Abstract
Background:
Diabetic kidney disease (DKD) is the most common and deranging microvascular complication of diabetes mellitus (DM). Podocytopathy is a key component of glomerular damage in DKD. Micro RNA-21 (miRNA-21) is an epigenetic regulator that plays a role in podocyte damage; however, the results of previous studies have not resolved the controversy about the role of miRNA-21 in the pathogenesis of DKD.
Objective:
The objective was to investigate the correlation between miRNA-21 levels and urinary nephrin, podocin, and urinary albumin-creatinine ratio (UACR) in patients with type 2 DM and albuminuria.
Design:
This is a cross-sectional study.
Setting:
This study was carried out in internal medicine outpatient clinic of Cipto Mangunkusumo Hospital Jakarta, Indonesia.
Patients:
This study consisted of 42 adults with type 2 DM and albuminuria.
Measurements:
The measurements include (1) Serum miRNA-21; (2) urinary podocin, nephrin, and albumin-creatinine ratio; and (3) serum miRNA-21 correlated to urinary podocin, nephrin, and albumin-creatinine ratio.
Methods:
The Spearman bivariate analysis to assess the correlation of miRNA-21 with nephrin, podocin, and UACR.
Results:
The mean relative expression of miRNA-21 was 0.069 (0.024), the median for nephrin, podocin, and UACR was 35.5 (15.75-51.25) ng/mL, 0.516 (0.442-0.545) ng/mL, and 150 (94.56-335.75) ng/mL, respectively. A correlation between miRNA-21 and nephrin was observed (r = 0.598; P < .0001). There was a correlation between miRNA-21 and UACR (r = 0.604; P < .0001). No correlation was found between miRNA-21 and podocin.
Limitations:
A lack of non-DM and non-albuminuric control population and small sample size. We could not exclude concurrent disease, and all other potential confounding variables, particularly those related to inflammation.
Conclusions:
The miRNA-21 can be considered an early biomarker for podocytopathy and albuminuria in DM, highlighting its potential for early diagnostic and therapeutic interventions. Further research is required to confirm these findings and explore their clinical applications, which could significantly alter management strategies for DKD.
Introduction
Diabetic kidney disease (DKD) is the most common microvascular complication in patients with diabetes mellitus (DM). This complication affects over 30% to 40% of patients with DM. 1 According to data collected by the Indonesian Renal Registry in 2018, DKD is the second most common cause of end-stage kidney disease (ESKD) patients undergoing dialysis, with the number of patients reaching 28% of all cases. 2 Although the current management of DKD which includes lifestyle modifications, intensive control of glycemic status, blood pressure, lipid profile, and albuminuria has yielded promising results in relation to DM complications, DKD remains a major risk factor for the development of ESKD. 1 The 5-year survival rate in late-stage DKD patients is only around 20%. 3 The pathogenesis of DKD is known to involve various metabolic, hemodynamic, and inflammatory pathways caused by persistent hyperglycemic conditions. Evidence has demonstrated that podocytopathy plays an important role in the early pathogenesis of DKD and is a major factor in glomerular damage, ultimately resulting in proteinuria and further renal damage. However, the molecules that modulate podocytopathy in DKD have not yet been fully clarified. 4 Therefore, identifying key molecules involved in podocyte damage will provide new insights regarding the pathogenesis of DKD.
