Abstract
Case Description:
A 38-year-old man presented with NELL-1 positive membranous nephropathy (MN). Initial treatment with rituximab and later cyclosporine failed to result in sustained clinical improvement. Upon further review, the patient had been applying a skin fairness cream nightly for 2 years preceding diagnosis. The cream was discontinued, and follow-up testing confirmed markedly elevated serum mercury levels of 66.8 µg/L (normal less than 20 µg/L), and 24-hour urine mercury of 103.9 nmol/d. Two rounds of chelation therapy were arranged, the first with dimercaptosuccinic acid (DMSA) and the second with dimercaptopropane-1-sulfonic acid (DMPS) given cost and availability. Repeat mercury levels normalized but follow up kidney biopsy confirmed persistent immune complex glomerulonephritis. The patient was subsequently treated with prednisone followed by additional rituximab resulting in improvement of proteinuria and stabilization of kidney function.
Rationale/Teaching Points:
This case reinforces the risk of mercury containing topicals and is the first to report systemic absorption of this magnitude. While chelation therapy is effective at improving systemic mercury levels, patients may require additional immunosuppression to treat immune complex-mediated MN following exposure.
Introduction
Neural epidermal growth factor-like protein 1 (NELL-1) has been associated with both primary and secondary membranous nephropathy (MN) and is thought to be the second most common MN-associated antigen after Phospholipase A2 Receptor (PLA2R). 1 Specific to secondary NELL-1 MN, multiple potential culprit drugs have been reported including lipoic acid, non-steroidal anti-inflammatory drugs and traditional medicines. 2
Elevated urine and serum mercury levels have been reported in previously published cases of drug-induced secondary MN, ranging from 16 to 183 µg/L. 3 Heath Canada recommendations suggest blood and urine mercury levels less than 20 µg/L are considered to be in the normal range. 4 Mercury exposure can occur from a variety of sources, including fish consumption and smoking, however mean total blood mercury levels in adults typically range between 0.5 and 4.1 µg/L. 5
Mercury has been found to be present in products worldwide, and recently multiple published cases of NELL-1 MN have been attributed to mercury-containing skin fairness creams, primarily from India. 6 Of these cases, few met sufficient serum mercury levels to warrant chelation therapy. Patients responded with skin fairness cream cessation, the introduction of renin-angiotensin-aldosterone system inhibitors and in certain cases immunosuppression, with a mean time to complete remission of 11 months.
Presenting Concerns
We report a case of a 38-year-old previously healthy man who initially presented to a nephrology clinic in India with new onset proteinuria, lower extremity edema, and weight loss. Urinalysis was significant for greater than 3 g/L proteinuria and 25 to 50 RBCs/hpf. He was found to have an albumin of 25 g/L and 24-hour urine protein measurement of 14.3 g/24 h. Peak serum creatinine at that time was 167 µmol/L, previously known baseline creatinine was less than 100 µmol/L. Kidney biopsy and serological workup confirmed NELL-1 positive MN, and he was treated with 2 doses of rituximab (500 mg IV). He demonstrated partial improvement, later prompting additional treatment with maintenance cyclosporine.
Clinical Findings
The patient relocated due to employment and his care was taken on by another nephrologist. At that time, creatinine was 135 µmol/L, albumin 28 g/L and urine protein to creatinine ratio 925 mg/mmol, indicative of worsening proteinuria. In addition, he was noted for occasional chills, ongoing non-productive cough, and no features of nephrotic syndrome.
Diagnostic Focus and Assessment
Repeat serological assessment for secondary causes of glomerulonephritis was unremarkable, including negative viral serology, normal serum complement, and no evidence of plasma cell dyscrasia with serum protein electrophoresis and free light chains. Given ongoing treatment failure and concern for a secondary cause of MN in the setting of respiratory symptoms, the patient was arranged for a CT chest scan which demonstrated multifocal patchy branching nodular opacities that were concerning for tuberculosis (TB). Interferon gamma release assay testing at that time was positive, despite the patient describing previously being screened as a child for TB with negative skin testing and chest x-ray. Bronchoscopy ultimately ruled out TB with negative mycobacterial cultures. CT abdomen and pelvis did not identify an underlying occult malignancy.
Further detailed review of the patient’s medication found that the patient had been applying a skin fairness cream most nights for the past 2 years (preceding his initial diagnosis). Given potential association with mercury intoxication, the topical was discontinued. There were no additional herbal supplements or medicines used at the time with an association to MN or mercury intoxication. Twenty four hour urine mercury was elevated at 103.9 nmol/d, random urine 54.7 nmol/L (5.47 µg/L). Serum mercury levels were also elevated at 334 nmol/L (66.8 µg/L, normal = less than 20 µg/L). Serum creatinine rose to 180 µmol/L. The patient was subsequently treated with chelation therapy in the form of oral dimercaptosuccinic acid (DMSA) 10 mg/kg 3 times per day for 5 days, followed by 2 times per day for 14 days. Treatment was tolerated well; serum mercury levels decreased to 59 nmol/L and repeat 24-hour urine mercury fell to 70.9 nmol/d; however, serum creatinine increased from 180 to 222 µmol/L, albumin remained 29 g/L and repeat urine PCR 890 mg/mmol. Despite no improvement in kidney function or proteinuria, the patient described feeling much better on chelation therapy, noting improvement in energy levels as well as muscle aches.
