Abstract
Background:
Chronic kidney disease following liver transplantation is a major long-term complication. Most liver transplant recipients with kidney failure will be treated with dialysis instead of kidney transplantation due to noneligibility and shortage in organ availability. In this population, the role of peritoneal dialysis (PD) as a modality of kidney replacement therapy (KRT) remains unclear.
Objective:
To determine the feasibility regarding safety, technique survival, and dialysis efficiency of PD in liver transplant recipients requiring KRT for maintenance dialysis.
Design:
Systematic review.
Setting:
Interventional and observational studies reporting the use of PD after liver transplantation.
Patients:
Adult liver transplant recipients with kidney failure treated with maintenance KRT.
Measurements:
Extracted data included eligibility criteria, study design, demographics, and PD modality. The following outcomes of interest were extracted: rate of peritonitis and microorganisms involved, noninfectious peritoneal complications, technique survival, and kidney transplantation-censored technique survival. Non-PD complications included overall survival, liver graft dysfunction, and hospitalization rate.
Methods:
The following databases were searched until July 2020: MedLine/PubMed, EMBASE, CINAHL, and Cochrane Library. Two reviewers independently screening all titles and abstracts of all identified articles. Due to the limited sample size, observational designs and study heterogeneity expected, no meta-analysis was pre-planned. Descriptive statistics were used to report all results.
Results:
From the 5263 identified studies, 4 were included in the analysis as they reported at least 1 outcome of interest on a total of 21 liver transplant recipients, with an overall follow-up duration on PD of 19.0 (Interquartile range [IQR]: 9.5-29.5) months. Fifteen episodes of peritonitis occurred in a total cumulative PD follow-up of 514 patient-months, representing an incidence rate of 0.35 per year. These episodes did not result in PD technique failure, mortality, or impairment of liver graft function.
Limitations:
Limitations include the paucity of studies in the field and the small number of patients included in each report, a risk of publication bias and the impossibility to directly compare hemodialysis to PD in this population. These results, therefore, must be interpreted with caution.
Conclusions:
Based on limited data reporting the feasibility of PD in liver transplant recipients with kidney failure, no signal was associated with an increased risk of infectious complications. Long-term studies evaluating this modality need to be performed.
Registration (PROSPERO):
CRD42020218374.
Introduction
In advanced liver disease, liver transplantation (LT) is the treatment of choice in eligible patients. Over time, graft and patient survival have improved significantly with the development of potent immunosuppression, including calcineurin inhibitors (CNIs). 1 The development of posttransplant chronic kidney disease (CKD) is a major long-term complication of LT, with stage-3 CKD occurring in 56.8%, stage 4 in 6.1%, and stage 5 in 2.3% of LT recipients 10 years posttransplant.2,3 Since the introduction of the Model for End-stage Liver Disease (MELD) allocation system in 2002 that prioritizes LT candidates with pre-existing kidney dysfunction, the incidence of CKD after LT has progressed, where stage 4 or 5 occurred in up to 22% of all LT recipients 5 years after transplantation.4,5 Among LT recipients who survived the first 3 months post-transplantation, 4% will eventually require maintenance dialysis. 1
Post-transplant CKD has various causes, including pretransplant insults such as infection, renal hypoperfusion, perioperative renal insults, and finally, postoperative offenses such as CNI toxicity. 6 The occurrence of CKD after nonkidney organ transplantation is not without consequence, with an increased risk of death when compared to solid-organ transplant recipients who have not developed CKD. 1
Kidney transplantation confers a significant survival benefit in nonkidney solid-organ transplant recipients when compared to remaining wait-listed and on dialysis.5,7-9 However, due to the shortage in organ availability as well as the noneligibility of a large proportion of these patients, most LT recipients who develop kidney failure need to initiate dialysis. 8 Currently, hemodialysis (HD) is the most common kidney replacement therapy (KRT) modality used in LT recipients suffering from kidney failure. 8 These patients represent 3% to 5% of the prevalent long-term HD population in the United States. 1
In the general population of patient undergoing KRT, peritoneal dialysis (PD) and HD appear to be equivalent in terms of patient survival. 10 However, the role of PD in patients having received previous LT remains unclear. Possible concerns about technique success and infectious risks may explain the limited use of PD in this patient population. 11 Although a few publications have reported the success of the PD technique in LT recipients progressing to kidney failure, no study has systematically analyzed the feasibility of PD in this clinical context.
