Abstract
Rationale:
Imatinib is used in the treatment of Philadelphia chromosome positive (Ph+) leukemias and has been reported to have a direct effect on bone physiology.
Presentation:
To report on a child with Ph+ acute lymphoblastic leukemia who presented with bilateral flank pain and gross hematuria.
Diagnosis:
She was diagnosed with obstructive kidney stones 101 days after commencing daily oral imatinib. Stone analysis revealed the presence of calcium phosphate.
Interventions and outcome:
The patient passed the stones spontaneously with medical therapy that included the use of thiazide, allopurinol, and potassium citrate, but she required temporary insertion of a double-J stent to relieve an obstruction.
Novel findings:
Imatinib inhibits receptor tyrosine kinases and stimulates the flux of calcium from the extracellular fluid into bone, resulting in hypocalcemia with a compensatory rise in parathyroid hormone that may result in phosphaturia and the formation of calcium phosphate stones. Given that kidney stones are rare events in children, we believe that monitoring for kidney stone formation needs to be performed in children receiving imatinib.
Introduction
Imatinib mesylate (IM) is a small-molecule tyrosine kinase inhibitor that was designed to treat leukemias. 1 Many leukemias, including chronic myeloid leukemia (CML), acute lymphoblastic leukemia (ALL), and rarely acute myelogenous leukemia (AML), harbor a genetic abnormality, called the Philadelphia chromosome (Ph) in which a reciprocal chromosomal translocation arises between the Abelson leukemia virus oncogene (ABL1) on chromosome 9 and the breakpoint cluster region gene (BCR) on chromosome 22. The fusion of these genomic regions results in the production of a new protein, BCR-ABL1, which causes activation of intracellular signal transduction pathways that lead to an unstable genome, abnormal cellular proliferation, and amplification of leukemic white blood cell clones. While imatinib was specifically designed to target BCR-ABL1, it has been shown to be capable of binding to the ATP-binding site of additional receptor tyrosine kinases including c-kit and platelet-derived growth factor receptor (PDGFR), which have been implicated in the pathogenesis of gastrointestinal stromal tumors and myeloproliferative disorders, respectively.
Most adverse effects from imatinib occur within the first 2 years of therapy and are reversible after temporarily interrupting or stopping drug administration. 2 Some patients may develop skin hyper or hypopigmentation that is thought to be related to inhibition of c-kit in the skin. 2 Other side effects include hematological toxicities, bone pain, myalgias, abnormal liver enzymes, nausea and vomiting, and fluid retention. 2 In the largest pediatric study of imatinib that included 156 patients, 50% experienced significant growth impairment. 3 No kidney stones were reported. One study in adults, however, did report the development of symptomatic kidney stones in 7 of 177 patients (~4%) treated with imatinib for CML. 4
The objective of this article is to report the clinical course of a child with Ph+ ALL who received imatinib and developed renal calculi composed of calcium and phosphate.
Clinical History
Case
A 7-year-old girl with Ph+ ALL was in the consolidation phase of her treatment and receiving intrathecal methotrexate and cytarabine, vincristine, cyclophosphamide, mercaptopurine, and asparaginase along with oral imatinib at daily dose of 340 mg/m2/d. Her initial white blood cell count was 9.6 × 109 per liter and there were no biochemical markers of tumor lysis syndrome. She had no previous history of kidney stones. Ninety days after starting imatinib, she presented with fever, abdominal pain, vomiting, and diarrhea. On examination, she was febrile, tachycardic, and hypotensive. There was epigastric and right lower quadrant tenderness with decreased bowel sounds. Abdominal computed tomography (CT) scan was suggestive of typhlitis (pancolitis). Importantly, there was no evidence of kidney stones or hydronephrosis on the CT scan. She was managed in the intensive care unit due to septic shock (Enterobacter cloacae bacteremia) secondary to the bowel inflammation. There was no evidence of urosepsis. She was treated with IV meropenem, tobramycin, and vancomycin. Imatinib therapy was held on admission to the intensive care unit, and she was found to have a low serum phosphate level at 0.67 mmol/L, with a normal total calcium level of 2.1 mmol/L. Her serum creatinine was 35 µmol/L, which gave her an estimated glomerular filtration rate (GFR) using the Schwartz equation of 139 mL/min/1.73 m2, consistent with normal renal function.5 Blood parathyroid hormone and Vitamin D levels and urine electrolytes were not assessed at the time hypophosphatemia was noted. Due to excessive fluid accumulation, she was treated with daily intravenous furosemide. Eleven days after her admission to the intensive care unit, which was 101 days after starting imatinib, she developed bilateral flank pain and gross hematuria. There was no family history of renal stones.
