Abstract
Rationale:
Podocyte infolding glomerulopathy (PIG) is a newly described condition with only 37 cases reported worldwide. Due to its rarity, the pathogenesis and evolution of this disease is unclear. This case report contributes to our collective knowledge about the clinical and histological progression of this disease.
Presenting concerns of the patient:
Over the course of a year, a 52-year-old Malaysian woman with no known prior medical history developed progressively worsening edema and other findings consistent with nephrotic syndrome.
Diagnosis:
Unlike most patients with PIG, this patient did not have any autoimmune disease. She was Hepatitis B core antibody positive with a Hepatitis B surface antibody >1000, suggesting prior Hepatitis B infection with immunity. A renal biopsy was performed which was consistent with PIG. A second renal biopsy was done 2 years later which again showed characteristic findings of PIG with worsened podocyte effacement but no interval change in chronicity.
Interventions:
The patient was treated with blood pressure control and renin-angiotensin-aldosterone system (RAAS) blockade with irbesartan and spironolactone. She was also treated with prednisone at 1 mg/kg for 2 months followed by a taper for a total of 7 months of prednisone treatment.
Outcomes:
The patient had a partial response to a course of prednisone. However, since stopping steroids, her proteinuria and renal function has been gradually worsening.
Teaching points:
PIG is mostly found in patients of East Asian descent. It presents as proteinuria and is often associated with autoimmune disease but can be idiopathic. It is characterized on renal biopsy by infolding or protrusion of podocyte cytoplasm into glomerular basement membrane, as well as intramembranous cytoplasmic microspherules or microtubules. Atypical membranous nephropathy should be ruled out prior to diagnosis. Unlike membranous nephropathy, PIG usually responds at least partially to steroid monotherapy. To our knowledge, this is the first reported case of PIG from North America. Furthermore, it is the first case of PIG with repeat biopsy showing interval worsening of PIG rather than either resolution of PIG or transformation of PIG to a different diagnosis.
Introduction
Podocyte infolding glomerulopathy (PIG) is a condition first described in 2008 1 with only 37 cases reported worldwide. It has been mainly described in Japanese literature 2 and usually presents as subnephrotic proteinuria, although nephrotic syndrome occurs in approximately a third of patients. It is characterized on renal biopsy by infolding or protrusion of podocyte cytoplasm into the glomerular basement membrane, as well as intramembranous cytoplasmic microspherules or microtubules. 1 We report a case of a 52-year-old woman who developed nephrotic syndrome with 2 renal biopsies performed 2 years apart, both of which confirmed PIG.
Presenting Concerns
A 52-year-old Malaysian woman with no significant past medical history and baseline urine albumin-creatinine ratio (ACR) of < 1 mg/mmol only a year prior was noted by her family physician to have mild pedal edema associated with elevated urine ACR of 146.3 mg/mmol. Her creatinine at the time was normal (61 µmol/L). In the following months, she developed new onset dyslipidemia and hypertension. Approximately 9 months later, she presented to hospital with anasarca, hypoalbuminemia, and a constellation of findings consistent with nephrotic syndrome.
Clinical Findings
At hospital, the patient was hypertensive with a blood pressure of 158/97 mmHg. Her lungs were clear with no crackles or wheezes. She was noted to have 2+ pitting edema up to her mid thighs. There were no stigmata of connective tissue disease.
Diagnostic Focus and Assessment
At hospital, her creatinine was elevated at 111 µmol/L. Her albuminuria was markedly elevated with urine ACR of 1600 mg/mmol. Albumin was 23 g/L. Urinalysis revealed >3.0 g/L protein, and moderate hematuria with 11 to 20 red blood cells (RBCs) per high-power field. Antinuclear antibody (ANA) was initially weakly positive, with normal C3 and C4. She was assessed by a rheumatologist and found to not have any connective tissue disease, and a repeat ANA was later negative. Furthermore, no antibodies to double-stranded DNA, SSA, SSB, Smith, RNP, Scl-70, Jo-1, histone, or centromere were found. Neutrophil cytoplasmic antibody was negative. She was found to be Hepatitis B core positive with Hepatitis B surface antibody >1000, suggesting prior Hepatitis B infection with immunity. Hepatitis C serology was negative. Hemoglobin A1C was 6.2%. Serum and urine protein electrophoresis was normal. Renal and pelvic ultrasounds were also normal.
