19 hemodialysis patients involved in a clinical intradialytic cycling program found that participants experienced a 30% lower mean number of cardiac RWMA during peak hemodialysis stress when they exercised for 20 to 60 minutes than during a hemodialysis session without exercise. 34 Another study of 18 prevalent, exercise naïve people receiving hemodialysis, demonstrated exercise during hemodialysis (compared with a hemodialysis session with no exercise) was associated with fewer RWMA at peak hemodialysis stress (mean 5 vs 7 RWMAs, respectively). 26 These two small studies observed no differences in LV GLS, cardiac output, and blood pressure between control and exercise sessions.26,34 In cross-sectional studies, intradialytic cycling has also improved LV mechanics, such as circumferential strain and torsion. 35
In addition, intradialytic exercise seems to improve clinical outcomes associated with hemodialysis-related myocardial stunning, including muscle cramping, symptoms of depression, and restless legs syndrome.36,37 In a pilot randomized controlled trial (RCT) with 13 participants, cycling during hemodialysis for three months mitigated declines in cognitive function seen in the control group. 38 Finally, in a cluster randomized trial of intradialytic cycling for six months, individuals who cycled thrice weekly had a statistically significant 11.1 g decline in LV mass compared with controls who performed no intradialytic exercise. 39 These findings suggest that aerobic exercise during hemodialysis may decrease hemodialysis-related myocardial stunning through an ischemic preconditioning effect and can provide protection from hemodialysis-induced ischemia of vital organs, improve dialysis-related symptom burden, and preserve long-term cardiac function.
The evidence for harm related to hemodialysis-related myocardial stunning is compelling, and the role of intradialytic exercise in mitigating this harm is promising. Individuals receiving hemodialysis, researchers, and clinicians have identified reducing CV risk as a research priority, and the effects of exercise on CV outcomes are an important knowledge gap.40-42
In this RCT, our primary objective is to evaluate the effect of intradialytic aerobic exercise on hemodialysis-related myocardial stunning as assessed by the number of LV RMWA measured with echocardiography. Secondary objectives include correlating the magnitude of hemodialysis-induced myocardial stunning with an established biomarker for myocardial injury, hsTnT, characterizing the effect of intradialytic cycling program on post-dialysis fatigue and symptom burden, and exploring the “off treatment effect” of exercise on hemodialysis-induced myocardial stunning four weeks after an intradialytic cycling program is completed. Finally, we will conduct an implementation process evaluation 43 alongside the trial to assess the fidelity of intradialytic cycling program delivery at each site and identify barriers/enablers experienced by staff to inform optimization of program delivery for clinical programs and future trials. This study expands on our exploratory work and is the first to use adequately powered RCT methodology to determine the longitudinal effect of aerobic exercise during hemodialysis on hemodialysis-related myocardial stunning.
Methods
Design and Setting
This is a multicenter, assessor-blinded RCT with 1:1 parallel group design, comparing change in hemodialysis-induced myocardial stunning in individuals receiving hemodialysis participating in 12 weeks of cycling during hemodialysis compared with individuals receiving standard hemodialysis care (N = 160) (Figure 2). Participants will be recruited from seven facility-based hemodialysis units in 6 centers, including two hemodialysis units in Winnipeg, Manitoba; 1 hemodialysis unit in London, Ontario; one hemodialysis unit in Calgary, Alberta; one hemodialysis unit in Edmonton, Alberta, onehemodialysis unit in Vancouver, British Columbia; and one hemodialysis unit in Adelaide, Australia. The 12-week intervention period will be followed by a four-week washout period for a total of 16 weeks. The RCT protocol was developed in accordance with the Consolidated Standards Of Reporting Trials (CONSORT) checklist . 44

Trial of intradialytic cycling as kidney exercise rehabilitation for cardiac stunning in hemodialysis (TICKERS) study design schematic.
Ethical Issues
This study complies with International Council for Harmonization of Technical Requirements and the Declaration of Helsinki. 45 The study protocol and informed consent forms have been approved at the coordinating site by the University of Manitoba Biomedical Research Ethics Board No. HS24857 (B2021:039), the Central Adelaide Health Network Human Research Ethics Board 2022/HRE00015, and by local ethics boards at each of the 4 other study sites. The trial is registered at the US National Institutes of Health (ClinicalTrials.gov) No. NCT04877041. All participants will provide informed consent, acknowledging their voluntary participation and the right to withdraw at any time without penalty.
