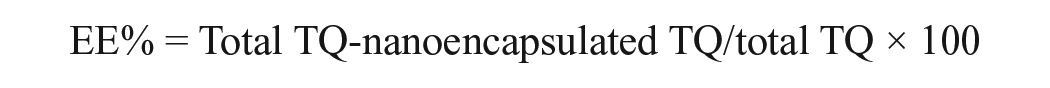Abstract
Background:
Cisplatin (Cis) is potent chemotherapy used to treating already many different types of cancer; however, it is found to correlate with nephrotoxicity and other adverse health consequences. Thymoquinone (TQ) is an antioxidant and anti-inflammatory molecule that may defend against the consequences of different chemotherapies. Thymoquinone uses, although, are negatively impacted by its weak solubility and inadequate biological availability.
Objectives:
This investigation examined the efficacy of a new nanoparticle (NP) absorbing TQ in an Ehrlich Ascites Carcinoma (EAC) mice model to address its low solubility, enhance its bioavailability, and protect against Cis-induced nephrotoxicity.
Methods:
Following 4 treatment groups were included in this research: (1) control, (2) EAC, (3) EAC + Cis + Thymoquinone nanoparticle (TQ-NP) treated, and (4) EAC + Cis-treated.
Results:
The study revealed that TQ-NP was efficacious in avoiding Cis-induced kidney problems in EAC mice, as well as restoring kidney function and pathology. Thymoquinone nanoparticle considerably reduced Cis-induced oxidative damage in renal tissue by augmenting antioxidant levels. According to tumor weight and histological investigation results, TQ-NP did not impair Cis’s anticancer efficacy.
Conclusion:
Thymoquinone nanoparticle might be used as a potential drug along with Cis anticancer therapy to reduce nephrotoxicity and other side effects while maintaining Cis anticancer properties.
Introduction
Cancers of many different sorts are treated with cisplatin (Cis). Based on its propensity to induce deoxyribonucleic acid (DNA) and trigger cytotoxic DNA damage, Cis can annihilate cancer cells. 1 However, it generates apoptosis and diminishes the effectiveness of cell healing. 2 Myelosuppression, nausea, and vomiting are among the unfavorable side effects of Cis use. 3 Rapid deterioration in the kidney function is considered to be a significant deleterious reaction of using Cis as well. 4 In laboratory animals, the first nephron segment after the glomerulus is primarily seriously affected by Cis. 5 The notable histopathological hallmarks of Cis-induced nephrotoxicity in animal research include extensive necrosis and progressive degeneration of renal proximal tubular epithelial cells. 6 The underlying mechanism by which Cis-induced nephrotoxicity caused is heavily contested. Numerous processes, including apoptosis, free radicals, inflammation, and hypoxia, have been postulated. 7
When used with traditional remedy, complementary therapy can produce excellent outcomes with fewer side effects and improved therapeutic resistance. Black cumin or black seed, also known as thymoquinone (TQ), a monoterpene, is the predominant effective component of Nigella sativa. 8 It contains antiphlogistic and antioxidizing qualities and can prevent a variety of malignant cancers. 9 Thymoquinone has been shown to decrease leukemia cell proliferation and induce apoptosis.10-12 Several studies have found that a high dosage of TQ exacerbated the nephrotoxic harmful effects of Cis in cancer-free female rats. 13 On the contrary, numerous different studies have concluded that minimal concentrations of TQ protect rats against Cis-induced nephrotoxicity.13-17 Another recently published research study found that combining TQ with curcumin at high doses protects against Cis-induced nephrotoxicity. 18 Thymoquinone-chemotherapeutics amalgamation boosted the antitumor effect of numerous chemotherapeutic drugs while mitigating their harmful effects on normal cells and tissues due to their antioxidant properties. 19 The disadvantages of using TQ as a primary pharmaceutical drug are connected to its low solubility, which impairs its bioavailability.20,21
The Ehrlich carcinoma model is widely used in cancer research as it easily grows in the peritoneal cavity. It has also, shown to proliferate and metastases violently in the absence of the estrogen hormone. 22 Thus, this study elected to focus specifically on male mice for better outcomes to achieving the aims of the study. The planned study would assess the potential protective advantages of TQ.
