Abstract
Background:
Recent atrial fibrillation guidelines recommend shared decision-making between clinicians and patients when choosing stroke-prevention therapies. Although decision aids improve patients’ knowledge and decisional conflicts, there is no decision aid for stroke-prevention strategies in people with atrial fibrillation receiving hemodialysis.
Objective:
The objective was to develop and field test the first decision aid for Atrial Fibrillation in HemoDialysis (AFHD-DA) for stroke prevention in atrial fibrillation and hemodialysis.
Design:
This is a sequential 3-phase mixed-methods study following the International Patient Decision Aid Standards and the Ottawa Decision Support Framework.
Setting:
This study was conducted in 2 ambulatory hemodialysis centers in Montreal and Laval (Canada).
Participants:
Adults with atrial fibrillation receiving hemodialysis and clinicians (physicians, pharmacists, or nurse practitioners) involved in their care.
Methods:
In phase 1, we conducted systematic and 2 rapid reviews and formed the steering committee to pilot the first version of AFHD-DA. In phase 2, we refined the AFHD-DA through 4 rounds of focus groups and interviews, using a qualitative analysis of transcripts and a descriptive analysis of acceptability and usability scores. In phase 3, we field-tested the decision aid during 16 simulated clinical consultations. We assessed decisional conflict and patient knowledge using before-and-after paired t-tests and compared the proportion of patients with high decisional conflict using McNemar’s test. We used the Ottawa Hospital preparation for decision-making scale and participants’ feedback to evaluate how AFHD-DA facilitated shared decision-making.
Results:
We enrolled 8 patients and 10 clinicians in phase 2. The predefined usability and acceptability thresholds (68 and 66, respectively) were reached. Theme saturation was achieved in the fourth round of focus groups and interviews. Four major themes emerged: acceptability, usability, decision-making process, and scientific value of the decision aid. Sixteen patients and 10 clinicians field-tested the decision aid in phase 3. In clinical settings, AFHD-DA significantly decreased the mean decisional conflict score from 41.0 to 13.6 (P < .001) and the proportion of patients with decisional conflicts from 81.3 to 18.8% (P = .002). It improved the patients’ mean knowledge score from 62.7 to 76.6 (P = .001), and 81% of patients and 90% of clinicians felt highly prepared for decision-making. Clinical consultations lasted, on average, 21 minutes (standard deviation = 8).
Limitations:
The main limitations were the low quality of existing literature, the small number of participants, and the absence of a control group.
Conclusions:
The decision aid facilitated time-efficient shared decision-making between clinicians and patients, improved patients’ knowledge, and reduced decisional conflict around selecting a stroke-prevention strategy for patients with atrial fibrillation receiving hemodialysis.
Introduction
Atrial fibrillation (AF) is highly prevalent among people receiving dialysis, affecting as many as one in every 4 patients.1,2 People with AF receiving dialysis have a 3 times higher risk of stroke and a 2 times higher risk of mortality than those receiving dialysis without AF. 2 However, recent guidelines could not reach a consensus for stroke prevention in this population due to conflicting data on the risks and benefits of anticoagulation for AF in people receiving dialysis.3-6 This lack of consensus translates to fewer anticoagulation prescriptions and more heterogeneous management across medical specialties, countries, and even medical centers within the same region.7-10
Shared decision-making (SDM) is a process in which patients and clinicians discuss possible options, their respective benefits and risks, the goals of care, and the patient values to make a partnership health-related decision.11,12 Decision aids (DAs) support and enable SDM between patients and clinicians. Their development and implementation in clinical practice have resulted in substantial improvements in patient-important outcomes, such as patient knowledge regarding therapeutic options and satisfaction with treatment. 13 The SDM is particularly critical when decisions are complex and rely heavily on patient preferences and values.3,14
Recent Canadian and US guidelines on AF promote SDM for stroke prevention, leading to the development and sharing of many DAs.3,4 Although DAs have demonstrated positive effects in adults with AF, to our knowledge, no DA has been developed to support SDM on anticoagulation for people with AF receiving dialysis.15,16 Considering the conflicting evidence and lack of consensus, there is an urgent need for a DA in this population. The SIMPLIFY-HD study addresses this gap by developing and evaluating the first decision aid for Atrial Fibrillation in HemoDialysis (AFHD-DA).