The patient’s renal biopsy sample contained up to 29 glomeruli by light microscopy. The renal biopsy showed a glomerulopathy with rarefaction (irregular attenuation and pinhole changes) without epimembranous spike formation on Jones-methenamine silver-stained sections. Replication of basement membranes was not present either. Up to 25% of sampled glomeruli showed global glomerulosclerosis and approximately one third of them showed segmental sclerosis (Figures 1A and 1B). Chronic tubulointerstitial injury (fibrosis and atrophy) was seen in 20% of the sampled cortex. Direct immunofluorescence showed no significant staining for IgG, IgM, IgA, C1q, C3, kappa and lambda light chains, fibrin/fibrinogen, or albumin. Immunofluorescence on paraffin-embedded tissue after pronase digestion was negative as well and no evidence of bias toward either light chains was seen in the glomeruli, tubular basement membranes, or tubular reabsorption droplets (Figures 1C and 1D). On electron microscopy, there was diffuse foot process effacement, without typical immune-type subepithelial electron dense deposits (Figures 2A and 2B). However; there were subepithelial/intramembranous microspherules and microtubules which ranged in diameter from 25 to 80 nm (average 40 nm) with glomerular basement membrane remodeling (Figure 2C). Focal invagination of podocyte foot processes into the glomerular basement membrane (Figure 2D) was seen. Podocytes showed scattered cytoplasmic vacuoles, which were occasionally associated with mitochondrial cristae (Figure 2E). Endothelial cells showed preservation of fenestrae in the majority of capillary loops and there was no evidence of intracytoplasmic tubuloreticular inclusions (Figure 2F). Given the characteristic findings on renal biopsy, a diagnosis of PIG was favored over a diagnosis of atypical membranous nephropathy with spherules.