Thymoquinone nanoparticles (TQ-NPs) research plans to work on TQ’s restorative characteristics, like its targeting specificity, bioavailability, and potential for unspecific binding. Thymoquinone nanoparticles surpassed free TQ in studies considering different TQ-NPs compositions in contrast to renal harm and cancer. 23 Niosomal, liposomal, polymeric, solid lipid nanocarriers (SLNs), and nanostructured lipid carriers (NLCs) were contained in these formulations. 24 Thymoquinone is a potential anticancer medication that smothers cancer cell development and its progression in in vitro experimental animal model. Also, using of NP formulation against Ehrlich Ascites Carcinoma (EAC) might lead to renal failure in male mice. 25 Regardless of TQ’s huge pharmacological properties, its clinical interpretation is hampered by its narrow therapeutic index and hydrophobicity. 25 Accordingly, we and others implanted TQ in NPs to increase efficiency and limit anticancer activity. When these TQ-NP formulations were equated to Cis, a chemotherapeutic therapy used to treat kidney cancer, the former had predominant anticancer and mitigating properties.24-26 We give a depiction of the numerous TQ-NP formulations, underline their unprecedented efficiency, and examine conventional procedures to help TQ bioavailability and anticancer movement, expanding the chance of clinical therapy.
Materials and Methods
Protocols
Initially, Swiss albino healthy male mice of 8 weeks age and 25 g body weight, were either injected with saline or the Ehrlich tumor cells (1 × 104-107 in 25 µL) intraplantar (i.pl.). Between 0 and 12 days, assessments of paw edema/tumor development, mechanical and thermal hyperalgesia, and overt pain-like behavior were recorded. The dose of 1 × 106/paw of tumor cells was determined for the subsequent studies of mechanical and thermal hyperalgesia, paw edema/tumor development, and histological examination at the designated time points based on the results. For tests involving overt discomfort, 1 × 107 tumor cells/paw and assessment on day 8 following inoculation were selected. After 12 days of tumor injection, paw specimens were collected for histological and microscopic examinations. To determine the hyperalgesic action of cellular fragments, Ehrlich tumor cells were inactivated and administered i.pl., then compared to saline and active Ehrlich tumor cell groups. The computations were measured on days 0 to 12. To assess the pharmacological modulation of Ehrlich tumor–induced pain-like behavior, mice were treated with Cis (5 mg/kg) i.pl. injection and TQ-NP treatment was given orally with a dosage of 10 mg/kg on the eighth day after Ehrlich tumor cells treatment, and quantifying paw edema/tumor growth and mechanical and thermal hyperalgesia.
Thymoquinone Nanoparticles
Thymoquinone nanoparticles were prepared with poly lactic-co-glycolic acid (PLGA) using the solid/oil/water (S/O/W) solvent evaporation method as previously described. 26 An optimized TQ-NP formulation in the form of oil-filled nanocapsules (F2-NC) was obtained with a mean hydrodynamic diameter of 117 nm, (polydispersity index) PDI of 0.16, about 60% loading efficiency, and sustained in vitro drug release. Briefly, PLGA (Sigma, St. Louis, Missouri) was dissolved in high-performance liquid chromatography (HPLC)-grade dichloromethane (DCM) as an oil phase for 12 hours to obtain uniform solution followed by the addition 5 mg TQ. The suspension was sonicated for 2 minutes to generate S/O primary emulsion that was emulsified with an aqueous phase of 20 mL saline with polyvinyl alcohol (1%) to form S/O/W emulsion using a magnetic stirrer for about 7 rcf. The mixture was vortexed for 10 seconds at a high setting followed by ultrasonication (20 KH2) for 3 minutes to generate the final S/O/W emulsion. The organic solvent was evaporated using a rotary evaporator at 50°C. Following centrifugation for about 9000 rcf for 20 minutes at 4°C, the synthesized material was collected and resuspended in 2% sucrose. The shape and size of the particles were confirmed using transmission electron microscope (TEM) (TEM is described in details in the literature27,28). The suspension of TQ-PLGA NPs exhibited turbid white color, and the NPs were spherical in shape and 20 nm (10-30 nm) sized with 80% encapsulation efficiency. Nanoparticles suspension was subjected to centrifugation for about 80 000 rcf for 15 minutes. The supernatant was removed, and 1 mL of methanol was added to the sediment which was treated with sonication for 5 minutes and then injected to HPLC to measure the amount of TQ.29-31 The EE was 80% as calculated using the following equation:
Cell Line Tested and Authenticated [from the source]
The non-human cell line identification is used. Mouse cell line authentication method was adopted to test cells. A multiplex polymerase chain reaction assay targeting 9 tetranucleotides short tandem repeat (STR) markers in the mouse genome. Mouse cell line authentication test in India is the most common model system and we perform STR profiling following ISO9001 and ISO/IEC17025 quality standards. Short tandem repeat for validated mouse cell lines at 18 STR markers. Consensus allele calls (greater than 98%). Primers were used to sequence STR regions to determine actual number of repeats (alleles) and correlate with fragment length obtained using capillary electrophoresis.