Albuminuria is currently the most important clinical biomarker for the early detection of DKD. However, its usefulness is limited in that not all diabetic patients with microalbuminuria will advance to end-stage DKD. As many as 30% of patients with DKD have normal urinary albumin levels. 5 Nephrin and podocin are transmembrane proteins found in podocytes, which play an essential role in the glomerular filtration barrier. If podocytes are damaged, these proteins are released from the glomerular basement membrane. Consequently, nephrinuria and podocinuria can be detected. 6 Elevated urinary levels of nephrin and podocin may be a more specific and sensitive early marker than albuminuria for the early detection of DKD.7,8
Recent research has investigated novel concepts regarding the pathophysiology of DM and DKD, including the contribution of epigenetic modifications. 9 One of the epigenetic modifications that has attracted attention is related to the role of microRNA (miRNA). Persistent hyperglycemia may result in the increased expression of miRNA. The miRNA that has been most commonly studied in relation to DKD is miRNA-21. 10 In the examination of genomic miRNA expression analysis, miRNA-21 exhibits the highest upregulation in the kidney. 11 Previous studies have revealed that both the increased expression and suppressed expression of miRNAs exert significant effects on podocyte damage. 12 The precise molecular mechanism responsible for miRNA-induced podocyte injury in DKD has yet to be fully elucidated. Previous research results remain controversial regarding the involvement of miRNA-21 in podocyte damage. To date, no research has been conducted regarding the relationship between miRNA-21 levels and urinary podocyte damage markers, including nephrin and podocin, which are earlier markers than albuminuria in individuals with DKD. Therefore, this cross-sectional study aims to establish the role of miRNA-21 and podocyte damage in the early pathogenesis of clinical DKD, marked by albuminuria in type 2 diabetic patients. We hypothesized that there is miRNA-21 that plays a part in podocyte damage, as shown by change in urinary nephrin, podocin, and albumin-creatinine ratio.
Methods
Study Design and Setting
This cross-sectional study was conducted at the nephrology and endocrinology outpatient clinic of Internal Medicine Department, Cipto Mangunkusumo Hospital, Jakarta, Indonesia (The National Referral Hospital Teaching Hospital). The recruitment period was from July 2022 through August 2023.
Inclusion and Exclusion Criteria
The inclusion criteria for this study were adult aged 18 to 70 years who had been clinically diagnosed with type 2 DM with albuminuria (urine albumin-creatinine ratio of ≥30 mg/g), who had given their consent to participate in the research. The exclusion criteria for this study pertained to patients suffering from other diseases that cause proteinuria, such as urinary tract infections, leukocyturia, hematuria, or urolithiasis. We also eliminated patients who were suffering from acute illnesses, such as acute complications of diabetes, acute coronary heart disease, acute cerebrovascular disease, acute infection, systemic infection, or malignancies; those who engaged in tobacco use or who had a previous smoking history; and those who were pregnant. Study participants were consecutively recruited.
Patients screening, recruitment, and data collection procedure
We screened 45 participants with clinical DKD, manifested as type 2 DM with albuminuria. Two samples were excluded due to hematuria and leucocyturia. One blood sample lysed during the laboratory check; hence, a total of 42 participants were analyzed. Primary and secondary data were obtained from all samples. Primary data from history taking and laboratory check of blood and urine. Clinical characteristics (eg, sex, age, duration of diabetes, previous medical history and medication used, duration on medication) were obtained from patient interview and the electronic health records.
Screening included history taking, physical examination, urine examination to determine albuminuria and blood examination to see estimated glomerular filtration rate (eGFR), calcium and phosphate to assess blood calcium and phosphate levels, and HbA1c to determine whether DM is controlled. Participants and their families received an explanation about the purpose, method of research, and the right to stop or refuse to take part in the research. All participants agreed and signed an informed consent to enroll in the study.
Urine sample collection and processing
Upon agreeing to participate in the study, 20 mL of urine sample was taken from the midstream urine in a sterile container for examination of urinary albumin-creatinine ratio (UACR), podocin, and nephrin.
Podocin was examined using human podocin/PDCN (NPHS2) enzyme-linked immunosorbent assay (ELISA) Kit, based on a sandwich enzyme-linked immunosorbent assay technology. An antibody is pre-coated onto a plate. Standards, test samples, and biotin-conjugated reagent are added to the wells and incubated. The horseradish peroxidase (HRP)-conjugated reagent is then added, and the whole plate is incubated. Unbound conjugates are removed using wash buffer at each stage. TMB (3,3’,5,5’-Tetramethylbenzidine) substrate is used to quantify the HRP enzymatic reaction. After TMB substrate is added, only wells that contain sufficient NPHS2 will produce a blue colored product, which then changes to yellow after adding the acidic stop solution. The intensity of the yellow color is proportional to the NPHS2. The concentration of NPHS2 was calculated based on the optical density.
