Abstract
Background:
Corona Virus Disease 2019 (COVID-19), an infection caused by the SARS-CoV-2 virus, has been the largest global pandemic since the turn of the 21st century. With emerging research on this novel virus, studies from the African continent have been few. Corona Virus Disease 2019 has been shown to affect various organs including the lungs, gut, nervous system, and the kidneys. Acute kidney injury (AKI) is an independent risk factor for mortality and increases the health care burden for patients with persistent kidney dysfunction and maintenance dialysis. Sub-Saharan Africa has a high number of poorly controlled chronic illnesses, economic inequalities, and health system strains that may contribute to higher cases of kidney injury in patients with COVID-19 disease.
Objectives:
The objective of this study was to determine the incidence, associated factors, and outcomes of AKI in patients hospitalized with COVID-19 in Kenya.
Methods:
This retrospective cohort study included 1366 patients with confirmed COVID-19 illness hospitalized at the Aga Khan University Hospital in Nairobi, Kenya, between April 1, 2020 and October 31, 2021. Data were collected on age, sex, the severity of COVID-19 illness, existing pregnancy and comorbid conditions including human immunodeficiency virus (HIV), diabetes mellitus, hypertension, and functioning kidney transplant patients. Univariate analysis was carried out to determine the association of clinical and demographic factors with AKI. To determine independent associations with AKI incidence, a logistic regression model was used and the relationship was reported as odds ratios (ORs) with a 95% confidence interval (CI). The outcomes of AKI including the in-hospital mortality rate, renal recovery rate at hospital discharge, and the duration of hospital stay were reported and stratified based on the stage of AKI.
Results:
The median age of study patients was 56 years (interquartile range [IQR] = 45-68 years), with 67% of them being male (914 of 1366). The AKI incidence rate was 21.6% (n = 295). Patients with AKI were older (median age = 64 years vs 54 years; P < .001), majority male (79% of men with AKI vs 63.6% without AKI; P < .001), and likely to have a critical COVID-19 (OR = 8.03, 95% CI = 5.56-11.60; P < .001). Diabetes and hypertension, with an adjusted OR of 1.75 (95% CI = 1.34-2.30; P < .001) and 1.68 (95% CI = 1.27-2.23; P < .001), respectively, were associated with AKI occurrence in COVID-19. Human immunodeficiency virus, pregnancy, and a history of renal transplant were not significantly associated with increased AKI risk in this study. Patients with AKI had significantly higher odds of mortality, and this effect was proportional to the stage of AKI (OR = 11.35, 95% CI = 7.56-17.03; P < .001). 95% of patients with stage 1 AKI had complete renal recovery vs 33% of patients with stage 3 AKI. Of the patients with stage 3 AKI (n = 64), 10 underwent hemodialysis, with 1 recovery in renal function and 3 patients requiring ongoing dialysis after discharge.
Conclusions:
This study was conducted at a single private tertiary-level health care facility in Kenya and only up to the time of hospital discharge. It is one of the first large studies from sub-Saharan Africa looking at the associated factors and outcomes of AKI in COVID-19 and forms a foundation for further analysis on the long-term consequences of COVID-19 on the kidneys. A major limitation of the study is the lack of baseline pre-admission creatinine values for most patients; thus, the impact of chronic kidney disease/baseline creatinine values on the incidence of AKI could not be established.
Introduction
Corona Virus Disease 2019 (COVID-19), a novel infection caused by the SARS-Cov-2 virus, was first reported in the Wuhan area of China in late 2019 and rapidly spread worldwide. On March 11, 2020, COVID-19 was declared a global pandemic by the World Health Organization. 1 Three years in, global reported infections have surpassed 700 million, with almost 7 million related deaths. 1 Despite daily emerging evidence on this novel virus, there has been a paucity and lethargy of studies from the African continent. Kenya announced its first case of confirmed COVID-19 on the March 13, 2020. Three years later, the country had reported 350 000 confirmed cases, with 5700 related deaths. 2 This number is, however, likely to be higher given the poor health-seeking behavior and a lack of confirmatory tests for COVID-19 in most hospitals across the country.
