Abstract
Purpose of review:
Glomerulonephritis refers to a rare group of diseases characterized by glomerular inflammation, which collectively are a common cause of kidney failure. Until recently, there was a lack of high-quality clinical trials to inform the care of patients with glomerulonephritides. We identified examples of successful translational research spanning from basic science to clinical applications, and highlight gaps in implementation science.
Sources of information:
The focus of our review was derived from discussions between health care professionals, researchers, and patient partners. We also performed literature searches pertaining to the treatment of glomerulonephritis in PubMed and Google Scholar.
Methods:
Examples of successful knowledge translation were generated through review of new evidence in the past 5 years and by iterative discussions by the authors. We then conducted a narrative review of several themes related to knowledge translation in glomerulonephritis. This was complemented by an interview with a patient partner to provide an example of a patient’s perspective living with glomerulonephritis.
Key findings:
We summarized selected recent advances in glomerulonephritis and its knowledge translation in the following domains: (1) identification of auto-antibodies in membranous nephropathy and minimal change disease; (2) clinical trials of novel targeted therapies for IgA nephropathy and lupus nephritis, which have led to approval of new treatments; (3) developments in research networks and clinical trials in glomerulonephritis; (4) recognition of the importance in developing standardized patient reported outcome measures in clinical trials; and (5) barriers in knowledge translation including access to medication.
Limitations:
A systematic search of the literature and formal assessment of quality of evidence were beyond the scope of this review.
Introduction
Glomerulonephritis (GN) is a group of rare kidney diseases that collectively account for 10% of incident cases of end-stage kidney disease (ESKD).1,2 Impressive advances have occurred in the field of GN spanning from basic sciences to novel therapies resulting in the revision of the KDIGO guidelines for GN in 2021. 3 Knowledge translation (KT) refers to processes facilitating synthesis, sharing and application into clinical use or health decision-making. 4 In this review, we discuss examples of successful translational research spanning from biomarkers, novel therapies, clinical trials, and the call to incorporate patient-reported outcome measures (PROMs) as key outcomes in trials for patients living with GN. We also highlight a patient partner’s perspective as she navigated through the diagnosis, treatment of GN, and participation in research to advance GN care.
A Patient’s Story
Kate has been living with IgA nephropathy (IgAN) for more than 12 years and kindly shared her perspective on GN clinical research and care. Despite being involved in the kidney community and research studies for over 10 years, surprisingly she was not aware until recently that IgA nephropathy was considered a type of GN.
Diagnosis of Glomerulonephritis and Novel Biomarkers and Antibodies
Challenges may occur at multiple stages in the care of patients living with GN, which may begin with delays in referral to nephrology, precipitating potential downstream consequences. 5 This was the case of our patient partner Kate who stated that there were some “hiccups” in her journey including being diagnosed quite late with IgAN at which point she had advanced chronic kidney disease (CKD). In a cross-sectional study from Ontario, 40% of patients living with GN reported delayed diagnosis and 45% were diagnosed after progression to advanced kidney disease nearing ESKD. 5 In particular, given the protean manifestations of GN, obtaining a diagnosis can be challenging, and the discovery of novel biomarkers or antibodies may serve as alternatives to kidney biopsy in some circumstances and may support the diagnosis of specific GNs such as membranous nephropathy.
Discovery of Antigens Responsible for Membranous Nephropathy
Membranous nephropathy (MN) is among the most common causes of nephrotic syndrome in adults. 6 While MN was first identified as an autoimmune disease in 1959, 7 it was not until 2009 that M-type phospholipase A2 receptor 1 (PLA2R) antibodies were identified to be causative in 70% of primary MN. 8 In their landmark study, Beck and colleagues 8 demonstrated PLA2R positivity in 70% of individuals with primary MN, and 58% of all patients with MN in this study. This has translated into clinical practice where PLA2R antibodies can now be measured routinely.
