Abstract
Background:
Kidney transplant recipients are uniquely exposed to the disordered bone metabolism associated with chronic kidney disease beginning before transplantation followed by chronic corticosteroid use after transplantation. Previous efforts to synthesize the rapidly accruing evidence regarding estimation and management of fracture risk in kidney transplant recipients are outdated and incomplete.
Objective:
To synthesize the evidence informing the overall incidence, patient-specific risk prediction, and methods of prevention of fractures in patient living with a kidney transplant.
Design:
Three systematic reviews will address the following questions: What is the overall incidence of skeletal fracture after kidney transplantation (review 1)? Which prediction models and individual prognostic factors predict fracture in kidney transplant recipients (review 2)? and How effective are different antifracture interventions at preventing fracture or improving surrogate markers of bone health in kidney transplant recipients (review 3)?
Setting:
Cohort studies (reviews 1 and 2) and randomized trials (review 2) with a mean/median follow-up ≥12 months beginning after transplant. Review 3: randomized trials or new-user cohort studies with concurrent controls evaluating the effect of antifracture interventions including bisphosphonates, calcium supplementation, cinacalcet, denosumab, parathyroid hormone analogues, parathyroidectomy, raloxifene, romosozumab, steroid withdrawal or minimization protocols after kidney transplant, vitamin D (both active and nutritional), other antifracture interventions.
Patients:
Adult kidney transplant recipients in studies published after the year 2000.
Measurements:
Review 1: incidence rate or cumulative risk of fracture. Review 2: For prediction models, measures of discrimination (eg, c-statistic), calibration (calibration curves, observed:expected ratios), and net benefit (ie, from decision curve analysis); for individual prognostic factors, relative measures of association with fractures. Review 3: measures of treatment effect on fractures and on surrogate markers of bone health (eg, bone mineral density, trabecular bone score).
Methods:
We searched MEDLINE, Embase, and the Cochrane Library using subject headings and keywords related to kidney transplant and fractures. Pairs of reviewers will screen records independently in duplicate to identify studies relevant to one or more of the 3 reviews and categorize each study accordingly. Single reviewers will extract data and evaluate risk of bias for each included study using one of the following tools as appropriate: the Quality of Prognostic Studies tool, the Prediction model Risk Of Bias ASsessment tool, the Risk Of Bias In Non-randomised Studies—of Interventions tool, and the Cochrane Risk of Bias 2.0 tool. A second reviewer will independently verify. We will synthesize study-level summary estimates by random-effects meta-analysis for review 1, by vote counting and random-effects meta-analysis in review 2, and by random effects pairwise and, if feasible, network meta-analysis in review 3. We will summarize findings according to latest guidance of the Grading of Recommendations Assessment, Development, and Evaluation Working Group applicable to each review.
Limitations:
Reliance on published studies is susceptible to publication bias, particularly in studies of prediction (review 2) and of treatment effects (review 3).
Conclusions:
This review will provide an evidence update on 3 topics of relevance to patients, clinicians, guideline developers, and researchers.
Introduction
Kidney transplantation improves the duration and quality of life of people with end-stage kidney disease (ESKD) compared with maintenance dialysis. 1 Kidney transplant recipients are more likely to sustain a skeletal fracture than the general population.2,3 This may be due to a several factors, including the longstanding effects of mineral and bone disorder of chronic kidney disease, common comorbidities such as diabetes and the impact of post-transplant immunosuppressive therapies, including corticosteroids.4-7 The outcomes after a fracture in this population are also poor, with increased mortality, prolonged hospitalization, increased risk of graft loss, and higher health care costs compared with those who do not fracture.8-10
Post-transplant fracture risk estimates vary widely across studies, ranging from 3.3 to 99.6 per 1000 patient years in one systematic review, likely due to heterogeneity in patient populations and in the definitions of fractures. 11 Changes in immunosuppression and in the age and comorbidity profiles of recipients compared with the early years of kidney transplantation 12 call for more contemporary estimates of fracture risk. 13 Risk assessment tools (eg, the Fracture Risk Assessment Tool) 14 lack kidney disease and transplant specific variables and are poorly studied in this population. 15 The authors of the Kidney Disease: Improving Global Outcomes (KDIGO) 2017 guidelines made weak recommendations based on low-quality evidence for measurement of bone mineral density if it will change management, and treatment with calcium, activated vitamin D or—in those with an eGFR ≥30 mL/min per 1.73 m2—antiresorptive therapies. 16 These were limited to the first 12 months post-transplant with no evidence to make recommendations after the first year post-transplant. Several studies that may inform management have been published since.