The SARS-CoV-2 virus has been shown to bind to receptors in the lungs, gut, nervous system, and kidneys, causing symptoms related to these organs.3-5 Studies suggest that up to 75% of patients with COVID-19 have a derangement in their kidney function.6,7 Acute kidney injury (AKI) is associated with increased mortality rates and a longer duration of hospital stay, with higher odds noted in COVID-19 patients.8-11 Acute kidney injury in COVID-19 may result in poor renal recovery rates and a faster progression into chronic kidney disease, which places an additional burden on a country’s health care system.12-14
The incidence of AKI has varied greatly across populations, ranging from 9% to 55% depending on the geographical area, differences in COVID-19 severity, race, age, and the socio-economic status of the patient population.10,15-20 Being of African descent in particular has been identified as an independent risk factor for AKI, with reasons ranging from socio-economic advantages limiting access to health care services, to poor health seeking behavior, the presence of comorbid conditions, and genetic factors increasing the AKI risk.10,11,21-23 The emergence of different variants of the SARS-CoV-2 virus, including the Delta and Omicron variants, in the background of vaccine introduction in late 2020 to 2021, has altered the course and severity of COVID-19 illness and its related complications.24,25
This study aims to assess the impact of COVID-19 infection on the kidneys in patients hospitalized at a tertiary-level facility in Nairobi, Kenya, in the course of 4 COVID-19 waves. We determined the incidence and the factors associated with AKI. We also looked at the outcomes of AKI at the time of hospital discharge, which include the mortality rate, the duration of hospital stay, and the status of renal recovery.
Methods
Study Population
This retrospective cohort study included all patients hospitalized at the Aga Khan University Hospital, Nairobi (AKUHN), Kenya, between April 1, 2020 and October 31, 2021. The AKUHN is a tertiary-level 260-bed private teaching hospital located in Nairobi, Kenya, that also admits patients from across Kenya and sub-Saharan Africa as witnessed during the COVID-19 pandemic. Patients with a COVID-19 diagnosis were identified from the Electronic Health Records (EHR) database unique to and maintained by AKUHN, using the International Classification of Diseases 10th Edition (ICD-10) 2005 version diagnostic codes B97.2 (“Coronavirus as the cause of diseases classified elsewhere”) and B34.2 (“Coronavirus infection: unspecified site”). Due to the strain on hospital resources at this time, only patients who met a pre-set admission criteria that included a requirement for oxygen supplementation to maintain oxygen saturation (spO2) above 94%, or the presence of comorbid conditions in stable patients, were hospitalized (see Supplemental Information). Patients requiring in-patient care for other ailments, but with an incidental finding of COVID-19, were also coded as COVID-19 positive and included in the study.
Inclusion Criteria
All patients above the age of 18 years with a confirmed COVID-19 diagnosis were said to meet the inclusion criteria. The diagnosis of COVID-19 infection was confirmed based on a positive RT-PCR (reverse transcription-polymerase chain reaction) test of nasopharyngeal swabs performed on all patients on the day of admission. The hospital laboratory utilizes 2 analyzer machines that identify 2 of the genes within the SARS-CoV-2 virus (Allplex SARS-CoV-2 assay, Seegene RealStar SARS-CoV-2 RT-PCR kit 1.0, Altona Diagnostics, Germany). The RT-PCR test for COVID-19 infection has been shown to have a 99% sensitivity.26,27
In addition, patients who may have tested negative on RT-PCR but had a high clinical suspicion for COVID-19 infection were included in this study based on chest computed tomography (CT) scan findings suggestive of COVID-19 (24 of 1366). The COVID-19 Reporting and Data System (CO-RADS) classification system was used in reporting images, where patients with a CO-RADS 4 and 5, corresponding to high and very high suspicion for COVID-19 infection, were included in this study. The CO-RADS classification system allows for standardized reporting of chest images in suspected COVID-19 cases, with utility reported in various studies, including 1 study done at the AKUHN.28-31 The hospital uses a 256 slice multidetector dual-energy and dual-source CT scan machine, with chest images reviewed by 2 qualified hospital radiologists.