Participants
Adults 18 years or older, no upper age limit, receiving facility-based maintenance hemodialysis three times per week for at least three months with no planned or expected change in dialysis modality, elective surgery, or relocation during the 16-week study period who can communicate in English and provide written informed consent; and are assessed to be safe and able to exercise by the hemodialysis unit nephrologist (Supplementary Material 1) are eligible for inclusion in this study.
Exclusion criteria include acute coronary syndrome in the past three months, unstable arrhythmia, shortness of breath at rest or with minimal activity (NYHA class 4), symptomatic hypoglycemia more than twice in the week before enrollment, and having participated in an intradialytic exercise program within the last 3 months.
Recruitment
Voluntary written informed consent to contact will be obtained from all participants. Following consent to contact, potential participants will be contacted by study staff to arrange an initial meeting. At this meeting, the study will be briefly described, and the inclusion/exclusion criteria will be reviewed with the potential participant to assess their study eligibility cursorily. If eligibility criteria are met after this cursory eligibility review and the individual is interested in proceeding, study staff will proceed with the informed consent process. Participants will be given verbal and written information detailing the study and provided at least 48 hours to consider the risks and benefits of participation. Once informed consent is obtained, the baseline assessment at the individual’s hemodialysis unit will be arranged. At this initial assessment, all participants will receive standardized physical activity counseling (usual care) and undergo baseline testing. This baseline appointment services as a “run-in phase.” Individuals who do not attend two scheduled baseline visits are not enrolled.
In previous exercise studies in hemodialysis, females have been under-represented, and CV disease presents differently in women.46-48 To address this, the study actively aims to recruit at least 25% female participants. This proportion of female participants approaches the less than 35% midlife and older female representation typically seen in exercise-related trials, which is even lower than the 40% female participation level seen in dialysis-related RCTS and reflects the collective experience with exercise-related RCTs in people receiving hemodialysis.39,49-51
Randomization
To ensure allocation concealment, balance and to minimize bias, randomization will be performed by a third-party team member. The Research Electronic Database Capture (REDCap) study database held at the University of Manitoba will deliver block randomization with randomly permuted block sizes of 2, 4, and 6, stratified by study site with overall 1:1 allocation. Study-group assignment will occur after baseline exercise counseling and outcome assessment. Due to the nature of the intervention in this trial, the study principal investigator, hemodialysis unit clinical staff, and study participants cannot be blinded. Outcome assessors and the statistician are blinded to the study group.
Intervention and Control Groups
As recommended in clinical practice guidelines for individuals with chronic kidney disease, all study participants will receive standardized exercise counseling at baseline.52,53 Standardization of exercise counseling at all study sites will be achieved through coordinating site kinesiologists’ or exercise physiologists’ review of counseling session plan at each study site.
Although not prohibited from exercising outside the study protocol, participants in the control group undergo no formal structured exercise intervention. They are asked to refrain from intradialytic cycling during the 16-week study period. Exercise activity is tracked via weekly self-reported log sheets and accelerometry at the 3 assessment time points. The study kinesiologist collects log sheets every week from study participants in both groups during the study period. This is done to control for the “personal trainer effect,” that is, visits with the kinesiologist can influence the participant’s exercise performance and motivation to participate in exercise.
Participants in the intervention arm participate in a supervised 12-week intradialytic cycling program. This exercise programming duration has improved health parameters in our previous studies.9,54-57 A maximal incremental cycle test (to volitional exhaustion) is performed before intervention start using bedside ergometers to standardize exercise intensity. 58 The type of ergometer and participant comfort can be adapted to needs of the individual and each study site’s dialysis beds or chairs. At each study site, all ergometers used in the study are calibrated with power meter pedals using a standardized protocol developed by the coordinating site. Study kinesiologists instruct participants to target an exercise intensity of 50% to 60% of maximal workload, as determined by the initial incremental cycling test.59,60 Due to autonomic dysfunction and high prevalence of beta blockers in the hemodialysis population, percent heart rate reserve will not be used to target intensity. 61 Study kinesiologists are trained regarding standard operating procedures (SOPs) for the exercise program through video conferencing organized with the Winnipeg site. Regular monthly virtual meetings to support participating sites facilitates adherence to all study SOPs.