While a kidney biopsy is the gold standard for diagnosis of GN, and the rate of kidney biopsies increased from 10.8 to 18.2 per 100,000 person years from 1985 to 2014 in Alberta, Canada, with glomerular diseases being the most common diagnosis, 9 they are not without risks. 10 The discovery of markers such as the PLA2R antibody in MN offers an alternative approach to diagnosis. Positive PLA2R serology in some cases may obviate the need for a kidney biopsy for diagnosis. 11
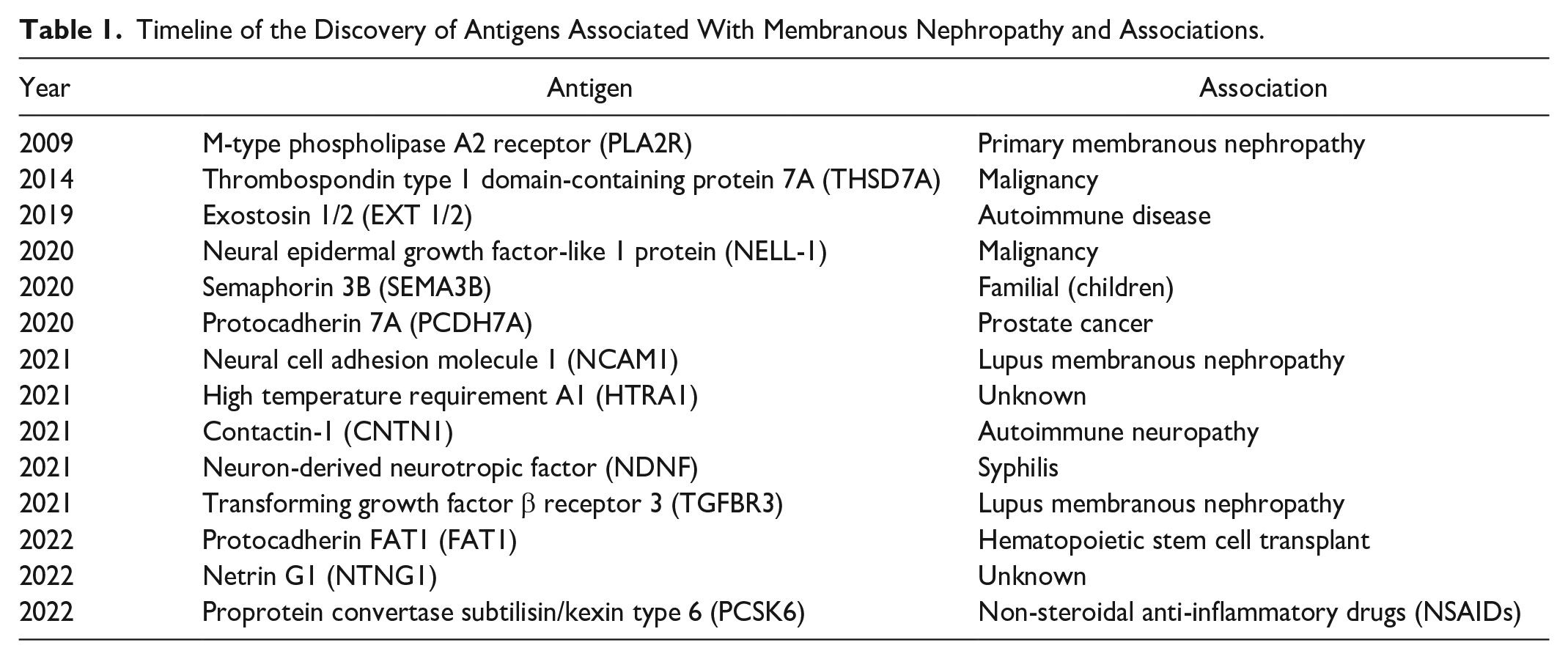
This was followed by the discovery of thrombospondin type 1 domain-containing 7A (THSD7A) as a second antigen causing MN in 2014. 12 Utilizing advances in laser microdissection and mass spectrometry, new antigens have been discovered at a rapid pace with specific disease associations including exostosin 1/exostosin 2 (EXT1/2; autoimmune disease), 13 neural epidermal growth factor-like-1 (NELL-1; associated with malignancy), 14 protocadherin FAT1 (FAT1; hematopoietic stem cell transplant), and neuron-derived neurotropic factor (NDNF; syphilis), contactin-1 (CNTN; polyneuropathy), proprotein convertase subtilisin/kexin type 6 (PCSK6; NSAID usage), semaphorin-3B (familial), 15 and protocadherin 7a (PCDH7), 16 which are expanding our understanding and classification of different subtypes of MN (Table 1). With the expanding number of antigens discovered, less than 10% of cases of MN still do not have an identifiable causative antibody.
Timeline of the Discovery of Antigens Associated With Membranous Nephropathy and Associations.