Nephrin was measured using Human Nephrin ELISA (Exocell) kit. The assay is conducted in an indirect competitive mode. The mouse anti-human nephrin is added to the plate. Antibody interacts and binds with the stationary-phase or fluid-phase nephrin (competitive binding). A subsequent reaction with anti-mouse IgG-HRP conjugate labels the probe with enzyme. After washing, only the antibody-conjugate reacting with the anti-nephrin antibody bound to nephrin antigen of the stationary phase remains in the well, and detected using a chromogenic reaction. Color intensity in nephrin ELISA is inversely proportional to the logarithm of nephrin concentration in the fluid phase.
The urine creatinine-albumin ratio was determined through the sandwich immunometric assay technique.
Blood sample collection and processing
Ten mL of venous blood sample were collected from each participant at the same time of blood sample collection. After centrifugation at 1900 × g for 10 minutes at 4°C using swinging bucket rotor, plasma serum was separated to a new container, where it was stored in cryovials at 70°C until assays were performed.
The total RNA was extracted from serum using reagent mIRNeasy Serum/Plasma Advanced Kit (Cat. No. 217204, Qiagen, Hilden, Germany) with spin column method. The extracted RNA was reverse-transcripted to complementary DNA (cDNA) using TaqMan MicroRNA Reverse Transcription Kit (Cat. No. 4366596, Applied Biosystem, Thermo Scientific, Waltham, Massachusetts) with standard protocol as follows: reverse transcription in 16°C and 42°C for 30 minutes each, then the reaction is stopped at 85°C for 5 minutes, then held at 4°C. The miRNA-21 value was assessed using Real-Time Polymerase Chain Reaction (RT-PCR) technique using TaqMan Universal Master Mix. The reaction was done twice for each sample, using standard protocol: enzyme activation at 95°C for 10 minutes, denaturation 95°C for 15 seconds, and anneal 60°C for 1 minute. The expression of miRNAs was given as Ct value, where the cycle threshold (Ct) is defined as the number of cycles necessary for the fluorescent signal to cross the threshold of RT-PCR. The miRNA-16 was used as reference. The expression (F) of miRNA-21 was counted using relative quantitative method by Schmitgen and Livak: F = 2-ΔXτ; ΔCt: Ct miRNA-21 (−) Ct miRNA-16.
Biochemical analysis
Plasma fasting blood sugar, serum urea, and creatinine were estimated using automated chemistry analyzer. The estimated glomerular filtration rate was calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation.
Ethics
The Ethical Committee of the Faculty of Medicine, Universitas Indonesia, approved this study (ref: 176/UN2.F1/ETIK/PPM.00.02/2022).
Statistical Analysis
A data analysis was performed using the SPSS 20 software. For normally distributed numerical data, the mean and standard deviation were reported, whereas the median and interquartile range were reported for non-normally distributed data. Categorical data were presented as percentages. A Pearson correlation test was used to analyze the association between miRNA-21 and the nephrin, podocin, and UACR if the data were normally distributed. A Spearman analysis was used if the data were not normally distributed.
Results
Forty-two participants, with a mean age of 57.7 (standard deviation [SD] = 6.67) years were included in this analysis. Of them, 47.6% were male, and 52.4% were female. The duration of their DM diagnoses varied from 1 to 23 years (median 6 years, with an IQR of 3-15.5 years). Hypertension was detected in 73.8% of the participants, and 66.7% of them were treated with angiotensin-converting enzyme inhibitor/angiotensin receptor blocker (ACE-i/ARB) therapy. Furthermore, 54.8% of participants had dyslipidemia, with 45.2% receiving statins. Obesity was observed in 26.2% of the study participants, with a mean body mass index (BMI) of 26.85 (SD = 4.7) kg/m2. Of the study participants, 45.2% had HbA1c levels exceeding 7%, with a mean HbA1c value of 6.78% (SD = 0.75). Our participants had a median eGFR of 70 (56.85-90.77) mL/min/1.73 m2. Most of the study participants (66.7%) had an eGFR equal to or greater than 60 mL/min/1.73 m2. The median neutrophil-lymphocyte ratio (NLR) for this study was 2.77 (IQR = 2.03-3.48). The study found a mean relative expression of serum miRNA-21 of 0.069 (SD = 0.024), with a median urinary nephrin level of 35.5 ng/mL (IQR = 15.75-51.25) and a median urinary podocin level of 0.516 ng/mL (IQR = 0.447-0.620). The median UACR was 16.95 mg/mmol (IQR = 10.69-37.94). The demographic and clinical characteristics of the participants are presented in Table 1.