The target exercise duration will be 60 minutes in the first half of each hemodialysis session (usual duration of four hours), three times per week for 12 weeks, with rest periods as needed for participants unable to complete 60 consecutive minutes of exercise.56,60 Total exercise time, self-reported intensity (Borg Rating of Perceived Exertion [RPE]) and average workload (Watts) are recorded for each session. Target RPE is 13/20, which is the equivalent of moderate intensity. Ergometer resistance is adjusted over time by study kinesiologists as triggered by a one-point RPE change. The kinesiologists closely supervise participants during the initial three intradialytic exercise sessions. After three sessions (or more if requested by participant or bedside hemodialysis staff), hemodialysis unit and research staff will assist with cycle ergometer set up and monitoring during exercise sessions, and kinesiologists will check in with participants weekly. This exercise training protocol was developed to assess study quality and reporting in exercise training studies (TESTEX) reporting standards for studies with exercise interventions. 62
Study Outcomes
The primary outcome of this study is change in hemodialysis-induced myocardial stunning from baseline to 12 weeks as measured by change in number of RWMAs determined by intradialytic echocardiography at peak hemodialysis stress (30 minutes before the end of hemodialysis session) between baseline and post-cycling exercise. Echocardiogram acquisition are completed using standard LV apical 2, 3, and 4 chamber views collected by trained operators at each study site three times during the mid-week hemodialysis session at each outcome assessment time point as follows: baseline (pre-hemodialysis), post-cycling exercise (or mid-dialysis in controls), and at peak hemodialysis stress (~30 minutes prior to end hemodialysis) (Figure 3). Image analysis is performed in London, Ontario using automated speckle-tracking analysis (EchoPAC-PC V. 110.1.3; GE Healthcare).34,56 Three cardiac cycles are analyzed for each image and RWMA will be defined as a > 20% decrease in segmental longitudinal strain (SLS) from baseline to peak stress as per our previous studies.18,34 To ensure accuracy and standardized image capture at all sites, a level 3 certified echocardiologist will provide remote quality assurance support and pilot echocardiography images will be submitted to London, Ontario from each collecting site for assessment and recommendations to ensure optimal image acquisition. See Table 1 for secondary and exploratory outcomes.
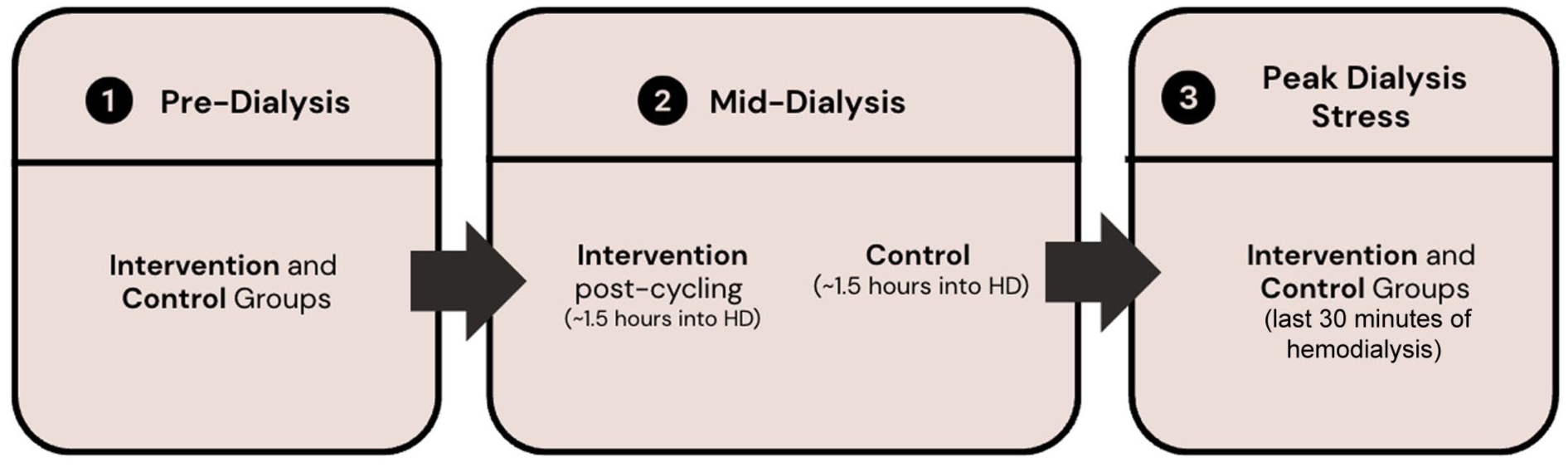
Timing of echocardiograms during dialysis at each assessment point.