Methods
Theoretical Frameworks and Development Process
We developed the methodology in accordance with the International Patient Decision Aids Standards (IPDAS) guidelines and the Ottawa Decision Support Framework (ODSF).17-20 We used the SUNDAE and DEVELOPTOOLS checklists to design and report this study (Supplemental Appendix A).17,21 The DA development process consisted of 3 phases adapted from the IPDAS guidelines: initial prototype development, refinement with user feedback, and field testing (Figure 1).17,18

DA development process.
A steering committee composed of the investigators and a patient partner who had received dialysis treatments for at least 1 year was established to oversee all stages of AFHD-DA development. 17 The patient partner was involved in each phase of the study. All steering committee decisions were unanimous.
Phase 1: Develop Initial Prototype
In phase 1, we developed the first iteration of the DA. The AFHD-DA is a web-based DA that incorporates the 6 key elements of SDM: situation diagnosis, choice awareness, choice clarification, discussion of harms and benefits, deliberation of patient preferences, and decision-making in partnership.16,22 Its target audience is all clinicians (eg, physicians, pharmacists, nurse practitioners) involved in discussing AF anticoagulation-related risks and benefits with patients receiving HD. The AFHD-DA’s initial content was based on the preliminary results from our systematic review (now published and updated, see sister publication). 23 Ischemic strokes and major bleeding events were considered to be the most clinically relevant outcomes to be included in the DA. 24 Mortality was not retained in the DA due to its high heterogeneity across studies. We ruled that warfarin, apixaban, rivaroxaban, and no treatment had sufficient evidence to be included in the DA. The DA version that was piloted reported a 23% decreased risk of ischemic strokes and 40% increased risk of major bleedings with warfarin, rivaroxaban, or apixaban compared with no treatment, based on preliminary results from our systematic review. Supplemental Appendix B describes the rationale for this decision. The final version of AFHD-DA will be updated to include the results of AXADIA-AFNET 8 and SAFE-D trials, which were not published at the time the DA was piloted. 23
We also conducted 2 rapid reviews (unpublished) on the predictive value of thromboembolic and bleeding risk scores in dialysis, following the Mayo Clinic’s method to operationalize evidence. 24 We used the CHA2DS2-VASc score to calculate the participants’ baseline risk of ischemic stroke.25,26 Despite some publications calling for caution in using the CHA2DS2-VASc score for patients receiving HD, most validation studies in this population showed similar c-statistics to those found in the general population.27-31 The steering committee decided not to provide individual estimations of the bleeding risk due to the low predictive power of bleeding risk scores (ie, HAS-BLED score) in the population receiving dialysis.32,33 Instead, the DA pilot provided an average historical risk of major bleeding in the hemodialysis population of 8 events (95% confidence interval [CI] = 7.14 to 8.10) per 100 persons-year. 34 Clinicians could estimate the patient’s individual bleeding risk by assessing their overall number of risk factors for bleeding (derived from the HAS-BLED score) compared with the provided average risk.
The AFHD-DA’s initial format and visual aspects were based on the ODSF eTraining, reviews conducted under the IPDAS Collaboration and existing DAs in AF.15,19,35-40 During phase 1, all steering committee members, including the patient partner, provided ideas and feedback on the content and format of AFHD-DA.
Study Participants and Recruitment
Study participants needed to (1) be 18 years and older, (2) have documented AF, (3) be receiving HD treatments, and (4) have sufficient knowledge of French for reading and writing. Participants were excluded if they (1) received HD treatments for less than 3 months, (2) had valvular AF or a mechanical valve, (3) were unable to give informed consent, or (4) had vision or hearing impairments preventing them from using the DA. Eligible participants were identified through chart review. Recruitment took place during dialysis treatments.
Participating clinicians were heart or kidney physicians, pharmacists, and nurse practitioners with a valid professional permit. We recruited clinicians during 2 information sessions and through emails.
