Abstract
Acute kidney injury (AKI) in non-Hodgkin lymphoma has diverse etiologies. We report a case in which AKI due to light chain cast nephropathy was the initial manifestation of extranodal marginal zone lymphoma, occurring without systemic symptoms. A 64-year-old male presented with severe AKI without other symptoms. His physical examination and renal ultrasound were unremarkable. Renal biopsy revealed light chain cast nephropathy, and a subsequent bone marrow biopsy confirmed marginal zone lymphoma. The patient received R-CHOP chemotherapy (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisolone) for a total of six cycles. The patient had a partial response to lymphoma. However, his renal function did not improve, and ultimately he progressed to end-stage kidney disease, requiring maintenance hemodialysis. This case highlights extranodal marginal zone lymphoma presenting as AKI, emphasizing its unique renal-limited manifestation in the absence of systemic symptoms and the critical role of renal biopsy in diagnosing unexplained AKI.
Introduction
Non-Hodgkin lymphoma (NHL) is a diverse group of hematologic malignancies that includes both indolent and aggressive subtypes. Marginal zone lymphoma (MZL), an indolent B-cell lymphoma, has three main types: extranodal (EMZL), nodal (NMZL), and splenic (SMZL). EMZL constitutes about 8% of all B-cell lymphomas, with common sites of involvement including the gastrointestinal tract, ocular adnexa, and lungs. 1 Renal complications in lymphoma are common, with acute kidney injury (AKI) significantly increasing the morbidity and mortality in patients. 2 AKI in lymphoma often arises from multifactorial mechanisms such as tumor lysis syndrome, paraproteinemia, or glomerulonephritis. 2 AKI due to light chain cast nephropathy, resulting from excessive light chain production is rare in non-myeloma B-cell lymphomas. We report an unusual case of a patient who presented with AKI as the initial manifestation, in the absence of anemia, lymphadenopathy, or B symptoms, and was subsequently diagnosed with EZML.
Presenting Concerns
A 64-year-old male presented to Lampang Hospital in September 2021 with persistent, non-radiating bilateral flank pain. He reported no fever, dysuria, hematuria, or edema and denied systemic symptoms such as weight loss or night sweats. His medical history included hypertension and gout, and he regularly used non-steroidal anti-inflammatory drugs (NSAIDs). His medications included allopurinol, colchicine, diclofenac, paracetamol combined with orphenadrine, omeprazole, furosemide, calcium carbonate, and enalapril.
Clinical Findings
The patient’s blood pressure was 145/89 mm Hg, with no signs of anemia, lymphadenopathy, or edema. Initial laboratory tests showed a significantly elevated serum creatinine level of 860.0 μmol/L, up from a prior baseline of 97.2 μmol/L. His electrolyte levels were as follows: sodium 136 mmol/L, potassium 4.7 mmol/L, chloride 105 mmol/L, bicarbonate 13 mmol/L, calcium 2.1 mmol/L, phosphate 2.1 mmol/L, uric acid 535.0 μmol/L, albumin 34 g/L, and globulin 32 g/L. Liver function tests were within normal limits. Complete blood count showed a hemoglobin level of 119 g/L, leukocyte count of 7.6 × 109/L, and platelet count of 136 × 109/L. Serology testing for antineutrophil cytoplasmic antibodies was negative. Urinalysis revealed albumin at 1+ and trace blood. A 24-hour urine collection showed significant proteinuria at 2.7 grams per day. Microscopy showed 0-1 red blood cells per high-power field (HPF) and 1-2 white blood cells/HPF, with no casts present in the sediment. Renal ultrasound revealed normal-sized kidneys with increased parenchymal echogenicity but no signs of stones, solid masses, hydronephrosis, or perinephric collections.
Diagnostic Focus and Assessment
The patient was diagnosed with NSAID-induced AKI, felt to be related to acute tubular necrosis (ATN). Diclofenac and enalapril were discontinued. After 4 weeks of supportive treatment and regular follow-up, his AKI showed no improvement, and hemodialysis was initiated. Given the lack of improvement after NSAID discontinuation, a course incompatible with the typical recovery timeline of ATN, and the presence of subnephrotic-range proteinuria, a renal biopsy was performed.
