Abstract
Background:
Sodium-glucose co-tranporter-2 inhibitors have been shown to be safe and effective in patients with type 2 diabetes for improving glycemia. Furthermore large, randomized control trials have shown cardiovascular and renal benefits. However, limited safety and efficacy data is available in kidney transplant patients with diabetes.
Objective:
To investigate the safety and efficacy of SGLT2i use on stability of renal function in adult kidney transplant recipients (KTR) with type 2 diabetes mellitus (DM2) or New Onset Diabetes After Transplantation (NODAT).
Design:
We performed a single center, retrospective cohort study pre- and post-SGLT2i exposure.
Patients:
Adults with DM2 or NODAT who received a living or deceased kidney transplant (Tx) and started on an SGLT2i post-Tx were reviewed. Patients who had type 1 diabetes were excluded.
Measurements and Methods:
The baseline was the SGLT2i start date. We reviewed available data from 24 months (M) before and after SGLT2i initiation. The primary endpoints were the effects of SGLT2i use on stability of renal function using serum creatinine and eGFR, change in urine albumin excretion(uACR), and glycosylated hemoglobin (A1C). Secondary endpoints compared blood pressure, body mass index and adverse reactions at baseline and quarterly after SGLT2i initiation.
Results:
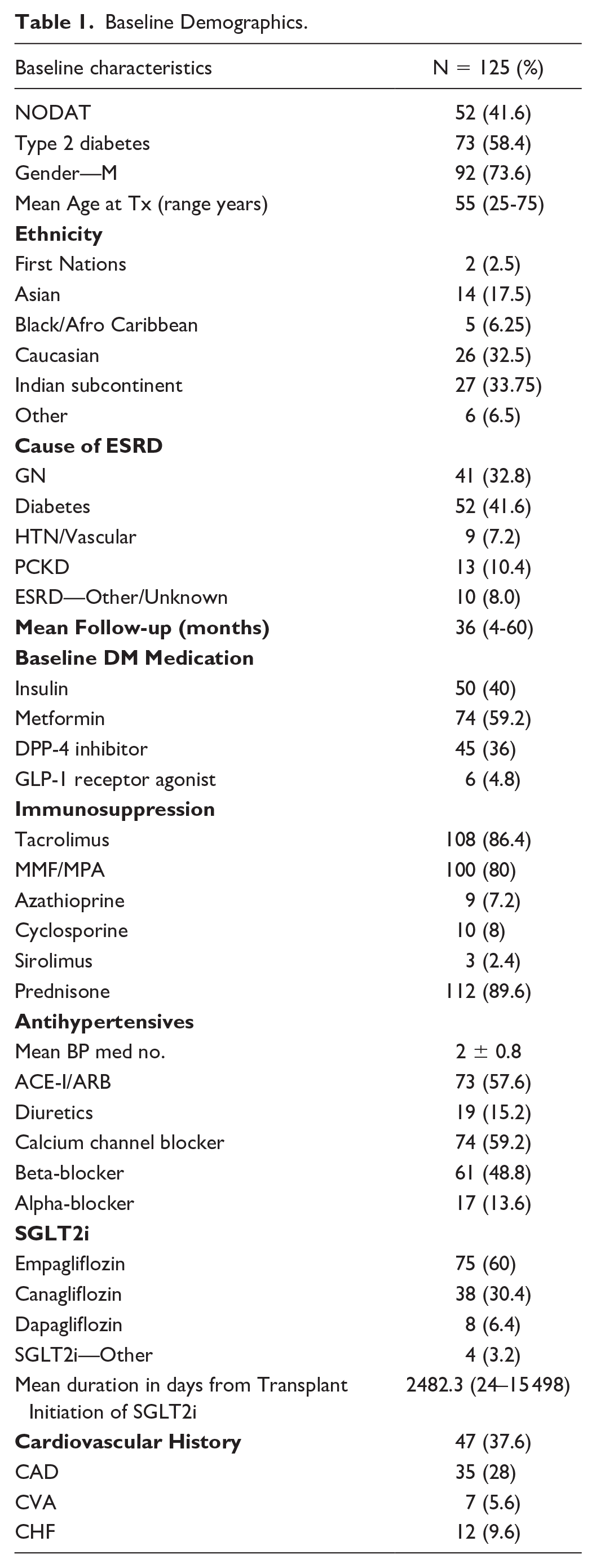
125 KTRs were included in cohort: NODAT (52, 42%), DM2 (73, 58%); female (33, 27%); mean age at Tx 55 years (25-75); LD (56, 45%), DD (69, 55%); mean duration of Tx (6.8 years, 0.1-42.5); study follow-up (1.8 years, 0.3-4.9).
