Abstract
Background:
Despite atrial fibrillation affecting nearly 25% of patients receiving dialysis, conflicting study findings and guideline recommendations continue to make individual treatment decisions challenging.
Objective:
The objective was to examine the efficacy and safety of stroke-prevention interventions in patients with atrial fibrillation receiving dialysis.
Design:
A systematic review of randomized controlled trials and observational studies.
Patients:
Adult patients with non-valvular atrial fibrillation receiving any type of dialysis.
Measurements:
Ischemic strokes or systemic embolism, major bleeding, and all-cause mortality.
Methods:
We searched Medline, Embase, Cochrane Central Register of Controlled Trials, and the gray literature from inception to February 16, 2023. We selected all studies comparing antiplatelet, anticoagulant agents or left atrial appendage closure to placebo, no treatment, or an active comparator (most often vitamin K antagonists). Two reviewers independently screened and included the studies, extracted the data and assessed the risk of bias using the Cochrane Risk of Bias tool (version 1) and the Newcastle-Ottawa Scale. When randomized controlled trials were available, we pooled the risk ratios using fixed-effect meta-analyses. Results from observational studies were described narratively. We appraised the certainty of evidence (CoE) of the findings using GRADE.
Results:
Of the 8526 citations identified, we included 50 studies (4 randomized controlled trials and 46 observational studies) involving 155 058 participants. Our meta-analysis of 4 randomized controlled trials of direct oral anticoagulants (apixaban or rivaroxaban) compared with vitamin K antagonists (486 participants) suggested that the effect of direct oral anticoagulants on ischemic strokes or systemic embolism is very uncertain (risk ratio [RR] = 0.52; 95% confidence interval [CI] = 0.22-1.20; very low CoE). Direct oral anticoagulants may decrease major bleeding (RR = 0.67; 95% CI = 0.44-1.03; low CoE) and may result in little to no difference in mortality (RR = 0.89; 95% CI = 0.70-1.13; low CoE). Our narrative review of the observational evidence suggests that, compared with no treatment, vitamin K antagonists may decrease ischemic strokes or systemic embolism (18 studies, low CoE) and likely increase major bleeding (12 studies, moderate CoE), whereas their effect on mortality is very uncertain (16 studies, very low CoE). Compared with no treatment, the effect of direct oral anticoagulants on all outcomes is very uncertain (2 studies, very low CoE). We also found that the effect of antiplatelet agents, left atrial appendage closure, and heparin-related therapies is very uncertain on all outcomes (very low CoE), except for antiplatelet agents that may increase major bleeding (1 study, low CoE) compared with no treatment.
Limitations:
Most of the included studies were observational and retrospective, resulting in low or very low certainty for nearly all of the outcomes. The included interventions were highly heterogeneous, which precluded meta-analysis of the results in all cases except for direct oral anticoagulants.
Conclusion:
In patients with atrial fibrillation receiving dialysis, vitamin K antagonists may decrease ischemic strokes or systemic embolism compared with no treatment at the cost of an increased risk of major bleeding. Direct oral anticoagulants (apixaban or rivaroxaban) may also lead to less major bleeding than vitamin K antagonists. The overall low to very low certainty of the evidence emphasizes the importance of engaging in shared decision-making when selecting a strategy for stroke prevention for atrial fibrillation in people receiving maintenance dialysis.
PROSPERO registration ID:
CRD42022307009.
Introduction
Atrial fibrillation (AF) affects nearly 25% of patients receiving dialysis and is associated with an increased risk of ischemic stroke and mortality.1 -4 Stroke prevention is complex in this population at higher risk of both ischemic and bleeding events.4,5
Previous meta-analyses of randomized controlled trials (RCTs) in the general population have suggested warfarin reduces stroke risk by two thirds compared with placebo and direct oral anticoagulants (DOACs) by a further 19% compared with warfarin.6,7 The DOACs lead to a 52% lower risk of intracranial hemorrhages in the general population and a 21% lower risk of major bleeding in the population with chronic kidney disease (CKD).7,8 However, patients receiving dialysis were excluded from these large-scale RCTs, thus creating a knowledge gap for patients and clinicians. Recent Canadian, European, and US guidelines have yet to reach a consensus on whether to recommend DOACs, vitamin K antagonists (VKAs) or no anticoagulation for patients with AF receiving dialysis.9 -12
The VKAs were historically the first anticoagulants used for stroke prevention in AF. However, many studies demonstrated an increase in major bleeding and conflicting results on the reduction of ischemic strokes and systemic embolism in people receiving dialysis.13,14 Furthermore, VKAs may contribute to vascular calcifications and calciphylaxis, leading many clinicians to be cautious of VKAs in this population.15 -17 Although warfarin remains the preferred anticoagulant agent (89%) for AF in patients receiving dialysis in Canada, the prescription of apixaban and rivaroxaban has increased substantially in this population despite lacking high-quality evidence.18 -20 In the meantime, left atrial appendage closure (LAAC) has emerged as a non-pharmacological stroke-prevention intervention, but with minimal evidence in the dialysis setting. 21
Therefore, the risk-benefit ratio of usual stroke-prevention interventions for patients with AF receiving dialysis remains to be determined. Several systematic reviews addressed this question, but their conclusions were limited by conflicting results, high study heterogeneity, lack of control over essential confounders and, for some of them, the inclusion of patients not receiving dialysis.13,14,22 -30 Furthermore, an updated systematic review was needed to incorporate new evidence from RCTs on DOACs, to clarify the increasingly numerous observational studies on VKAs and to assess the risk-benefit ratio of other treatment strategies studied in the dialysis setting (eg, LAAC, antiplatelet agents). This systematic review aims to assess the efficacy and safety of all stroke-prevention interventions studied in adults with AF receiving dialysis to help inform shared decision-making with patients.