Abdominal ultrasound showed left-sided grade 2 hydronephrosis with obstructive stones measuring 12 mm × 6 mm within the left renal pelvis (Figure 1A). The right kidney displayed 2 large non-obstructive calyceal stones measuring 18 mm × 5 mm and 12 mm × 6 mm, respectively (Figure 1B). Because of the large size of the stones, a double-J stent was inserted in the left kidney. A metabolic stone workup was performed 6 days after the development of flank pain while she was off imatinib and revealed normal serum phosphate of 1.24 mmol/L, normal total calcium of 2.33 mmol/L, and normal bicarbonate of 24.8 mmol/L. Spot urine analysis revealed hypercalciuria (urine calcium/creatinine ratio: 3.5 mmol/mmol), hyperuricosuria (urine uric acid/creatinine ratio: 0.77 mmol/mmol), normal urinary citrate (urine citrate/creatinine ratio: 0.15 mmol/mmol), and urine pH 6.5. The girl was managed with hydrochlorothiazide (1.85 mg/kg/d), fluid therapy, and analgesia. Due to the presence of hyperuricosuria, potassium citrate (1.85 mEq/kg/d) and allopurinol (10 mg/kg/d) were added to the management. She passed stone fragments 16 days after development of flank pain, and when analyzed using infrared spectrophotometry, they were composed of calcium and phosphate. Four weeks after the development of the renal stones and 10 days after resuming imatinib, her parathyroid hormone (PTH) level was measured and found to be suppressed at 0.9 pmol/L with a normal 25-hydroxy Vitamin D (25(OH)D) level at 75 nmol/L. Forty-five days after presentation with renal colic, a CT scan of the abdomen and pelvis confirmed that her stones had disappeared.
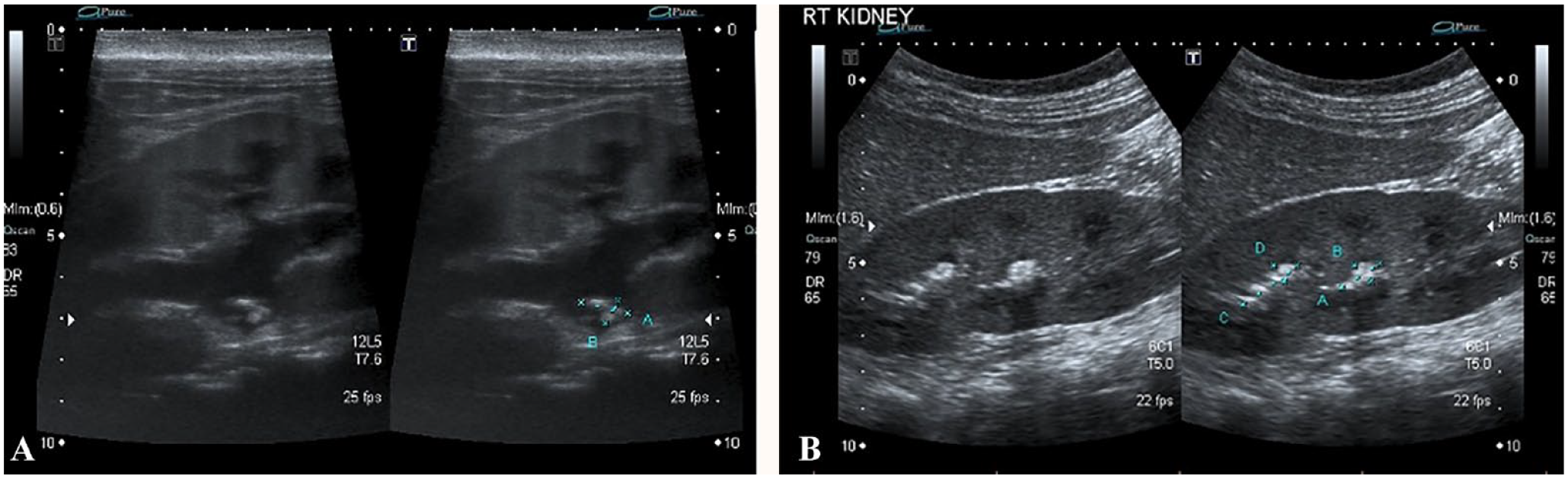
(A) Split-screen ultrasound (US) image shows left grade 2 hydronephrosis with a cluster of obstructive stones. (B) Split-screen US image shows stone burden at the mid- and upper poles of the right kidney. The stones themselves appear echogenic with posterior acoustic shadowing, more apparent at the mid pole stone.
Discussion
Kidney stones are relatively rare events in children with the incidence varying from 12 to 18 per 100 000 children depending on the study and the geographical region sampled. 6 We report on a child who developed calcium phosphate kidney stones 101 days after starting oral imatinib. Imatinib use was associated with a low serum phosphate.