Mouse multiplex polymerase chain reaction (PCR) assay is as follows:
Forward Primer (5’-3’)
TCTTTCTCCTTTTGTGTCATGC
Reverse Primer (5’-3’)
GTTTCTTGCTAAATAACTAAGCAAGTGAACAGA
Cell Line Preparation Before Inoculation
Ehrlich’s Ascites Carcinoma (EAC) cell lines were received since 2/2021. Cells were tested at 3/2021 and then we immediately used them in experimental models. Cells were provided by the Indian Institute of Chemical Biology (IICB), Kolkata, India (EAC strain 7 transplantable mouse carcinoma sources). The IICB followed UKCCCR Guidelines for the Use of Cell Lines in Cancer Research. 32 To check the in vitro cell viability before inoculation into the experimental mice groups we applied MTT assay (MTT Assay Kit MyBiosource Catalog No: MBS2540588). The MTT cell viability measurements protocol for tumor growth was conducted according to standard manufacturers’ description. To observe the EAC cells, the ascitic fluid obtained from treated mice was stained with DAPI fluorescent dye and kept in dark condition at 37°C for 15 minutes, and then to measure cellular metabolic activity as an indicator of cell viability, proliferation, and cytotoxicity readings absorbance at 570 nm. Protocol was applied once before the inoculation of cancer cells to ensure cell lines are adequate for the planned mice experiments (Figure 1).
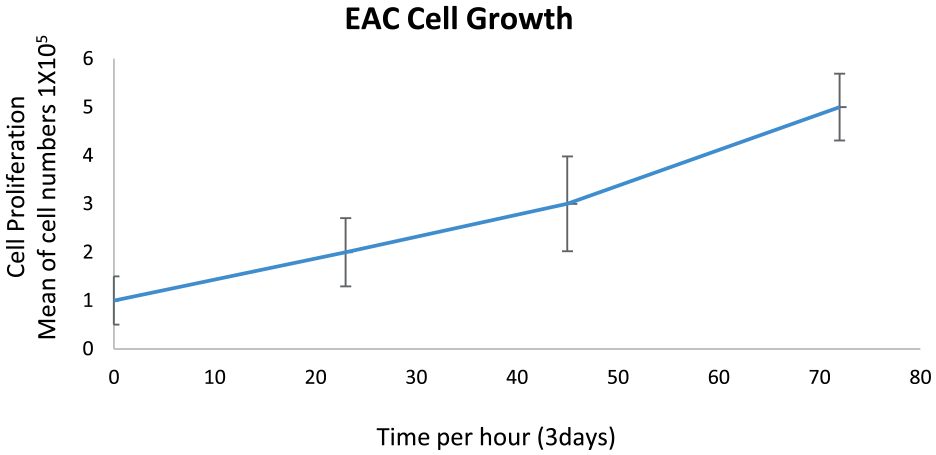
Initial evaluation of in vitro cell viability, this has been measured to determine cell line viability and tumor growth rate prior to the inoculation of the EAC into the planned 40 mice experimental animals.