Methods
We conducted and reported this systematic review in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Supplementary Table S1). 31 The protocol was registered on PROSPERO (CRD42022307009). 32
Search Strategy
We searched Medline, Embase, and Cochrane Central Register of Controlled Trials from inception to February 16, 2023. Literature search strategies were adapted from the latest Cochrane search strategies in the fields of AF, anticoagulation and dialysis (Supplementary Table S2).33 -35 We searched unpublished trials using the International Clinical Trials Registry Platform and ClinicalTrials.gov. We searched the gray literature using Cochrane Kidney, Science.gov, Science.org, MedRxiv, and Google Scholar. Reference lists of eligible studies and recent systematic reviews were also screened for additional citations.
Eligibility Criteria
We formulated the eligibility criteria using the PICO (Population, Intervention, Comparator and Outcome) approach.
Patients
We included studies enrolling adult patients with non-valvular AF receiving any type of dialysis. Studies recruiting a broader population were included if they provided the results for the subset of participants meeting our eligibility criteria.
Interventions
We included any intervention to reduce stroke risks (antiplatelet agents, anticoagulants, LAAC). 36
Comparison
In a comprehensive approach, we included studies comparing the interventions to placebo, no treatment or all active comparators.
Outcomes
The outcomes of interest were SSE events (ischemic strokes or systemic embolism), major bleeding, and all-cause mortality. Systemic embolism is generally the primary outcome found in the literature on anticoagulation in AF and includes ischemic strokes, transient ischemic attacks, and arterial embolism. When systemic embolism data were not available, we used data for ischemic strokes. The definition of major bleeding varied across studies. We considered major bleeding that satisfied the International Society on Thrombosis and Haemostasis definition, which includes fatal bleeding, symptomatic bleeding in a critical area or organ, bleeding causing a 20 g/L fall in hemoglobin, or requiring 2 or more blood unit transfusions. 37
Other
We included RCTs and observational studies. Studies published in abstract form were included when at least 1 outcome of interest was reported quantitatively. The language was restricted to English and French. We contacted the corresponding authors to clarify the missing data where necessary.
Study Selection
Three authors (O.M., N.M., G.D.) independently screened all titles, abstracts and, when needed, full text for eligibility. Disagreements were resolved through discussion.
Data Extraction and Risk of Bias Assessment
Two authors (all authors, except A.B.-J.) independently extracted the data using a predefined extraction form and assessed the risk of bias. Disagreements were resolved through discussion, and a third author (G.D, N.D.) was available for persisting disagreements. For RCTs, we extracted the number of events and calculated risk ratios (RRs) with 95% confidence intervals (CIs). For observational studies, we extracted the number of events per 100 patient years and the maximally adjusted treatment effect with 95% CIs.
We assessed the risk of bias for RCTs using the Cochrane risk-of-bias tool (version 1). 38 We considered an RCT with any domain judged at high risk of bias as having an overall high risk. We assessed the risk of bias for observational studies using the Newcastle-Ottawa Scale (NOS). 39 We converted the NOS scores to low, moderate, or high risk of bias using the Agency for Healthcare Research and Quality Standards. 40
We also used the GRADE approach and its summary of findings table to report the certainty of evidence (CoE).41 -43 The GRADE assessment was conducted by 2 authors (O.M., G.D.) and approved by all authors. When no pooled estimates were available, we used a narrative description of the size effects in the summary of findings table.44,45 When RCTs were found, we prioritized their findings in our GRADE assessment but also considered complementary observational studies. 46
Data Synthesis
We used fixed-effects meta-analyses to pool the results of RCTs. We assessed heterogeneity by visual inspection of the forest plots and the I 2 statistic. No subgroup analysis was performed due to the small number of studies. Statistical analyses were performed on the RevMan platform v 5.3.