Baseline Characteristics.

Note. ACE- i/ARB = angiotensin converting enzyme inhibitor/angiotensin receptor blocker; OHA = oral hypoglycemic agent; HbA1c = hemoglobin A1c; LDL = low-density lipoprotein; HDL = high-density lipoprotein; GFR = glomerular filtration rate; NLR = neutrophil-lymphocyte ratio; UACR = urinary albumin-creatinine ratio; SD = standard deviation; IQR = interquartile range; BMI = body mass index; SBP = systolic blood pressure; DBP = diastolic blood pressure.
Correlations Between miRNA-21, Nephrin, Podocin, and Urinary Albumin-Creatinine Ratio
The results of the analysis demonstrated that there was a moderate positive correlation between miRNA-21 and nephrin (r = 0.59, P < .0001). We found no significant correlation between miRNA-21 and podocin (r = 0.22, P = .17). There was a moderate positive correlation between miRNA-21 and UACR (r = 0.60, P < .0001). Table 2 and Figures 1 to 3 present the results of the bivariate analysis between the study variables.
Correlations Between miRNA-21, Nephrin, Podocin, and UACR.

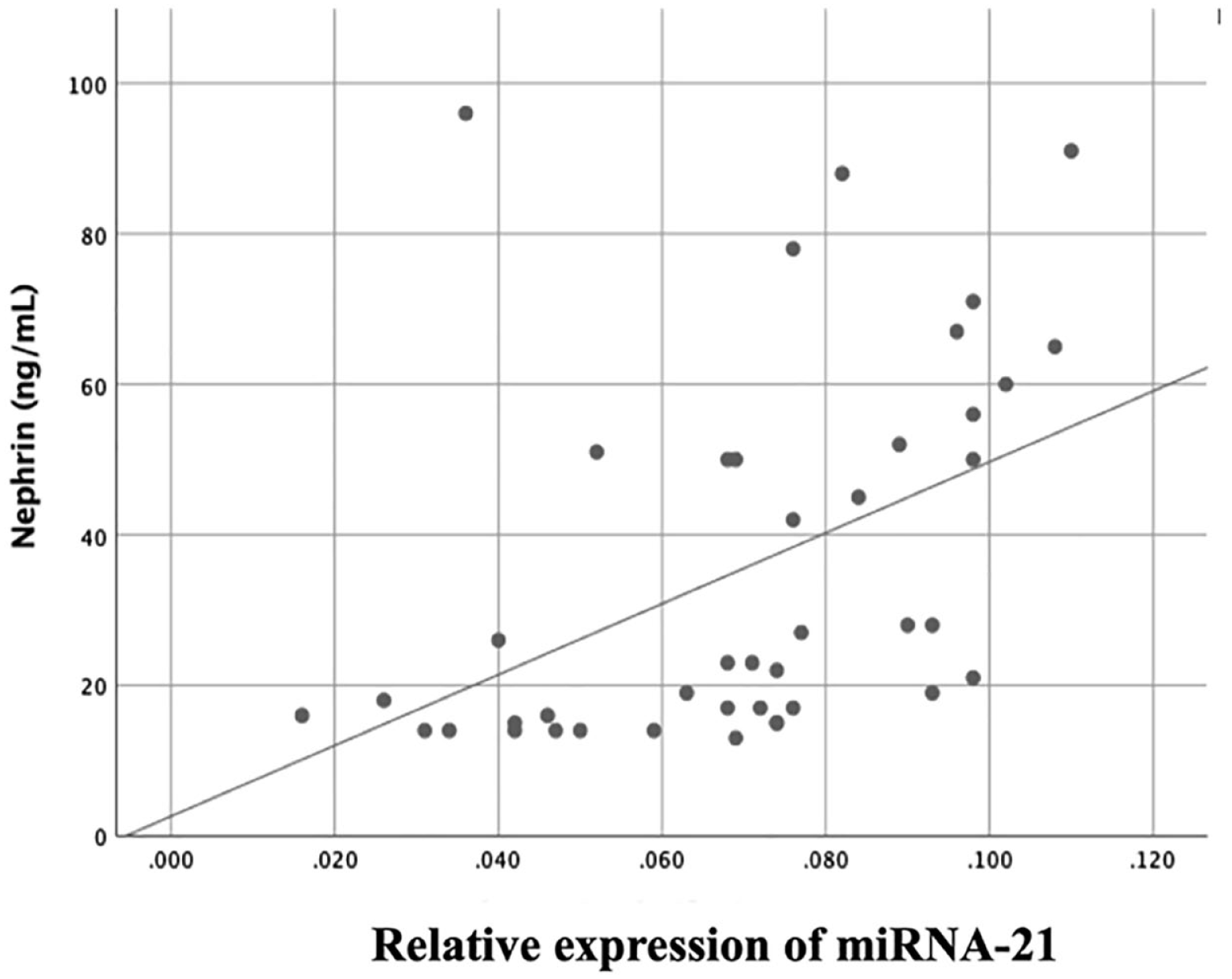
Scatter diagram illustrating the correlation between miRNA-21 and nephrin.