Trial of Intradialytic Cycling as Kidney Exercise Rehabilitation for Cardiac Stunning in Hemodialysis (TICKERS_HD) Study Primary and Secondary Outcomes.

Note. DSI = Dialysis Symptom Index; GLS = global longitudinal strain; HD = hemodialysis; hsTnT = high-sensitivity troponin T; ISWT = incremental shuttle walk test; LV = left ventricular; RWMA = regional wall motion abnormality; SLS = segmental longitudinal strain;
Data Collection Procedures and Timing
Demographic data are collected by a blinded assessor at baseline via patient interview and hemodialysis chart review and will include age, self-reported sex, ethnicity, kidney failure cause, comorbidities (diabetes, hypertension, smoking, cardiac disease/arrhythmia, cerebrovascular disease/stroke/transient ischemic attack, lung disease, peripheral vascular disease/lower extremity amputation, and arthritis), date of dialysis start, and other relevant medical history.
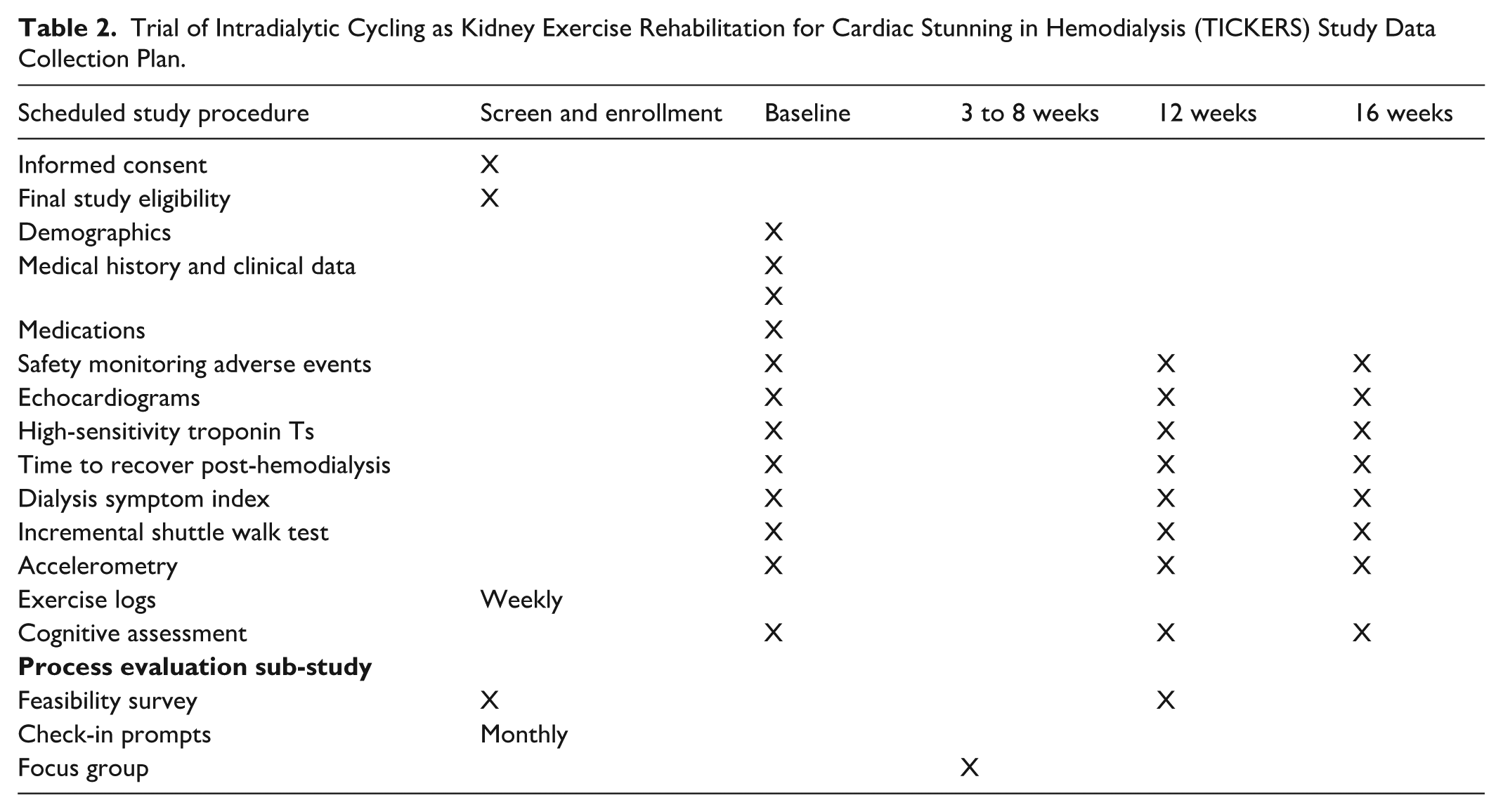
Clinical data collected from hemodialysis chart includes height, weight, body mass index, heart rate, systolic and diastolic blood pressure, dialysis prescription, including time, dialysis adequacy, weekly intradialytic fluid gains, and duration of hemodialysis sessions (Table 2).
Trial of Intradialytic Cycling as Kidney Exercise Rehabilitation for Cardiac Stunning in Hemodialysis (TICKERS) Study Data Collection Plan.
Laboratory data collected from hemodialysis chart include serum hemoglobin, calcium, albumin, and phosphate.
Outcome Assessments
The study kinesiologist conducts baseline outcomes assessments. Twelve- and 16-week assessments (within +/–10 days of this time point) are conducted by a blinded assessor. They are performed pre-dialysis on a mid-week dialysis day to minimize effects of fluid overload and dialysis fatigue, but maximize convenience and adherence to the assessment schedule. All demographic, clinical, laboratory, and outcome assessment data are entered into a secure REDCap database 63 hosted at the University of Manitoba, Canada. Refer to Table 2 for details on the outcome assessment.
Sample Size and Power