Histopathology findings revealed tubular injury with contained periodic acid-Schiff stain (PAS)-negative casts with a sharp-edged, fractured appearance, surrounded by mononuclear cells. Extensive interstitial fibrosis was noted, along with moderate interstitial inflammation, but no glomerular pathology. Congo red stain was negative, and no abnormal plasma cells were detected. Immunoperoxidase staining was negative. The findings were consistent with cast nephropathy (Figure 1). Following the biopsy results, further investigation to identify the cause of light chain cast nephropathy was undertaken. Serum protein electrophoresis showed polyclonality. In addition, serum testing revealed elevated kappa free light chain levels of 4836.0 mg/L (normal range: 3.3-19.4 mg/L), normal lambda light chain levels (20.2 mg/L), and a kappa/lambda ratio of 239, suggestive of monoclonal gammopathy. A subsequent bone marrow biopsy confirmed EMZL. Immunohistochemistry staining was positive for CD20, BCL2, and kappa light chain, while negative for CD3, CD5, CD10, CD23, and lambda light chain. Notably, there was no evidence of plasma cells (Figure 2). A computed tomography (CT) scan showed mild lymphadenopathy in the chest and abdomen. A positron emission tomography (PET) scan was not performed due to financial constraints. The patient was ultimately diagnosed with stage IVe EMZL with light chain cast nephropathy.

Renal histology.
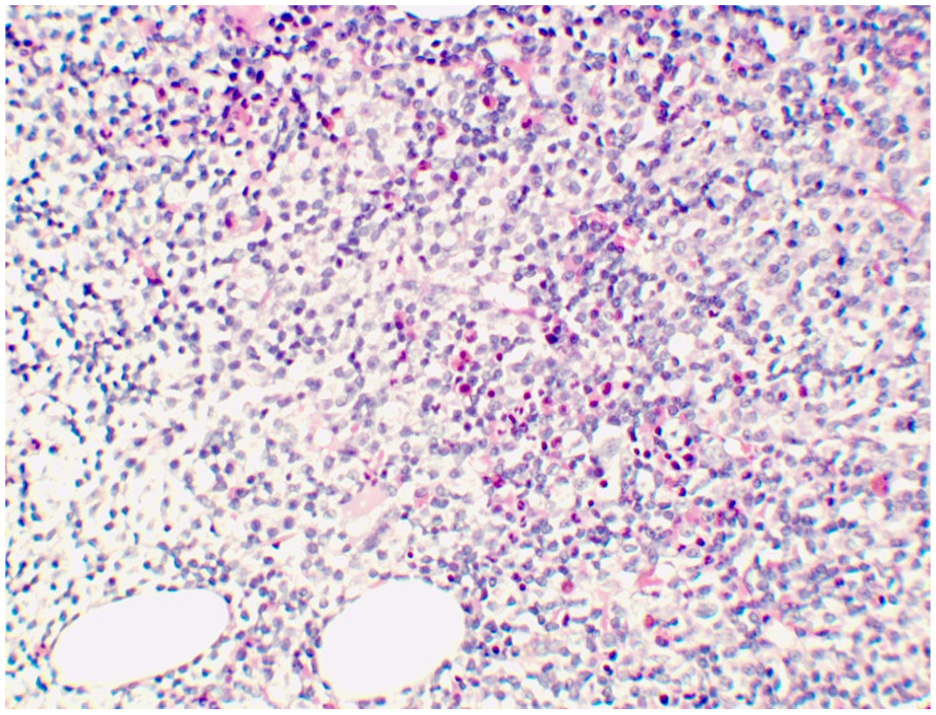
Bone marrow histology.
Therapeutic Focus and Assessment
The patient began treatment with R-CHOP chemotherapy (rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisolone) for a total of 6 cycles.
Follow-up and Outcomes
Following chemotherapy, a follow-up CT scan showed a reduction in the size of multiple paraaortic lymph nodes, with the largest decreasing from 1.4 cm to 0.7 cm. Other mild lymphadenopathy remained stable. A repeat bone marrow biopsy showed no tumor infiltration, and immunohistochemistry for CD20 was negative, with no evidence of myeloma. Serum free light chain levels decreased in accordance with the treatment timeline (Figure 3). Although his lymphoma partially responded, his renal function did not recover, and he progressed to end-stage kidney disease (ESKD), necessitating maintenance hemodialysis.

Timeline of treatment.