Scatter diagram indicating the correlation between miRNA-21 and podocin.

Scatter diagram illustrating the correlation between miRNA-21 and UACR.
Correlations of Nephrin, Podocin, and Urinary Albumin-Creatinine Ratio With Confounding Variables
A bivariate analysis was performed to determine the correlations between the levels of nephrin, podocin, and UACR and the confounding factors. The results obtained are presented in Tables 2 and 3. Nephrin levels was associated with hypertension, with its value found to be higher in those with hypertension (P = .04). There was also a statistically significant association between nephrin levels and NLR, with higher nephrin values in those with NLR > 3 (P = .04). Higher podocin level was observed in participants aged 60 years and above, revealing a statistically significant association between podocin levels and age (P = .02). A significant association was observed between podocin levels and eGFR, wherein higher podocin values were found in the eGFR < 60 mL/min/1.73 m2 group (P = 0.01). There was a statistically significant association between UACR levels and eGFR, in which UACR values were higher in the eGFR < 60 mL/min/1.73 m2 group compared with the eGFR ≥60 mL/min/1.73 m2 group (P = .01). No significant correlation was found for other confounding variables (Table 4).
Correlations of Nephrin and Podocin, With Confounding Variables.

Note. SD = standard deviation; IQR = interquartile range; DM = diabetes mellitus; NLR = neutrophil-lymphocyte ratio.
Correlations of UACR With Confounding Variables.

Note. UACR = urinary albumin: creatinine ratio; DM = diabetes mellitus; NLR = neutrophil-lymphocyte ratio; IQR interquartile range.
Discussion
Our study revealed a moderate and statistically significant positive correlation between serum miRNA-21 levels and urinary nephrin levels in patients with type 2 DM and albuminuria. This suggests that the process of podocytopathy that occurs in DKD may involve the epigenetic mechanism of miRNA-21. These results are consistent with previous studies that examined the role of miRNA-21 in podocyte damage in DKD. Wang et al conducted a study to investigate the effect of miRNA-21 on the podocyte dedifferentiation process in the podocyte cells of diabetic nephropathy mice model. The results demonstrated that miRNA-21 expression in podocyte activates transforming growth factor beta 1 (TGF-β1), which increases the expression of Smad3 but suppresses the expression of Smad7. 13
The presence of nephrin in urine indicates podocyte injury in early DKD development. Changes in the regulation of nephrin may result in impairments in the size selectivity of the diaphragm gap that lies between the podocyte foot processes. Normoalbuminuric patients with type 1 and type 2 diabetes exhibited increased levels of nephrinuria, a phenomenon suggesting that nephrinuria may precede microalbuminuria in the progression of DKD. 14 According to the literature, miRNA-21 plays a role in the development of DKD in the glomerulus. It has a strong correlation with hyperglycemia, resulting in an increase in renal cell hypertrophy and matrix expansion. 15 The miRNA-21 most likely undergoes similar activity in podocytes, as indicated by the significant correlation with nephrinuria levels found in this study.