The mean eGFR remained stable pre-SGLT2i at 64.6 mL/min/1.73m2, vs post at 64.3 mL/min/1.73m2. There was no difference in mean A1C after SGLT2i initiation. The slope of uACR using natural log transformation pre-SGLT2i compared with post-SGLT2i slope reduced from +0.7 (0.03, 0.11) to -0.04 (-0.01, -0.35) mg/mmol/3mths (P = .002). The risk of developing new genital mycotic infections among all patients was 4% (95% CI 1.3%-9.1%) While there was no significant difference in UTI before (13.6%) and after (12%) SGLT2i use (P = .68), there was a higher risk of UTI seen in patients with a previous history of UTI (23.5%) vs no previous history (10.2%) post initiation. There was no significant increase in AKI pre 8%, post 10.4%, P = .51. There was a single DKA event pre- and post-SGLT2.
Limitations:
The limitations of this study include its retrospective nonrandomized nature.
Conclusion:
In this retrospective analysis, SGLT2i use in KTR appears to be safe and efficacious with stable renal function and glycemic control, alongside improvements in uACR. There was a low risk of new genital yeast infections after SGLT2i start. UTI occurrence was higher in patients with a previous history of UTI compared with those with no previous history.
Introduction
Diabetes is the leading cause of end stage kidney disease (ESKD), requiring dialysis or transplant, in Canada. 1 Kidney transplantation is the treatment of choice in patients with ESKD affording better quality of life and decreasing mortality risk compared with dialysis. However, new onset diabetes after transplant (NODAT) occurs in up to 45% of kidney transplant recipients (KTR) who are at a higher risk of infection, rejection, and graft loss as well as diabetes associated macrovascular and microvascular complications. 2 The prevalence of NODAT at our institution is approximately 20% by 2 years posttransplant.
Sodium glucose transport 2 inhibitors (SGLT2i) have changed the landscape for the treatment of type 2 diabetes in patients with established cardiovascular disease (CVD) or high risk for CVD and diabetic kidney disease. Large cardiovascular and renal outcome trials (CVOTs), namely EMPA REG outcome, EMPA-Kidney CANVAS program/CREDENCE, DECLARE-TIMI 58, DAPA-CKD, and VERTIS CV have shown that SGLT2i not only improves glycemia and are safe, but also have cardiovascular and renal benefits.3-7
SGLT2i work by blocking glucose reabsorption in the S1 segment of the proximal convoluted tubule, resulting in glycosuria which can exceed 100g/day in patients with type 2 diabetes leading to improved glycemic control of up to 1% reduction in A1c. 8 Furthermore, SGLT2i have been shown to have additional beneficial effects, such as weight loss and blood pressure reduction.3-5,7 The main adverse events include euglycemic DKA, mycotic genital infections, urinary tract infections, and lower limb amputations. 8
However, kidney transplant recipients (KTR) were not included in these large trials. Therefore, SGLT2i raise concerns for urinary tract infection and mycotic genital infections in view of surgically altered urinary tract and immunosuppression. Previous studies in non-KTR have shown that the incidence of UTIs can be up to 34% compared with placebo and non-SGLT2i. 9 Furthermore, mycotic genital infections were 3.3 times higher in patients on SGLT2i. 9 Conversely, a Canadian population study found an increased risk of UTIs were not associated with SGLT2i use compared with other antidiabetic medications, specifically DPP-4i, SU, GLP-1 RA, and TZD. 10
Thus, we sought to examine the safety and efficacy of SGLT2i in KTR. Their metabolic advantage favors KTR as they are more at risk for metabolic dysfunction—specifically NODAT due to secondary effects of immunosuppressants. Cardiorenal benefits have been shown in limited KTR cohort studies. 11 However, there is a paucity of data to demonstrate a significant difference in eGFR, serum creatinine, uACR in KTRs on SGLT2i. This study is the largest single-center cohort study looking into the safety and efficacy of SGLT2i in KTR and offers the longest follow-up after receiving SGLT2i. 11