Saikia et al reported an association between the development of renal calculi and imatinib therapy in adult patients with CML. In their observational study, 7 out of 177 adult patients with CML developed renal calculi as early as 1 month after treatment with imatinib. Analysis of renal stone composition was reported in only 1 patient and revealed the presence of calcium phosphate and oxalate crystals. 4 No systematic screening for metabolic abnormalities in blood levels of phosphate or calcium was performed.
Berman et al noted hypophosphatemia in some of their adult patients with CML that were treated with imatinib. 7 They observed hypophosphatemia in 51% (25/49) of patients between 9 and 132 days after treatment with imatinib. The patients with a low serum phosphate, defined as less than 0.6 mmol/L, were younger (median age 50 vs 63 years), they received a higher dose of imatinib (median daily dose 600 vs 350 mg), and they had higher PTH levels (median 84 vs 42 pg/mL, 8.9 vs 4.4 pmol/L) than the normal-phosphate group, defined as serum phosphate level greater than 0.8 mmol/L. Importantly, both the low-phosphate and normal-phosphate patients had elevated levels of urinary phosphate excretion when compared with the control group of healthy adult volunteers that did not receive imatinib. 7 The patients with hypophosphatemia also had significantly lower serum calcium levels compared with the control group. During a limited follow-up period of 15 months, there were no reported renal stones or bone fractures. Ultrasound imaging of the kidneys was not reported. Similarly, Saglio et al reported on adverse effects during 12 months of imatinib in 280 adults with CML. Overall, 45% of them had hypophosphatemia (serum phosphate less than 0.8 mmol/L) and 8% had grade 3 or 4 hypophosphatemia (defined as serum phosphate less than 0.6 mmol/L). 8 Kantarjian et al reported on a similar number of adults who received imatinib versus dasatanib for CML: they observed that the imatinib-treated group had a 21% incidence of grade 3 or 4 hypophosphatemia versus 4% in the dasatinib-treated group. 9 Pediatric studies of imatinib have described hypophosphatemia in 4.6% to 18% of children.10,11 Severe hypophosphatemia, grade 3 or 4 hypophosphatemia, was observed in 0% to 2.5%.