As per the IPDAS guidelines, participants could only be enrolled in 1 phase to avoid contamination across study phases. 17 We conducted the study in 2 ambulatory hemodialysis centers in Quebec, Canada. The local Research Ethics Board at both participating centers approved the study protocol. All participants provided written consent before participation.
Phase 2: Refine With User Feedback
Study design and setting
Phase 2 followed a convergent parallel mixed-methods design with a qualitative analysis of focus groups and semi-structured interviews with patients and clinicians and a descriptive analysis of acceptability and usability scores. 41 Therefore, quantitative and qualitative data were collected simultaneously, analyzed independently, and interpreted together a posteriori in the final stage of the research.
Following Krueger and Casey’s guide, we conducted focus groups and individual semi-structured interviews, face-to-face or via videoconferencing software, lasting approximately 60 minutes.42,43 Two pharmacy residents (N.M., C.T., Y.H., C.M.) without prior experience in qualitative research conducted the focus groups and semi-structured interviews. Each focus group had a minimum of 5 participants with at least 1 clinician and 2 patients to bring together different points of views and experiences. We also conducted semi-structured interviews to reach in-depth opinions on specific aspects of the DA. 42 The steering committee developed the first iteration of the interview guide (identical for focus groups and interviews) to focus on participants’ needs for decision support, values, preferences, and interests, as per ODSF recommendations. 19 After each round of focus groups and/or interviews, 1 investigator (either N.M., C.T., Y.H., or C.M.) briefly analyzed the transcript to refine the DA and interview guide. 43
Outcomes
The phase 2 outcomes were participants’ experience with the DA, acceptability, and usability. All phase 2 questionnaires are available in Supplemental Appendix C.
We conducted focus groups and interviews until saturation was achieved, ie, minimal new content (particularly new themes or suggestions) emerged from the focus groups or interviews. 44 Before the focus groups or interviews, the patients also completed a questionnaire developed by our team exploring their values and priorities around stroke prevention and anticoagulation, the results of which were included in the qualitative analysis.
Acceptability was defined as the DA’s comprehensibility and suitability for decision-making. 45 Participants assessed the DA’s acceptability after each session using the Ottawa Hospital acceptability questionnaires. Usability was defined as the DA’s usage convenience and was evaluated after each session using the System Usability Score (SUS). 46 We determined thresholds a priori for transition to phase 3: a score of 66 for acceptability as proposed by experts in SDM from the Ottawa Hospital and a score of 68 for usability as proposed by the SUS manual.46-48
Data collection and analysis
After transcription of the recorded focus groups and interviews, a single investigator (O.M.) with limited experience in qualitative analysis analyzed the transcripts with Dedoose v9.0.46 using a general inductive approach and a thematic analysis. 49 This design was deemed more appropriate by the steering committee, as this approach entails identifying themes and suggestions (an important step to refine the DA) without a preconceived theory (to let the data guide this refinement). A second investigator (C.T.) with no experience in qualitative analysis revised the analyses to ensure all themes and codes were identified. Acceptability and usability scores were reported with means and standard deviations.
Phase 3: Field Testing
Study design and setting
We used a before-and-after quasi-experimental design to assess AFHD-DA’s effectiveness in facilitating SDM in clinical settings. Clinicians and patients met one-on-one to discuss stroke-reduction strategies using the DA. The consultations were simulations of SDM and were not intended to alter the patient’s treatment. We did not ascertain whether the patient had previously discussed the risk of stroke and bleeding with their treating physician at the moment of AF diagnosis.
Outcomes
The phase 3 outcomes were decisional conflict, content knowledge, and preparation for decision-making. All phase 3 questionnaires are available in Supplemental Appendix D.
Decisional conflict was defined as the patients’ uncertainty about their decision. 50 Patients completed the Decisional Conflict Scale (DCS) immediately before and after the consultation with the clinician. We planned to recruit 14 patients to achieve 80% power to detect a minimum difference of 21 points on the DCS using a 2-tailed alpha of .05. 51
We assessed patient knowledge using a true or false questionnaire developed by our team and administered before and after the consultation. It was developed based on the Ottawa Hospital knowledge questionnaire and the AF Knowledge Scale.52,53 Results were reported as the mean number of correct answers.