We summarized the results of the observational studies narratively as the very high clinical, methodological, and statistical heterogeneity prevented meta-analysis of the results. We followed the Economic and Social Research Council (ESRC) methodology and the Synthesis Without Meta-analysis (SWiM) guidelines (Supplementary Table S3).47,48 We considered the observational studies’ findings, their risk of bias, the degree of heterogeneity, their congruence with RCTs’ results, and the presence of potential confounders to reach a consensus statement among authors. For the VKA comparisons, we synthesized the results by providing the number of studies showing benefits vs the number of studies showing harm. We pooled the outcomes for VKAs against no treatment a posteriori to facilitate the interpretation of VKAs’ potential effects and harms and appraise the statistical heterogeneity. We assessed publication bias by visual inspection of the funnel plots whenever a comparison contained more than 10 trials. 49
Results
Study Selection
We identified 6775 unique citations (8526 before removal of duplicates) (Figure 1). After screening titles and abstracts, we reviewed 302 citations in full text and included 50 studies involving 155 058 participants. We included 1 supplementary RCT whose results were presented at the American Society of Nephrology Annual Meeting after the completion of the search. 50
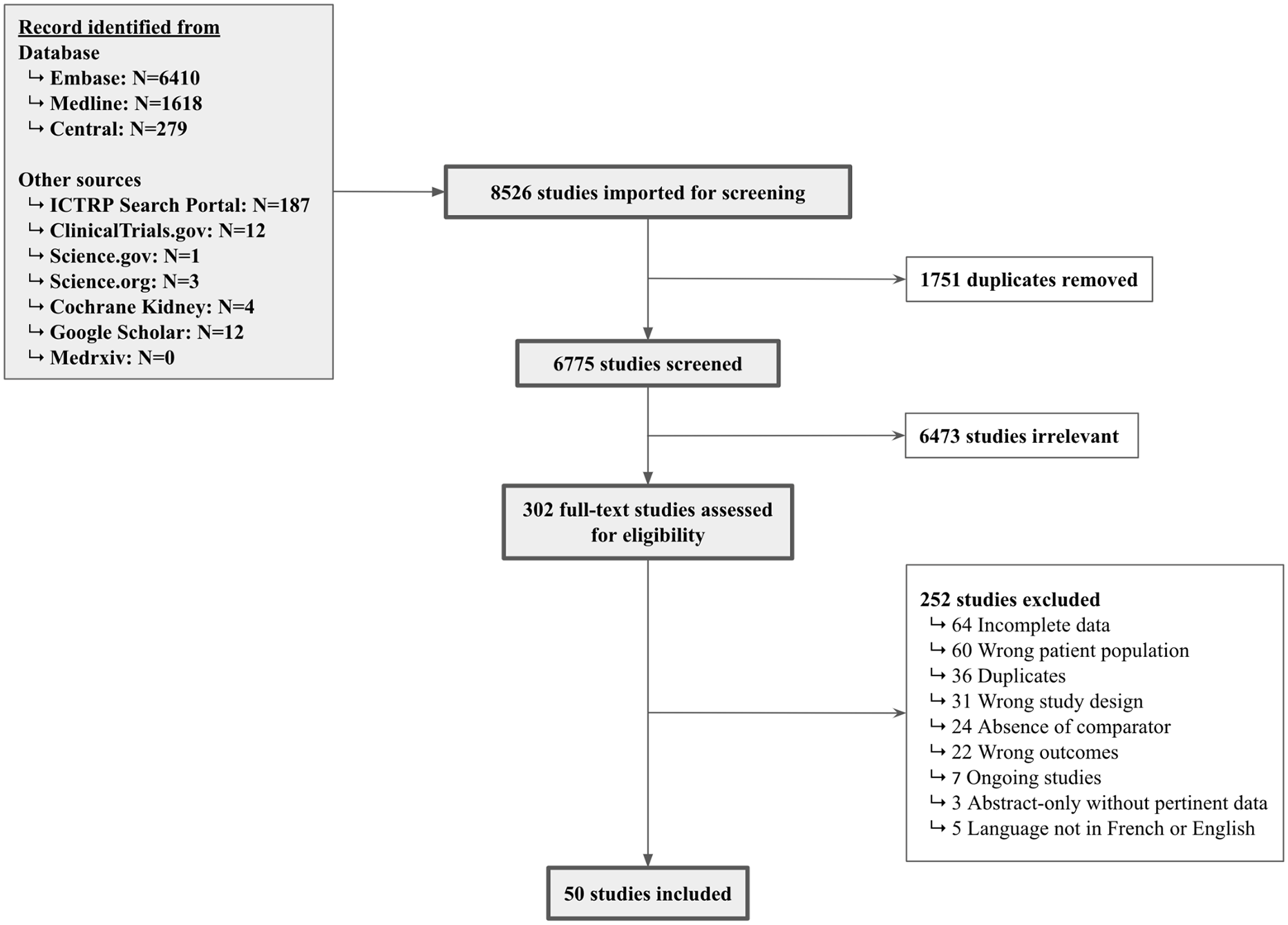
PRISMA study flow chart.
Study Characteristics
Table 1 summarizes the characteristics of the included studies. Overall, we identified 4 RCTs,50 -53 6 prospective cohort studies,54 -59 and 40 retrospective cohort studies.29,60 -98 Six studies were only published as abstracts.50,62 -66 Eight compared more than 2 groups.50,58,59,63,65,67,69,70 Two included only patients receiving peritoneal dialysis,72,80 and 6 recruited both patients receiving peritoneal dialysis or hemodialysis.54,69,85,88,94,96 The mean follow-up duration varied considerably across studies, ranging from 6 months to 5 years.
Characteristics of Included Studies.a,b

Note. ASA = aspirin; CLOP = clopidogrel; DOAC = direct oral anticoagulation; HEP = heparin; LAAC = left atrial appendage closure; LWMH = low-weight molecular heparin; NT = no treatment; OAC = oral anticoagulation; PC = prospective cohort; RCT = randomized controlled trial; RC = retrospective cohort; VKA = vitamin K antagonists.
When multiple arms were included in 1 study, we combined characteristics for the pertinent arms, whenever possible. If not possible, we presented the characteristics for the full cohort.
If available, we presented the characteristics for the adjusted population after propensity score weighting.