The Ehrlich Tumor Cells Inoculation
Ten days following the introduction of the tumor, Ehrlich tumor cells were taken from the ascitic fluid in the peritoneal cavity of mice. The ascitic fluid was 3 times rinsed with phosphate-buffered saline (PBS), pH 7.4, subjected to centrifugation (200 g, 10 minutes), and rinsed 3 times with PBS. The Neubauer chamber was used to test the cell viability using the 0.5% trypan blue exclusion method, steps were applied according to the Trypan Blue Exclusion Method as previously described. 33 In 25 µL of saline, the Ehrlich tumor cells were diluted to final concentrations of 1 × 104, 1 × 105, 1 × 106, and 1 × 107. Measurements were conducted before and following the injection of cancer cells on days 0 through 12.
Experimental Design
Forty Swiss albino male mice, 8 weeks adults, body weight 25 g supplied from animal holding facility, within the university pharmacy veterinary laboratory, were acclimatized, and divided into 4 groups, 10 mice in each group (Table 1). The assessment of blood biomarkers for the renal function test of urea and creatinine to evaluate kidney oxidative damage as reflected in glutathione (GSH) level, and furthermore, tumor growth and inflammation impact was also assessed by measuring the tumor necrosis factor (TNF-α) levels. At the end of experiments and before mice scarification and biopsied for histological analysis, serum samples were obtained from across the experimental mice groups. 1.0 mL of whole blood collected and allowed to clot, then centrifuged for about 90 rcf for 10 minutes to separate the serum to be stored at −20°C until biochemical tests performed.
Animal Grouping and Experimental Design.

Statistical Analysis
Data analysis was done by using SPSS (version 26.0; SPSS Inc, Chicago, Illinois) for Microsoft Windows. Results were calculated as mean ± standard deviation (SD) and the group variables were given as numbers and percentages (%). An unpaired t-test was used for comparing 2 means, whereas chi-square was achieved for comparing the frequency of 2 groups or more. The strength of a relationship between 2 variables was evaluated using the Pearson’s correlation test. P < .05 is the measure of significance. Each value represents the mean ± SEM (standard error of the mean) of the group using 1-way analysis of variance (ANOVA) test.
Histological Preparation and Interpretation
Experimental mice were sacrificed and both kidneys from across the experimental mice were biopsied and fixed in 10% neutral buffered formalin solution and then samples were processed into a paraffin wax blocks, a 4 μm thick paraffin sections were obtained. The sections were stained for histological hematoxylin and eosin (H&E) to observe and report pathological changes throughout the experimental mice. Anonymously and blindly histopathological analysis has been conducted across all samples and in a systematic fashion. The findings were reported blindly regardless of treatment group by experienced pathologist and later reports identified and compared to mice groups. After each mice group and the relevant treatment exposure was confirmed, findings were correlated to their specific groups, a representative histology for each of the 4 mice experimental groups was selected and shown in Figure 2.
