Abstract
Introduction:
Tracking the evaluation process of living kidney donor candidates facilitates benchmarking and can inform process redesign to improve experiences with the evaluation and enable more living donor kidney transplantation.
Methods:
We reviewed the medical records for all living donor candidates who were actively undergoing evaluation at any time between January 1, 2013, and December 31, 2016, at the London Health Sciences Centre in London, Ontario, Canada. We abstracted information on demographic factors, the evaluation process, reasons for a delayed evaluation, reasons for an evaluation termination (eg, donation, decline, withdrawal, loss to follow-up), frequency and timing of evaluation testing, and recipient dialysis status.
Results:
Over time, the number of living donor kidney transplants increased from 22 in 2013 to 32 in 2016 (18% and 34% of which were pre-emptive, respectively). The median number of candidates coming forward doubled from 167 in 2013 (2 candidates per recipient) to 348 in 2016 (4 candidates per recipient). Median time from first contact until donation decreased from 12.8 months in 2013 to 7.1 months in 2016 (a 45% reduction). The time from computed tomography (CT) angiography until donation (n = 74) was a median of 75 (interquartile range [IQR] = 36, 180) days, the longest single step in the evaluation. Common reasons for delay included waiting for the referral of their intended recipient for transplant evaluation (11% of candidates) and a need for the donor candidate to lose weight (8% of candidates). Donors completed the main evaluation tests on a median of 5 different dates. Thirty-six recipients started dialysis after their living donor candidates’ evaluation had been underway for at least 3 months.
Conclusion:
Tracking the steps and reasons for an inefficient living kidney donor evaluation process can be used for quality improvement, and efficiency improvements are expected to translate into improved outcomes and experiences.
Introduction
Transplant centers must thoroughly evaluate potential living kidney donor candidates to determine whether they are eligible to donate. 1 The evaluation process includes counseling the candidate on what to expect during the donation process, and a series of steps to minimize the risk of adverse medical and psychosocial outcomes following donation.2-4 However, a prolonged evaluation is associated with donor candidate attrition, a lower likelihood of living donor transplantation, and a lower likelihood of a pre-emptive transplant.5-9
Given the impact of the length of evaluation time on donation, tracking the process and outcome indicators is critical to evaluate the performance of the current donor evaluation process. 10 Process indicators use well-defined time points as anchors to measure the efficiency of the evaluation and can identify preventable bottlenecks. Outcome indicators measure the effect of a change in a process (eg, the number of living donor transplants and the number of potential pre-emptive transplants lost). 10 Both process and outcome indicators can provide important information to transplant centers on candidates who ultimately donate and those who do not proceed.
We conducted a detailed retrospective cohort study reviewing the medical records for all living donor candidates who contacted a single medium-sized transplant center in Ontario, Canada. We demonstrate how key process and outcome indicators can be used to track the performance of transplant centers and identify potential opportunities for quality improvement. 10
Methods
This study included all persons who contacted or were actively being evaluated at the London Health Sciences Centre (LHSC) living kidney donor program (London, Ontario, Canada) for some or all of the study period (January 1, 2013, to December 31, 2016). This is considered to be a medium-sized program in Canada.
Medical Record Review
De-identified information from medical records was retrieved and analyzed. Medical records included clinic notes, diagnostic and laboratory test results, results of the preliminary screening checklist, living donor coordinator notes, and a self-reported multi-paged medical-social questionnaire (MSQ). The MSQ asked candidates to provide information about their medical history, family history, travel history, and anticipated time of donation if eligible. Data on donor candidate demographics, social habits (eg, smoking history, alcohol use), and medical characteristics were also abstracted. Blood pressure and body mass index (BMI) were typically self-reported and values were averaged from 1 to 5 measurements. In order to determine rural or urban residence, we used the second digit of the Canadian postal code (rural if zero; urban otherwise). Persistent hematuria was defined as the definitive presence of blood in the urine in at least 50% of urine samples (Supplemental Appendix 1 for further detail).