We extracted some data in a previous systematic review. 23
Risk of Bias and Quality Assessment
All the included RCTs were considered at high risk of bias (Supplementary Table S4) for either being open-label, providing incomplete information on allocation concealment or having limited sample sizes. The risk of bias was considered low for 25 (54%) cohort studies29,54,55,57 -59,61,67,68,70,75 -77,81,83 -87,89,92,93,95 -97 and moderate to high for the remainder (Supplementary Table S5). Only one third of the studies controlled for both concurrent antiplatelet agents and CHA2DS2-VASc or CHADS2 scores.57 -59,67,68,70,73,77,83 -85,87,92,95 -97 Furthermore, observational studies considered at low risk of bias according to the NOS scores remained prone to numerous biases, such as confounding, unexplained heterogeneity, and imprecision of results. Ultimately, we considered the evidence from the included RCTs to be of higher quality than those from the included cohort studies. A funnel plot could only be performed for observational studies of VKAs, and it did not suggest the presence of a publication bias (Supplementary Figure S2).
Findings
Table 2 summarizes the systematic review’s findings, and Table 3 provides the extracted results for individual study outcomes for VKAs and DOACs.
Summary of Findings.
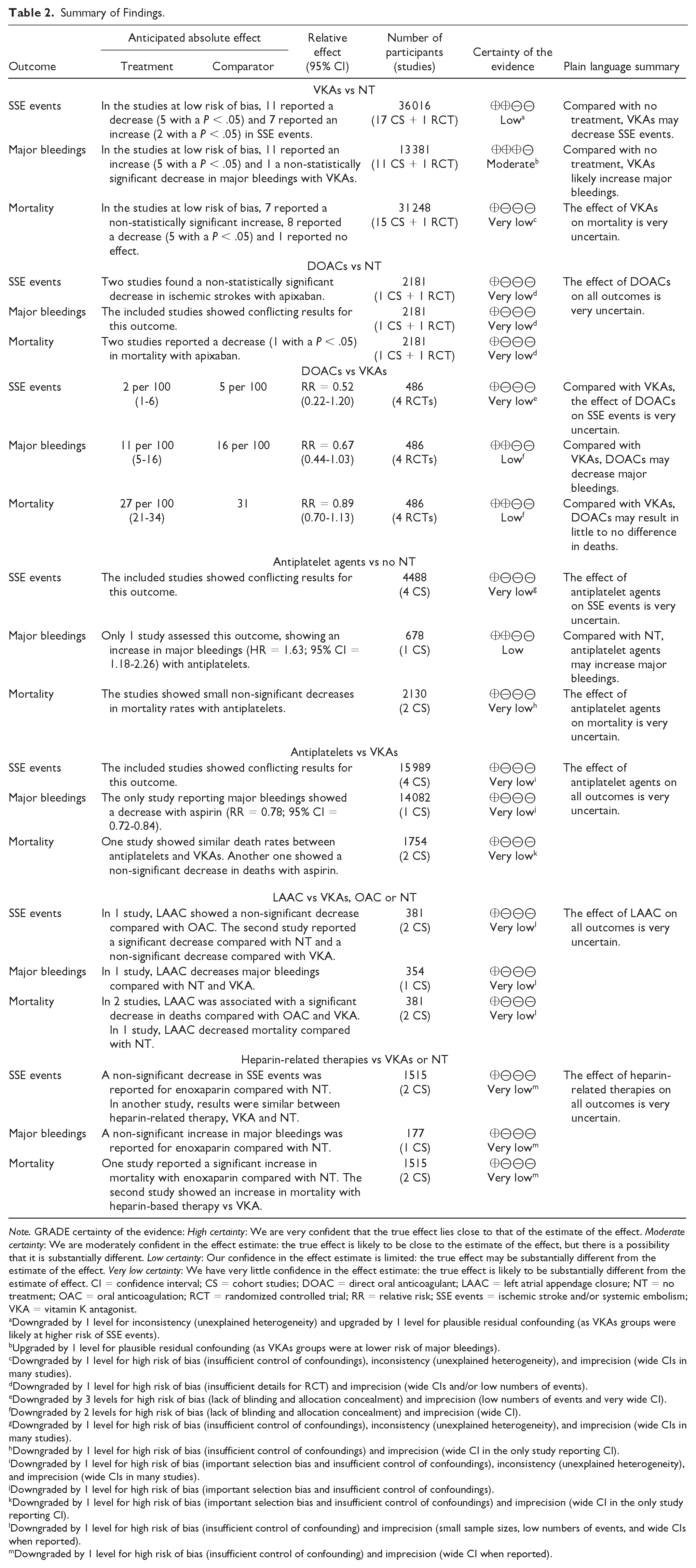
Note. GRADE certainty of the evidence: High certainty: We are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: We are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: Our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: We have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. CI = confidence interval; CS = cohort studies; DOAC = direct oral anticoagulant; LAAC = left atrial appendage closure; NT = no treatment; OAC = oral anticoagulation; RCT = randomized controlled trial; RR = relative risk; SSE events = ischemic stroke and/or systemic embolism; VKA = vitamin K antagonist.
aDowngraded by 1 level for inconsistency (unexplained heterogeneity) and upgraded by 1 level for plausible residual confounding (as VKAs groups were likely at higher risk of SSE events).
bUpgraded by 1 level for plausible residual confounding (as VKAs groups were at lower risk of major bleedings).
cDowngraded by 1 level for high risk of bias (insufficient control of confoundings), inconsistency (unexplained heterogeneity), and imprecision (wide CIs in many studies).
dDowngraded by 1 level for high risk of bias (insufficient details for RCT) and imprecision (wide CIs and/or low numbers of events).