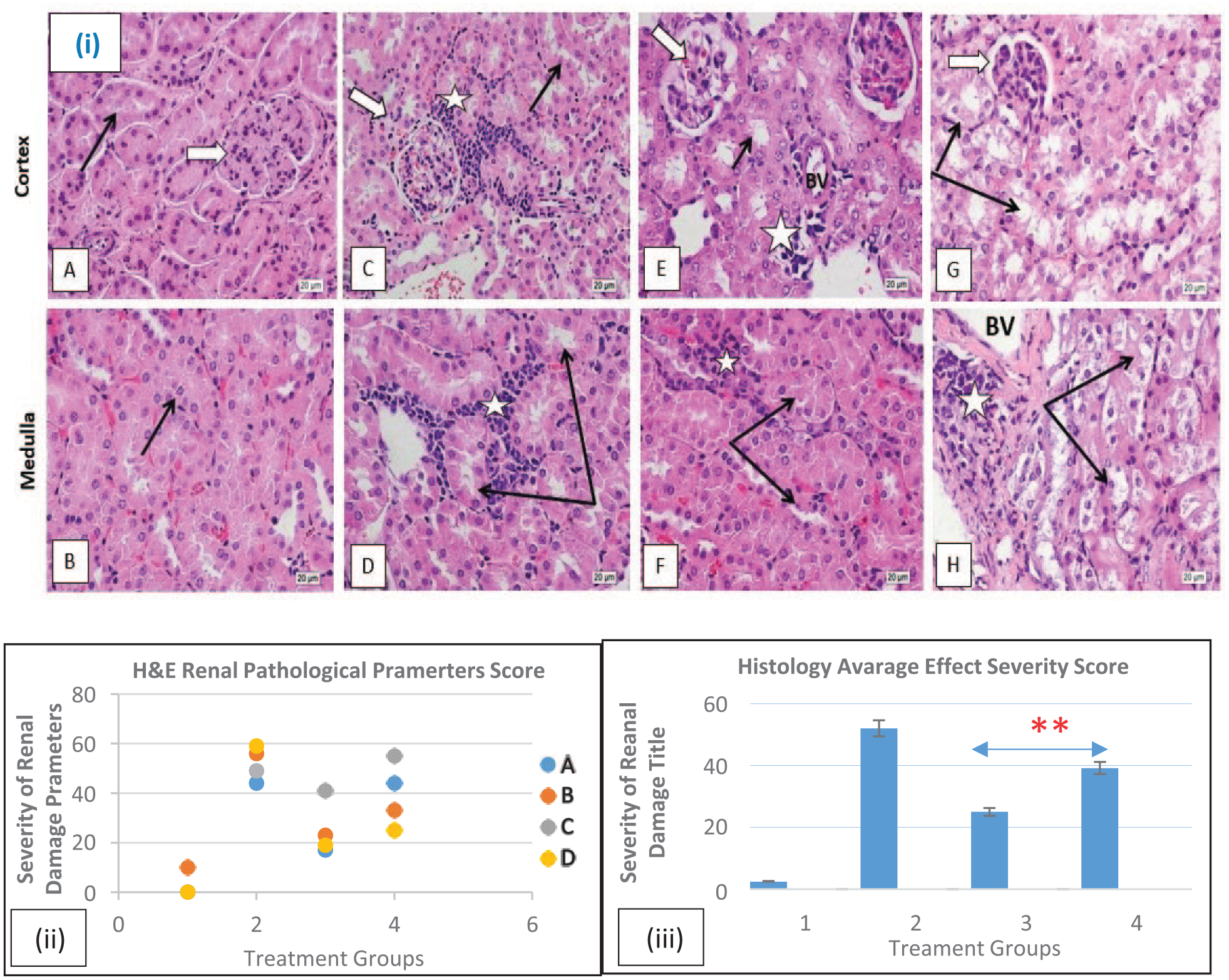
Histology study experimental design of 40 mice treated in 4 different groups. Shown here are the representative micrographs of H&E-stained histology sections (4 µm) (magnification: ×40) mice kidney cortex and medulla to show renal corpuscles (white arrows) glomerular capillaries (black star), kidney tubules (black arrows) and neoplastic cells (white star) histological analysis revealed:
Furthermore, histopathological quantitative analysis was applied to establish average scores and semi-quantitative assessment applied to identify more subtle differences in kidney injury and to strengthen our previous reported pathological findings. Each mice group (of 10) average score was calculated and analyzed to quantitively assess the degree of severity for the histological/morphological changes in each treatment group of the mice. A previously described semi-quantitative method for severity of histological/pathological scoring was used with minor modifications.36,37 The severity scores are often recorded using scale of [0-3], with [0] indicating no pathological effect or semi normal histological morphology, [1] indicating minimal pathological effect or morphological change, [2] moderate pathological effect, and [3] for severe effect. Fisher’s exact test was applied to compare between the 2 groups in question: treatment in combination with QT-NP and without QT-NP.