Discussion
Renal involvement is common in lymphoproliferative diseases, with AKI occurring in up to 60% of patients with lymphoma. AKI in such patients is particularly concerning due to its association with increased mortality and prolonged hospital stays. The etiology of AKI in lymphoma varies and may include tumor lysis syndrome, volume depletion, direct tumor infiltration, and intrarenal mechanisms. 2 Given this complexity, it is critical to accurately identify the cause of AKI in lymphoma, as recovery of renal function largely depends on treating the underlying condition.
In this case, the patient presented with AKI and a history of NSAID use, suggesting a possible drug-related nephrotoxic injury. Despite discontinuing NSAID and enalapril, and providing supportive treatment, renal function did not improve, prompting a renal biopsy. Renal biopsy is recommended in cases of unexplained AKI to achieve a definitive diagnosis, guide treatment, and provide insight into prognosis. 3 The renal biopsy findings in our patient revealed diffuse tubular injury and PAS-negative casts with sharp edges, while intact glomeruli were characteristic of light chain cast nephropathy.
Light chain cast nephropathy has been recognized as a significant cause of AKI in plasma cell disorders, particularly in multiple myeloma where it is often termed “myeloma cast nephropathy.” 4 When free light chain production exceeds proximal tubular reabsorptive capacity, these chains accumulate in the distal nephron, bind to Tamm-Horsfall protein, and form casts that obstruct and damage the tubules, leading to AKI.5-7 Light chain cast nephropathy has been recognized as a myeloma defining event. In contrast, non-Hodgkin B-cell lymphomas, such as EMZL, are less frequently associated with cast nephropathy, making this case particularly unusual.4,8 Typically, patients with lymphoma-associated AKI present with systemic symptoms, including lymphadenopathy, hepatosplenomegaly, and cytopenias. 9 In contrast, our patient’s only initial symptom was severe AKI, without systemic manifestations, making the presentation atypical.
While serum and urine free light chain measurements, along with serum protein electrophoresis, serve as initial screening tools, a bone marrow biopsy and flow cytometry may be necessary to identify an underlying hematologic malignancy. 10 In our patient, renal biopsy was essential in diagnosing cast nephropathy, and a subsequent bone marrow biopsy confirmed EMZL. Prompt diagnosis is crucial, as early treatment of the hematologic malignancy may improve renal outcomes. Treatment for cast nephropathy involves reducing free light chain production, usually through chemotherapy, along with supportive measures such as hydration and correction of electrolyte imbalances. Renal outcomes closely depend on the reduction in serum free light chains in response to treatment, underscoring the need for early intervention. 10 Unfortunately, light chain cast nephropathy often has a poor prognosis for renal recovery, with many patients progressing to ESKD. 9
In summary, this case represents a rare and exceptional instance of EMZL manifesting solely as severe AKI due to light chain cast nephropathy, notably in the absence of systemic symptoms or typical extrarenal lymphoma features. This atypical presentation underscores two critical points: First, light chain cast nephropathy can be the sole initial manifestation of EMZL, even without extrarenal involvement, highlighting the unique pathophysiology of such cases. Second, renal biopsy remains crucial in cases of unexplained AKI, as it enables the identification of underlying hematologic malignancies that may not be apparent otherwise. Early biopsy and diagnosis are essential in guiding timely and effective treatment ultimately impacting patient outcomes.
Footnotes
Acknowledgements
We acknowledge Ms. Natasha Goga for English language editing.
Author Contributions
Thanawat Vongchaiudomchoke (TV) (corresponding author): conception and design, analysis and interpretation of data, drafting of the manuscript, final approval of the manuscript submitted; Naphasorn Naruemon (NN): conception and design, analysis and interpretation of data, drafting of the manuscript, final approval of the manuscript submitted; Piriyaporn Iamsai (PI): analysis and interpretation of data, final approval of the manuscript submitted; Piyapong Ounpanyo (PO): analysis and interpretation of data, final approval of the manuscript submitted; Boonyarit Cheunsuchon (BC): analysis and interpretation of data, final approval of the manuscript submitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study protocol was reviewed and approved by the Lampang Hospital Ethics Committee, approval number EC 091/67.
Informed Consent
Written informed consent was obtained from the patient who participated in this study for his participation in and publication of this study and any accompanying images. A written informed consent obtained from the patients is available from the corresponding author upon reasonable request.
Data Availability
Data can be accessed from the corresponding author upon reasonable request.