Our aim was to assess the feasibility regarding safety, technique survival, and dialysis efficiency of PD in patients who develop end-stage CKD after LT. To achieve this, we performed a systematic search of the literature to uncover the potential benefits and limitations of PD in this patient population.
Method
This systematic review is reported following the Preferred Reporting Items for Systematic Reviews and Meta-analyses guidelines. 12 The protocol was registered into the PROSPERO international prospective register of systematic reviews (CRD42020218374).
Eligibility Criteria
Eligible studies reported the use of PD for maintenance KRT in adult LT recipients. Studies addressing HD were included if data for at least one patient treated with PD were reported separately. Patients who received LT from living or deceased donors were included. Likewise, studies addressing maintenance KRT after various solid-organ transplantations were included if data for LT recipients were individually reported. No study design, date, or language limits were imposed on the search.
Literature Search
According to the predetermined protocol, a systematic literature search of the following databases was performed from inception until July 2020 in cooperation with a trained medical librarian: MedLine/PubMed, EMBASE, Cochrane Library, and CINAHL. The literature search strategy was developed using medical subject headings and text words related to PD and LT (Supplementary File, Table S1). To ensure literature saturation, we also scanned the references list of all eligible articles and searched the gray literature for all abstracts listed into the annual meetings archives of the American Society of Nephrology, the European Renal Association-European Dialysis & Transplant Association, the International Society of Peritoneal Dialysis, and the International Society of Nephrology. Finally, we circulated a bibliography of the included articles to all authors, to add any other relevant publications.
Study Selection
Eligible studies were clinical trials, cohort studies, case-control studies, case reports, and case series. Published abstracts were also eligible when at least one outcome of interest was described quantitatively. Literature search results were screened with Rayyan QCRI. Two reviewers (J.M.C. and I.E.) independently screened the titles and abstracts of all identified articles. Disagreements were resolved by consensus-based discussions.
Data Extraction and Quality Assessment
Extracted data included eligibility criteria, study type, demographics, PD modality, and results. The following prespecified outcomes of interest related to PD were extracted: rate of peritonitis and microorganisms involved, noninfectious peritoneal complications classified as abdominal hernia, exit-site or abdominal leak, or others; technique survival and kidney transplantation-censored technique survival. Non-PD complications included overall survival, liver graft dysfunction, and hospitalization rate.
Risk of bias was assessed using the Newcastle-Ottawa Score for cohort studies. Although not specified in the preplanned protocol, the Joanna Briggs Institute Tool was used to assess the risk of bias for case series. 13 When insufficient details were reported, the risk of bias was judged as unclear.
Statistical Analysis and Synthesis of Results
As most studies were expected to be observational, no meta-analysis was preplanned. Descriptive statistics were reported as median with interquartile range (IQR) for continuous variables and proportion for dichotomic variables (SPSS 26; Armonk, NY; IBM Corp.). No statistical hypothesis test was performed due to the limited sample size and various follow-up definitions. Heterogeneity across all included studies was described narratively. When quantitative synthesis was not possible, a narrative synthesis provided additional relevant information.
Results
Study Selection
Study selection is depicted in Figure 1. After removal of duplicates, we identified 5,263 studies. Of these, 5,251 were excluded after an initial screening of titles and abstracts. A total of 12 unique studies were deemed eligible for full-text screening8,11,14-23 from which 4 presented quantitative results for at least 1 prespecified outcome.15,16,20,21 The remaining studies were excluded based on reasons specified in Figure 1. The studies by Shukla et al 22 and Cornelis et al 17 were excluded because cases reported were already described in the more recent publication by Saiprasertkit et al. 20

Flow chart of included studies.