Based on pilot data, we predict the mean number of RWMAs will decrease by 2 from a baseline mean of 6 to 4 RWMA (SD = 3) at peak hemodialysis stress. 34 A target sample size of 160 (80 participants per study arm) is proposed with the following assumptions: conservative 40% dropout based on our previous RCT experiences (acute illness, modality change, move, and disinterest),26,54,56,64 90% power, alpha 0.05, and the number needed in each study group is 48. We have power to detect reference change values (≥25%) in hsTnT levels. Study sites have 2070 prevalent patients and 1020 incident patients per year. We will recruit 160 participants (less than 10% of the potentially eligible population). In our previous study, we successfully recruited 20% of the population, demonstrating the feasibility of recruiting 160 participants (10%) for this study. 56
Statistical Analysis
Statistical analysis plan will be finalized before locking the final study database. Analysis will be performed in an intention-to-treat manner on a case available basis. We will compare baseline descriptive variables between study groups using independent 2-tailed t tests, Mann-Whitney U tests, and chi-square tests, as appropriate. Similarly, baseline descriptive variables of individuals who complete the study protocol will be compared with those who do not. Missing data (if < 25%) will be imputed according to current Food and Drug Administration National Research Council recommendations using multiple imputation by chained equations.65-67
Changes in the number of RWMAs at peak hemodialysis stress from baseline to 12 weeks will be compared between study groups using an independent 2-tailed t test. We will perform a secondary analysis for the primary outcome using a Poisson generalized linear mixed model with study group as a fixed effect and time and mean ultrafiltration rate as random effects. We will also perform Poisson regression to assess difference in RWMA count at peak hemodialysis stress between baseline and 12 weeks, adjusted for baseline RWMA count at peak hemodialysis stress and baseline ultrafiltration rate. The correlation of change in number of RWMAs and hsTnT levels will be analyzed using logistic regression whereby change in number of RWMAs will be used to predict a 25% change (reference change value) in hsTnT. Additional variables known to affect study outcomes, including fluid removal rate during hemodialysis, diabetes, blood pressure at time of peak hemodialysis stress, age, sex, and comorbidities will be added to these models in secondary analyses, with a minimum of 10 events required for each additional variable added to the model. P value of < .05 will be statistically significant for our primary outcome and we will use Bonferroni adjustments for our secondary outcomes.