Exclusion Criteria
Patients in whom a diagnosis of AKI could not be made due to lack of serial creatinine levels and those on maintenance dialysis were excluded from this study.
Data Collection
Data were manually extracted from the hospital EHR system by the primary investigator and independently reviewed for accuracy by other authors (J.S., C.G., A.S.). These records included uploaded discharge summaries, clinical notes, patient vital signs, and laboratory investigations. Information was collected on demographic data (age and sex), comorbid conditions including human immunodeficiency virus (HIV), diabetes mellitus, hypertension, post-renal transplant and pregnancy states, and laboratory values including creatinine values, HIV viral load, and glycated hemoglobin levels.
Description of Patient Characteristics and Outcomes
Acute kidney injury is defined using the 2012 Kidney Disease: Improving Global Outcomes (KDIGO) criteria as an increase in serum creatinine by 26.53 micromole/L (0.3 mg/dL) or more within 48 hours or a 50% or more increase in serum creatinine within the last 7 days. 32 Preadmission baseline creatinine values were missing for most patients; the baseline creatinine was taken to be the median of creatinine values during admission (used to calculate AKI based on 7-day definition). Urine output was not used in this study to diagnose AKI due to missing data for most patients. The incidence of AKI was assessed, with occurrence within or after 48 hours of admission based on an early (admission) elevated creatinine value. Using the KDIGO criteria, AKI was further characterized into stages based on severity: stage 1 as an increase in serum creatinine level by 1.5 to 1.9 times baseline, stage 2 as an increase in serum creatinine by 2.0 to 2.9 times the baseline, and stage 3 as an increase in serum creatinine level to 3.0 times the baseline or greater, or an increase by 353.68 micromole/L (4.0 mg/dL) or greater, or the initiation of renal replacement therapy (RRT).
Once a diagnosis of COVID-19 was made, patients were then classified using the National Institute of Health (NIH) clinical spectrum of SARS-CoV-2 infection, adapted by the Kenya Ministry of Health, as mild to moderate disease, severe disease, or critical illness. 33 Patients with mild to moderate disease have clinical and/or radiological features of pneumonia and SpO2 at or greater than 94% on room air. Severe illness is defined as an SpO2 less than 90% on room air, and critical illness as the presence of respiratory failure, septic shock, or multiorgan dysfunction in severely ill patients.
Hypertension is defined based on the European Society of Cardiology (ESC)/European Society of Hypertension (ESH) guidelines as a blood pressure greater than 140/90 mm Hg in 2 or more readings taken 4 or more hours apart. 34 Patients were said to be hypertensive if they were on treatment for hypertension prior to admission or were newly diagnosed based on in-patient blood pressure readings keeping with this definition.
A new diagnosis of diabetes mellitus was made based on the 2017 International Diabetes Federation (IDF) Clinical Practice Recommendations for managing type 2 diabetes 35 which have been adopted in the Kenya Diabetes Management guidelines and include a fasting plasma glucose level of 7 mmol/L (126 mg/dL) or higher or a random plasma glucose of 11.1 mmol/L (200 mg/dL) or higher in a patient with classic symptoms of hyperglycemia, or a hemoglobin A1c (HbA1c) level of 6.5% (48 mmol/mol) or higher. All admitted patients had an HbA1c level and a random/fasting plasma glucose level done at the time of admission.
The study assessed outcomes during the hospital stay, up until the time of in-hospital mortality or hospital discharge. These outcomes include the mortality rate, the rate of renal recovery, and the RRT rate. In-hospital mortality rate was calculated as the number of patients with AKI who died during the hospital stay. The definition of recovery of kidney function is derived from 2012 KDIGO AKI guidelines as independence from dialysis therapy or a return to baseline kidney function in patients younger than 65 years. 32 Partial renal recovery is defined by consensus in KDIGO guidelines as recovery in kidney function by >50% without a return to initial baseline values. Patients were categorized as having either complete or partial renal recovery at hospital discharge based on the latest creatinine values. Transient AKI was recovery in renal function within 48 hours of an AKI diagnosis.