Anti-Nephrin Antibodies in Minimal Change Disease
Minimal change disease (MCD) is the most common cause of nephrotic syndrome in children.17,18 Despite corticosteroids classically resulting in rapid resolution of MCD suggesting an autoimmune etiology, the underlying cause of MCD remained elusive for decades. 19 However, the discovery of novel antibodies against nephrin, a component of the slit diaphragm, in 29% of adults and children with MCD using samples from the NEPTUNE cohort has advanced our understanding of the pathogenesis of MCD. 20 While the role of nephrin in MCD remains to be further investigated, this finding may have future clinical applications analogous to the discovery of PLA2R wherein anti-nephrin antibodies could be used for diagnosis, monitoring response to therapy, and potentially aid in the development of targeted therapies for MCD.
Emerging Therapies in GN
Patients living with GN may consider entering clinical trials after exhausting multiple lines of immunosuppressive therapy. Improved understanding of the pathogenesis of glomerular disease has led to a proliferation of clinical trials evaluating promising therapies for GN. While new therapies have become available for a number of glomerulonephritides, we have focused on Immunoglobulin A nephropathy (IgAN) and lupus nephritis (LN) due to the remarkable success of recent clinical trials for these diseases leading to the approval of new treatments for these conditions.
IgA Nephropathy
IgAN is the most common GN worldwide. 21 The pathogenesis of IgAN is complex but can be summarized by the 4-hit hypothesis (Figure 1). 22 Supportive care consisting of renin-angiotensin-aldosterone system inhibition (RAASi), blood pressure optimization, and lifestyle modification have been the backbone of management of IgAN. 23 Given the autoimmune etiology of the disease, patients at high risk of disease progression, defined as proteinuria >1g/day despite supportive therapy have traditionally been treated with immunosuppression including corticosteroids, a therapy which remains controversial. 24 Our patient partner Kate was initiated on a brief course of corticosteroids prior to her diagnosis of IgAN but this was discontinued following her kidney biopsy, which demonstrated advanced glomerulosclerosis that precluded significant therapeutic benefit from corticosteroids. The STOP-IgAN study found that the addition of steroid therapy to supportive care did not attenuate estimated glomerular filtration rate (eGFR) decline. 25 Moreover, the TESTING trial was terminated early due to risk for serious infection in participants who received corticosteroid therapy. 26 Thus, there is a keen interest to expand the armamentarium of IgAN-specific therapies. Our improved understanding of the pathophysiology of IgAN has led to the development of novel therapies which target different points in the disease. We will discuss the recent advances in gut-directed therapy with local steroid delivery to the Peyer’s patches, endothelin receptor antagonism, complement inhibitors and other novel therapies that target various steps in the pathway leading to glomerular and tubulointerstitial injury in IgAN.

Pathogenesis of IgA nephropathy and targets of novel therapies.
Gut-directed therapy
Galactose-deficient IgA1 (GdIgA1) immune complex deposition in the mesangium incites glomerular and tubulointerstitial injury, precipitating kidney impairment. 21 The Peyer’s patch, located in the ileum approximating the ileocecal junction, contains aggregates of lymphoid follicles with the largest concentration of mucosal B cells that produce Gd-IgA1 that underlies the gut-kidney axis.21,27,28 Nefecon is a gut-directed budesonide formulation designed for targeted-release on the Peyer’s patches where it is hypothesized to suppress Gd-IgA1 synthesis through more targeted delivery of corticosteroid.27,29 This an attractive alternative to traditional systemic corticosteroids such as prednisone or methylprednisolone, as the local delivery mitigates systemic adverse effects due to the first-pass metabolism of gut-directed steroids. 21
Efficacy and Safety of Nefecon in Patients With Primary IgA Nephropathy (NefIgArd) was a phase 3 randomized controlled trial (RCT) that compared Nefecon to placebo in adults with biopsy proven primary IgAN and optimized RAASi.
27
After 9 months of therapy, the Nefecon group experienced a 27% reduction in proteinuria compared with placebo group (
Endothelin receptor antagonists
Endothelin within the kidney results in cell proliferation and hypertrophy which has been implicated in CKD progression, fibrosis, and proteinuria. Endothelin receptor antagonists (ERAs) have been shown to promote regression of glomerulosclerosis and attenuate albuminuria in experimental models of CKD. 31 Therefore, 2 ERAs are currently under investigation to assess their efficacy in IgAN.
Sparsentan blocks the endothelin type A (ETA) receptor and angiotensin II type 1 receptor.
29
On February 17, 2023, sparsentan received accelerated approval from the U.S. FDA as the first non-immunosuppressive IgAN therapy based on interim results of the PROTECT trial which demonstrated a 49.8% reduction in proteinuria amongst patients receiving sparsantan compared with 15.1% reduction in the comparison group (
Summary of Ongoing Clinical Trials of Targeted Therapies for IgA Nephropathy.