Results
Histological Results
Final representative histology of induced leg solid Ehrlich carcinoma in a mice model were found to be transformed into aggressive infiltrating cells with highly characteristic pleomorphic nuclei associated with numerous mitotic figures. It massively infiltrated the underlying leg muscles and was found to metastasize to distant organs including the kidney, often via blood circulation, where it continues to proliferate near large BVs, and peritubular capillaries and tissues around. A single administration of Cis was found to moderately decreases the size of the growing tumor based on measuring mice leg weight and histologically findings of a decreasing the frequency of the presence of neoplastic cells deep in kidney tissue and their deformative appearance. However, Cis nephrotoxicity was observed in the kidneys of treated mice (Figure 2).
Discussion
The ongoing research demonstrated that renal function was impaired in EAC-bearing mice, as exemplified by higher serum urea and creatinine levels (Figures 3 and 4). In addition to these observations, different vascular, degenerative, and inflammatory pathological alterations were detected in the renal tissue of EAC-bearing mice, as well as a substantial infiltration of neoplastic cells. Abd Eldaim et al, 38 Donia et al, 39 and Hashem et al 40 are in agreement with these findings. Following that, the increase in kidney functional biomarkers could be compared to renal damage sustained by tumor metastasis and cancerous cells infiltration in renal tissue. Thus, resulting in abnormal proximal tubules reabsorption and glomerular filtration rate, plus a decrease in urea and creatinine excretion, contributing to higher blood levels. 41 A significant evaluated renal function test urea and creatinine recorded kidney oxidative damage as demonstrated by substantial decreases in the reduced GSH. A precursor for tumor progression that ultimately decreases the cellular antioxidants and, subsequently, induces renal tissue damage. Oxidative stress is well known to be one of the most contributing factors to cancer initiation and progression. It is also implicated as a potential mechanism of EAC-induced renal damage. 40 On the contrary, mice groups (G3 and G4) have been treated with Cis chemotherapy similarly. Thus, in fact, it would also impact these histological findings.4-6,42-44 Certainly, treatment was equally introduced onto both groups, therefore, this does not exclude the impact of this well-known aggressive chemotherapy implications on histological changes; however, this study was not aiming to study this indicator as far as to soften and minimize the severity of histological damage that caused by either EAC or Cis treatment by applying TQ-NP (G3) and compare the findings with the current approach in clinical practice of today (G4).
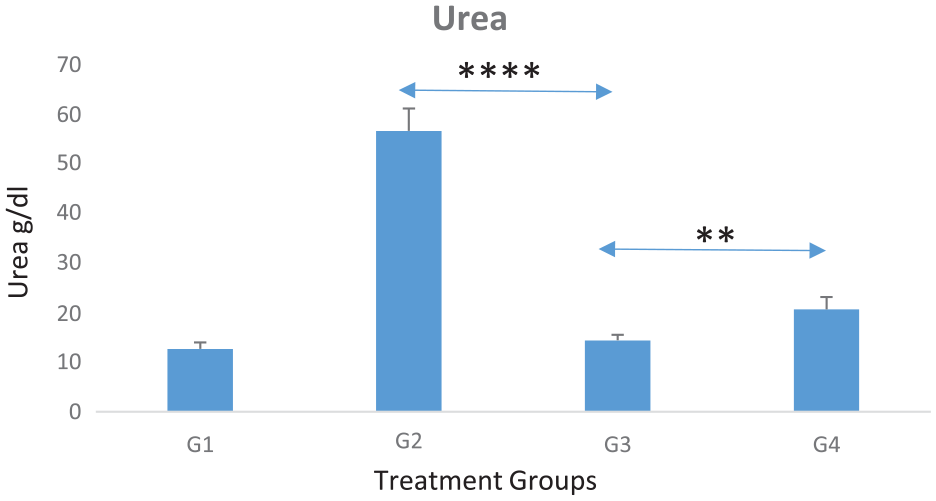
Graph shows average serum urea levels for experimental groups (each of 10 mice). Noticeable increased levels in most affected nephrotoxicity in untreated group (G2) by neither therapies Cis nor Cis + TQ-NP), whereas the nephrotoxicity was significantly decreased in those treated with combined (Cis + TQ-NP) therapy (G3), when compared with untreated group (G2) (P = .0000117). Also, when comparing between Cis and Cis + TQ-NP groups (G4 and G3), nephrotoxicity was significantly decreased, this was shown in urea biochemical level, results were lower than Cis only treated group (G4) (P = .01).