Multiple linear and logistic regressions (as appropriate) will be performed to determine intradialytic exercise’s role and RWMA’s severity on absolute change in post-dialysis recovery time and cognitive function and symptoms relative to minimal clinically important difference (MCID). These models for each outcome will include a binary variable indicating whether or not the absolute change met the MCID threshold. Study group and other key predictors of these outcomes, including baseline physical function and physical activity level, will be tested in a hierarchical, stepwise selection manner with a P value of < .05 for entry criteria and < .10 for exit criteria. The remaining predictors in the stepwise model will be considered statistically significant. Statistical analysis will be performed using SAS 9.4 (Cary, NC).
Sub-group analyses are pre-specified and minimized in the statistical analysis plan according to best practices and based on the interaction term with the intervention.
We report sex-stratified results for efficacy and safety as per the sex and gender equity in research guidelines. 68
Additional planned exploratory analyses include the effect of the severity of RWMA on change in intradialytic blood pressure, cognitive status, and post-dialysis fatigue, change in RWMA by quartile of baseline physical activity (an important potential confounder) as measured by accelerometry, and analysis of the primary outcomes “on protocol” defined as completion of 60% or greater of exercise sessions during the intervention period.
Data Safety Monitoring Board
A Data Safety Monitoring Board (DSMB) reviews safety data yearly and recommends whether the study should continue as is, be modified to protect subjects’ safety, or be terminated. Serious adverse events, including all-cause death and hospitalizations, fall with fracture, and tendon rupture will be recorded and reported to the DSMB (Supplementary Material 2). No formal interim analysis for efficacy, safety, or futility will be performed due to short duration of the trial and demonstrated safety of the intervention in multiple previous trials. The DSMB works independently from the trial and did not participate in developing the statistical plan.
Process Evaluation Sub-Study
To better understand how the cycling intervention may be impacted by the hemodialysis unit context at each of the six sites and guided by implementation scientists (J.P.), we conduct a mixed-methods process evaluation with hemodialysis unit staff from sites participating in the trial that consists of three components: a pre-post intervention survey (Supplementary Material 3); check-in prompts during monthly site meetings, and focus groups with hemodialysis staff during the intervention period at each site (Supplementary Material 4).
Discussion
We designed an RCT to determine the effect of intradialytic exercise on hemodialysis-related myocardial stunning and a validated surrogate outcome of cardiac morbidity and mortality in the hemodialysis population. 18 Previous cross-sectional and pilot studies have shown potential effects of exercise on myocardial stunning.26,34 In cross-sectional studies, intradialytic cycling has improved LV mechanics, such as circumferential strain and torsion. 35 This study provides an opportunity to build on exploratory work, increasing our understanding of the effects of intradialytic exercise on myocardial stunning.
Intradialytic exercise has been shown to improve clinical outcomes associated with hemodialysis-related myocardial stunning, including muscle cramping, depression, cognitive function, and restless legs syndrome.36-38 This may be explained by decreases in hemodialysis-related multi-organ stunning through an ischemic preconditioning effect of aerobic exercise, leading to protection from hemodialysis-induced ischemia and decrease in symptom burden associated with hemodialysis treatments.
This study is the first to use adequately powered RCT methodology to determine the longitudinal effect of aerobic exercise during hemodialysis on hemodialysis-related myocardial stunning and related clinically important outcomes, such as symptoms, physical function, and cognition. Another strength of this study is the process evaluation sub-study, which will help further understand and provide potential solutions to the barriers (and enablers) to intradialytic exercise interventions in clinical trials. This will help inform the design and implementation of future intradialytic exercise clinical programs and trial interventions. Additional strengths include selecting pragmatic and clinically relevant eligibility criteria that maximize representativeness and generalizability of study results.