Statistical Analysis
Baseline characteristics were reported using medians and interquartile ranges (IQRs) for continuous variables and counts and percentages for categorical variables. Acute kidney injury incidence was calculated as the fraction of patients with AKI to all admitted COVID-19 patients and stratified according to the severity of COVID-19 disease. Chi-square test and Mann-Whitney tests were used for comparison between percentages and medians, respectively. The relationship between comorbid conditions and AKI was assessed by age-sex adjusted models using logistic regression models. A P < .05 was considered statistically significant. Results of significant relationships were reported as odds ratios (ORs) with a 95% confidence interval (CI). The outcomes of AKI, including the in-hospital mortality rate, RRT rate, and renal recovery rate, were calculated and stratified based on AKI severity. The median duration of hospital stay was calculated for each stage of AKI. Statistical analysis was performed using SPSS (IBM version 20).
Results
Patient Demographic Characteristics
A total of 1366 patients were included in the final analysis after application of inclusion and exclusion criteria as shown in Figure 1.

Patient flow chart showing the patient selection process, number of patients with AKI diagnosis, and their AKI severity staging.
The median age of study patients was 56 years (IQR = 45-68), with 34 patients aged 30 years and below and 9 patients above the age of 90 years (see Table 1 below). Two thirds of all the patients were male (n = 914). Fifteen patients were pregnant on admission (3.3% of females). Of the 1366 admitted patients, slightly more than half had mild to moderate disease (n = 791), whereas 199 (15%) patients required mechanical ventilation. The most common comorbid conditions in this study were hypertension and diabetes mellitus, present in 32% (n = 441) and 40% (n = 548) of all admitted patients, respectively. Five patients admitted with COVID-19 illness had undergone a kidney transplant in the past. Approximately 50% of all admitted patients were not tested for HIV, either due to a lack of provider-initiated testing and counseling or due to test decline. Of the 24 patients with an HIV-positive diagnosis, 7 were newly diagnosed (0.01% of all tested). Of the 17 patients already on treatment, 15 were virally suppressed.
Patient Characteristics.

Note. AKI = acute kidney injury; CT = computed tomography; IQR = interquartile range; MRI = magnetic resonance imaging.
AKI Incidence and Factors Associated
The AKI incidence rate in our study is 21.6% (n = 295); 170 patients (57.6%) had stage 1 AKI, 61 (20.7%) had stage 2 AKI, and 64 (21.7%) had stage 3 AKI. There was a higher AKI occurrence during the fourth COVID-19 wave characterized by the global emergence of the Delta SARS-CoV-2 variant, as shown in Table 2 and Figure 2.
Incidence of AKI According to the COVID-19 Waves.
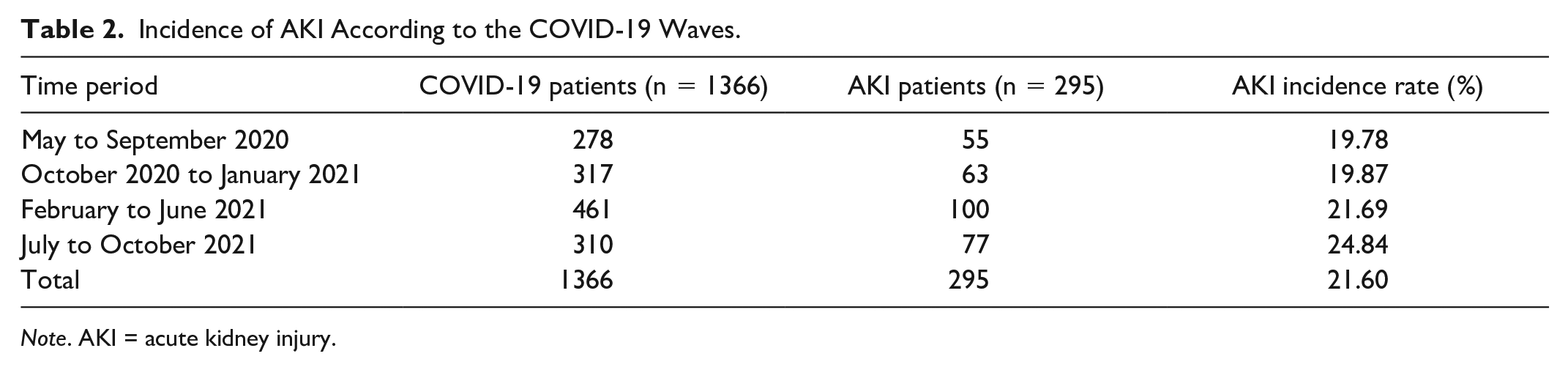
Note. AKI = acute kidney injury.