Here, we prospectively report our methods for a family of 3 systematic reviews to address the following questions:
Methods
Protocol and Registration
We registered our review protocol with PROSPERO (CRD 42024536926) and present it here according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Protocols (PRISMA-P) extension statement. 17
Overarching Concepts
Each question is addressed in a dedicated review conducted by a team of methodologists and nephrologists. This overarching protocol describes the methods of all 3 related reviews. Figure 1 summarizes the review strategy.
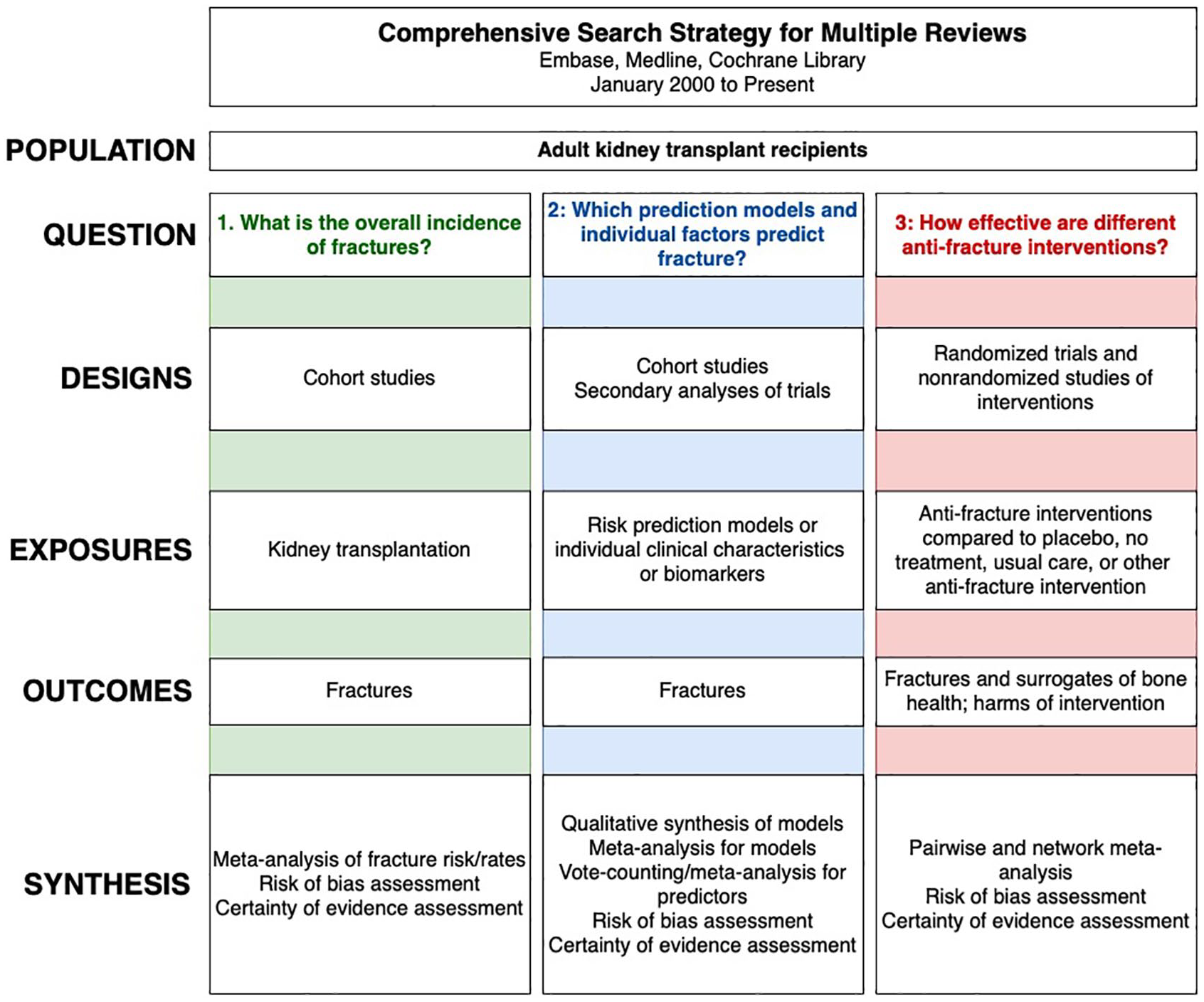
Review strategy.