Recipient data for the same study period were obtained from a local transplant database, including the date a recipient started dialysis (if applicable), the date a transplant candidate was referred for transplant evaluation, and the date candidates were placed on the deceased donor waitlist. During the study period, the living donor program would wait for the general nephrologist to complete initial testing for the intended recipient before starting the donor work-up.
Statistical Analysis
Abstracted data were recorded using the Research Electronic Data Capture (REDCap) tool. 11 Results are reported using appropriate descriptive statistics, including proportions, mean (standard deviation [SD]) and/or median (interquartile range [IQR] 25th, 75th percentile). Analyses were performed using Statistical Analysis Software (SAS Institute Inc, Cary, North Carolina).
Results
Donor Evaluation Activity
During the study period, 103 living donor transplants were performed and 939 living donor candidates contacted the program. With follow-up until December 2017, 95/939 (10%) of these contacts donated. When restricted to candidates with a known recipient (n = 860), the median number of candidates coming forward for the same recipient was 3 (IQR = 1, 7), ranging from 1 to 24; mean = 5.5 (SD = 6.10).
Donor Candidate Characteristics
Most of the 939 living donor candidates who contacted the transplant center during the study period did so by phone (65%) followed by e-mail (31%). Candidates were predominantly female (n = 614; 67%), age = 45.6 (SD = 13.8) years, Caucasian (n = 424; 85%), lived a median driving distance 125 (IQR = 50, 207) kilometers from the LHSC, and intended to donate to a friend or non-relative (n = 181, 21%), distant relative (n = 175, 20%), sibling (n = 167, 19%), spouse (n = 98, 11%), parent (n = 89, 10%), child (n = 81, 9%), or non-directed (n = 67, 8%) (Table 1). A total of 37 candidates were flagged as having been enrolled in the Kidney Paired Donation (KPD) program.
Sociodemographic Characteristics of Candidates Contacting the Living Donor Program Between 2013 and 2016.

Follow-up to establish whether donation occurred was until December 31, 2017.
One non-donor candidate and 1 donor had to remove their kidney for their own health care needs.
Smoking status at the time the evaluation started, determined using the clinical documentation or candidate self-report on the medical-social questionnaire.
SD = standard deviation; IQR = interquartile range.
Flow of Candidates
Figure 1 describes the flow of living kidney donor candidates through the evaluation process, along with the activities that occurred in these phases. Of 939 contacts, almost half (n = 427; 45%) did not complete the self-reported 12-page medical-social questionnaire needed to begin the evaluation. Of the 512 candidates who completed the questionnaire, 205 (22%) did not proceed past the advanced screening phase. The most common reason was ineligibility, the evaluation was placed on hold (152 candidates), donor lost to follow-up (24 candidates, described below), or the evaluation was still active or ongoing as of December 31, 2016 (27 candidates).

Flow of candidates through the evaluation process.
Reasons for Attrition
Attrition was considered if there was documentation of a cessation of the evaluation process for that candidate, or no response from the candidate after 3 months since the last contact (loss to follow-up). Living donor coordinators would attempt to contact the potential donor by email or voice message with a gentle reminder to contact us if still interested. If there was no response after 3 months, then the file would be closed and there were no additional contact attempts. Donor lost to follow-up was the most common reason for attrition (Table 2). Other reasons for the donor not proceeding with the evaluation included the donor being deemed medically unsuitable, other donors being prioritized, and donor needing to change lifestyle (eg, smoking cessation is a requirement). The 2 most common reasons for attrition due to the recipient included the recipient not being ready for transplant assessment or the recipient receiving another transplant (living or deceased). Recipient death or development of a condition that made them transplant-ineligible occurred in only in 6 patients, 4 of whom had a donor who had passed the advanced screening phase (Table 2).
Reasons for Attrition During Evaluation.