Study and Patient Characteristics
An overview of study characteristics is presented in Table 1. All included studies, published between 2004 and 2018, were observational. One study had a comparator group of PD patients with no history of transplantation. 16 Two studies described cases from Canada, while the two others were from France and the United States, cumulating 21 LT recipients treated with PD as a modality of KRT. Patients’ median age was 60 (IQR: 57-66) years and 43% of patients were female. The most common cause of liver failure leading to LT was viral hepatitis (63%), while CNI toxicity, based on clinical judgment, was involved in the cause of kidney failure for 81% of reported patients. Only one patient started KRT following acute kidney injury in the postoperative LT period and remained dialyzed for 7 months. 15 For the remaining, the time between LT and kidney failure requiring KRT initiation ranged from 2 to 24 years (median: 10.0; IQR: 7.1-15.8). At the time of PD initiation, 38% of LT recipients had diabetes, and only 1 patient was anuric. The PD modality chosen was automated peritoneal dialysis (APD) for 11 patients and continuous ambulatory peritoneal dialysis (CAPD) for another 11 patients. For 19 patients (90%), the first modality of KRT received was PD, while 2 patients were previously treated with HD before transferring to PD. The overall follow-up duration on PD was 19.0 months (IQR: 9.5-29.5).
Characteristics of Included Studies and Patients.
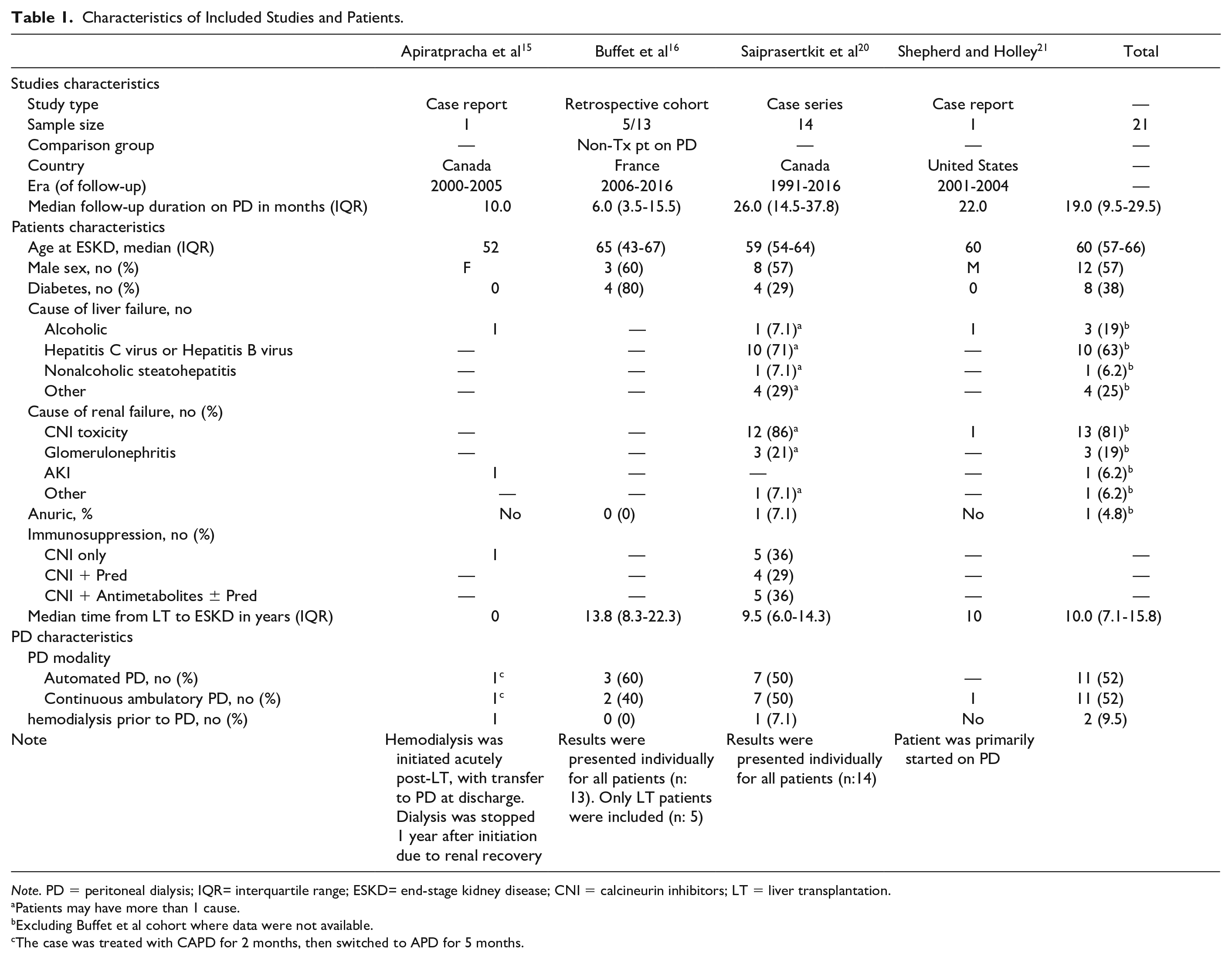
Note. PD = peritoneal dialysis; IQR= interquartile range; ESKD= end-stage kidney disease; CNI = calcineurin inhibitors; LT = liver transplantation.
Patients may have more than 1 cause.
Excluding Buffet et al cohort where data were not available.
The case was treated with CAPD for 2 months, then switched to APD for 5 months.
Risk of Bias and Quality Assessment