eDowngraded by 3 levels for high risk of bias (lack of blinding and allocation concealment) and imprecision (low numbers of events and very wide CI).
fDowngraded by 2 levels for high risk of bias (lack of blinding and allocation concealment) and imprecision (wide CI).
gDowngraded by 1 level for high risk of bias (insufficient control of confoundings), inconsistency (unexplained heterogeneity), and imprecision (wide CIs in many studies).
hDowngraded by 1 level for high risk of bias (insufficient control of confoundings) and imprecision (wide CI in the only study reporting CI).
iDowngraded by 1 level for high risk of bias (important selection bias and insufficient control of confoundings), inconsistency (unexplained heterogeneity), and imprecision (wide CIs in many studies).
jDowngraded by 1 level for high risk of bias (important selection bias and insufficient control of confoundings).
kDowngraded by 1 level for high risk of bias (important selection bias and insufficient control of confoundings) and imprecision (wide CI in the only study reporting CI).
lDowngraded by 1 level for high risk of bias (insufficient control of confounding) and imprecision (small sample sizes, low numbers of events, and wide CIs when reported).
mDowngraded by 1 level for high risk of bias (insufficient control of confounding) and imprecision (wide CI when reported).
Outcomes of Included Studies.

Note. ASA = aspirin; CLOP = clopidogrel; DOAC = direct oral anticoagulant; DPL = dose per label; HEP = heparin; HR = hazard ratio; IRR = incidence rate ratio; LAAC = left atrial appendage closure; LWMH = low-weight molecular heparin; LL = low label; NT = no treatment; OAC = oral anticoagulation; PY = patients-years; RR = risk ratio; VKA = vitamin K antagonist.
Incidence of events was preferably expressed in /100 PY when available, except for the randomized controlled trials (presented in absolute numbers).
We extracted relative effects from the maximally adjusted statistical model.
We extracted some data in a previous systematic review. 23
Vitamin K antagonists
We included 1 RCT (1 comparison, 100 participants) 50 and 39 observational studies comparing VKAs with no treatment (41 comparisons, 90 492 participants, 31% receiving a VKA).60,54 -57,59,61 -65,68 -94,98 Of these 40 studies, 34 studies used warfarin,60,50,55 -57,61 -63,68 -80,82 -94,98 3 did not specify the VKA agent,54,64,65 1 used acenocoumarol, 81 and another one used phenprocoumon. 59 One study with a treatment group of both DOACs and VKAs was classified in the VKA treatment group, as 92.8% of the participants were taking warfarin, and no subgroup analysis was provided. 63 The participants’ mean age ranged from 52 to 77 years old. Only 13 studies reported the mean or median CHA2DS2-VASc score (range = 3.1-6.0),50,56,62,65,72,73,80,81,82,86 -89 and 3 reported the mean or median CHA2DS2 score (range = 1.6-2.5).56,88,91 Nineteen studies reported the mean or median use of antiplatelet agents, which ranged from 7.3% to 88.4%.55 -57,59,70,73,75,76,79 -81,83 -85,87,88,91 -93 Use of antiplatelet agents was frequent, with 50% of studies reporting a mean or median number of participants under antiplatelet agents of more than 40%. Eleven studies reported mean or median international normalized ratio (INR) (range = 1.5 to 2.3) or time in therapeutic range (TTR) (range = 33.3% to 65%) in the VKA group.50,55,56,59,61,79 -81,87,89,91
There were insufficient events in the single included RCT (SAFE-D) to conclude on the effect of VKAs on the outcomes of interest. 50 All 39 observational studies were highly heterogeneous for the 3 outcomes assessed (Supplementary Figure S1). We therefore conducted a subgroup analysis of only the studies at low risk of bias for each comparison.
Thirty-seven studies (87 476 participants) reported SSE events.60,50,55 -57,59,61 -65,68 -74,76 -90,92 -94,98 The single included RCT was inconclusive on the effect of VKAs on SSE events (0 vs 2.1%; RR = 0.31; 95% CI = 0.01-7.39). 50 In the 18 studies (36 016 participants) at low risk of bias, 11 reported a decrease50,57,70,76,77,84 -87,89,92 (5 with a P < .05)70,77,84,86,89 and 7 reported an increase55,59,61,68,81,83,93 (2 with a P < .05)61,68 in SSE events. Visual inspection of the forest plot suggested a downward trend in ischemic events. Because heterogeneity remained high, we further explored these findings within the 13 studies at low risk of bias that adjusted for the CHA2DS2-VASc or CHADS2 scores.50,55,57,59,70,76,83,84 -87,89,92 Ten reported a decrease50,57,70,76,84 -87,89,92 (4 with a P < .05)70,84,86,89 and 3 reported a non-statistically significant increase55,59,83 in SSE events. Therefore, we concluded that VKAs may decrease SSE events (18 studies, low CoE) in people with AF receiving hemodialysis.
Twenty-one studies (59 203 participants) reported major bleeding events.50,55 -57,59,63,64,68,70,73,74,76,77,79,81 -83,85,88,89,91 The single included RCT was inconclusive for the effect of VKAs on major bleeding (7.7 vs 4.2%; RR = 1.85; 95% CI = 0.35-9.63). 50 Among the 12 studies at low risk of bias (13,381 participants), 11 reported an increase50,57,59,68,70,76,77,81,83,85,89 (5 with a P < .05)59,68,77,83,85 in major bleeding with VKAs. Only 1 study reported a non-statistically significant decrease in major bleeding with VKAs. 55 Based on this subgroup analysis, we concluded that VKAs likely increase major bleeding (12 studies, moderate CoE).