After the consultation, we assessed the DA’s perceived usefulness using the preparation for decision-making scale. 25 We also collected participants’ verbal and written feedback.
Data collection and analysis
The consultations between patients and clinicians took place at the hospital and were not audio recorded. After completing the study questionnaires, participants were invited to a short interview with 1 investigator where their comments were collected to enrich our assessment of the DA’s impact on SDM.
We reported all scores using means and standard deviations. We compared the DCS and knowledge scores before and after using the DA using paired t-tests and the proportion of patients with a high decisional conflict (defined as a DCS score greater than 25) using McNemar’s tests (with exact P-value). 50 Statistical analyses were performed with SPSS Statistics v28.0.1.0 using a 2-sided significance level of .05.
Results
Participants’ Demographics and Clinical Characteristics
Between March and October 2022, we screened 543 patients, of which 114 (21%) had a diagnosis of AF and 50 (9.2%) met the eligibility criteria (Figure 2). Eight patients were enrolled in phase 2, and 16 patients in phase 3. Ten clinicians agreed to participate in phase 2, and 10 others participated in phase 3.

Screening of patients.
Patients mainly were white (75%) taking warfarin (54%) with post-secondary education (50%), a mean age of 74 years (SD = 8.0) and a mean CHA2DS2-VASc score of 4.4 (SD = 1.4). Clinicians were mainly pharmacists (50%) and kidney physicians (30%). All clinicians reported limited to no experience with DA. Tables 1 and 2 summarize the characteristics of the patients and clinicians, respectively.
Patient Characteristics.

Clinician Characteristics.

Phase 2: Refine With User Feedback
Acceptability and usability scores
The mean acceptability and usability scores (SD) were 82 (15) and 73 (18), reaching the predefined thresholds of 66 and 68, respectively. The clinicians’ mean scores were higher than those of patients, particularly for the usability score. Table 3 summarizes the results from phase 2.
Acceptability and Usability Score Results.
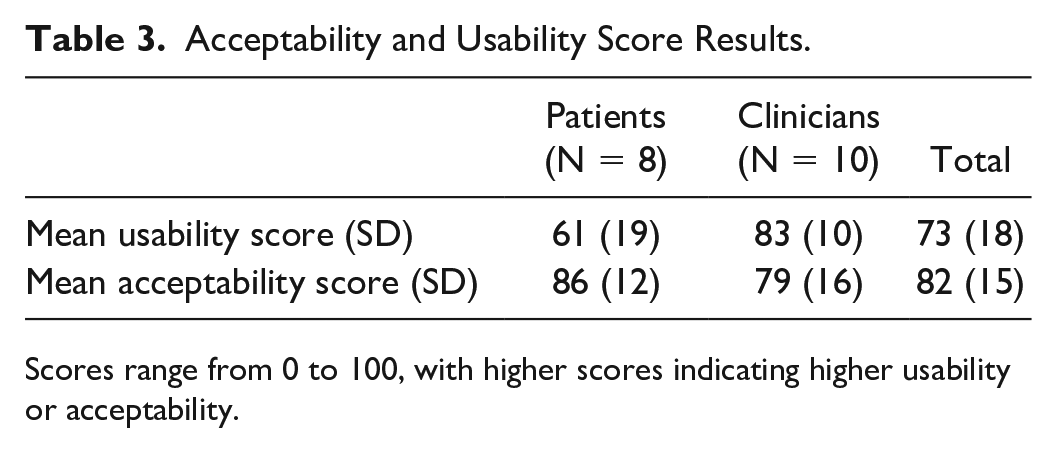
Scores range from 0 to 100, with higher scores indicating higher usability or acceptability.
Qualitative analysis
We conducted 4 rounds of focus groups and interviews with 8 patients and 10 clinicians. Supplemental Appendix E describes the DA’s development process. The qualitative analysis led to 8 themes, 85 codes, and 449 code occurrences. Saturation was reached in the fourth round. Supplemental Appendix F lists the themes and corresponding codes. Hereby, we present the qualitative analysis of the 4 main themes.