The primary objective of this study was to understand and confirm the safety and efficacy of SGLT2i use and the effects on the stability of kidney function in KTR with type 2 diabetes or NODAT. The primary outcome was to determine the effects of SGLT2i use on the stability of kidney function using measures of serum creatinine, eGFR and uACR. We compared BP, weight, glycemic control (A1C), lipids and adverse reactions at SGLT2i initiation and quarterly post-SGLT2i initiation as secondary outcomes.
Methods
This was a single-center pre- and postexposure retrospective cohort study. St. Michael’s hospital, Unity Health Toronto is a major kidney transplant center in Toronto, that provides care to approximately 1900 adult kidney transplant recipients of diverse ethnicities. We identified a cohort of all participants who received a living donor or deceased donor kidney transplant with type 2 diabetes or NODAT and started on an SGLT2i on or before December 31, 2020. One of the authors T.M.-A. is a nurse practitioner in our transplant clinic who specializes in diabetes management and prescribed the majority of patients in this study with the SGLT2i. The initiation of the SGLT2i was individualized and based on A1c or uACR. Data was collected from both our electronic kidney transplant database, Claris FileMaker Pro and Soarian electronic records until March 31, 2021, allowing for at least 3 months of posttransplant data. We retrospectively collected demographic and baseline data spanning 24 months prior to SGLT2i initiation and up to 24 months following initiation. The date of SGLT2i initiation served as the baseline date for each participant. Subsequently, data was collected at quarterly intervals from this baseline date for as long as it remained available, with a maximum post-SGLT2i time frame of 24 months, totaling a maximum of 48 months observation period. Research Ethics Board approval was obtained.
Clinical Data
Demographic data collected included age, gender, and ethnicity. Past medical history specifically diabetes, renal disease, cardiovascular disease, and transplant were collected. Anthropometric and clinical information such as weight, height, body mass index, seated and standing blood pressure, heart rate, current medications (i.e. immunosuppressive medications, antihypertensive medications, lipid lowering medications, diabetes medications, diuretics) were collected from electronic charts. SGLT2i information such as, name and dose, date of initiation, date of discontinuation, time of first dose from transplant, were also collected.
Adverse Events (AEs)
Euglycemia diabetic ketoacidosis, urinary tract infection, genital/yeast infection, volume depletion, hypotension, hypoglycemia, hyperkalemia, AKI, allergic reaction, cardiovascular complications, and amputations were collected starting from the date of most recent transplant up to March 31, 2022.
Statistical Analyses
Data for eGFR, hemoglobin A1c (A1C), uACR, blood pressure (BP) and body mass index (BMI) were obtained at 3-month intervals during 24 months before and after SGLT2i initiation periods. To determine whether changes in eGFR, A1C, BP and BMI were different between periods, we used the piecewise linear mixed effects regression model with random intercepts and slopes. For uACR, we chose a model that considered zero values for samples under the detection limit of 0.2 (left censoring), and the uACR skewed distribution of positive values. We fitted a model that combined the piecewise linear mixed model with random intercepts, with log transformed positive uACR and censoring due to values below the limit of detection 12
All models followed the parametrization provided in Singer and Willett, 13 accounted for the repeated measurements, and estimated intercepts and slopes in each period, as well as the change in intercepts and slopes with 95% confidence intervals (details for both models are provided in Supplemental Material 1). Because we assumed that there were no differences by sex at birth, and because of over one-third of missing data for ethnicity, we did not adjust for these variables in our models.