Eligibility Criteria
Review 1: overall incidence
We will include cohort studies that (1) enrolled at least 50 human recipients of a kidney transplant; (2) had a mean/median follow-up duration of ≥12 months, with time zero for the follow-up defined as the day of kidney transplant or thereafter; (3) evaluated fractures occurring after the kidney transplant; and (4) reported the incidence rate or cumulative risk of fracture or provided sufficient data to calculate them. If follow-up time is not defined, we will contact the authors for clarification. We will exclude studies that are highly selective and not representative of the broader, typical kidney transplant population. This includes studies where all participants receive specific treatments for bone health beyond standard care (eg, all using denosumab), have all undergone specific bone-related testing beyond standard care radiographic screening for nonclinically apparent fractures (as these will result in the inclusion of prevalent fractures), or are restricted to patients with a history of fracture.
Review 2: patient-specific prediction
We will include cohort studies or secondary analyses of randomized trials that enrolled at least 50 human recipients of a kidney transplant and had a mean/median follow-up duration of ≥12 months and either (1) describe the development or validation of a multivariable prediction model or (2) report adjusted measures of association (eg, hazard ratio [HR], odds ratio [OR], risk ratio [RR]) between potential predictors and incident fracture. We will extract all predictors from multivariable models regardless of statistical significance.
Review 3: effectiveness of antifracture interventions
We will include randomized controlled trials (RCTs) or nonrandomized studies (prospective or retrospective) with a concurrent control group in a new-user design that (1) examined the effect of antifracture therapy following kidney transplantation as part of their study objectives, (2) reported fracture-related outcomes or surrogate markers of bone health, or (3) (if nonrandomized) provided adjusted estimates of treatment effect. We use the term adjustment to describe a variety of methods that aim to reduce bias from confounding; these may include regression adjustment, matching on variables, methods operating on a propensity score (regression adjustment by the propensity score, stratification, matching, or inverse probability of treatment weighting), instrumental variable methods, simple restriction of a study sample, or any combination of these. The interventions include cinacalcet, parathyroidectomy, denosumab, bisphosphonates, raloxifene, parathyroid hormone analogues, romosozumab, calcium supplementation, vitamin D (both active and nutritional) and steroid withdrawal or minimization protocols after kidney transplant, and other antifracture therapy currently in use. The comparator can be placebo, no treatment, standard care, or a different antifracture therapy. Predesigns and postdesigns and historical control studies are excluded. Due to challenges in detecting small effects and distinguishing prescribed from over-the-counter use, which introduces heterogeneity, we will exclude nonrandomized studies when evaluating vitamin D or calcium.
For all reviews, in studies involving multiple organ transplants, we will include those with kidney transplant subgroup data or at least 80% kidney transplant patients. Studies with >50% patients younger than 18 years of age at baseline will be excluded. However, we will include studies of patients who received a kidney transplant as a child but were studied as adults.
Information Sources and Search Strategy
The Supplemental Appendix contains our search strategy and results from its execution on May 11, 2024. We conducted a preliminary search on February 13, 2024, in Epistemonikos to identify existing systematic reviews11,18 and relevant search filters 19 to inform the development of our search strategy. Following this, in consultation with a librarian, we developed a single comprehensive search strategy using the following electronic databases: Medline, Embase, and the Cochrane Library, aiming to identify published articles addressing one or more of our research questions from January 2020 to the present. Due to significant overlap, the search concepts for the overall prognosis and prognostic factor reviews shared the same terms: kidney transplantation and bone or fracture. The set of search concepts for the effectiveness review included kidney transplantation and antifracture therapies, including steroid withdrawal or minimization. To ensure feasibility while maintaining a comprehensive scope, we applied observational study 20 and RCT 21 filters to this set. We used Boolean operators to combine synonyms for each concept with OR and the main concepts with AND. Finally, the sets of concepts were combined using the OR operator. We limited the studies to those published after 2000 to align with current transplant practices and use of antifracture interventions. We did not restrict language. In addition, we will use Citationchaser 22 for forward and backward citation chasing, finding records citing or referenced by included studies and existing reviews.