Acceptability
Acceptability was the most recurrent theme in both focus groups and interviews. Participants were most often concerned with comprehensibility and information quantity, and their comments suggested these aspects improved as we refined the DA. All participants wanted the DA to focus exclusively on elements directly impacting the decision at stake. Other elements, such as mechanisms of action, were erased from the DA. All clinicians emphasized patients’ understanding of stroke and bleeding risks as critical to decision-making, pointing out that this information should be included at the beginning of the DA. In the first version, a clinician rightfully pointed out that the no anticoagulation option was receiving less space and attention while being a valid treatment option. Based on feedback from the first 2 focus groups, we rapidly updated the DA to be used exclusively with the assistance of a clinician to reduce patients’ fears and support their understanding. A clinician reported that “the patient and the clinician must be together in front of the screen [the DA].” Clinicians were all enthusiastic about the DA and SDM, but some patients expressed fear of making a decision beyond their knowledge and understanding, thereby doubting the relevance of their involvement in the SDM process.
Usability
Discussions on the DA usability led to more synthesized and relevant content. Sentence complexity, text quantity, time to use the DA, and the balance between figures and texts were subthemes extensively explored early in phase 2. For example, patients expressed that the content needed to be more user-friendly: “For the patients, it seems to me that the content is very academic. It needs to be more concise. To make it more useful too . . .” (patient 2). Contrastingly, by the end of phase 2, a patient commented: “It was not long . . . It’s very complete . . .” (patient 13). Nearly all clinicians were concerned about the time necessary to complete the DA and the change of practice in using SDM. This concern was also expressed in the last interview with a clinician but with a more positive outlook: “It takes some time . . . But regarding the length, we keep it as simple as possible. We have patients with different levels of understanding.”
Decision-making process
Participants identified many elements as relevant in choosing a treatment for stroke prevention (prior experience with a medication, a personal history of bleeding or stroke, pill burden, patient preferences and values, clinician opinion, availability of antidote, etc), which were then added to the DA. Overall, we identified 2 key points: (1) the decision-making process is complex with several conflicting considerations and (2) the DA should support a broader discussion on the implications of embolic prevention therapies going beyond the basic health information provided. The patients were mostly interested in the DA exploring the relative level of concern on bleeding and stroke, reporting: “My biggest concern for me, I saw what happened to my dad with strokes, so my priority is to protect myself from strokes.” Two patients expressed the desire to be informed of the therapies’ rare but serious adverse effects, which were then added to the DA. One patient said: “I want to know it all . . . I did chemotherapy. In one of the chemotherapy drugs, one of the uncommon adverse effects is to cause bone pain. Since then, I’ve been having a lot of back and hip pain. I had this rare consequence. I want to know it all.”
Decision aid’s scientific value
Although a central concern for all clinicians, patients less often explored this theme. Clinicians wanted the evidence included to be relevant to people on dialysis: “Are there any risk calculators for people on dialysis? Because I don’t think that we can use the HASBLED or the CHADS . . . The problem is really in dialysis . . . For many reasons, it’s not at all the same risk, as much for the bleeding risk as for stroke risk . . .” (clinician 4). Clinicians also regarded the quality of the underlying evidence as critical for the DA’s acceptability and usability. They insisted on providing the scientific rationale for them to be confident in using the DA. The AFHD-DA first 2 versions represented the quality of the underlying evidence using a star system, but many clinicians disagreed with this decision: “For the clinicians, I think the level of evidence needs to be accessible, but not with stars like you did . . . We need more details . . . For example with a pop-up page.” In the end, we removed the star rating system and relied on clinicians to communicate the quality of the evidence to the patient. To do so, we provided them with an evidence summary accessible within the DA application. Nearly all clinicians questioned the inclusion of aspirin and left atrial appendage closure in the first version of the DA, and they were then removed.