AEs were collected before and after SGLT2i initiation were summarized as counts and percentages. Due to very low rate of most AEs, McNemar’s test was performed to compare incidence in the 2 periods for only a few of the AEs.
All analyses were conducted using SAS 9.4 (SAS Inc. Cary, NC, USA) and statistical significance was defined when p-values are less than 0.0. Detailed results are presented in Supplemental Material 1 and summarized in Table 2.
The study was approved by the local REB.
Results
129 KTR met initial criteria of which 4 were excluded. Of those excluded, 1 had type 1 diabetes, 1 we could not confirm date and type diabetes, 1 was prescribed an SGLT2i but never started, and 1 did not have diabetes. The remaining 125 KTR who received a transplant and started an SGLT2i on or before December 31, 2020, were included in the analysis. Each patient served as their own control as they each individually contributed to data 24 months before their SGLT2i initiation date and 24 months after their SGLT2i initiation date. The majority of patients were either on Empagliflozin (60%) or canagliflozin (30%) reflecting the available evidence on nontransplant renal patients. Fifty eight percent of patients were on either an ACEi or ARB. Patient demographics are shown in Table 1.
Baseline Demographics.
Results From Linear Piecewise Mixed Effects Models for Primary and Secondary Endpoints.

Primary Endpoints
eGFR
The mean eGFR at 24 months before SGLT2i initiation was 64.6 (95% CI 61.1-68.2) mL/min/1.73m2 The mean eGFR immediately post-SGLT2 was not different, at 64.3 (60.3, 68.2) mL/min/1.73m. 2 Post-SGLT2, the rate of change per every 3 months of eGFR decreased faster to -0.43 (95% CI -0.80 to -0.05), from -0.16 (95% CI -0.53 to 0.21) mL/min/1.73m2 per 3 months. However, our data did not provide evidence for a significant change, -0.26, 95% CI (-0.80 to 0.27), P = .3310, (Figure 1)

eGFR 24 months pre- and post-SGLT2i.
Hemoglobin A1c
There were no differences in the mean A1c at 24 months before and immediately after (change 0.001, 95% CI -0.001 to 0.004). Our data suggest a difference in rates of change per 3 months between pre (0.0002, 95% CI -0.0002 to 0.0007) and post (-0.0006, 95% CI -0.0012 to -0.0001) periods, resulting in a difference of -0.0009, 95% CI -0.0017 to -0.0001, P = .0332.
uACR
Of all uACR measurements, 17.2% were censored (coded as zero because under the detection limit of 0.2). In addition, only 10.5% of all uACR measurements were ≥30mg/mmol. Of importance in these results is the fact that the pre-SGLT2i log(uACR) slope was positive, at 0.07 (0.03, 0.11) per every 3 months but that after receipt of SGLT2i, the trend switched direction to -0.04 units of log(ACR). The change in slopes post minus pre slopes was -0.11, 95% CI -0.16 to -0.05, P = ..0002 (Figure 2).

Predicted mean (95% CI) natural log transformation of uACR (mg/mmol) trajectory between 24 months pre- and 24 months post-SGLT2 initiation.
Secondary Endpoints
Blood pressure
Our data suggest stability of systolic blood pressure, with no evidence for significant change in mean SBP immediately after initiation of SGLT2i (change in level -1.7, 95% CI -5.5 to 2.2, P = .3929) or in change in slopes -0.4, 95% CI -1.4 to 0.7, P = .4770. Diastolic blood pressure pattern was likewise unchanged.