Steps implemented to mitigate potential challenges include: a conservative estimated drop-out rate of 40%, blinded outcome assessors at all assessment time points and blinded study statistician to minimize bias, exercise supervision to ensure target intensity is achieved, and facilitate participation completion of exercise sessions, and incorporation of physical activity level as measured objectively by accelerometry into outcome analyses to control for potential confounding by this modifier.
In conclusion, we anticipate that cycling during hemodialysis, compared with usual care, will decrease hemodialysis-induced myocardial stunning, resulting in reduced symptom burden and improved functional and cognitive status in the intervention group compared with controls. This RCT will provide information regarding the role of exercise on patient-important outcomes, including CV risk (myocardial stunning), symptom burden, and cognition. Findings from this RCT and the implementation sub-study will inform the design of large future exercise-related studies looking at long-term clinical outcomes, including CV events and mortality. Such evidence and knowledge is required for widespread clinical exercise adoption in the hemodialysis setting and has been identified as an important evidence gap.34,41
Supplemental Material
sj-docx-1-cjk-10.1177_20543581251391112 – Supplemental material for Randomized Controlled Trial of Intradialytic Cycling as Kidney Exercise Rehabilitation for Cardiac Stunning in Hemodialysis (TICKERS_HD): A Clinical Research Protocol
Supplemental material, sj-docx-1-cjk-10.1177_20543581251391112 for Randomized Controlled Trial of Intradialytic Cycling as Kidney Exercise Rehabilitation for Cardiac Stunning in Hemodialysis (TICKERS_HD): A Clinical Research Protocol by Paul N. Bennett, Jarrin D. Penny, Christopher W. McIntyre, Jessica Vanderlinden, Megan S. Borkum, Gisell Castillo, David Collister, Bonnie Corradetti, Todd A. Duhamel, Haley Farion, Emilie Ford, Eric Garcia, Ruth Getachew, Shilpanjali Jesudason, Mercedeh Kiaii, Richard Le Leu, Justin Presseau, Claudio Rigatto, Ashley K. Seitz, Mitra Shirazi, Anita Soni, Krista Stewart, Brett Tarca, Karthik Tennankore, Nancy Verdin, Reid Whitlock, Ken Wilund, Stephanie Thompson, Jennifer M. MacRae and Clara Bohm in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
Monica Sharma, MD, University of Manitoba, for assistance with developing protocol and early phases of the study and Dr Tania Salehi, Jing Zhang, and Bronwyn Hockley, Royal Adelaide Hospital, for support at the Australian study site.
ORCID iDs
Author Contributions
The study concept and design were conceived by C.B., C.M., J.P., T.D., P.B., J.M.M., S.T., K.W., K.T., J.V., and J.P. First draft of the manuscript prepared by P.N.B., C.B., E.F., and H.F. All authors provided edits and critiques of the manuscript for intellectual content.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work i supported by a Canadian Institute for Health Research Project Grant No. PJT 175258 and the University of Manitoba University Research Grants Program UM Project #56980 .
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.D.P., C.W.M., J.V., M.B., G.C., B.C., T.A.D., H.F., E.F., E.G., R.G., S.J., M.K., R.L.L., J.P., C.R., A.K.S., M.S., A.S., K.S., B.T., N.V., R.W., K.W., and S.T., and J.M.M. report nothing to disclose. C.B. reports grant funding from CIHR, KFOC, Research Manitoba, Seven Oaks Hospital Foundation, and Fresenius, and honoraria from Vantive. C.B. is site co-investigator for clinical trials funded by Amgen, Alexion, Astra Zeneca, Eli Lilly, and CSL Vifor. P.N.B. has received honoraria from Seqirus, Vifor, and Vantive. D.C. reports grant funding from CIHR, KFOC, Research Manitoba/Boehringer Ingelheim, ACT, and National Leader funds directed from CSL Behring and AstraZeneca. K.T. reports consultancy with Otsuka Canada, Vifor, and Virtual Hallway.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