Complement pathway inhibitors
Given the involvement of the alternative and lectin complement systems in the pathogenesis of IgAN as depicted in Figure 1 (Hit 4), evidenced by C3 deposition in the mesangium,40-42 monoclonal antibodies have been developed to target these pathways. Narsoplimab is a MASP-2 inhibitor that acts on the lectin binding pathway to inhibit activation of C3 convertase, 43 while iptacopan is a small molecule inhibitor of factor B, preventing C3 convertase formation, thereby inhibiting alternative complement activation. 29 The efficacy and safety of iptacopan in adult patients with IgAN are being evaluated in APPLAUSE-IgAN 37 and narsoplimab in ARTEMIS-IgAN. 36
BAFF and APRIL inhibitors
The cytokines B-cell activating factor (BAFF) and a proliferation-inducing ligand (APRIL) have been implicated in the initiation formation of GdIgA1 (Figure 1, Hit 1). 21 BAFF and APRIL elevations are associated with kidney dysfunction and proteinuria.44-46 New therapies directed at these pathways including blisibimod (BAFF inhibitor) and sibeprenlimab (monoclonal antibody against APRIL) are currently under evaluation in phase 1 to 3 trials (Table 2).
Lupus Nephritis
Lupus nephritis (LN) is one of the most severe end-organ complications of systemic lupus erythematosus (SLE) and occurs in 50% of patients. 47 After a long history of negative clinical trials in LN, the fortunes have reversed with the recent U.S. FDA approval of belimumab and voclosporin for LN management. We have focused on these 2 new agents in this review. Other promising therapies are also under evaluation (Table 3), although there is heterogeneity in the outcome measures used across trials. 48
Selected Summary of Clinical Trials for Lupus Nephritis.
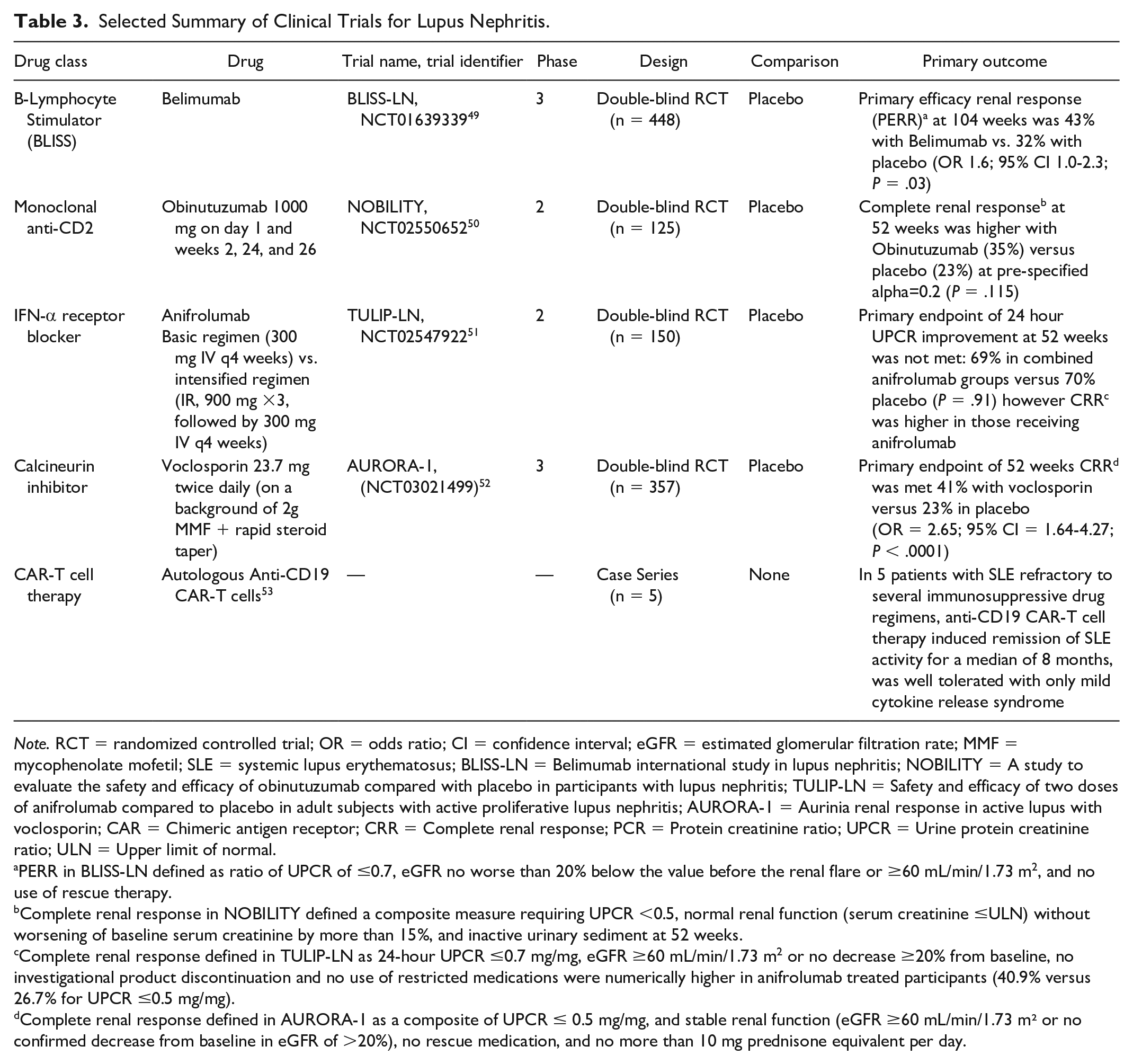
PERR in BLISS-LN defined as ratio of UPCR of ≤0.7, eGFR no worse than 20% below the value before the renal flare or ≥60 mL/min/1.73 m2, and no use of rescue therapy.
Complete renal response in NOBILITY defined a composite measure requiring UPCR <0.5, normal renal function (serum creatinine ≤ULN) without worsening of baseline serum creatinine by more than 15%, and inactive urinary sediment at 52 weeks.
Complete renal response defined in TULIP-LN as 24-hour UPCR ≤0.7 mg/mg, eGFR ≥60 mL/min/1.73 m2 or no decrease ≥20% from baseline, no investigational product discontinuation and no use of restricted medications were numerically higher in anifrolumab treated participants (40.9% versus 26.7% for UPCR ≤0.5 mg/mg).
Complete renal response defined in AURORA-1 as a composite of UPCR ≤ 0.5 mg/mg, and stable renal function (eGFR ≥60 mL/min/1.73 m² or no confirmed decrease from baseline in eGFR of >20%), no rescue medication, and no more than 10 mg prednisone equivalent per day.
Belimumab