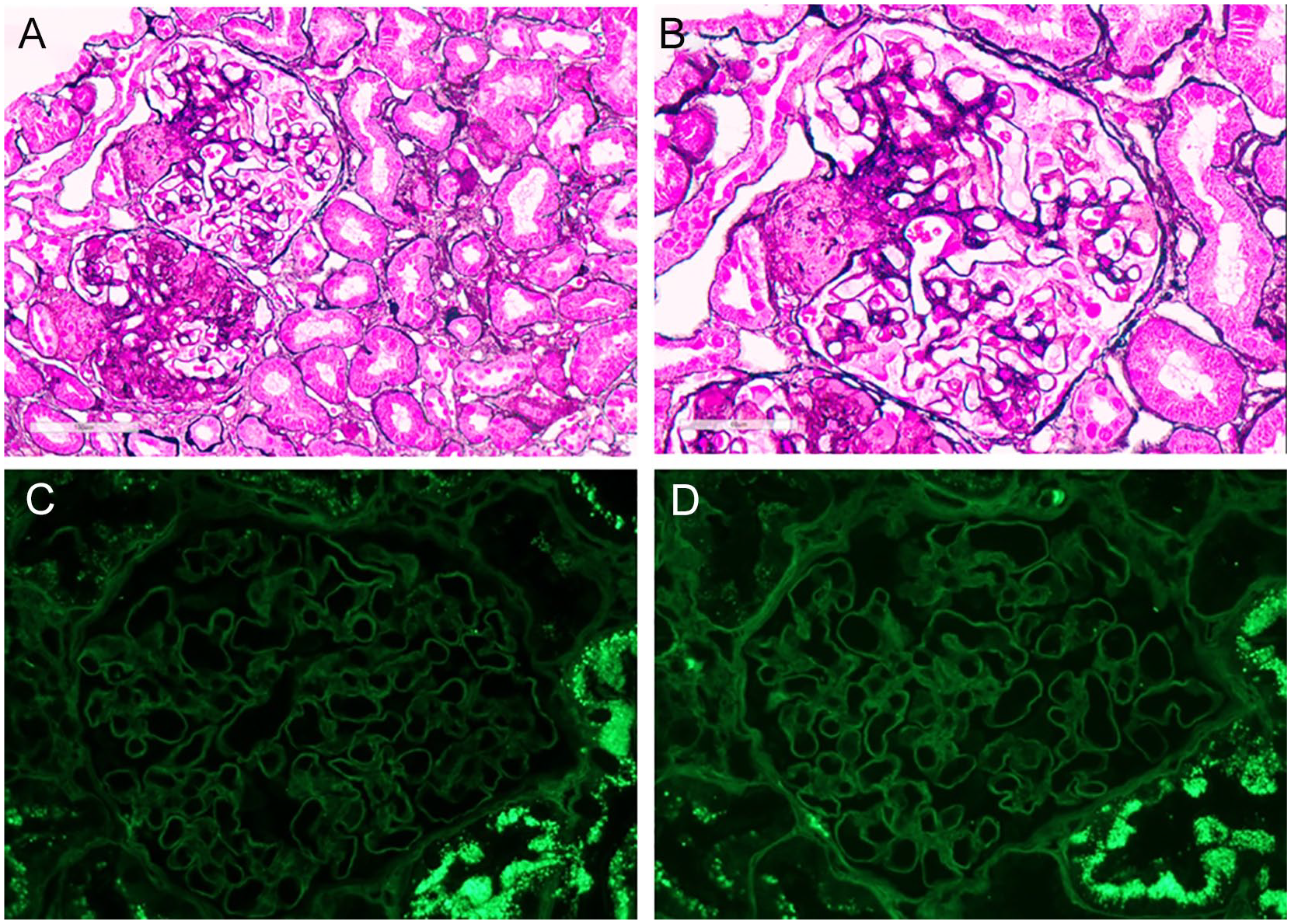
(A, B) Jones methenamine silver-stained sections of renal cortex show architecturally well-preserved tubulointerstitium. Two glomeruli are present one of which exhibits segmental sclerosis with segmental occlusion of capillary lumen by hyaline with adhesions to adjacent Bowman’s capsule (A). The other glomerulus shows mild rarefaction of glomerular basement membranes (with irregular attenuation and occasional pinholes) without replication or epimembranous spike formation (B). (C, D) Immunofluorescence staining on paraffin-embedded tissue shows no staining of glomeruli for kappa (C) and lambda (D) light chains. Tubular reabsorption droplets stain for both light chains without any bias for either light chains.
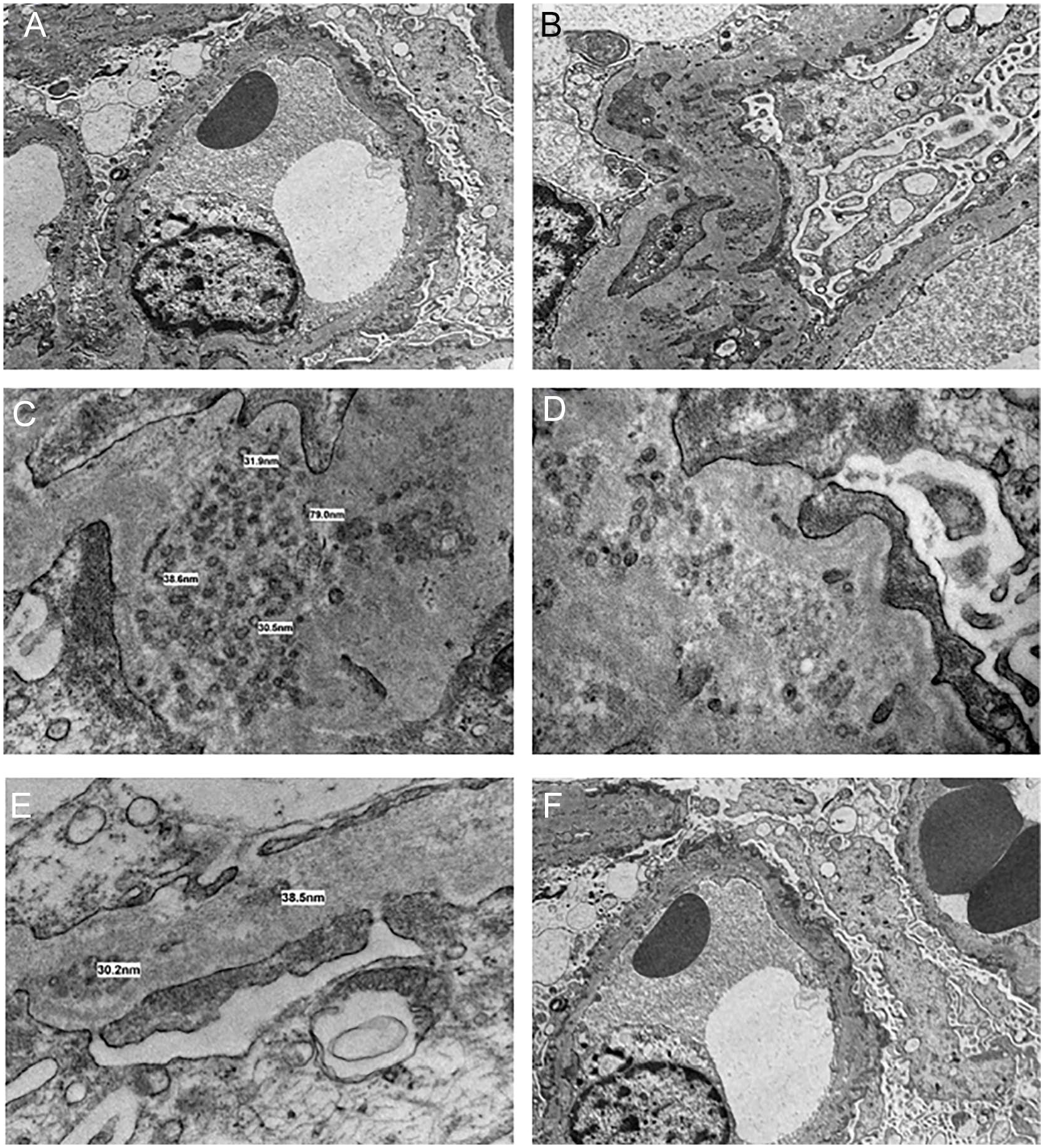
(A, B) Prominent podocyte foot process effacement. (C) Ultrastructural finding of intramembranous microspherules, 25-80 nm. (D) Invagination of foot processes into glomerular basement membrane and adjacent microtubules with scattered microspherules. (E) Podocyte with intracytoplasmic vacuole containing remnants of mitochondrial cristae. (F) Endothelial cells are flat with preserved fenestrae with no evidence of tubuloreticular aggregates.
Therapeutic Focus and Assessment
The patient was treated with irbesartan 300 mg and spironolactone 25 mg per day. She was also treated with prednisone for 2 months at 1 mg/kg, then had her dose tapered over the following 5 months. She had a partial response to steroids with a nadir in her urine ACR of 101.2 mg/mmol and creatinine of 71 µmol/L (Figure 3). Her serum albumin was 36 g/L and her nephrotic symptoms had resolved. However, after stopping steroid treatment her albuminuria partially rebounded with urine ACR of 282 mg/mmol and creatinine of 93. At the time, her blood pressure ranged from 108/70 to 143/79 in the office and was felt to