Final phase 2 decision aid
Upon reaching theme saturation and the acceptability and usability thresholds, the steering committee approved the transition to phase 3. Before field testing, an investigator (O.M.) analyzed the DA readability using Scolarius to confirm the text’s understandability to low-grade readers.54,55 On average, the information could be understood by people with an elementary-level education.
Phase 3: Field Test
Questionnaires
Clinical consultations lasted 21 (SD = 8) minutes on average. Table 4 compares the outcomes before and after the DA. The mean DCS score was significantly lower after using the DA (41.0 vs 13.6, P < .001). The DA decreased the proportion of patients with high decisional conflict (81% vs 19%, P = .002). The DA also significantly improved the mean patients’ knowledge score (62.7 vs 76.6, P = .001).
Decisional Conflict and Patient Knowledge Scores.

Scores range from 0 to 100, with higher scores indicating higher decisional conflict or knowledge.
The mean score of the preparation for decision-making scale was 81 (95% CI = 73 to 88) and 90 (95% CI = 87 to 93) for patients and clinicians, respectively. The highest-rated item among patients was the DA’s helpfulness in thinking about the pros and cons of anticoagulation. The highest-rated item among clinicians concerned how the DA helped patients make more informed decisions. Complete results regarding preparation for decision-making are available in Supplemental Appendix G.
User feedback
Most patients mentioned that AFHD-DA facilitated SDM and helped them better understand AF and anticoagulation implications. Patients thought choosing specific anticoagulants remained difficult, largely due to insufficient information about the effects of therapies. Many patients questioned the relatively small benefits of anticoagulation compared with the increased risk of major bleeding. Clinicians’ comments were very positive regarding the DA’s content and usability in clinical settings, except for 1 professional finding the summary table on anticoagulants too complex. Another clinician found individualizing the bleeding risk challenging. Screenshots of the updated version following phase 3 are available in Supplemental Appendix H.
Discussion
The approach in health care has changed from paternalistic to patient-centered and now patient-directed. 56 However, patient participation in health care is limited by a need for knowledge and understanding of treatment implications. Although multiple DAs exist for patients with AF, to our knowledge, AFHD-DA is the first DA for AF management in patients receiving HD. It was iteratively developed and field-tested following the IPDAS stepwise approach. The DA reached the predefined threshold for usability and acceptability among patients and clinicians. The AFHD-DA’s performance was consistent with previous clinical trials of SDM interventions in easing decision-making and improving decisional conflict and content knowledge.13,15,16 The AFHD-DA probably did not lengthen the discussion, considering consultations lasted 21 minutes (SD = 8) on average. Although we could not compare to a control group, it was quicker than the mean time to evaluate stroke-prevention strategies in AF (31 minutes, SD = 17) in a recent large-scale randomized controlled trial. 57
Available findings on the risks and benefits of warfarin for patients receiving HD with AF are conflicting and based on low-quality evidence, thereby limiting the level of certainty of the DA’s scientific content. However, SDM is most necessary in contexts of gaps in scientific knowledge, and DAs can help communicate to patients the uncertainty around the available evidence. 58 The IPDAS suggests using the GRADE system to explain the level of confidence around scientific evidence to patients. 36 In the first version of AFHD-DA, the GRADE rating for each treatment comparison was visually represented to patients using stars, but focus group participants chose to remove it from future versions. Instead, it was decided to make the GRADE rating available only to clinicians in a separate section.
The SDM literature for populations with kidney failure (KF) primarily focuses on dialysis initiation, timing, and modality. It provides crucial insights into patients’ perspectives, where longevity is important but not the sole consideration, as the quality of life and functional status are as essential. 59 Similarities in factors enhancing patient involvement in decision-making can be drawn between people with KF and people with AF. These factors include interpersonal relationships (eg, provider, family opinions), how well medical recommendations match the patient’s health goals, maintaining the current well-being, the impact on daily life, the desire for control, and the personal significance of the decision’s benefits and risks.56,60 These issues were frequently raised by participants in the focus groups and guided the conception of AFHD-DA.