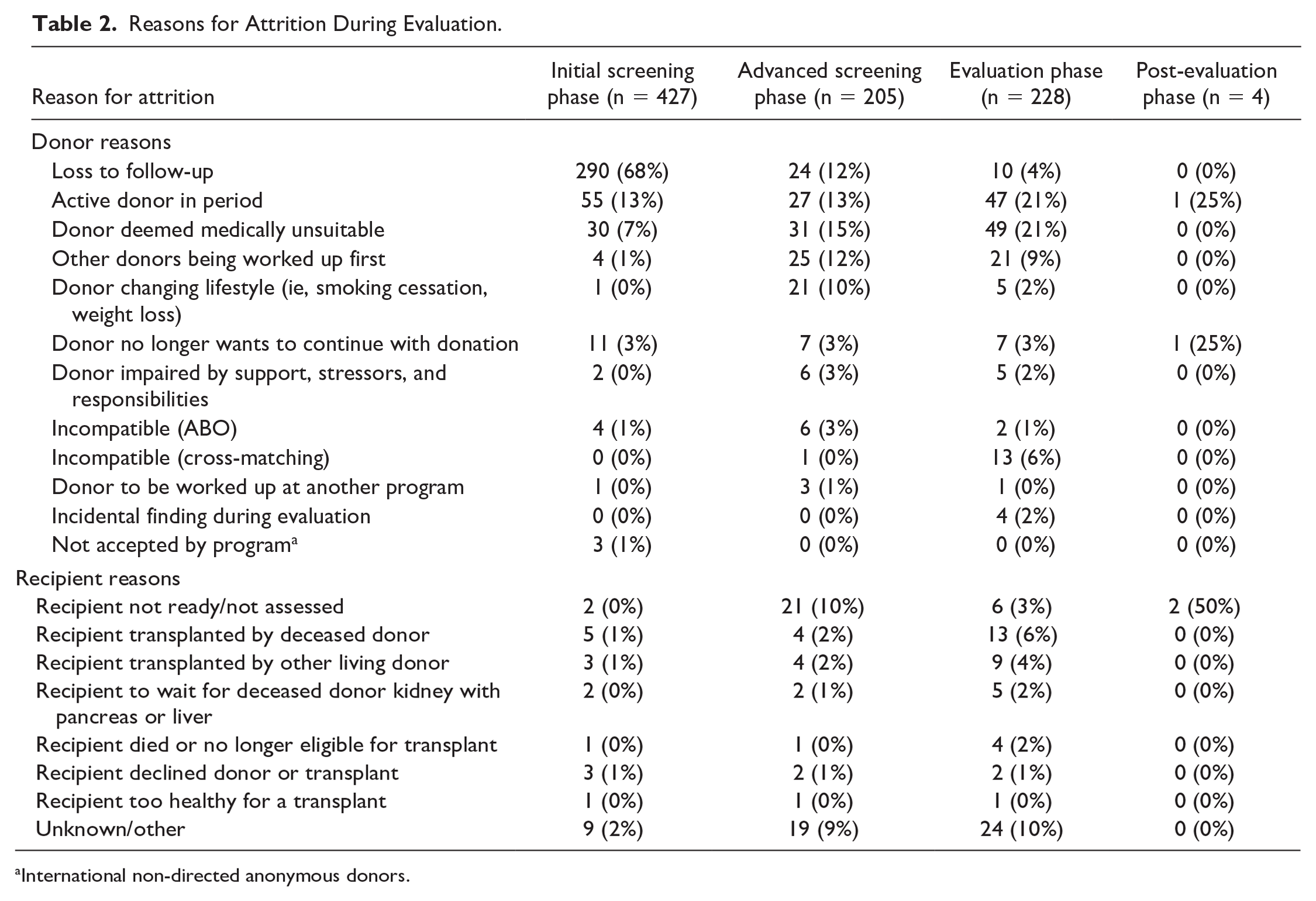
International non-directed anonymous donors.