BMI
Our data do not support evidence for change in pattern of BMI post-SGLT2i initiation compared with preinitiation.
Adverse Events
As shown in Table 3, There were no differences in incidence of any adverse event pre- and post-SGLT2i initiation.
Adverse Events Pre- and Post-SGLT2i Initiation (N = 125).

Events too rare to compare.
Discussion
Our cohort of 125 patients followed up retrospectively for a maximum of 48 months presents the largest single center SGLT2i cohort study and longest follow-up time study on KTR. The eGFR did not change, and although the uACR was not very high pre-SGLT2 initiation, in those with uACR >30 mg/mmol there was a reduction posttreatment. A1c only slightly decreased, suggesting it is not as proficient at glycemic control than originally thought. The cardiovascular benefits were evident in this study with the improvement of BP and the reduction of the probability for CVD. UTI were not different; however, this is most likely due to confounding with other treatments. Furthermore, AKI was not different Overall, our data, which has the longest follow-up time, further suggests that SGLT2i are safe and effective as an anti-diabetic in KTR.
Renal function represented by eGFR remained stable post-SGLT2i initiation compared with pre-SGLT2i. However, the eGFR mean rate of change increased slightly. This eGFR dip is a known effect postinitial start likely due to vasoconstriction of the afferent arteriole. 14 However, evidence shows this initial dip is not associated with long term adverse kidney outcomes or AKI in non KTR. 13 We found no significant increase in AKI pre- (8%) vs. post- (10.4%) initiation (P = .51).
There was no significant difference in mean A1c observed. SGLT2i reduction of HbA1c is dependent on eGFR. 15 Halden et al 15 demonstrate that the reduction in HbA1c is greater in patients with eGFR > 60 mL/min per 1.73m2, which favors the mechanism of insulin-dependent reduction through renal glucose excretion. Interestingly, in our data our model of unadjusted A1c was not different when we adjusted the A1c model for eGFR as a time varying covariant. Anemia could potentially affect level of A1c, but this was not accounted for in this study.
Along with stabilizing eGFR, SGLT2i offers protective affects against proteinuria. uACR post-SGLT2i slope compared with pre-SGLT2i slope reduced by -0.11 log units (95% CI -0.16 to -0.05, P < .01). Our first analysis of the pre-SGLT2i log (ACR) slope was positive, and there was a reduction in the post-SGLT2i. However, only 10% of patients had macroalbuminuria at baseline as a majority had already been on renin/angiotensin/aldosterone inhibitors.
The cardiorenal benefits of SGLT2i are well documented in literature. The EMPA-HEART studies showed great success in the regulation of BP and heart health in non-KTR patients. 4 In our cohort of KTR we found that SBP improved. In addition, probability of CVD was lower after initiation of SGLT2i. However, the probability of developing CVD is higher for patients who already had a cardiovascular event before SGLT2i initiation than those who did not have one. Interestingly, in spite of SGLT2i promoting increased EPO, hemoglobin was not statistically significant before and after baseline. 16
The CANVAS and CANVAS-R trials showed an increased risk for amputation among SGLT2i users. 4 SGLT2i promotes a diuretic effect in patient with diabetes, who are already at risk for low hemoconcentration. 17 Thus, it can increase the risk of peripheral artery disease and consequently peripheral amputation. 17 However, subsequent cohort studies have not shown this. 11 Our finding confirmed that there was no increased risk of amputation. A systematic review by Ujjawal et al 11 found that in 9 published studies with 182 KT4 in 8 different counties; there was no reported amputation after SGT2i initiation.