The average daily new COVID-19 cases in Kenya (April 2020 to October 2021).
Patients with AKI were older (median age = 64 years vs 54 years; P < .001) and more likely to be male (P < .001). After adjustment for age and sex, patients with severe COVID-19 disease were 1.92 times more likely to develop an AKI compared with patients with mild to moderate disease (95% CI = 1.38-2.68; P < .001), with the odds increasing to 8.03 with critical disease (95% CI = 5.56-11.60; P < .001). The incidence rate ratio for AKI in hypertension was 1.68 (95% CI = 1.27-2.23; P < .001) and 1.75 in diabetic patients (95% CI = 1.34-2.30; P < .001) as shown in Tables 3 and 4. Human immunodeficiency virus, pregnancy, and a history of renal transplant were not significantly associated with a higher AKI incidence.
Factors Associated With AKI in Univariate Analysis.

Note. AKI = acute kidney injury; CI = confidence interval; HRCT = high-resolution computerized tomography; IQR = interquartile range; OR = odds ratio.
Age-Sex Adjusted Model for Significant Factors Associated With AKI.
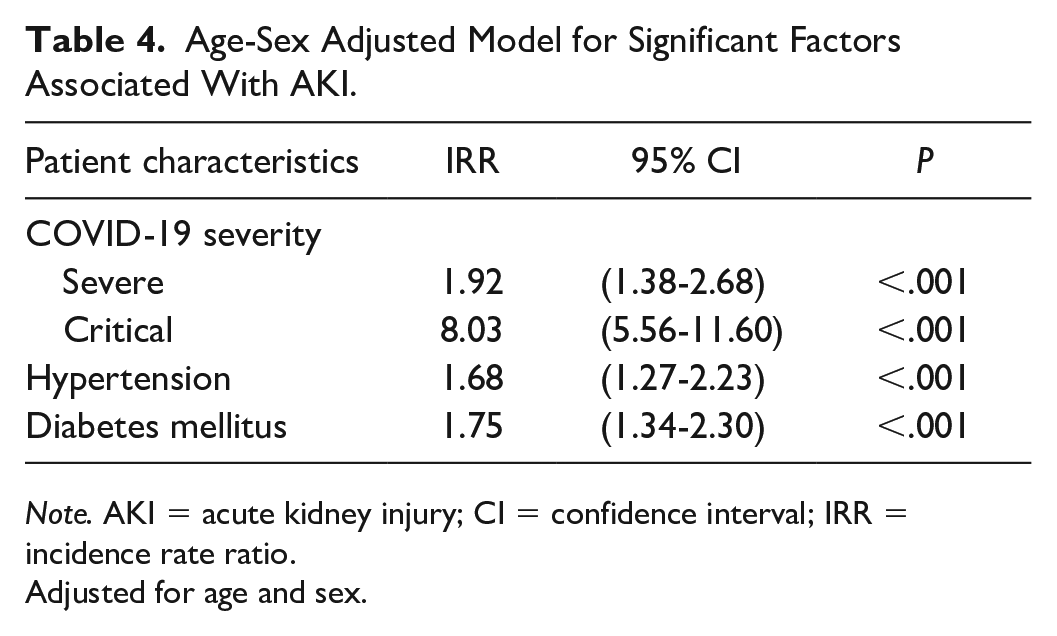
Note. AKI = acute kidney injury; CI = confidence interval; IRR = incidence rate ratio.
Adjusted for age and sex.