Reasons for Delay
A delay in the evaluation was considered if there was documentation of a circumstance that stalled the evaluation with the intent that it resumes after the circumstance was resolved. The most frequently cited reason for delay was that the recipient was not assessed yet (n = 99; 10.5%), which typically occurred early in the donor evaluation process (Supplemental Appendix 2). Other early delays included that the donor had to lose weight (n = 74; 7.9%) or quit smoking (n = 30; 3.2%). Delays occurring in the middle of the process include incidental findings during donor evaluation (n = 37; 3.9%) and the recipient was not ready to proceed (n = 17; 1.8%). A total of 35 candidates were waiting, as other donor candidates were prioritized for the intended recipient (n = 35; 3.7%).
Timing of Donor Evaluation Process
Among donors who donated between 2013 and 2016, the time from first contact until donation was a mean of 13.7 (SD = 15.7) and median of 9.2 (IQR = 6.1, 14.0) months. For candidates who had a date of withdrawal or decline available (n = 290, excluding those lost to follow-up), the total evaluation time was a mean of 6.2 (SD = 6.1) and median of 4.3 (IQR = 1.4, 9.1) months. A total of 667 recipients were referred to the transplant center a median of 9 (IQR = −108, 104) days after the donor candidate’s first contact date. Of these, 532 were activated on the deceased donor waitlist after the candidate’s first contact a median of 175 (IQR = 63, 306); mean = 127 (SD = 572) days.
The transplant program received the completed MSQ a median of 19 days after it was sent to the candidate (email, or hard copy mail), and the time from first contact until the center reviewed the questionnaire took a median of 40 (IQR = 22, 90) days (Supplemental Appendix 3). Since delays in this step may be out of the control of the transplant center, we use the date the center received the completed questionnaire as the point of reference when expressing the time to complete various aspects of the evaluation (Figure 2). 9 The renal ultrasound, chest X-ray, and electrocardiogram were completed a median of 40 days after this time point; the initial crossmatch, psychosocial evaluation, and nephrology consult after a median of 50 to 57 days; and the pelvic/abdominal computed tomography (CT) scan, nuclear renogram, and surgical consult after a median of 82 to 89 days. Donors completed these tests on a median of 5 different days (range = 2-9; mean = 4.7 [SD = 1.4]). For candidates who completed a nephrology, urology, and psychosocial assessment (n = 127), the time between consults was a median of 27 (IQR = 4, 69) days. A nuclear renogram occurred at a median of 0 (−35, 4) days following the CT scan (when done, the result is a negative value when the renogram preceded CT angiography). The time from CT until donation (n = 74) was a median of 75 (IQR = 36, 180) days.

Timing of milestones in the living donor evaluation process. The date the medical-social questionnaire was reviewed by the program was used as the start of the evaluation (time 0). Median (25th, 75th) percentile is reported.
The first recipient serum collection used for crossmatch testing was a median of 19 (−43, 102) days after the first donor candidate contact. The initial crossmatch was performed a median of 118 (63, 203) days after the first contact (n = 186), with the final crossmatch conducted a median of 95 (56, 133) days afterwards. Other tests required on an individual-level basis included ambulatory blood pressure monitoring (n = 54), echocardiography (n = 20), stress tests (n = 19), and 24-hour urine analysis for kidney stones (n = 13). The transplant center organized additional consultations with other health care professionals on an individual basis, including dietitians (n = 20), obstetricians/gynecologists (n = 11), transplant infectious disease specialists (n = 11), cardiologists (n = 9), hepatologists (n = 5), gastroenterologists (n = 5), and respirologists (n = 5).