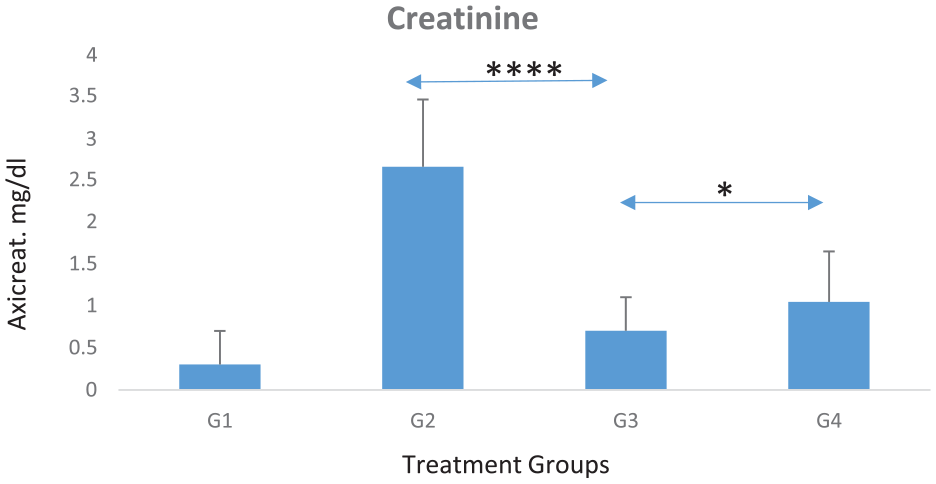
Graph shows average serum creatinine levels for each experimental group (of 10 mice). Noticeably increased creatinine levels in most affected nephrotoxicity untreated group (G2) and significantly decreased in those treated with combined (Cis + TQ-NP) therapy (G3) (P = .00005). When comparing between Cis and Cis + TQ-NP groups (G4 and G3), nephrotoxicity was significantly decreased, this was represented in creatinine biochemical level, results were slightly lower than Cis only treated group (G4) (P = .03). The differences between (G1 and G3) and (G1 and G4) groups were noted; certainly, this is reflecting the none-fully restoration of kidneys functionality at this point of experimental window timeline, but a great significant restoration was reported as above and confirmed later in histology (Figure 2).
Effect of TQ-NP on Ehrlich tumor induced tumor and inflammation impact on serum TNF-α levels in experimental mice. Statistical analysis when comparing the EAC tumor in untreated with Cis nor QT-NP group (G2) with the third experimental group (G3) (Cis + TQ-NP) dose treated shows a significantly decreased TNF-α levels (P ≤ .0007). Also, analysis shows a significant increase in glutathione (GSH) levels (P ≤ .00001). However, there was no significance differences in TNF-α and GSH between the Cis only treatment and Cis combined TQ-NP treatment groups (G3 vs G4) (P = .09 vs P = .06, respectively) (Figures 5 and 6). Interestingly, previous research also found some of the renal biochemical markers have no significant differences in a related nephrotoxicity study in brown Norway rats, as in Cis-treated mice in developing a new platform for early detection in nephrotoxicity and in acute nephrotoxicity.42-44

This graph shows GSH antioxidant factor across the 4 mice experiments groups (total of 40 mice studied). A significant negative correlation between the (Cis + TQ-NP) of 10 mice treated (G3) group, and the EAC tumor 10 mice (G2) group (P ≤ .00001109), this confirming a significant decreased GSH levels in the (10 mice) group (G2) untreated. However, there was no significant difference in GSH levels between G3 and G4 groups (P = .06).