The hypophosphatemia reported from the use of imatinib could be related to its effects on bone physiology. In a pediatric study, 7 of 32 (22%) children who received imatinib for greater than 12 months had significant impairment in longitudinal growth and 6 of 32 had decreased bone mineral density. 12 Another pediatric study reported growth delay in 51 of 108 children who received imatinib for at least 12 months. 3 In adult studies, the effect of imatinib on bone health appears to be favorable. Jonsson et al reported that imatinib-treated adults (n = 17) had no signs of osteoporosis or osteomalacia: indeed, they had higher volumetric bone mineral density compared with controls. 13 Similar results were reported by Fitter et al who found that 8 of 17 imatinib-treated adults had increased trabecular bone volume from bone biopsies that exceeded the upper limit of normal for their age. 14 O’Sullivan et al reported on 9 adults who received imatinib over a 2-year period. Despite sustained mild secondary hyperparathyroidism, bone mineral density was stable or increased over the observation period. 15
Imatinib has short- and long-term effects on bone physiology. During the early stages of therapy, imatinib directly stimulates bone formation through its effects on osteoblasts, 2 resulting in a net flux of calcium from the extracellular fluid compartment into bone, which leads to a decreased serum calcium level, and a compensatory rise in PTH. Hyperparathyroidism results in phosphaturia and hypophosphatemia. The long-term effects of IM therapy on bone remodeling have been studied by Fitter et al and appear to result in suppression of both bone formation and resorption. 14 They proposed that imatinib decreases bone resorption by inhibiting osteoclasts. Osteoclasts are dependent on macrophage colony-stimulating factor (M-CSF), which binds to its receptor c-Fms to promote bone differentiation. Imatinib binds to the kinase domain of c-Fms and inhibits both osteoclast differentiation and function.16,17 The effects of imatinib on bone physiology are complex because it binds to several receptor tyrosine kinases (c-Kit, PDGF-R, c-Fms, and c-Abl). Indeed, the proliferation and differentiation of osteoblasts is stimulated by PDGF-R and c-Abl. Taken together, imatinib appears to have a biphasic effect on bone physiology that merits further study in the growing skeleton of children.
The effects of imatinib are not only limited to the bone. Francois et al reported a case of partial Fanconi syndrome induced by imatinib in a patient with CML. 18 They noted acute kidney injury with proximal tubular dysfunction including increased urinary excretion of phosphate, uric acid, and glucose which were associated with hypophosphatemia, hypouricemia, and normoglycemia, respectively. However, serum bicarbonate and urinary excretion of amino acids were within normal ranges and thus they defined the tubular disease as a partial Fanconi syndrome. Interestingly, the PTH level was normal which suggests that hypophosphatemia was caused by primary renal phosphate wasting rather than secondary to hyperparathyroidism. The exact mechanism by which imatinib causes acute kidney injury and proximal tubular dysfunction is not well understood. In patients with acute tubular necrosis, proliferation and regeneration of proximal tubular cells depends on PDGF signaling with activation of PDGFRß.18,19 In addition to PDGFRß, c-kit is also expressed in the proximal tubule. Both tyrosine kinase receptors are targets of imatinib and together may predispose to proximal tubular injury. 19
The case reported here suggests that there may be an association between imatinib and the development of renal calculi in children. Alterations in bone and mineral metabolism and/or renal tubular leak secondary to the use of imatinib can cause changes in the serum levels of calcium, phosphate, and PTH and the development of calcium phosphate renal stones. It is important to perform monitoring of serum levels of calcium, phosphate, PTH, and urinary phosphate and calcium excretion during imatinib therapy to identify any abnormalities and to intervene accordingly. Medical therapy should be attempted to manage stone disease from imatinib given the fact these patients are frequently thrombocytopenic and at risk for bleeding with any surgical intervention to remove stones. Similarly, many of these patients are neutropenic and thus have a higher risk for infection post-procedure.
Footnotes
Ethics Approval and Consent to Participate
The patient and their parents provided consent to publish this anonymized case report, and this was approved for submission by our local research ethics board.
Consent for Publication
Consent for Publication was obtained from the family.
Availability of Data and Materials
Availability of Data and Materials: Not applicable for this case report.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