Donor Trends Over Time
The proportion of living donor transplants that were pre-emptive increased over time: 18% (4/22) in 2013, 27% in 2014 (6/22), 38% in 2015 (9/24), and 34% (11/32) in 2016. Throughout the study period, a total of 359 (42%) intended recipients were on dialysis before their donor candidate started their evaluation, 316 (37%) were never on dialysis, 91 (11%) started dialysis after their candidate completed evaluation, 58 (7%) started dialysis within the first 3 months of their candidates’ evaluation start date (eg, before the living donor evaluation could be expected to be completed), and 36 (4%) started dialysis after the living donor evaluation was underway for at least 3 months. Among those starting dialysis, dialysis started a median of 351 (IQR = 243, 419) days after the donor candidates’ first contact date. Among donors who could have donated pre-emptively, 8 (20%) of their intended recipients started dialysis before donation and after the evaluation was underway for at least 92 days.
Discussion
Our study demonstrates that it is possible to track both process and outcome measures for living kidney donor evaluations. We found that nearly half of initial contacts did not proceed past the initial screening phase and that the most common reason for attrition among potential donors was loss to follow-up. We also found that delays in the donor evaluation were common. Missed opportunities of pre-emptive living donor kidney transplantation could have been avoided with a more streamlined evaluation process or enhanced communication/coordination between the transplant recipient and living donor teams.
Owing to the length of the donation process, a non-trivial number of intended recipients could have received a pre-emptive transplant but instead started dialysis. The number of times an intended recipient started dialysis, became ineligible for transplant, died, or received a deceased donor transplant before their donor candidate completed their evaluation occurred often enough to warrant concern. Our results are comparable to other studies, and quality improvement efforts should attempt to reduce these occurrences whenever possible.6,7,12
We also noted that some of the reasons for a delayed living donor evaluation are modifiable. For example, some donor candidate evaluations were delayed because the intended recipient was not assessed, yet 73% of these evaluations resumed. If the candidate’s evaluation was not delayed, we would expect some living donor kidney transplants to occur earlier, in turn, potentially improving outcomes and reducing health care costs attributable to dialysis. 7 Another potentially modifiable reason for the delay was the prioritization of other donor candidates. In some cases, it may be more cost-effective to evaluate multiple candidates simultaneously if the cost attributable to the recipient’s disease during this delay (ie, dialysis costs) is greater than the cost associated with evaluating all donor candidates simultaneously during this time. 13
Rather than reporting a single metric (eg, the time until donation), we reported several measures that have been deemed important through a previously conducted modified Delphi. 10 The results of this approach can be used by programs to identify specific bottlenecks for focused quality improvement efforts. Using the LHSC program as an example, improvements may include facilitating earlier recipient transplant referrals (a commonly cited delay for the donor evaluation); conducting initial crossmatch testing earlier (a late test that could have been performed 2 months earlier); expanding operating room availability (little donor evaluation activity was observed between the final test and the pre-operative assessment period); or evaluating multiple candidates simultaneously.
Despite bottlenecks observed, the number of living donor transplants at our center increased over time. This may be explained by a rise in the number of candidates coming forward for the same recipient, a reduction in the evaluation time, or both. If this trend continues, this will also increase the strain on resources available to the living donor program. One potential solution is to provide the MSQ on the program’s website; a practice used by several programs in the United States and Canada (Toronto, Hamilton). 12 Anecdotally, reviewing the MSQ requires around 1 hour of focused living donor coordinator time and is done for all candidates. As most candidates were lost to follow-up during the initial screening phase, providing the MSQ online with the ability to complete it electronically may ease the burden on the program and facilitate prioritization of candidates using pre-scored instruments and flags, freeing coordinators to focus on key issues.