Thirty-two studies (71 474 participants) reported all-cause mortality.60,50,54 -57,61,62,65,71,73 -76,79,80,84,85,87 -91,93,94 The single included RCT was inconclusive on the effect of VKAs on mortality (17.3 vs 8.3%; RR = 2.08; 95% CI = 0.68-6.31). 50 In the 16 studies (31 248 participants) at low risk of bias, 7 reported a non-statistically significant increase,50,54,59,61,76,84,93 8 reported a decrease57,75,77,81,85 -87,89 (5 with a P < .05),75,77,86,87,89 and 1 reported no effect. 55 Therefore, we concluded that the effect of VKAs on mortality is very uncertain (16 studies, very low CoE).
Direct oral anticoagulants
We included 1 RCT 50 (1 comparison, 99 participants) and 1 observational study 95 (1 comparison, 2082 participants) comparing apixaban with no treatment, 4 RCTs (4 comparisons, 486 participants) comparing DOACs with VKAs,50 -53 and 5 observational studies (9 comparisons, 55,205 participants) comparing DOACs with VKAs.29,63,67,96,97 The mean age varied between 66 and 80 years old. Patients were at high ischemic stroke risk with a mean CHA2DS2-VASc between 4.0 and 5.2. The use of antiplatelet agents was frequent in the RCTs (from 34.0 to 40.9%) and heterogeneous in the cohort studies (from 3.2% to 23.0%). The Valkyrie study randomized participants to a reduced dosing regimen of rivaroxaban (10 mg daily). 51 The RENAL-AF randomized the apixaban group to the usual dosing regimen (5 mg twice daily or 2.5 mg twice daily if at least 2 of the following criteria are met: age ≥ 80 years, body weight ≤ 60 kg, or serum creatinine ≥ 133 μmol/L), AXADIA-AFNET 8 to the reduced dose (2.5 mg twice daily), and SAFE-D permitted both regimens.50,52,53 The observational studies did not report the TTR for the VKAs group, but the RCTs reported a low TTR (between 44% and 51%).53 -55
Direct oral anticoagulants vs no treatment
The single included RCT was inconclusive on the effect of apixaban vs placebo on SSE events (2.0% vs 2.1%; RR = 0.94; 95% CI = 0.06-14.63), major bleeding (3.9 vs 4.2%; RR = 0.94; 95% CI = 0.14-6.42), and mortality (3.9 vs 8.3%; RR = 0.47; 95% CI = 0.09-2.45). 50 The observational study found no significant difference in ischemic strokes with apixaban (hazard ratio [HR] = 0.85; 95% CI = 0.36 to 1.98) compared with no treatment but showed an increase in major bleeding (HR = 2.76; 95% CI = 1.38 to 5.52) and a decrease in mortality (HR = 0.58; 95% CI = 0.43-0.78). 95 Considering the overall risk of bias and wide CIs, the effects of DOACs vs no treatment on all outcomes remain very uncertain (very low CoE).
Direct oral anticoagulants vs vitamin K antagonists
Four RCTs contributed to the meta-analyses (Figure 2).50 -53 The evidence comparing VKAs with DOACs regarding SSE events is very uncertain (RR = 0.52; 95% CI = 0.22-1.20; very low CoE). The DOACs may decrease major bleeding (RR = 0.67; 95% CI = 0.44-1.03; low CoE) but may result in little to no difference in mortality (RR = 0.89; 95% CI = 0.70-1.13; low CoE). In these RCTs, only apixaban and rivaroxaban were used in people receiving dialysis.

Effect of DOACs against VKAs for (A) SSE events, (B) major bleedings, and (C) mortality.
Five observational studies reported SSE events and found no statistically significant differences between DOACs and VKAs.29,63,67,96,97 Five observational studies also reported major bleeding.29,63,67,96,97 One study reported an increase in major bleeding with dabigatran (HR = 1.48; 95% CI = 1.21-1.81) and rivaroxaban (HR = 1.38; 95% CI = 1.03-1.83). 63 However, we noted disproportionately high rates of major bleeding in this study (68.4-83.1 events/100 patients-years compared with 1.6-22.9 in the remaining studies). Two studies reported a significant decrease in major bleeding (HRs of 0.67 and 0.72).96,97 One study found similar major bleeding risks with multiple DOACs and VKAs. 29 Another study showed a signal toward a reduction in major bleeding with apixaban (HR = 0.29; 95% CI = 0.07-1.18) and rivaroxaban (HR = 0.69; 95% CI = 0.17-2.87) and an increase with dabigatran (HR = 1.82; 95% CI = 0.67-4.98). 63 Finally, the 2 observational studies reporting mortality data showed a decrease in deaths only with the usual dosing regimen of apixaban in one study (HR = 0.85; 95% CI = 0.78-0.92) and a non-statistically decrease in another study (HR = 0.85; 95% CI = 0.71-1.01), which may be the result of a selection bias.96,97 Therefore, the observational evidence did not alter our conclusions from the trial-based evidence.
Antiplatelet agents
We included 4 observational studies (4 comparisons, 4488 participants) comparing antiplatelet agents with no treatment65,69 -71 and 4 observational studies (4 comparisons, 15 989 participants) comparing antiplatelet agents with VKAs.65,67,71,72 The mean age was 72 years old. Two studies reported mean CHA2DS2-VASc scores of 3.9 and 4.0,65,72 and 1 study reported a mean CHA2DS2 score of 2.4. 67 No studies that included participants on VKAs reported the TTR.