Therapeutic Focus and Assessment
As follow up mercury levels remained elevated, a second round of chelation therapy was arranged. For cost purposes, the second chelation regimen consisted of oral dimercaptopropane-1-sulfonic acid (DMPS) 100 mg 3 times per day for 14 days. Serum mercury level improved to 23 nmol/L and urine mercury improved to 16.4 nmol/d. Creatinine stabilized at 180 µmol/L and albumin improved to 33 g/L, but PCR remained elevated at 800 to 900 mg/mmol. The patient was also arranged for repeat kidney biopsy, which confirmed an immune complex glomerulonephritis with both membranous and focal segmental proliferative features, 2 cellular and 1 fibrocellular crescent (Figure 1). Twenty-five out of 56 glomeruli were globally sclerosed and there was a background of a moderate patchy interstitial lymphoid infiltrate with plasma cells and marked interstitial fibrosis and tubular atrophy. Immunohistochemical staining was positive for NELL-1 in the glomerular basement membrane and negative for anti-PLA2R.
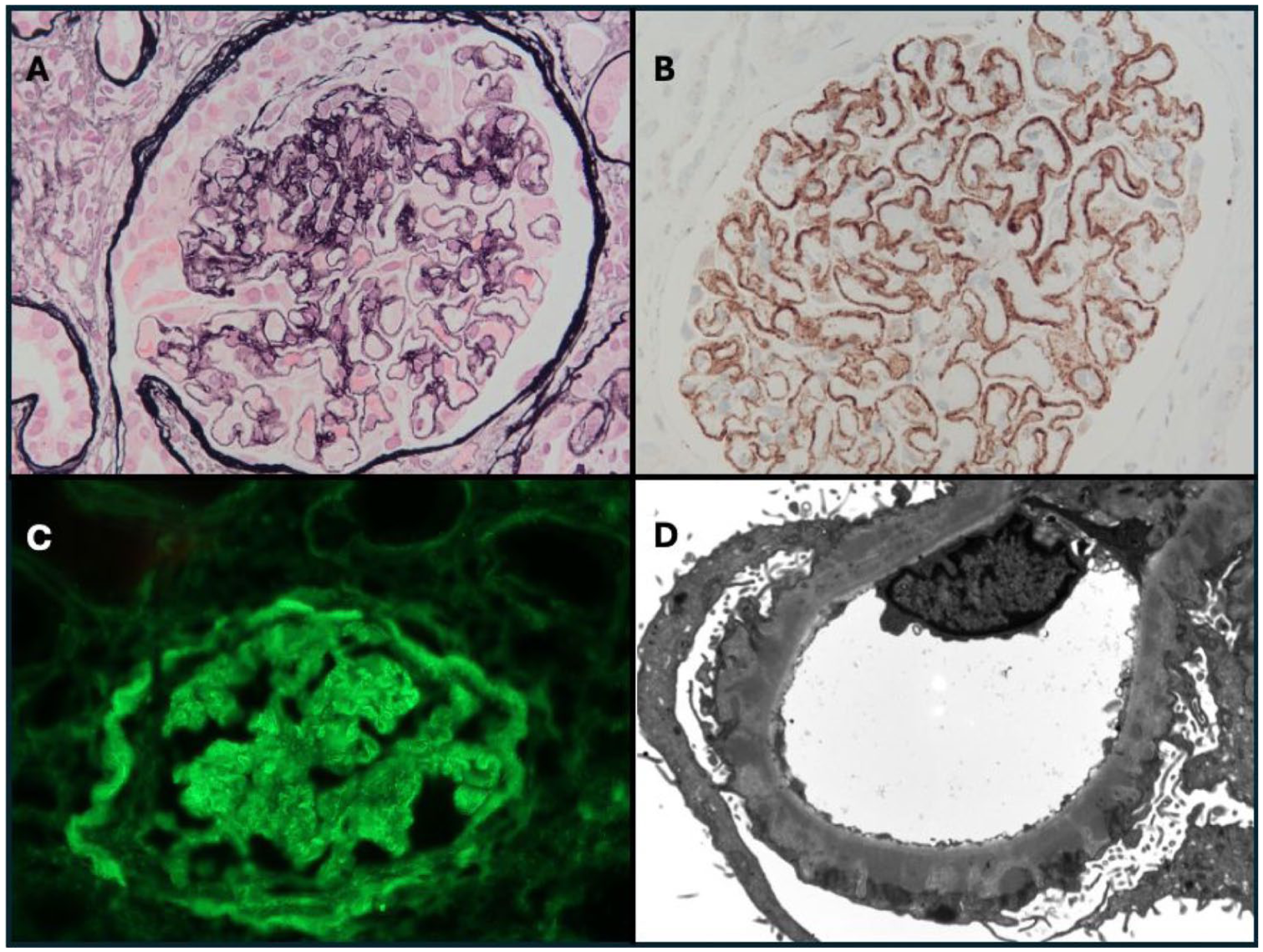
Kidney biopsy findings. (a) Glomerulus with basement membrane spikes and chains, segmental hypercellularity and a cellular crescent. Jones silver stain. 400x. (b) Glomerulus with positive staining for NELL-1 along the peripheral basement membrane. Immunohistochemistry. 400x. (c) Glomerulus with 3+ peripheral granular staining for IgG. Immunofluorescence. 400x. (d) Subepithelial electron dense deposits in glomerular capillary basement membrane, and foot process effacement. Electron microscopy. 4000x.