The risks of bias for cohort studies and case series are presented in Supplementary Tables S2 and S3. As all studies included were either retrospective cohort studies, case series, or case reports, the overall quality of evidence resulting from this systematic review was low. Considering the limited sample size and the rare use of PD in LT recipients, there is a risk of publication bias, where only positive experiences regarding PD after LT might have been reported in the medical literature.
Outcomes
Peritonitis
When combining all studies, 15 episodes of peritonitis occurred in a total cumulative PD follow-up of 514 patient-months, representing an incidence rate of peritonitis of 1 for every 34 patient-months or 0.35 episodes per patient-year. Clinical evolution and response to antimicrobial therapy were reported as similar to PD-related peritonitis in nontransplanted patients. Microorganism identification was specified in 2 studies16,20 and is detailed in Table 2. Staphylococcus sp. were the most frequently reported. The comparison between the rate of peritonitis for these LT recipients with the complete local PD program for Saiprasertkit et al 20 is presented in Table 3. The study by Buffet et al 16 showed no difference in the peritonitis-free survival when comparing the whole cohort of solid-organ transplant recipients treated with PD to nontransplant recipients (hazard ratio [HR]: 0.91; 95% CI: [0.37, 2.22]). One patient died after hospitalization for peritonitis-associated sepsis. 20 No study reported the antimicrobial regimen used for these peritonitis episodes.
Outcomes Reported in Included Studies.

Note. PD = peritoneal dialysis.
Including only liver transplanted patients on PD from the cohort.
Multiple organisms can be implicated for a single peritonitis episode.
Defined as inadequate PD leading to transfer to hemodialysis.
Risk of Peritonitis in Liver Transplant Recipients Compared to Nontransplanted Patients on Peritoneal Dialysis.

Note. PD = peritoneal dialysis; ? = data not reported; CI = confidence interval.