Study Selection
We will export citations to Covidence systematic review software for deduplication and screening. Our team will develop and pilot test title/abstract and full-text screening forms. After calibration, reviewers will independently screen titles and abstracts in duplicate. They will then retrieve and screen full-text articles in duplicate. Reviewers will categorize full texts according to the review(s) to which they pertain and record the reasons for exclusion specific to each review. They will resolve any conflicts through consensus or by consulting a third reviewer. For articles without accessible full texts, we will use interlibrary loans. If we cannot obtain full texts, we will exclude the citations and label them accordingly. In cases of duplicate publication, where multiple reports cover the same population, we will extract data from the report with the largest sample size, most comprehensive data, longest follow-up, and most recent publication date.
Data Collection and Items
Single reviewers will perform data extraction and risk of bias assessment for each article using a piloted spreadsheet with verification by a second reviewer. We will collect the following information: study design, participant characteristics (such as demographic and eligibility criteria), predictor characteristics (index factor, reference factor, definition), intervention characteristics (dose, regimen, duration of treatment), risk of bias assessment criteria, outcomes (eg, definition/assessment, intention to treat data, per-protocol data, length of follow-up, measures of central tendency and dispersion, cumulative risk, incidence rate, measures of association, treatment effect as applicable). For studies of prediction models, we will extract details of methodology and performance using the CHecklist for critical Appraisal and data extraction for systematic Reviews of prediction Modelling Studies (CHARMS) tool and the Prediction model Risk Of Bias Assessment (PROBAST) tool.23-26 We will categorize outcome data as reported from short-term (1-5 years), medium-term (>5-10 years), and long-term (>10 years) intervals. In cases where studies report outcome data at multiple time points (eg, at 2 and 3 years) within each interval category, we will extract data from the longest time points. We will use software to extract numerical data when only presented in graphical form (https://automeris.io/WebPlotDigitizer).
Studies evaluating individual predictors (review 2) and treatment effects (review 3) may present several adjusted estimators (eg, from several regression models); we will extract data from the estimator we judge to provide the lowest risk of bias. Multiple factors may influence this selection. When some measures of fit are available (eg, information criteria, log-likelihood, [pseudo] R2, Brier score, c-statistic, degree of minimization of a standardized mean difference in potential confounders between study groups), we will choose the model with the best performance. If these are not presented, we will extract the estimator that accounts for the largest number of covariates (which will also consider stratification or restriction in the ways one might account for a covariate). If the same number of covariates is accounted for in multiple estimators, then we will choose the one in which the point estimate of interest is closest to the null value.
For studies presenting multiple prediction models (review 2), we will select the model to review based on the following order of priority: the most robustly validated model (where external geographic validation > external temporal validation > internal/external validation > internal validation with resampling > split sample internal validation > apparent performance only), the best performing model, and finally, if there is no appreciable performance difference between 2 models that have undergone validation of similar rigor, we would choose the simplest model.
Outcomes
Our outcomes of interest are skeletal fractures and surrogate markers of bone health. For all reviews, fracture-related outcomes include clinical or radiographic fractures at various sites (eg, hip vs vertebral) and fractures leading to hospitalization.
Review 1 (overall incidence) will consider fracture risk (probability) or incidence rates for all reported types of fractures. Review 2 (patient-specific prediction) will include only fractures and not surrogate markers of bone health. We will use the study-defined primary outcome in our analyses if that outcome is fracture, regardless of site. If this primary criterion cannot be met (ie, no primary outcome is defined, multiple fracture outcomes are designated as primary, or all fracture outcomes are designated as non-primary), we will select the fracture outcome highest on the following hierarchy: any fracture > hospitalization for any fracture > major osteoporotic fracture > hospitalization for major osteoporotic fracture > hip fracture > hospitalization for hip fracture > vertebral fracture > hospitalization for vertebral fracture > lower limb fracture > hospitalization for lower limb fracture > fracture at other sites > hospitalization for fracture at other sites. The hierarchy prioritizes more inclusive fracture composites over more specific ones.