B-cells are involved in the pathogenesis of LN thereby representing a therapeutic drug target. This understanding led to the development of belimumab, which is a human monoclonal IgG1-λ antibody that inhibits soluble B-cell activating factor (BAFF, also referred to as B-lymphocyte stimulator, BLyS) and B-cell survival. While prior trials demonstrating efficacy of this agent in SLE excluded patients with LN, the BLISS-LN trial was a 2-year phase 3 multinational, multicenter, randomized, double-blind, placebo-controlled RCT in 448 patients with lupus nephritis.49,54,55 In this trial, more patients who received belimumab had a primary efficacy renal response than those who received standard therapy alone (43% vs. 32%; odds ratio, 1.6 (95% confidence interval [CI]: 1.0-2.3;
Voclosporin
Calcineurin inhibitors are known to have direct anti-proteinuric effects by stabilization of podocytes, but also reduce T-cell activation. Voclosporin is a novel calcineurin inhibitor which does not require therapeutic level monitoring which was evaluated in AURORA-1. In this trial, 357 patients with LN, where voclosporin was added on a background of a rapid steroid taper and mycophenolate mofetil (MMF) therapy.
52
The primary endpoint was complete renal response at 52 weeks which was met with 41% receiving voclosporin versus 23% in placebo obtaining a complete renal response (odds ratio [OR] = 2.65; 95% CI: 1.64-4.27;
Research in GN: Challenges and Progress
Research Networks
In 2010, a review bemoaned the dearth of high-quality clinical trials for patients with glomerular disease. Reasons for this disparity included the rarity of GN, varied clinical presentations, heterogeneity in clinical endpoints, challenges in patient recruitment, lack of collaborative efforts, and high costs of RCTs. 59
Recognition of the need to invest in medical research in rare diseases led to the creation of the Vasculitis Clinical Research Consortium (VCRC) and the Nephrotic Syndrome Study Network (NEPTUNE). The VCRC is a patient registry which includes individuals who self-identify as having vasculitis 60 while NEPTUNE is a North American multicenter consortium which has created infrastructure for translational research in patients with nephrotic syndrome (www.neptune-study.org) including a registry of patients living with GN with detailed clinical, molecular, histologic information, and patient-reported outcomes (PROs). 61 Furthermore, the Canadian Glomerulonephritis Registry (CGNR) has established a national patient-centered, multidimensional web-based clinical database and federated virtual biobank. 62 The BC GN Network and Registry leverages existing health infrastructure to capture population level data on all patients with GN in the province which had 3473 GN cases from 2000 to 2012. 63 Together these networks have resulted in the infrastructure and connections necessary to conduct high quality research in GN. Our patient partner Kate also expressed her interest in participating research as she has been involved in research studies throughout her care journey. She did note that in some clinical trials she felt like just a subject for data. She emphasized the value of communicating effectively with patients in research, stating: “being able to know exactly how the data is going to help impact their disease or care, they [the research participants] may feel more involved.”