Noninfectious PD-related complications
Abdominal hernias were reported for 3 patients (14%) of which 2 were corrected surgically with patients resuming PD after surgery15,20 One was medically managed with conversion from CAPD to APD with dry day to minimize intra-abdominal pressure. 15 Exit-site leak was reported for 1 patient who required a temporary conversion to HD before resuming PD once the exit-site had healed. 20 Catheter dysfunction occurred in 2 patients, but no additional information regarding its impact was reported. 16
Dialysis adequacy and technique survival
The overall dialysis adequacy was considered within current standards for all included studies. One patient (4.8%), from the Saiprasertkit et al study, 20 was transferred to HD after 16 months due to poor ultrafiltration, reaching the endpoint of PD failure. The remaining LT recipients terminating PD either underwent kidney transplantation (n = 6, 29%) or died (n = 7, 33%) during follow-up. The median time from PD initiation until death was 10 months (IQR: 5-33, n = 7).
Liver graft dysfunction
No liver graft dysfunction or rejection was observed in any study. One group reported their home protocol for liver biopsies for PD patients. 20 The procedure was done in the absence of intraperitoneal PD solution, with antibiotic prophylaxis consisting of oral amoxicillin and intravenous tobramycin. Patients were allowed to resume PD exchange 48 hours after the procedure.
No data regarding the prespecified outcome of hospitalization rate were reported in the selected studies.
Discussion
As LT recipient survival increases, CKD has become a major complication of LT, leading to increased morbidity and mortality. Most CKD risk factors in this population, such as the use of CNI, need for contrast procedures, or pre-existent CKD, cannot be easily mitigated. 24
The impact of the dialysis modality on LT recipients who require KRT remains unknown. However, having the opportunity to offer a home-based KRT modality can be attractive for this patient population as LT recipients are on average younger that the general dialysis population. 17 Potential benefits of PD include reduction of the risk of hepatitis virus transmission, optimization of hemodynamic stability, decreasing risk of catheter-related bloodstream infection, and preservation of residual kidney function and vascular access.11,20,25-27 However, infectious and anatomical concerns regarding the feasibility of PD in patients with an intra-abdominal transplant and remodeled abdomen may deter nephrologists from choosing PD when LT recipients develop end-stage CKD.
Here, we identified four studies reporting quantitative data for 21 patients concerning our prespecified outcomes of interest. The paucity of reports of PD use in LT recipients probably reflects the fear of an increased risk of peritonitis and associated complications in this immunocompromised population. Two studies allowed direct comparisons of the rate of peritonitis in LT patients to that observed in cohorts of nontransplanted PD patients (see Table 3).16,17,20 When pooling the reported experience from this systematic review, an overall peritonitis incidence rate of 1/34 patient-months was found, which represents 0.35 infections per patient-year. This rate is slightly higher than that recently reported for general PD population in Canada (0.27; 95% CI: [0.25, 0.30]) and Australia/New Zealand (0.29; 95% CI: [0.26, 0.32]) but similar to that observed in the United Kingdom (0.35; 95% CI: [0.30, 0.40]). 28
Immunosuppression is a known risk factor for peritonitis and other intra-abdominal infections, and this risk cannot be easily minimized following solid-organ transplantations.29,30 However, observational data from Australia and New Zealand showed that patients with ESKD who started PD after failed kidney transplants—most of whom were still receiving immunosuppressive therapy—had similar peritonitis-free survival (adj. HR: 0.92, 95% CI: [0.72, 1.16], P = .44) to those starting PD with native kidneys. 31 In addition, other studies have reported incidence rates of peritonitis of 0.36 episode per patient-year following heart and lung transplantation, 32 and 0.41 episode per patient-year following nonrenal solid-organ transplant in a second. 17 These figures are similar to the incidence rate we derived in this systematic review of LT recipients (0.35 episode per patient-year). However, when compared to nontransplanted PD populations receiving immunosuppressive therapy, the risk of peritonitis in LT patients was lower than for PD patients with lupus (0.68 episode per patient-year) but similar to PD patients with primary long-term glomerulonephritis (0.29 episode per patient-year). 33 Thus, in this review, no clinically significant trend of an increased risk of peritonitis episodes in LT patients receiving PD has been identified, when compared to nontransplanted patients receiving immunosuppression or to patients transplanted with solid organs other than the liver.