Follow-up and Outcomes
Given the cellular crescents and ongoing elevation in creatinine, the patient was treated with immunosuppressive therapy. Due to concerns for reactivation of TB, prednisone 1 mg/kg x4 weeks was started after treatment with isoniazid for latent TB (identified during work-up) with a plan to introduce Rituximab after several weeks of isoniazid. Unfortunately, the patient developed Varicella-Zoster infection, prompting discontinuation of treatment. After a course of valacyclovir and continued prophylaxis he was reinduced with rituximab (1g IV X2 doses separated by 14 days). Serum creatinine subsequently improved to a low of 160 µmol/L over the following month and urine PCR decreased to 497 mg/mmol.
Timeline
A timeline of presentation and treatment is summarized in Figure 2.

Timeline of patient presentation and management.
Discussion
We report a case of biopsy-proven secondary NELL-1 MN in the setting of systemic mercury intoxication related to skin fairness cream. This case highlights the importance of comprehensive medication review in patients presenting with glomerular disease. Skin fairness creams have now been implicated in cases of MN, minimal change disease and IgA nephropathy. 7 While chelation therapy may improve detectable levels of heavy metals, immunosuppression may be indicated to ultimately reduce immunologically mediated inflammation. Long-term mercury exposure and untreated glomerular disease can result in chronic changes on kidney biopsy that can be difficult to interpret, re-emphasizing the importance of longitudinal follow up in the patients and a combination of clinic, biochemical and histological data to drive management decisions.
While treatment with mercury chelation may improve kidney function and proteinuria in select cases, in this case it also resulted in improvement of patient symptoms. Symptoms of mercury intoxication can be vague and vary from chronic fatigue and weight loss to neurological manifestations such as insomnia, headache, tremors and paresthesias. 8 Chronic exposure can also lead to interstitial lung disease and symptoms of bronchitis. Interestingly, in the case of our patient, there was some improvement in cough after mercury chelation, which may support the idea that the lung findings were in part due to inhaled mercury from the facial cream.
Chelation therapy specifically for mercury-associated membranous nephropathy has been previously reported, for example with 2 cases associated with herbal medicines. 3 In these 2 particular cases, treatment was based on urine mercury levels and consisted of DMPS. More broadly, DMPS has been used as chelation therapy in patients with acute kidney injury in the setting of mercury poisoning. A retrospective analysis from 2013 to 2019 in China included 46 patients with mercury poisoning and carrying degrees of kidney injury who underwent chelation therapy with DMPS. 9 Patients in this analysis experienced remission rates of greater than 90%, regardless of whether they received additional immunosuppressant agents.
The above case is the first publication to our knowledge describing the treatment of skin fairness cream-associated NELL-1 MN with DMSA. DMSA is a sulfhydryl-containing, water-soluble, orally administered heavy metal chelator that has been shown to similarly effectively reduce renal mercury content compared with DMPS, with advantages including its more reliable commercial availability and oral administration. 10 Medication adverse effects typically consist of gastrointestinal intolerance, skin reaction, elevated liver enzymes or neutropenia. Our patient did not report any of these adverse effects and overall, tolerated both rounds of chelation without significant adverse effects.
Patient Perspective
While ultimately satisfied with eventual improvement in his nephrotic syndrome, the patient’s primary takeaway from this case was the systemic improvement noticed after starting chelation therapy. Symptoms he had been experiencing for over a year, including fatigue, brain fog, cough and tremors, all improved following initiation of mercury chelation. Since stopping the skin fairness cream and mercury levels have normalized, these symptoms have not returned. Furthermore, the patient did not experience any adverse effects from either chelation agent.
The patient’s experience further emphasizes the importance of considering chelation therapy even in the absence of significant kidney function improvement. In this case, despite a need for eventual immunosuppression, the improvement of this patient’s symptoms with chelation therapy demonstrates its value in cases of heavy metal-related nephropathies.
In conclusion, we report the treatment of skin fairness cream-associated NELL-1 MN with concurrent immunosuppression and chelation therapy in the form of both DMSO and DMSA. The evolving published cases of skin fairness cream-associated MN supports the need for wider recognition of this phenomenon, in addition to available options for treatment in the appropriate context.
Footnotes
Informed Consent
Informed consent for publication of this case report was obtained from the patient prior to submission.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