Reducing Corticosteroid Exposure
Adverse effects related to corticosteroids including weight gain, fluid retention, deleterious effects on mood, and infection risk are outcomes highly prioritized by patients living with glomerular disease. 64 Therefore, there has been an interest in determining whether steroid dosing can be minimized or reduced in a number of types of GN including IgAN, LN, and anti-neutrophilic cytoplasmic antibody (ANCA) vasculitis. An example of success from recent clinical trials has addressed this patient-centered issue by examining reducing corticosteroid dosage in several types of GN.
AURORA-1 and -2 demonstrated the efficacy of voclosporin in inducing complete renal response while reducing cumulative steroid exposure in patients with LN.52,57 In IgAN, the recent TESTING trial demonstrated that reduced-dose methylprednisolone 0.4 mg/kg/day (previously 0.6-0.8 mg/kg/day), reduced the primary composite kidney outcome by 47% (hazard ratio [HR]: 0.53, 95% CI: 0.39-0.72). 65 The incidence of serious infections also diminished with corticosteroid dose reduction and addition of pneumocystis pneumonia prophylaxis. 65 Furthermore, the MAIN study demonstrated that MMF may be a steroid sparing strategy in IgAN, as MMF significantly reduced the composite kidney outcome of doubling of serum creatinine, kidney replacement therapy, or mortality compared with supportive care (HR: 0.23, 95% CI: 0.09-0.63). 66 Among patients with severe ANCA vasculitis, the PEXIVAS (Plasma Exchange and Glucocorticoids for Treatment of Anti-Neutrophil Cytoplasm Antibody - Associated Vasculitis) trial showed that a rapid tapering and reduced-dose steroid regimen was non-inferior to the usual dose steroid strategy. 67 This regimen resulted in approximately 50% reduction in steroid exposure and associated with lower incidence of serious infections (incidence rate ratio 0.69, 95% CI: 0.52-0.93). 67 The results of this study precipitated the subsequent guideline change to adopt this steroid tapering regimen. 3 The ADVOCATE (A Phase 3 Clinical Trial of CCX168 [Avacopan] in Patients With ANCA-Associated Vasculitis) study demonstrated the efficacy of avacopan, a C5a receptor inhibitor, in sustained disease remission compared with a steroid taper. 68 The success of ADVOCATE was incorporated into the KDIGO 2023 ANCA Guideline Update as an alternative therapy to glucocorticoids in the induction regimen. 69 Collectively, these studies provide evidence to reduce the dosage of steroids in IgAN, LN, and ANCA vasculitis, which is a welcome finding to both patients and clinicians.
Surrogate Outcomes Measures
A key advancement promoting clinical trials in GN has been the establishment of approved surrogate outcomes acceptable by the U.S. FDA for drug approval following a workshop between the National Kidney Foundation in Collaboration with the U.S. FDA and European Medicines Agency. At this workshop, newer proposed surrogates included albuminuria reduction of a geometric mean of 30% within 6 months or attenuation of eGFR slope of 0.5-1.0 mL/min/1.73 m2 over 2-3 years. 70 The Kidney Health Initiative established a work group to identify appropriate surrogate endpoints for glomerular disease including IgAN, MN, and Focal segmental glomerulosclerosis (FSGS) patients in clinical trials. This has resulted in the reduction in proteinuria being accepted as surrogate endpoints for IgAN and MN as opposed to the traditionally accepted outcome of 50% reduction in eGFR or progression to ESKD. Use of these surrogate outcomes measures will shorten trial duration and cost. Accelerated drug approval is now possible using these surrogate endpoints, however, benefit must be verified in postmarketing trials.71,72
While validated surrogate outcomes measures may facilitate the conduct of clinical trials, it is crucial that trials continue to be designed to rigorously evaluate hard outcome measures including kidney function, mortality, and need for renal replacement therapy which are the outcomes which ultimately matter most to patients. Thus, even drugs that have already been approved by the U.S. FDA or Health Canada based upon data from surrogate measures should be subjected to longer and large RCTs that definitively address the clinical benefit of new therapies for GN.