We found similar clinical evolution during treatment and no report of liver graft dysfunction following peritonitis episode. Some centers have already developed protocols to minimize the risk of infectious complications in patients requiring posttransplantation liver biopsy. 20 However, no international guideline has been published regarding the prophylactic management of such interventions in LT recipients on PD.
LT is a potential risk factor for impaired wound healing and postsurgery incisional hernia. The limited number of studies selected does not allow us to draw definite conclusions concerning the safety of PD with regards to noninfectious complications. Nevertheless, none of the patients who developed such complications had technique failure requiring permanent transfer to HD. To decrease intrabdominal pressure associated with PD exchange, LT recipients might be better candidates for APD then CAPD when possible. Careful planning of early PD catheter insertion in patients with progressive deterioration of kidney function may also reduce the risk of requiring urgent HD initiation prior to PD by permitting sufficient time for wound healing.11,20
Only 1 case report quantitatively mentioned dialysis adequacy using Kt/v. 21 However, all other studies reported that dialysis adequacy for these patients was within standard recommendations until the end of follow-up, death, or kidney transplantation, except for one. This patient, with ultrafiltration failure, was transferred to HD 16 months after PD initiation. 20 Overall, although LT is considered as major abdominal surgery, there is no evidence in this review showing any significant peritoneal membrane malfunction, lower PD efficiency, or limitation in the installation of a functioning catheter. However, due to the limited sample size, this review does not allow to definitely address this question.
On the contrary, the short median follow-up in these studies could have underestimated some late complications of PD such as sclerosing encapsulating peritonitis, for which both PD and LT patients are at increased risk.34,35
This review has several limitations. As the topic of PD after solid-organ transplantation is rarely described in the literature, a risk of publication bias exists, where it is possible that only positive experiences were published. In addition, the observational design of all included studies, as well as the small number of patients reported, limit the level of evidence resulting from this review. The level of details in the description of all cases reported varied across included studies. Finally, as no study directly compared patients treated with PD to similar patients treated with HD, we were unable to evaluate the impact of the dialysis modality itself in LT recipients requiring KRT.
Conclusion
With the caveat of limited data availability, our systematic review of the literature shows that PD is feasible in LT recipients who require KRT, where the risk of peritonitis reported was similar than that reported in comparable PD populations receiving immunosuppression. These episodes of peritonitis had no significant impact on clinical evolution, technique survival, and liver graft function. Due to observational data mostly coming from limited case series, no definitive recommendation regarding this modality can be formulated. However, despite the possibility of publication bias, we found no signal suggesting that undertaking PD following LT is putting the patient at an increased risk of infectious or technical complications. Large and long-term studies evaluating PD in this population need to be performed. Further collaborative multicenter studies may help to address this important question.
Supplemental Material
sj-pdf-1-cjk-10.1177_20543581211029722 – Supplemental material for Peritoneal Dialysis After Liver Transplantation: A Systematic Review
Supplemental material, sj-pdf-1-cjk-10.1177_20543581211029722 for Peritoneal Dialysis After Liver Transplantation: A Systematic Review by Jean Maxime Côté, Isabelle Ethier, Héloïse Cardinal and Marie-Noëlle Pépin in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors would like to thank Daniela Ziegler, biomedical librarian, Center hospitalier de l’Université de Montréal, for her assistance with the systematic literature search.
Ethics Approval and Consent to Participate
Not applicable for this type of study.
Consent for Publication
All authors consent to the publication of this study.
Availability of Data and Materials
Data are available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No specific funding has been received for this study. J.M.C. was supported by the Fonds de Recherche en Santé du Québec and Société Québécoise de Néphrologie. J.M.C. and I.E. were supported by a Fellowship grant from the Center Hospitalier de l’Université de Montréal and the Fondation du CHUM. H.C. is a Fonds de Recherche en Santé du Québec Junior 2 Scholar.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