be well-controlled overall. The significant worsening in albuminuria prompted a repeat renal biopsy 27 months after her initial renal biopsy to assess for disease change. This renal biopsy sample contained up to 23 glomeruli by light microscopy. As in the first biopsy, the repeat renal biopsy demonstrated features characteristic of PIG. The extent of chronic injury in the glomeruli (global glomerulosclerosis and segmental sclerosis) and tubulointerstitium (tubular atrophy and interstitial fibrosis) showed no interval increase. Immunofluorescence studies on frozen sections (direct) and paraffin-embedded tissue (with pronase digestion) were negative, consistent with prior biopsy. Additional indirect immunofluorescence for IgG subtypes, and phospholipase A2 receptor (PLA2R) were performed and returned negative. Therefore, a serum test for circulating anti-PLA2R levels was not done because the glomerular staining test for PLA2R has been shown to have superior sensitivity and specificity. 3 Congo red staining and immunohistochemical staining for DnaJ homolog subfamily B member 9 (DNAJB9, a specific and sensitive marker for fibrillary glomerulonephritis), 4 were also negative.

Creatinine (µmol/L) and urine albumin-to-creatinine ratio (mg/mmol) over time.
Follow-Up and Outcomes
Eleven months after being tapered off prednisone, the patient’s urine ACR was now 364.8 mg/mmol and creatinine was now 105 µmol/L. Her serum albumin was 40 g/L. Other than her significant proteinuria, she did not have other features of nephrotic syndrome. However, based on the repeat biopsy findings of worsened podocyte effacement with no increase in disease chronicity, the decision was made to re-treat her with a course of prednisone. Just 3 weeks after starting prednisone, her urine ACR decreased to 174.8 mg/mmol and her edema improved. Her creatinine remains stable at 108 µmol/L.