In review 3 (treatment effects), we expect that there will be few large studies to evaluate the effects on fractures, and that those large studies will likely be nonrandomized. For this reason, we will additionally include the following surrogate markers of bone health as secondary outcomes to assess effectiveness: bone mineral density, bone biopsy parameters, trabecular bone score, high resolution peripheral quantitative computed tomography, or other measures of bone quality or quantity. We anticipate that secondary measures such as trabecular bone score and bone biopsy will be less commonly reported (as compared with bone mineral density) and will only be included in a meta-analysis if more than 3 studies comparing the same treatments report the same outcome. Review 3 will also examine secondary safety outcomes: hypocalcemia, vascular calcification, renal allograft survival, any one or a composite of various cardiovascular events (myocardial infarction, stroke, coronary revascularization, unstable angina, heart failure events, or cardiovascular death), and mortality.
Data Synthesis
In review 1, we will pool estimates of cumulative risk and incidence rate by fracture location using random-effects meta-analyses. When possible, we will additionally pool estimates in the following categories: follow-up after kidney transplantation: up 5 years, >5 to 10 years, and >10 years. To assess heterogeneity, we will present 95% prediction intervals along with our summary point estimates and 95% CIs. We chose this approach because we expect that studies included in this review will provide precise within-study estimates of rates and proportions that result in extremely high I2 that no longer indicates important between-study heterogeneity. We will explore the following study-level sources of heterogeneity including year of publication, duration of follow-up, and risk of bias in subgroup analyses or (when possible) meta-regression. We recognize that there are patient-level sources of heterogeneity but highlight that these are predictors of fracture risk and are the focus of review 2 where they are more reliably assessed within rather than across studies.
In review 2, we will address prediction models and individual prognostic factors separately. For prediction model studies, we expect the synthesis of these studies to be mostly narrative. We will report an assessment of model performance for each study based on a measure of discrimination (most often the c-statistic), an assessment of calibration (based on calibration plots or observed:expected event ratios), and decision analytic net benefit (ie, from decision curve analysis) if provided. If 3 or more studies report external validation of the same prediction model, we will pool reported c-statistics and observed:expected ratios using random-effects meta-analysis.
For studies evaluating individual predictors, our interest is primarily whether a potential predictor associates with fractures; our secondary interest concerns the magnitude of the association. We expect substantial variability in the way predictor-outcome associations are calculated and reported across the literature. To accommodate this, we will use vote counting to summarize associations based on statistical significance and direction of association (ie, negative, null [not significant], positive). We will use the P-value threshold specified in each study as the threshold for statistical significance. To estimate the magnitude of associations, we will calculate pooled (adjusted) measures of association by random-effects generic inverse-variance meta-analysis for any predictor evaluated in 3 or more studies that provide appropriate data. Some examples that would preclude meta-analysis include inconsistent transformations or categorizations of continuous variables. The primary pooled measure will be the HR which we anticipate will be reported most. If studies instead report adjusted OR or RR, we will treat these as equivalent to HRs and pool them in the same meta-analysis because we expect the incidence of fractures to be sufficiently low (<30%) to support this approximation.
In review 3, whenever possible, we will perform a frequentist network meta-analysis (NMA) using a random-effects model to estimate the pooled network effects for all antifracture interventions. The common comparator will be placebo/no treatment. We will evaluate whether the characteristics of the studies are sufficiently similar across treatment comparisons using NMA-studio web application (https://www.nmastudioapp.com/). We will assess the consistency assumption through a global test for overall inconsistency and a local test to identify inconsistencies within each closed loop in the network.