Strategies to improve the efficiency of the living donor evaluation process have been discussed elsewhere.1,13 However, we believe the future of living donor programs is to enable a 1-day evaluation (or similar) for willing candidates, where following screening, most (if not all) testing is performed in 1 day and candidates visit the transplant center only once or twice (Figure 3). In our study, candidates interacted with the health care system a median of 5 times before the pre-operative assessment, a metric that can be minimized. 10 Several transplant centers already have such a strategy in place including a program in Ontario.5,13 While a step-wise evaluation reduces costs by avoiding over-testing, there is a risk of higher opportunity costs as a recipient may start or continue dialysis while waiting for their donor. 7 If successful, a 1-day evaluation should be completed in approximately 4 months: a realistic and optimal target that be more in-line with many candidates’ expectations.5,15 Additional opportunity for efficiency improvement implicates the time from CT angiography (one of the last tests) until donation. This was the longest component of the evaluation, a period requiring little-to-no donor work-up, suggesting substantial delay after the donor candidate is likely approved.9,16 This may be due to several factors, including donor and recipient readiness and scheduling challenges. Programs can explore how to maximize the available operating room time to avoid potential delays.

Proposed clinical pathway for a 1-day evaluation of living donor candidates.
Limitations
This study should be interpreted in the context of its limitations. Reasons for delay were underreported but were reported in increasing frequency over time as measures of evaluation time were increasingly being requested. Another limitation is the lack of data on the date the donor candidate was approved. This date is critical, as it separates the delay due to the donor evaluation itself from other factors related to scheduling the transplant. Barriers to an efficient living donor evaluation may vary from one center to another and can even change over time at the same center, requiring active monitoring of an array of process and process and outcome indicators. 10 This, however, creates a data burden problem due to the constant collection and analysis of performance data. Instead, data should be entered synoptically in a patients’ electronic medical record rather than as free text on paper files. This will enable data quality checks at the time of entry; can facilitate automated reporting in real-time; or can enable some natural language processing to extract meaningful features from the notes. Another limitation is the recency of the data. Although the data are not timely, the data reflect the pre-pandemic environment and are likely to generalize to the state of operations today. Given the extremely onerous and time-consuming nature of manual data collection, more recent data are not analytically ready for analysis, which represents an important constraint to evaluating future quality improvement strategies.
Another limitation is the lack of information about delays specific to participation in the KPD program. Key timepoints in the evaluation may be missing if performed elsewhere and therefore unavailable for analysis. Previous work has shown that the evaluation time for donors who participated in this program had a longer evaluation time by 6.6 months. 10 Delays specific to the KPD process should be a focus of future research because this program may experience different sets of challenges and delays. It would also be useful to indicate whether the KPD program was discussed with the candidate, regardless of compatibility, to ensure candidates are informed of this option.
Conclusion
The living donor evaluation process remains a challenging and resource-intensive process. Tracking process and outcome measures provides insight into the performance of a living donor program. Improving the efficiency and timing of living donor evaluation improves recipient outcomes while reducing costs to the health care system. Future studies may wish to explore the efficacy and cost of a 1-day evaluation relative to current practice as a solution to improve the living donor evaluation process, but the burden associated with data collection should be addressed.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581251323964 – Supplemental material for The Flow of Living Kidney Donor Candidates Through the Evaluation Process: A Single-Center Experience in Ontario, Canada
Supplemental material, sj-docx-1-cjk-10.1177_20543581251323964 for The Flow of Living Kidney Donor Candidates Through the Evaluation Process: A Single-Center Experience in Ontario, Canada by Steven Habbous, Beth Montesi, Christy Masse, Corinne Weernink, Sisira Sarma, Mehmet A. Begen, Ngan N. Lam, Christine Dipchand, Seychelle Yohanna, Dervla M. Connaughton, Lianne Barnieh and Amit X. Garg in Canadian Journal of Kidney Health and Disease
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by a grant from the Canadian Institutes of Health Research (CIHR).
Ethics Approval
From author: in the methods section we wrote: “The Research Ethics Board at the Lawson Research Institute approved the study (HSREB #107847) (individual patient consent was not required).”
Consent to Participate
Not applicable.
Consent for Publication
Consent for publication was obtained from all authors.
ORCID iDs
Data Availability
The data included are highly sensitive and can be used to re-identify individuals. Thus, to safeguard the privacy of patient data at this single center, we are not permitted to publish the data for secondary use.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