We found no significant correlation between serum miRNA-21 and urinary podocin levels in patients with type 2 DM and albuminuria. However, Sato et al 16 reported that compared with the initial phase of podocyte damage, there is increased podocin excretion as the podocyte damage progresses. Zeng et al found an association between podocin levels and reduced eGFR in people with DKD. Participants in the study had a mean eGFR of 41.4 (SD = 31.3) mL/min/1.73 m2. Median proteinuria was 2.5 (1.7-4.5) g per day. 17 In our study, the majority of participants exhibited normal levels of eGFR and were classified as having microalbuminuria, indicating a lack of advancement in DKD. This varying characteristic could be the reason for the absence of a notable correlation between miRNA-21 and podocin.
Based on the data analysis performed in this study, a statistically significant and moderately positive correlation was observed between serum miRNA-21 levels and the urinary creatinine-to-albumin ratio in patients with DKD. Research by Fouad et al found that miRNA-21 levels increased significantly in type 1 DM patients as early as 5 years after the onset of diabetes, before the onset of microalbuminuria, and that they continued to increase with the progression of DM, whereas eGFR levels decreased. In addition, the study found that microRNA-21 levels ≥0.01 could predict the occurrence of microalbuminuria. 18
Wang et al conducted a study using a mice model of DKD. Their findings revealed a positive association between miRNA-21 expression as well as both the UACR and the glomerular fibrosis index. However, they also found a negative correlation between miRNA-21 expression and creatinine clearance. 19 Chien et al conducted a study in Taiwan and reported higher miRNA-21 levels in DKD groups with macroalbuminuria compared to those with microalbuminuria or normoalbuminuria. The expression of miRNA-21 significantly increased in individuals with an eGFR < 60 mL/min/1.73 m2 compared to those with higher eGFR. 20 This finding is consistent with our current study, in which statistically significantly higher levels of miRNA-21 were found in the macroalbuminuria group compared to those with microalbuminuria.
The strength of this study is that, to our knowledge, it is the first study to examine the correlations between miRNA-21 levels and urinary markers of podocytopathy using nephrin and podocin. However, this study has several weaknesses that must be taken into account. There is lack of a control group to serve as control compared to the diabetic and albuminuric participants, and the sample size of study was relatively small. We could not exclude concurrent disease, and all other potential confounding variables, particularly those related to inflammation, due to limited time and resources. This research design is a cross-sectional study; thus, it does not explain the cause and effect relationship.
Conclusions
The miRNA-21 emerges as a crucial biomarker with the potential to transform the early detection and treatment of podocytopathy and albuminuria in diabetic patients. It is a promising avenue for timely intervention, enabling health care professionals to identify renal complications in their early stages and implement proactive therapeutic measures. Recognizing miRNA-21 as an early indicator not only underscores its diagnostic importance but also highlights its potential for tailored interventions aimed at slowing the progression of DKD. Comprehensive validation studies and further exploration of its applications across various clinical contexts are needed to reshape current approaches to managing DKD.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241260948 – Supplemental material for The Correlation of MicroRNA-21 With the Nephrin, Podocin, and Urinary Albumin-Creatinine Ratio in Patients With Type 2 Diabetes and Albuminuria: A Cross-Sectional Study
Supplemental material, sj-docx-1-cjk-10.1177_20543581241260948 for The Correlation of MicroRNA-21 With the Nephrin, Podocin, and Urinary Albumin-Creatinine Ratio in Patients With Type 2 Diabetes and Albuminuria: A Cross-Sectional Study by Pringgodigdo Nugroho, Tri Hadi Susanto, Maruhum Bonar, Aulia Rizka, Aida Lydia, Soekamto Koesno, Pradana Soewondo, Hamzah Shatri, Carissa Cornelia Chundiawan and Fidel Hermanto in Canadian Journal of Kidney Health and Disease
Footnotes
Author Contributions
PN designed the study, collected the data, performed the data analysis, and drafted the original manuscript. THS collected some of the data and helped draft the manuscript. CCC and FH helped the data collection and analysis process. MB, AR, AL, SK, PS, and HS supervised and gave input throughout the drafting and data analysis process. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