Antiplatelet agents vs no treatment
Compared with no treatment, the effect of antiplatelet agents on SSE events (4 studies)65,69 -71 and mortality (2 studies)65,71 was very uncertain (very low CoE), but they may increase the risk of bleeding (1 study, low CoE). 70 Two studies reported a non-significant decrease in SSE events (HRs of 0.88 and 0.83) with antiplatelet agents.69,70 One study reported fewer SSE events with aspirin or clopidogrel without providing a measure of association. 65 One showed a statistically significant increase in ischemic strokes (HR = 2.01; 95% CI = 1.04-3.88). 71 Only 1 study reported major bleeding and showed an increased risk with aspirin (HR = 1.63; 95% CI = 1.18-2.26). 70 This study was considered sufficiently at low risk of bias to conclude that antiplatelet agents may increase major bleeding. Finally, 2 studies reporting mortality data reported no statistically significant differences in mortality rates.65,71
Antiplatelet agents vs vitamin K antagonists
Compared with VKAs, the effect of antiplatelet agents on SSE events (4 studies),65,67,71,72 major bleeding (1 study), 67 and mortality (2 studies)65,71 was very uncertain (very low CoE). One study showed a decrease in SSE events (RR = 0.81; 95% CI = 0.66-0.99) and major bleeding (RR = 0.78; 95% CI = 0.72-0.84) with aspirin, but its high incidence of bleeding and lack of adjustment for important confounders call for caution in interpreting its results. 67 Another study showed an exaggerated decrease in ischemic strokes with VKAs compared with aspirin (HR = 0.16; 95% CI = 0.04-0.66), but the patients taking aspirin were older, sicker, and had higher thromboembolic risks. 72 Another study showed no statistical differences in ischemic strokes (HR = 1.58; 95% CI = 0.50-4.98) or mortality (HR = 0.90; 95% CI = 0.53-1.51) with aspirin. 71 The last study (abstract-only, NOS at very high risk of bias) showed similar SSE events and mortality rates when taking antiplatelet agents (aspirin or clopidogrel) or VKAs. 65
Left atrial appendage closure
We included 2 observational studies (3 comparisons, 381 patients) comparing LAAC with no treatment or oral anticoagulation and found that the effect of LAAC on all outcomes was very uncertain (very low CoE).58,66 The nonrandomized study design, small sample sizes and low number of events limited the results. The first study showed a non-statistically significant decrease in ischemic strokes (0% vs 7.7%) and mortality (22% vs 41%) compared with a historical cohort on oral anticoagulation (choice of anticoagulants unspecified). 66 The second one reported an increase in SSE events (10.8% vs 2.2%, P = .021), mortality (HR = 3.09; 95% CI = 1.59-5.98) and major bleeding (HR = 4.87; 95% CI = 1.08-21.97) of no treatment compared with LAAC. 58 Similar increased risks were seen when comparing warfarin with LAAC on mortality (HR = 2.76; 95% CI = 1.31-5.89), major bleeding (HR = 6.48; 95% CI = 1.32-31.72) and SSE events (7.0% vs 2.2%, P = .092). 58
Heparin-related therapies
We included 2 observational studies—one comparing enoxaparin (4000 units daily on non-hemodialysis days) with no treatment (1 comparison, 177 participants) 64 and one comparing heparin (unspecified agent) with no treatment and VKAs (2 comparisons, 1338 participants). 51 We found that the effect of heparin-related therapies on all outcomes was very uncertain (very low CoE). The first study reported a statistically significant increase in mortality (HR = 1.88; 95% CI = 1.20-2.92) and non-statistically significant differences for major bleeding (HR = 1.86; 95% CI = 0.81-4.28) and SSE events (HR = 0.49; 95% CI = 0.11-2.18). 64 The second study, published in abstract-only, reported an increase in mortality with heparin (1.7 years of survival) compared with VKAs (2.8 years) and similar survival times compared with no treatment (1.8 years) but a decrease in SSE events with heparin (1.2%) compared with VKAs (2.9%) and no treatment (2.6%). 51
Discussion
In this systematic review of thromboembolic prevention strategies for adults with AF receiving dialysis, we found that VKAs may be associated with a decrease in SSE events and an increase in major bleeding compared with no treatment. Previous systematic reviews in patients receiving dialysis found no clear association between VKAs and decreased SSE events.13,22 -24 The discordance with our findings might be explained by several factors: (1) previously published systematic reviews ended their literature search between 2015 and 2019; (2) they included between 9 and 14 studies reporting SSE events with VKAs, whereas our systematic review included 37; and (3) they gave equal weights to all included studies while we gave proportionally more importance to the results of studies at lower risk of bias, including the first RCT of VKAs against placebo. Furthermore, many of the VKAs studies had insufficient power to detect a significant change in SSE events, included a high proportion of participants with low or unreported CHA2DS2 or CHA2DS2-VASc scores, and reported frequent subtherapeutic INR or TTR among VKAs users. Others have suggested that vascular calcifications, promoted by VKAs, are the main driver of SSE events in this population, which may explain the lack of association between VKAs use and stroke incidence in people with AF receiving dialysis.14,99,100 We observed a decreased risk in SSE events within the studies at low risk of bias controlling for thromboembolic risk, which may suggest the findings of previous systematic reviews were influenced by studies at higher risk of bias or more prone to confounding. Although we concluded that VKAs may reduce SSE events, an individualized analysis and discussion of the benefits and risks of oral anticoagulation remain critical considering the substantial risk of major bleeding in this population. The major hurdle facing clinicians is to appropriately select patients at high thromboembolic risk, as validation studies in the dialysis setting showed inconsistent predictive value for the CHADS2 or CHA2DS2-VASc (c-statistics ranging from 0.61 to 0.88 with wide CIs).88,101,102 Given the quality of the evidence and the challenges in balancing risk of ischemic stroke and major bleeding, we can only recommend that clinicians engage in a shared decision-making process using a patient decision aid (such as the one being developed by our team), rely on their clinical judgment to individualize stroke and bleeding risks (without providing absolute thresholds for the CHADS2, CHA2DS2-VASc, or HAS-BLED), and openly discuss with patients the studies’ limitations in the dialysis setting.