Surrogate markers of bone health evaluated as continuous outcomes may be reported in several ways and we will attempt to accommodate as many as possible with generic inverse-variance meta-analysis that relies on the point estimates of treatment effects along with their standard errors. We will preferentially extract baseline-adjusted estimates of intervention effects (from a regression model or analysis of covariance) when available. When these are not available, we will extract between-group differences in change scores. For studies that provided baseline and follow-up values for surrogate markers of bone health without a patient-level calculation of change, we will estimate mean change scores and their standard error using correlation coefficients. 27 If we calculate widely inconsistent correlation coefficients across studies or if the correlation coefficients are consistently below .5, we will pool differences in postintervention measurements rather than change from baseline, as this approach is likely to provide more precise estimates of the intervention effect. If information sufficient to calculate a correlation coefficient between baseline and postintervention measures is not available, we will assume a correlation coefficient of .5. When baseline values are not available, we will instead calculate differences in the postintervention measurements (adjusted for potential confounders in nonrandomized studies).
We will pool RCTs and nonrandomized studies to enhance precision and network connectivity if we judge the certainty of evidence from nonrandomized studies to be at least as good as that from the RCTs for a given outcome. Results will be stratified by study design (RCTs vs nonrandomized studies), with pooled estimates provided for each subgroup separately, in addition to the overall results. Furthermore, the decision to combine or separate intervention nodes in a network will depend on expert opinion, network connectivity, and the transitivity assumption with careful consideration of whether pooling placebo, no treatment, and standard care groups is appropriate based on study design. If NMA is not feasible, we will perform pairwise random-effects meta-analyses and explore reasons for heterogeneity through sensitivity analyses, subgroup analyses, or meta-regression. In cases where data are too heterogeneous for pooling, we will also use narrative synthesis to describe trends and patterns across different interventions and outcomes.
We will perform subgroup analyses based on risk of bias and these will be our only prespecified subgroup analyses. We may additionally perform subgroup or meta-regression analyses based on patient characteristics (eg, age, diabetes, sex, dialysis vintage, fracture history, or time between transplant and treatment initiation) but these will be considered post hoc and purely exploratory. Our position is that important heterogeneity of treatment effects is exceptionally rare and is not reliably assessed at the aggregate level.
For all reviews, we will request additional data from study authors as needed. If studies reported medians and ranges or interquartile ranges, we will impute mean and standard deviation.27-29 For studies that did not report 95% CIs or standard errors for a measure of association but only reported a P value and point estimate, we will calculate the 95% CIs and standard errors using the P values. 27
Assessing the Certainty of the Evidence
We will use the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach to assess the certainty of the evidence for overall incidence (review 1), 30 for each prognostic factor (review 2), 31 and for each treatment effect estimate (review 3).32,33 In network meta-analyses, we will first evaluate the certainty of direct evidence based on risk of bias, inconsistency, indirectness, and publication bias. We will then assess the certainty of indirect evidence and intransitivity focusing on the most dominant first-order loop. Finally, we will evaluate imprecision of the network evidence using null value as the threshold and assess incoherence between direct and indirect evidence. If direct and indirect estimates are incoherent, we will use the estimates with the highest certainty. 33
The GRADE requires assessment of the risk of bias in individual studies. We will assess the risk of bias of included studies using Quality of Prognostic Studies (QUIPS) tool domains of Study Participation, Study Attrition, and Outcome Measurement for review 1. 34 For review 2, we will use the full QUIPS tool for prognostic factor studies and PROBAST for studies of prediction models. 23 For review 3, we will use the Cochrane Risk of Bias (RoB) 2.0 tool for RCTs 35 and Risk Of Bias In Non-randomised Studies—of Interventions (ROBINS-I) tool for comparative nonrandomized studies. 36
In review 1, no adjustment factors will be required. For the assessment of individual predictors in review 2, we will rate down for risk of bias if a study did not adjust for the same set of confounders except for all of sex and age. For nonrandomized studies of treatment effects in review 3, we consider it critical that adjustment be performed (through any one of a variety of appropriate methods) for at least a minimal set of confounders, including sex, age, diagnosis of osteoporosis or previous fracture, and the use of antifracture therapy. We will only consider denosumab use as a potential confounder in studies published in 2010 or later (when this medication was approved for use in many jurisdictions).
For review 1, we will assume low risk of publication bias. For review 2, we will assume high risk of publication bias. For review 3, we will base the assessment of risk of publication bias on visual inspection of funnel plots.