Previous studies like the CANVAS trials showed risks of DKA, 4 however in our cohort there was only a minimal risk of DKA with a 0.8% incidence rate, similar to pre-SGLT2i initiation. same incidence rate before initiation. Concurrently, Ujjawal et al 11 a found no cases of DKA in their population before or after initiation. This stresses the importance of patient-specific factors and careful monitoring in assessing the actual risk of DKA in clinical practice. Further research and larger studies are needed to fully understand the factors contributing to DKA incidence and its potential variation across different patient populations and treatment regimens.
New genital mycotic infections increased from a 0% to a 4% prevalence. UTI prevalence decreased in our cohort as well. These findings correlate to the reports from the CANVAS trial and other literature such Shuster et al 18 arguing that UTI’s are not as great of a risk than originally thought. 4 Despite the urine becoming a more favorable environment for bacteria, this is readily ameliorated by healthcare providers using preventative measures. This results in a misrepresentation in the numbers. Other possible reasons for the low risk of UTIs propose that the increased urinary flow and natriuresis effects of SGLT2 inhibitors are not conducive to bacterial colonization. 19
Retrospective observational cohort studies, while valuable for examining associations between exposures and outcomes, have limitations when patients are followed up for varying durations. Differential follow-up times can introduce survivor bias since it less likely for patients who have been followed for less time to experience events. In addition, varying follow-up durations may complicate comparison between groups, potentially confounding results and making it challenging to establish causality. Retrospective studies are also at risk to have incomplete data which can impact the statistical power of the study. In addition, such analysis can be confounded by selection biases. This was a single center study; however, our population was quite diverse.
Our study is an observational study that confirms the consensus that SGLT2i are safe as eGFR remains stable and protects against proteinuria. However, it did not reduce A1c as much as may have been anticipated. The gold standard of research is a randomized control trial, against other glycemic control medications like DPP-4i or GLP-1 RA, to more accurately conclude the cardiorenal, and glycemic control of SGLT2i on diabetic KTR. In the future, a greater focus on the effect of SGLT2i on nondiabetic KTR will help determine if it still offers protection against proteinuria, cardiorenal benefits, and stable eGFR. If similar result is seen, SGLT2i can be potentially used to stabilize renal control posttransplant, rather just for its anti-glycemic effect. Polypharmacy of SGLT2i needs to further be considered as SGLT2i are metabolized through the CYP3A4 pathway, the same utilized by calcineurin inhibitors. 11 Although no interactions of SGLT2i with calcineurin inhibitors and mycophenolate have been reported in the literature, there have been no studies evaluating these interactions specifically.
Conclusion
Our data shows that upon initiation of SGLT2i in diabetic KTR, eGFR and A1c remained stable, while uACR improved. In addition, there was a low risk at genital yeast infections and UTIs, however patients with previous UTIs were at a higher risk of developing a UTI upon SGLT2i initiation. The reduction in A1c levels was marginal, indicating it may not be as effective in managing blood sugar levels as initially believed. This adds to the growing body of evidence as outlining the safety and efficacy of SGLT2i in another at-risk population. However larger RCTs with longer follow-up time remain the gold standard for determining evidence-based conclusions.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241293202 – Supplemental material for Single Center Experience With Sodium-Glucose Co-Transporter-2 Inhibitors (SGLT2i) in Kidney Transplant Recipients With Diabetes
Supplemental material, sj-docx-1-cjk-10.1177_20543581241293202 for Single Center Experience With Sodium-Glucose Co-Transporter-2 Inhibitors (SGLT2i) in Kidney Transplant Recipients With Diabetes by Albi Angjeli, Tess Montada-Atin, Rosane Nisenbaum, Niki Dacouris, Michelle Nash, G.V. Ramesh Prasad and Jeffrey Zaltzman in Canadian Journal of Kidney Health and Disease
Footnotes
Authors’ Note
This work was presented as an oral abstract at the American Transplant Congress, San Diego USA, June 2023.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Approved by REB at St. Michael’s Hospital, Unity Health Toronto.
Consent to Participate
Not applicable.
Consent for Publication
All authors agree to publication.
Availability of Data and Materials
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