48% of the cases of AKI occurred in the first 48 hours of admission (n = 144); 71% of these were stage 1 AKI (n = 102), 17% of these were stage 2 AKI (n = 25), and 12% of these were stage 3 AKI (n = 17). A total of 148 patients developed AKI 48 hours after admission; it includes 59% of patients with stage 2 AKI and 73% of patients with stage 3 AKI and as shown in Table 5.
Incidence of and Time to Resolution of AKI.

Note. AKI = acute kidney injury.
Outcomes of AKI
The in-hospital mortality rate for COVID-19 patients with AKI in our study was 36.9% (n = 109). 18% of patients with stage 1 AKI (n = 34) died compared with 51% of patients with stage 2 (n = 33) and 64% of patients with stage 3 AKI (n = 42). Patients with AKI had 11.35 higher odds of mortality compared with non-AKI patients (95% CI = 7.56-17.03; P < .001). After adjustment for age, sex, COVID-19 severity, and comorbid conditions, the odds for mortality in AKI patients were 3.5 (95% CI = 3.5-9.53; P < .001) as shown in Table 6.
Associations Between AKI and Mortality Risk.

Note. AKI = acute kidney injury; CI = confidence interval.
Ten out of 64 patients with stage 3 AKI underwent hemodialysis, representing an RRT rate of 3.4%. Nine out of these 10 patients had an AKI diagnosis within 48 hours of admission and received dialysis shortly after admission. Out of all dialysis patients, 4 were discharged alive; 3 of the 4 patients required ongoing dialysis after hospital discharge. 90% of patients with stage 1 AKI who were discharged alive had complete renal recovery (n = 122), compared with 82% of stage 2 (n = 23) and only 32% of stage 3 patients (n = 7) (see Figure 3 below).

A graphical representation of the outcomes of AKI in COVID-19, including mortality, renal recovery, and dialysis rates stratified by stage of AKI.
The duration of hospital stay ranged from 1 to 79 days for patients admitted with COVID-19 infection. The median duration of hospital stay was higher in patients with AKI compared with non-AKI patients (8 days vs 10 days; P < .001); 9 days for stage 1 AKI (IQR = 6-13 days), 11 days for stage 2 AKI (IQR = 7-9 days), and 12 days for stage 3 AKI (IQR = 8-18 days) (see Figure 4 below).

The median duration of hospital stay with interquartile ranges for all study patients (COVID-19 positive patients with and without AKI).
Discussion
In our study that included 1366 patients admitted in the course of 4 country COVID-19 waves, the AKI incidence rate was 21.6%. Demographic and clinical factors like male sex, increasing age, diabetes, and hypertension were associated with an increased risk of AKI in COVID-19. Furthermore, the overall mortality rate was high at 36.9%, whereas many patients discharged alive did not fully recover renal function at the time of hospital discharge.
The AKI incidence in our study is lower than the 40% to 60% reported from studies with a similar socio-demographic population.11,18,21 That said, it is still higher than initial reports from China and Europe.9,20 Compared with pre-COVID-19 Kenyan studies on AKI incidence, 36 this figure is 5 times higher. The greater AKI rate in COVID-19-infected persons may be explained by the pathological effects of the SARS-CoV-2 virus on the kidneys, the immune system, and cardiovascular stability, manifesting as pre-renal and intrarenal causes of kidney injury. This argument is further strengthened by the rise in AKI cases with emergence of SARS-CoV-2 virus variants.24,25 The difference in AKI staging between our study and related studies, including comparably fewer stage 1 AKI patients, may be explained by the exclusion of patients without serial creatinine levels (repeat tests are not routinely done in relatively stable patients) and the hospital admission criteria that excluded “well” patients who may have had stage 1 AKI.