Our systematic review suggests that DOACs may be as effective as VKAs to reduce SSE events and may be associated with less major bleeding, but the quality of the evidence was low to very low. To our knowledge, we conducted the first meta-analysis of DOACs against VKAs to include the newest findings from the AXADIA-AFNET 8 and SAFE-D trials.50,52 Previously published systematic reviews of mostly observational studies concluded that apixaban may decrease25 -28 or have no effect29,30 on major bleeding compared with VKAs. Our findings support the inclusion of DOACs as an SSE event prevention strategy in the shared decision-making process with patients. The choice of DOAC in dialysis should be limited to apixaban and rivaroxaban, considering the absence of data for edoxaban and the worrying rates of major bleeding with dabigatran. 67 A reduced dosing regimen of rivaroxaban (10 mg daily) was used in the Valkyrie study and produced similar pharmacokinetics endpoints in dialysis compared with the standard dose in healthy patients.51,103 When selecting apixaban, the question of the dosing regimen remains controversial, as no dosing regimen was clearly superior in RENAL-AF, AXADIA-AFNET 8, and SAFE-D.50,52,53 Pharmacokinetic studies of repeated apixaban doses in hemodialysis inconsistently showed supratherapeutic levels with the usual dosage.53,104
This systematic review found the level of evidence for antiplatelet agents, heparin-related therapies, and LAAC to be very low. These therapies should, therefore, not be routinely used for stroke prevention in people with AF receiving dialysis. Our findings align with previous guidelines and findings from systematic reviews in the general population to prioritize oral anticoagulation over antiplatelet therapy.6,9 -12 The LAAC appears to be a potentially interesting alternative to oral anticoagulation in closely selected patients with prohibitively high thromboembolic and bleeding risks. Still, the risks of LAAC in the dialysis population (eg, pericardial effusion, device embolization, procedure-related thrombus) remain to be defined. 105 A prospective observational study (WATCH-HD) comparing LAAC with no treatment is underway. 106
To our knowledge, this is the first systematic review assessing all stroke-prevention interventions for patients with AF receiving dialysis. Its strengths include an extensive search strategy, independent double data extraction, and risk of bias assessment. We included subgroup analyses of data at low risk of bias for the VKAs group and only conducted meta-analyses when appropriate. However, this systematic review also has some significant limitations. The included studies were mostly retrospective cohort studies at moderate to high risk of bias, including classification mistakes (eg, in using procedure codes), confounding by indication (eg, patients receiving anticoagulation at higher thromboembolic risk), and residual confounding (eg, TTR for warfarin and over-the-counter use of aspirin). Indeed, only a third of the observational studies controlled for both antiplatelet agents and CHA2DS2-VASc or CHADS2 score, thus limiting our confidence in the effect size reported in many studies. We also encountered high statistical heterogeneity for the effect of VKAs on SSE events, which prevented robust conclusions on their benefit over no treatment in people receiving dialysis. Two upcoming RCTs comparing anticoagulation with placebo (DANWARD and AVKDIAL) may help answer this question.107,108 Finally, as per protocol, we did not compare the different DOACs’ dosing regimens, preventing us from providing definite conclusions on the optimal dosing.
Conclusion
Our systematic review showed that VKAs may reduce the risk of SSE events in people with AF receiving dialysis at the cost of an increase in major bleeding. The DOACs (apixaban or rivaroxaban) may be as effective as VKAs in reducing SSE events with possibly fewer bleeding events. However, all but 1 comparison had a low to very low CoE, reminding clinicians to openly discuss values and preferences with patients to guide clinical decisions. Until well-designed RCTs comparing DOACs, warfarin, and placebo help inform clinicians and patients, engaging in a shared decision-making process is the 1 intervention we can confidently recommend for managing AF-related ischemic events in people receiving hemodialysis.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241290316 – Supplemental material for Stroke Prevention in Patients With Atrial Fibrillation Receiving Dialysis: A Systematic Review
Supplemental material, sj-docx-1-cjk-10.1177_20543581241290316 for Stroke Prevention in Patients With Atrial Fibrillation Receiving Dialysis: A Systematic Review by Olivier Massé, Gabriel Dallaire, Noémie Maurice, Yu Hong, Claudia Mei Mercurio, Catherine Tremblay, Nicolas Dugré and Amélie Bernier-Jean in Canadian Journal of Kidney Health and Disease
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