Barriers to Knowledge Translation
While there is much to celebrate given the recent success in clinical trials for GN, there remain barriers to KT. For example, although the U.S. FDA has approved therapies such as neficon, sparsentan, belimumab, and voclosporin, these agents are not readily accessible in Canada. Furthermore, the approval of multiple disease-specific targeted therapies in GN highlights the need for personalized medicine to determine which therapy is appropriate for a particular patient.
A GN needs assessment conducted in Ontario, revealed that there are current gaps in the care of patients living with GN: in particular tools to support patients to make informed decisions regarding treatment choices are lacking; there is lack of standardization of GN protocols and resources available to patients, and variable data collection to inform research and health policy. Furthermore, patients access to appropriate medication is also variable. 5
Kate, our patient partner, expressed that she was fortunate to have private insurance which paid for medications not covered by the government. However, cost of medications is a substantial barrier, as patients living with GN are frequently young and may not have drug coverage. One solution for drug access that has been employed by the British Columbia, as part of BC Renal, an oversight organization, that has supported the development of a provincial GN drug formulary which improves patient and physician access to evidence-based immunosuppressive treatment in a cost-effective manner. This model which could potentially be extended to other jurisdictions. 73 Even with improved access, new agents will force us to confront cost-effectiveness, as for example belimumab costs $43,000 USD per patient per year while voclosporin costs $92,000 USD per patient per year. Thus, despite success in clinical trials, voclosporin may not penetrate into clinical practice as clinicians may opt to use tacrolimus which is familiar from use in kidney transplantation, readily available, and far less costly. 74
Additional barriers exist which prevent new therapies and tests from reaching the bedside. Publication of a study may not penetrate into clinical practice without implementation science efforts. For example, a survey of nephrologists demonstrated that 2 years after the publication of the 2012 KDIGO GN guidelines, 15% to 46% of Canadian nephrologists reported treatment strategies not in keeping with guideline recommendations. 75 Potential strategies that may provide more effective knowledge transfer have been suggested including computerized physician order entry, multimodal/faceted interventions, disease management pathways, real-time clinic reminders, and feedback in comparison to peers. 76 The standardization of care through specialized GN clinics, which has been adopted in Ontario, may serve as a possible solution to ensure that appropriate guideline-directed care is delivered. Evidence from other fields suggest that improvement in implementation can be achieved using specific benchmarks, for example, targeted vaccination rates. Some of this work is already underway with a goal of establishing minimum GN clinic requirements and instituting performance indicators to improve care. Examples of multifaceted educational interventions have been attempted in the dialysis population through cluster-randomization in Canada. 77 Similar strategies could potentially be used to rigorously explore whether such intervention may improve knowledge transfer and clinical outcomes and PROMS in patients living with GN.
Patient-Reported Outcomes (PROMS)
Another emerging priority in CKD and GN care is the inclusion of PROMs in routine clinical care which refer to “measurement tools that patients use to provide information on aspects of their health status that are relevant to their quality of life, including symptoms, functionality, and physical, mental and social health.” 78 The benefits of using PROMs include timely identification of gaps in care, improved patient well-being, and optimized clinical outcomes.79,80 While PROs have not historically been emphasized as key health outcomes, there have been recent efforts to prioritize PROs more highly in medical decision-making and research, which has spurred movements within nephrology to identify and monitor PROs that are important to patients. For example, the Standardized Outcomes in Nephrology (SONG) initiative has engaged patients, families, and clinical stakeholders with GN internationally and has revealed life participation, fatigue, and anxiety to be top-priority health outcomes of this population. 81 A number of initiatives are also addressing barriers, such as patient burden, workflow issues, technical constraints, and a lack of validated measures,82,83 to implementing PROMs in routine CKD care. For example, SONG is creating tailored PROMs developed with patient stakeholders that will have established validity, reliability, and clinical utility for people with CKD. 84 CureGN is a multicenter consortium which has established a database of patients with glomerular disease with MCD, FSGS, membranous nephropathy, or IgA nephropathy.