Discussion
There have only been 38 reported cases of PIG worldwide so far, including the present report. The most recent case review identified 27 cases from Japan, 2 cases from China, 1 case from South Korea, and 1 case from India. 2 Since then, there has been an additional 1 case identified from Japan, 5 3 cases from China,6-8 1 case from Germany, 9 and 1 case from Argentina. 10 To our knowledge, we are the first to report a case of PIG in North America.
Most patients with PIG have underlying autoimmune diseases such as lupus nephritis, Sjögren syndrome, mixed connective tissue disease, rheumatoid arthritis, and autoimmune thyroiditis, 2 all of which were ruled out in our patient. One patient with PIG also had multiple myeloma, 11 but serum and urine protein electrophoresis were both normal in our patient and there was no reason to suspect a lymphoproliferative disease otherwise. There have also been a few cases associated with hydronephrosis, 2 but our patient had renal imaging which ruled out hydronephrosis. One case has previously been associated with hepatitis B infection, 12 and 4 cases have been idiopathic. 2 The patient described in this case report does have a positive hepatitis B core antibody, but given that her hepatitis B surface antigen was negative at the time of disease presentation, her PIG is most likely idiopathic and unrelated to hepatitis B.
The histology of PIG can be challenging to differentiate from other glomerular diseases. Microstructures in the glomerular basement membrane are usually not seen in membranous nephropathy, but can be seen in stage 3 membranous nephropathy and rare cases of HBV-associated glomerulonephritis. 1 However, in these HBV variants of membranous glomerulonephritis, the microstructures are still highlighted on immunofluorescence for one or more immunoglobulins, predominantly IgG, 13 which was not seen on the renal biopsies from this patient. We considered the possibility of membranous nephropathy with masked IgG kappa deposits. However, in both biopsies conducted on this patient, masked epitopes were assessed for via immunofluorescence studies on paraffin-embedded tissue that uses a pronase digestion step and no immune deposition was observed, ruling out membranous nephropathy with masked deposits. We also considered a diagnosis of membranous nephropathy with spherules, but again, positive immune staining is usually seen in this disease, 14 which was not observed in our case. Furthermore, membranous nephropathy with spherules has distinct subepithelial deposits with spherule-like substructure, 14 whereas in our case the distribution of spherules was more scattered. The possibility of remote or resolving membranous nephropathy was also considered. However, the patient’s clinical worsening and repeat biopsy results 2 years after the original biopsy showing worsened podocyte effacement but no immune deposition and no increase in chronicity argues against this possibility.
Worldwide, only 4 cases of PIG have had repeat biopsies performed. Two patients had class II lupus nephritis which converted to PIG.15,16 One patient had PIG associated with vesicoureteral reflux (VUR) with hydronephrosis that resolved after Foley placement. 17 The fourth patient had PIG that became focal segmental glomerulosclerosis (FSGS) with non-specific staining for IgM, C3, and C1q. 18 The case presented in this report offers more insight into the pathological evolution of PIG in a woman with partial response to a course of steroids. To our knowledge, this is the first case with repeat biopsy showing interval worsening of PIG rather than either disease resolution or a new diagnosis. This helps support the hypothesis that PIG can be a distinct disease entity.