Progress to Date
As of July 29, 2024, we have conducted the electronic literature searches which returned a total of 9508 records after deduplication and have completed all title/abstract screening (640 records included, 6.7%) and 64% of full-text screening. Currently, 110 records are included with 36% left to screen. The flow chart in Supplemental eFigure 1 summarizes this literature review process. The review team meets weekly to discuss disagreements and to review the data extraction process, form design, and methodological concepts relevant to each of the 3 reviews.
Discussion
Our systematic review aims to examine the incidence of fractures in patients who have received a kidney transplant, identify and evaluate prognostic factors, and investigate the efficacy of antifracture interventions in this population.
Previous systematic reviews in this area have had some limitations. A review by Naylor in 2013 was constrained by the small number of studies at the time, making it challenging to draw robust conclusions about the risk of fracture. 11 Other reviews have focused solely on the question of effectiveness and limited their review to bisphosphonates or denosumab and did not explore other increasingly prescribed contemporary antifracture therapies, limiting its applicability to the current treatment landscape.37-39 A systematic review of RCTs by Palmer evaluated a broader range of antifracture therapies; however, it relied on a pairwise meta-analysis, preventing an assessment of their relative effectiveness, and did not include well-performed nonrandomized studies, which, given the paucity of evidence in this area, may provide valuable additional information. 40 Finally, a recent review by Jia performed both incidence and prognosis reviews but had unclear follow-up times, potentially impacting the accuracy and applicability of the findings. 41
Our review has several strengths. To our knowledge, this is the first meta-analysis to use a comprehensive approach addressing fracture prevention from incidence, risk factors, and effectiveness of antifracture therapies, including certainty of the evidence. We will conduct a thorough literature search that focuses on studies published after 2000, ensuring the inclusion of studies that reflect contemporary management practices in transplant. There are several potential limitations. Although our literature search will use electronic databases with extensive coverage and will check reference lists of included studies and relevant systematic reviews, we may miss studies that are not indexed or published online. The inclusion of nonrandomized studies of treatment effects in review 3 could introduce confounding. However, the limited guidance in this area will limit confounding by indication and this can be mitigated through methods of adjustment. The extent of adjustment will be included in our assessment of the certainty of evidence. Furthermore, heterogeneity across included studies may be a limitation, potentially affecting the generalizability and consistency of the findings.
These reviews will inform patient-clinician discussions about post-transplant fracture risk and prevention strategies, help identify research priorities, and potentially support the development of clinical practice guidelines in this area of post-transplant care.
Conclusion
Fractures among kidney transplant patients are common but poorly understood. This these reviews will provide patients, clinicians, guideline developers, and researchers with an evidence update regarding the overall incidence of fracture, methods for patient-specific risk prediction, and effects of antifracture interventions in this population.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241306799 – Supplemental material for Incidence, Prediction, and Prevention of Fractures After Kidney Transplantation: A Systematic Review Protocol
Supplemental material, sj-docx-1-cjk-10.1177_20543581241306799 for Incidence, Prediction, and Prevention of Fractures After Kidney Transplantation: A Systematic Review Protocol by Andrea C. Cowan, Karla Solo, Victoria Lebedeva, Yasaman Mohammadi Kamalabadi, Maha El-Shimy, Aayushi Joshi, Edith Ginika Olalike, Misa Tanaka, Adam G. R. Klotz, Hatoun Wahid Elazhary, Antonia Zhu, Adam Forster, Shafaz Veettil, Sachin G. Nair, Maria Fernanda Servin Martinez, Dweeti Nayak, V. Nikhila Priya, Catherine Wellan, Diana Maria Cespedes Arcani and Pavel S. Roshanov in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Materials
Not applicable.
Author Contributions
All authors made substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work. In addition, all authors participated in drafting the work or revising it critically for important intellectual content, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Western University Division of Nephrology Evidence Synthesis Grant. Dr Cowan and Dr Roshanov receive salary support from the Academic Medical Organization of Southwestern Ontario Opportunities award; Dr Roshanov also receives research support from the William F. Clark Chair in Nephrology. The funding sources had no role in the design of this study, its execution, analyses, interpretation of the data, or decision to submit results.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