Age and sex as factors associated with AKI are in keeping with previous studies by Fisher et al 10 and Hirsch et al. 11 The higher risk may be attributable to more men presenting with comorbid conditions and a higher COVID-19 disease severity compared with women. In our study, 70% of patients with diabetes and hypertension were male, whereas the men constituted 68% of patients with severe and critical COVID-19. Increasing age is accompanied by a higher incidence of chronic illnesses, which are poorly controlled in many areas of sub-Saharan Africa, Kenya included.37,38
In this study, the mortality rate for patients with AKI in COVID-19 was similar to that reported in 2 studies with similar socio-demographic characteristics,11,39 but higher than findings in an early meta-analysis that featured mainly studies from China. 40 When compared with the pre-COVID-19 era,41-43 the current rates in patients with COVID-19 have been on average 3 times higher. This higher mortality may be attributable to increased virus infectivity, a strain in the health system attributed to the COVID-19 pandemic, admission of rather sick patients who met the hospital admission criteria, and late presentation of critically ill patients to the hospital. The mortality rate was also noted to be higher with increasing severity of AKI; two thirds of the patients with stage 3 AKI had critical COVID-19 illness, a known risk factor.
Of the 4 patients who were discharged alive after dialysis, 3 required continuous renal replacement therapy (CRRT). This incidence is high compared with similar studies where only approximately 30% of patients required ongoing dialysis.44,45 That said, the hospital dialysis rates in our study were comparatively low at 3.4% of all AKI patients. Interestingly, 90% of patients who underwent dialysis had an AKI diagnosis within 48 hours of admission, which may point to a lapse in identification and early RRT in COVID-19 patients with a later AKI diagnosis, probably due to strains in the health system.
The strengths of our study include a large sample size of 1366 patients. This is also one of the first few studies from sub-Saharan Africa evaluating the impact of COVID-19 on the kidneys.
A few limitations exist. First, this study assessed the impact of COVID-19 on the kidneys in patients who were hospitalized at 1 private health facility based on pre-set admission criteria to ensure only sick high-priority patients would get admitted. In addition, 487 patients with COVID-19 infection were excluded from the analysis by the absence of serial creatinine results. Due to these 2 reasons, the study results are not generalizable. Second and importantly, most patients were admitted for the first time at our hospital during the COVID-19 pandemic, thus missing pre-admission baseline creatinine values. Due to this, the impact of chronic kidney disease/baseline creatinine values on the incidence of AKI could not be established. Finally, patients who were transferred out of the hospital to continue care at other facilities were said to be discharged alive, and the outcomes of AKI were assessed at the point of hospital transfer, which may not have been their ultimate outcome.
Conclusions
This study demonstrates the relationship between AKI and COVID-19 infection in Kenya. It assessed the AKI incidence and the association between AKI and demographic and clinical risk factors, with age, sex, hypertension, and diabetes being significantly associated with increased AKI risk in COVID-19 infection. In addition, the outcomes of AKI in COVID-19 infection were determined and noted to be more pronounced in AKI patients compared with non-AKI patients. These included a longer hospital stay, higher mortality rates, and lower renal recovery rate after dialysis.
Evaluation of the impact of early RRT on outcomes in patients with stage 3 AKI and COVID-19 infection can add to the knowledge of RRT timing. This study also forms a foundation for further research on the intermediate and long-term outcomes of COVID-19 infection on the kidneys. Information on renal recovery and progression in kidney disease is necessary to guide health policy formulation as countries across the globe shift focus to the long-term consequences of the COVID-19 pandemic.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581241227015 – Supplemental material for Associated Factors and Outcomes of Acute Kidney Injury in COVID-19 Patients in Kenya
Supplemental material, sj-docx-1-cjk-10.1177_20543581241227015 for Associated Factors and Outcomes of Acute Kidney Injury in COVID-19 Patients in Kenya by Susan Cheruiyot, Jacob Shabani, Jasmit Shah, Catherine Gathu and Ahmed Sokwala in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
This study conformed to the ethical guidelines of the 1975 Declaration of Helsinki. Consent for the study was sought from the Aga Khan University Institutional Scientific and Ethics Review Committee (2022/ISERC-01) and a research permit from the Kenya National Commission for Science Technology and Innovation (NACOSTI) before the commencement of the study (NACOSTI/P/22/15836).
Consent for Publication
Data for this study was obtained retrospectively from hospital records. Consent for conduction of the study and publication was obtained after ethical review from the Aga Khan University ISERC.
Availability of Data and Materials
The data underlying this article are available in the article and in its online supplementary material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