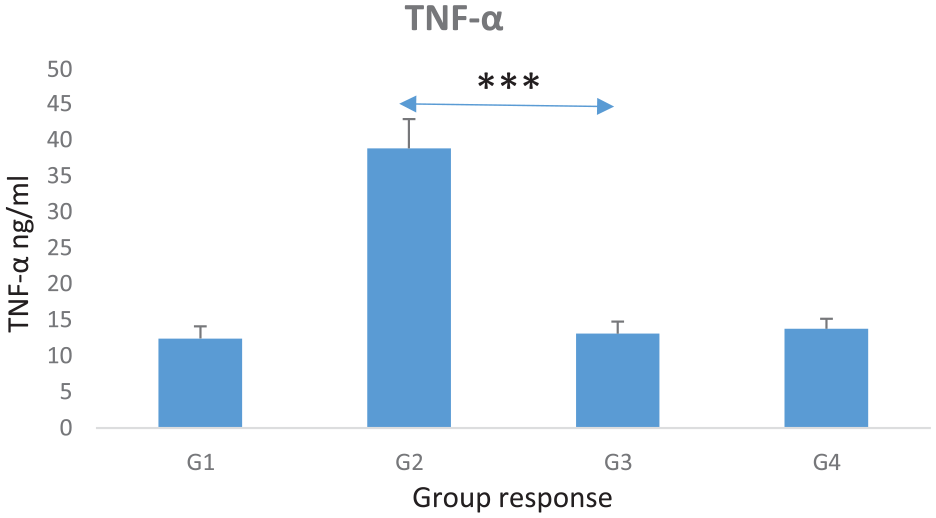
This graph shows TNF-α levels across the 4 mice experimental groups (total of 40 mice). The correlation between the (Cis + TQ-NP) treated 10 mice group (G3) and the EAC tumor group (G2) was statistically significant (P ≤ .0007), while slight but not significant TNF-α increase in G4 than G3; however, the effect of the TQ-NP on induced nephrotoxicity in experimental mice was significantly decreased. However, there was no significant difference in TNF-α levels between G3 and G4 groups (P = .09).
Conclusion
This study has revealed the combination of TQ-NP and cisplatin therapy could play a role in future innocuous cancer treatment design. Histological observations and histology quantitative analysis both have shown significantly a marked preserved normal kidney’s parenchyma and tubular morphology without interfering with the cisplatin drug bioactivity in defeating cancer cells. Levels of blood creatinine and urea biomarkers were significantly decreased reflecting healthier kidney function. Further studies are needed to investigate the mechanism of how TQ-NP would minimize the nephrotoxicity implication on kidneys.
Research Data
sj-odt-1-cjk-10.1177_20543581241258812 – Supplemental material for Thymoquinone Nanoparticles (TQ-NPs) in Kidney Toxicity Induced by Ehrlich Ascites Carcinoma (EAC): An In Vivo Study
sj-odt-1-cjk-10.1177_20543581241258812 for Thymoquinone Nanoparticles (TQ-NPs) in Kidney Toxicity Induced by Ehrlich Ascites Carcinoma (EAC): An In Vivo Study by Zakaria Eltahir, Maha Ibrahim, Muniera Y. Mohieldeen, Ammar Bayoumi and Samia M. Ahmed in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors are thankful to their universities for the continuous support of this study.
Author Contributions
All authors are responsible for the concept and design of the study. Z.E., M.I., A.B., and S.A. contributed to data acquisition. A.B., M.Y.M., Z.E., and S.A. contributed to data analysis. Z.E. and M.I. interpreted the results and drafted the manuscript. All authors contributed to the creation, critically revised, and approved the manuscript final version to be published, and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent for Publication
The authors agree to the final version of the paper.
Ethical Approval and Consent to Participate
Protocols for animal experiments were approved by the Animal Experimental Ethics Committee of the King Abdulaziz University (approval no. p745-2020), in compliance with the Saudi Law of Ethics of Research on Living Creatures guidelines for the care and use of laboratory animals. Methods were performed according to the recommendations of Good Clinical Practice and the Declaration of Helsinki (2013).
Consent for Publication
Not applicable.
Availability of Data and Materials
All relevant data are included in this published article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.