The CureGN initiative has introduced the possibility of using computer-adaptive testing (CAT) from the Patient Reported Outcomes and Measurement and Information System (PROMIS) in GN care, 85 which would allow for rapid assessment and uniform quality-of-life data that can be compared across chronic diseases. Several other initiatives within CKD are focused on electronic PROMs (ePROMS) to improve the efficiency, scalability, and integration potential of PROMs in clinical practice.86-88
In addition to recognizing and documenting patient concerns systematically, routine use of PROMs in GN care should provide an impetus for developing, testing, and implementing treatments that target PROs directly. 89 PROs such as life participation, symptoms, and mental health are complex health outcomes that call for a holistic approach to care, addressing the roles of biological, psychosocial, and/or environmental determinants. People with GN recognize holistic care as being important—in Ontario, 84% of people with GN ranked access to specialized, multidisciplinary care as very important or important, while 81% ranked education, counseling and support for patients and families as being very important or important. 5 These findings informed the development of a government-sponsored program in Ontario, that includes 6 GN specialty clinics fully staffed by nurses, pharmacists, social workers, pathologists, and nephrologists. While specialized clinics can enhance the level of support available for people with GN, there remains a lack of research or guidelines in GN to inform interventions that directly target life participation, fatigue, or other priority PROs. A secondary analysis of the CureGN data on 478 children and 1115 adults with GN identified edema as an independent risk factor for poor quality of life in GN, and found other factors such as immunosuppressive therapy, disease duration, and primary diagnosis were not significant. 90 However, studies that have investigated psychosocial or environmental mediators of quality of life and other PROs in GN remain lacking. Intervention studies targeting quality of life or other PROs are even more scarce; literature from other CKD and chronic disease populations on approaches such as exercise training, 91 psychotherapy, 92 and self-management education, 93 currently provide the best evidence to inform interventions in GN. However, there remains a need for greater research to understand PROs in the GN population specifically, so that routine PRO measurement can be paired with a nuanced and evidence-informed approach to treatment.
A Patient Partner’s Perspective Living With GN
Our patient patient partner Kate emphasized the importance of communication, compassion, and understanding in GN care. While she recognized that “healthcare workers are busy and not every patient is a self-advocate or knows to ask questions, educating and communicating hopefully helps both groups.” Kate did not receive immunosuppressive medications for her IgAN due to the late diagnosis but underwent pre-emptive living donor kidney transplantation 6 years following her GN diagnosis. She identified instances where communication could have been improved on her healthcare journey; for example, she was required to act as a bridge of communication between her CKD and transplant teams after not being monitored closely enough following a medication change which led to a decline in her kidney function.
While Kate has been quite involved in her own care and knew where to look for information she noted that “many people do not.” She emphasized that educating people about their disease, options for care, medication and peer engagement were important as many people do not know how to navigate the health system. She expressed that “patients have the lived experience that can help health care workers understand and help.”
Conclusion
In conclusion, exciting progress has been made in the field of GN spanning from basic sciences to new therapies. This review focuses on several examples of successes in research translation, and highlights some ongoing challenges in patient-centered care and knowledge translation. There is much work that lies ahead to ensure that new knowledge reaches clinical practice, and is effectively implemented. Through articulating the current state, and calling out those barriers, we hope that the Canadian GN community will be reinvigorated to collaborate on national and international efforts. Knowledge translation efforts using implementation science will enable uptake of new discoveries, but also ensure that proven therapies are accessible to all. Embracing the spectrum of bench to bedside, ensures that ultimately we address and achieve outcomes that matter most to patients.
Footnotes
Acknowledgements
The authors would like to thank our patient partner Kate for sharing their journey living with glomerulonephritis and the KRESCENT leadership committee for the valuable feedback and guidance. Figures were created using Biorender.com.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
All authors read and approved the final version of this manuscript.
Availability of Data and Materials
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: K.Y., C.W., R.A.B., and J.F.F. are funded by the Kidney Research Scientist Core Education and National Training (KRESCENT) Program through Post-Doctoral Fellowships (K.Y. and C.W.) and New Investigator Awards (R.A.B. and J.F.F.). The KRESCENT program is co-sponsored by the Kidney Foundation of Canada, the Canadian Society of Nephrology, and the Canadian Institute of Health Research. K.Y. is supported by the University of Toronto Department of Medicine Eliot Phillipson Clinician Scientist Training Program and a Banting and Best Diabetes Center Postdoctoral Fellowship. C.W. is supported by the Western University Clinician Investigator Program and PSI Foundation Research Trainee Award.