The pathogenesis of PIG remains poorly understood. Of note, the case we present showed segmental sclerosis. An association between PIG and segmental sclerosis has been reported in the largest case series and other case reports,2,8,10 with 21.6% of cases (8/37) having segmental sclerosis as the primary diagnosis by light microscopy. It is possible that PIG is a manifestation of a form of podocytopathy that leads to altered podocyte/basement membrane interactions with incorporation of podocyte cell membranes into the glomerular basement membranes. Ultrastructural studies showed that many podocytes contain cytoplasmic vacuoles, some with remnant cristae suggestive of mitochondrial origin. Prior research has proposed a prognostic value for cytoplasmic vacuolization of podocytes in pediatric population with FSGS. 19 However, whether this finding points to mitochondrial dysfunction as a cause or consequence of podocytopathy remains unknown. Similarly, whether the underlying pathology is genetic or molecular (posttranslational) can only be hypothesized at this stage. Xiong et al reported a case of PIG in a 4-year-old boy with Schimke immune-osseous dysplasia (SIOD) with a novel SMAR-Cal1 gene mutation. 8 Although the finding in the cited case report can represent a chance association rather a causal event, the report highlights the need and utility of genetic and molecular studies in case series to uncover pathogenic pathways in the development of this characteristic ultrastructural finding. On the other hand, the presence of microspherules in other conditions, such as in association with HBV and/or membranous nephropathy may suggest that some cases of PIG are driven or promoted by factors extrinsic to podocytes (such as immune complex deposition). In fact, some of the early cases described in the original reports also stain positively for immunoglobulins by immunofluorescence. 2 Whether these cases represent the collision of 2 separate entities (ie, membranous nephropathy and PIG) or the spherules are from organization of the deposits is not fully examined in literature and requires further understanding of the underlying pathogenesis of PIG. However, it is our opinion that cases with microspherules that co-localize to subepithelial immune deposits (with concomitant positive immunostaining) can be best categorized as membranous nephropathy with spherules. 14 Another possible hypothesis for the mechanism of PIG is that the microspherules may simply represent an exaggerated wear and tear response of podocytes. Overall, the best chance of shedding light on the pathogenesis of this heterogeneous group of disease would require an “omics” approach on a number of cases of PIG with genomics (such as whole genome sequencing) and proteomics (mass spectrometry) being among the chief methodologies utilized.
Proteinuria in PIG is in the subnephrotic range in the majority of cases,2,7 with 28.6% (10/35) that have proteinuria in the nephrotic range,2,8,9 and 27.7% (10/36) that are described as having nephrotic syndrome.1,8-10 Of the case reports that documented patient outcomes, 45.2% (14/31) described complete remission of proteinuria, defined as a 24-hour urine protein of ≤0.3 g/day.1,8,9,20 Another 45.2% (14/31) patients had partial improvement in proteinuria.1,2,11,18,21 Remission was usually in response to immunosuppressive therapy. However, there were 3 patients who had complete remission of PIG while not taking immunosuppressant medication.1,8 The remaining 9.7% of patients (3/31) described in the literature had either no change or worsening in proteinuria in response to immunotherapy. 1 There have been no documented cases of progression to end-stage renal disease or transplantation. The reportedly high rate of remission of PIG could be in part due to publication bias of successfully treated cases. Follow-up time is also variable, ranging from 1 month to 5 years. 1 As seen in the case of PIG we report here, the disease course fluctuates over time and remission in response to steroids may not be permanent, requiring repeat treatment. It has also been proposed that PIG responds better to steroid monotherapy than membranous nephropathy due to its association with autoimmune disease. 11 This is supported by the fact that out of the 5 idiopathic cases of PIG (including this one), only one patient achieved complete remission with steroids, which is a significantly lower remission rate than the overall remission rate in PIG. On the other hand, all 3 patients with PIG that did not respond to steroid treatment had a concurrent diagnosis of systemic lupus erythematosus (SLE). Ultimately, there are too few reported cases of PIG to draw any definite conclusions.
The vast majority of case reports of PIG come from Asia, although there are 2 case reports in people of white background, 1 from Germany 7 and the other from Argentina. 8 To our knowledge, we are the first to report a case of PIG in North America. Considering the diverse ethnic population found in North America, and in particular large Asian communities both in Canada and the United States, it is likely that the absence of case reports from North America represents under-recognition or under-reporting of this rare entity. By reporting our findings, we hope to raise awareness of this disease in North America, especially since treatment of PIG appears to differ from rival diagnoses such as atypical membranous nephropathy, in that PIG often responds to steroid monotherapy. 2 We are also the first to report a case of PIG with repeat biopsy showing interval worsening of PIG rather than either resolution of PIG or transformation of PIG to a different diagnosis. Thus, we offer insight into the clinical and pathological evolution of the disease and, with rigorous exclusion of other potential causes, support building evidence that PIG is a morphologically distinct entity on the spectrum of podocytopathy. Meta-analysis of existing case reports and genetic/molecular studies are required in the future to gain a better understanding of the pathogenesis and optimal management of this disease.
Footnotes
Ethics Approval and Consent to Participate
Informed consent was obtained from the patient for the publication of an anonymized case report, including renal biopsy images.
Consent for Publication
All authors have reviewed the manuscript and consented to publication.
Availability of Data and Materials
Request can be made to the corresponding author for access to the data and materials included.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