A recent systematic review of DAs in AF found them more likely to improve SDM when delivered before the consultation by giving patients more time to reflect on their decisions. 16 Still, the steering committee opted for AFHD-DA to be used during clinical encounters to minimize barriers to patient engagement. A clear consensus among focus groups supported this choice. Furthermore, the AFHD-DA allowed participants to postpone their decision if desired, ensuring they had sufficient time to decide. According to this systematic review, DAs incorporating more than a description of the patient’s problem (situation diagnosis) and the available options (option clarification) were more likely to lead to SDM. The AFHD-DA incorporated all 6 key elements of SDM and improved the decisional conflict and patient knowledge, thereby supporting this latter finding.16,22
Age has been identified as a barrier to the implementation of SDM in KF and AF.56,61,62 Older people with KF prefer their health care provider to make the decisions regarding their treatments in a greater proportion than younger individuals. 61 In our study, where the mean age was 74 years, some patients expressed discomfort with being involved in the decision-making process and preferred a more passive role. Although DAs can empower patients to get involved in SDM, we need innovative ways to engage the older generation in the SDM process.
This study had several strengths. We followed the updated IPDAS guidelines, the most rigorous reference for DA’s development. 17 A recent systematic review identified that DAs for AF stroke prevention lack adherence to IPDAS criteria and appropriate involvement of patients and clinicians in its development. The AFHD-DA development is characterized by its fidelity to IPDAS standards and by the involvement of patients and clinicians in all phases. The AFHD-DA scientific content was supported by a rigorous systematic literature review. 23 The effect sizes were large, with a Cohen’s d above 0.8 on decisional conflict and knowledge scores. 63 Moreover, we reached the target sample size with no missing data.
The study also had significant limitations. The lack of a control group likely inflated our results from phase 3, as patients did not systematically discuss the risk of stroke and bleeding as part of the study. A randomized controlled trial would provide a more accurate assessment of AFHD-DA’s benefits. However, this before-and-after study was a necessary first step to ensure the acceptability and usefulness of the DA before engaging in more extensive studies. Our participants were mostly white with a high level of education, which could have improved the performance of the DA, considering previous studies have shown low health literacy, low education level, and different cultural or linguistic backgrounds to be barriers to SDM in chronic kidney disease.64-66 Although the study questionnaires were validated in English, only the DCS was validated in French. The other questionnaires were freely translated into French without prior validation. Finally, the low-quality evidence informing the DA is certainly a limitation to the current DA.
Conclusion
In conclusion, AFHD-DA facilitates SDM between patients and clinicians and improves decisional conflict and content knowledge. Further studies are needed to assess whether improvement in decision-making outcomes translates into clinical benefits, such as medication adherence and reduction of ischemic events.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241311077 – Supplemental material for Shared Decision-Making Aid for Stroke-Prevention Strategies in Patients With Atrial Fibrillation Receiving Maintenance Hemodialysis (SIMPLIFY-HD): A Mixed-Methods Study
Supplemental material, sj-docx-1-cjk-10.1177_20543581241311077 for Shared Decision-Making Aid for Stroke-Prevention Strategies in Patients With Atrial Fibrillation Receiving Maintenance Hemodialysis (SIMPLIFY-HD): A Mixed-Methods Study by Olivier Massé, Noémie Maurice, Yu Hong, Claudia Mercurio, Catherine Tremblay, Lysane Senécal, Amélie Bernier-Jean, Nicolas Dugré and Gabriel Dallaire in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors are grateful to all participants, the patient partner (M. Pierre-Louis Leduc) and the staff at the participating ambulatory hemodialysis centers. The authors also thank Ricky Turgeon (
Ethics Approval
SIMPLIFY-HD received ethics approval from the CIUSSS du Nord-de-l’Île-de-Montréal (MP-32-2022-2418) and the CISSS de Laval (MEO-32-2023-941).
Consent to Participate
Informed consent was obtained from all participants.
Consent for Publication
All authors reviewed the final manuscript and provided consent for publication.
Availability of Data and Materials
The data presented in this study are available, from the corresponding author, on reasonable request pending approval by the ethic committee.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Faculty of Pharmacy of the University of Montreal provided funding for statistical analyses. This funding agency had no input into the study design, data collection and analysis, or redaction and publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
