Abstract
Background and Objective:
Bioimpedance technologies are increasingly used to determine fluid status in patients with chronic kidney disease and those with end-stage kidney disease on dialysis. We aimed to determine whether this technology improves clinical outcomes as compared with usual care.
Methods:
We performed a systematic review and meta-analysis of trials, comparing fluid management guided by bioimpedance technologies to standard of care in patients with chronic kidney disease. Our primary outcome was all-cause mortality. Secondary outcomes included blood pressure control, all-cause hospitalization, major adverse cardiovascular events, and change in left ventricular mass index.
Results:
Our search identified 819 citations of which 12 randomized controlled trials were included (2420 patients). No studies of non-dialysis-dependent chronic kidney disease patients met inclusion criteria. Mean age was 55 years and mean follow-up was 1 year. There was a statistically significant difference in all-cause mortality between both arms studied (risk ratio [RR] 0.64, 95% confidence interval [CI]: 0.44, 0.99). Better blood pressure control was observed in the bioimpedance arm of the included articles, weighted mean differences (WMD) −3.13 mm Hg (95% CI: −5.73, −0.53 mm Hg) for systolic blood pressure and WMD −2.50 mm Hg (95% CI: −4.36, −0.64 mm Hg) for diastolic blood pressure. No difference was observed concerning the other outcomes.
Conclusions:
Among patients on maintenance dialysis, bioimpedance-guided volume management showed decreased all-cause mortality and blood pressure but no significant difference in all-cause hospitalization, major adverse cardiac event, or change in left ventricular mass index. This may be due to a younger population sample than previous articles. Moreover, our study identified a knowledge gap by highlighting the lack of studies evaluating this technology in non-dialysis-dependent chronic kidney disease patients.
Key Learning Points
What was known before
Bioimpedance technologies are validated tools in accurate assessment of volume status.
Bioimpedance is effective at lowering blood pressure in patients on maintenance dialysis owing to its ability to tailor prescriptions of accurate ultrafiltration.
A systematic review and meta-analysis in the past has shown little to no impact on key clinical outcomes such as all-cause mortality and all-cause hospitalization.
What this adds
This study shows there is a reduction in all-cause mortality that has not been reported previously.
This study highlights a knowledge gap regarding the use of bioimpedance in the non-dialysis chronic kidney disease population.
What impact this may have on practice
Integration of this technology into clinical practice may improve important clinical outcomes.
More studies using this technology in the older, multi-comorbid maintenance dialysis and chronic kidney disease population are needed.
Introduction
Chronic kidney disease (CKD) is a significant health issue with a prevalence of 8% to 16% worldwide. 1 As CKD progresses to end-stage kidney disease (ESKD), treatment choices are limited to therapies such as dialysis, transplantation, or alternatively conservative care. Given limited access to life sustaining therapies and the associated morbidity, preventing CKD progression has become vitally important. While there have been some medical (sodium/glucose cotransporter 2 inhibitors) and technologic advances (biocompatible membranes) in the field, mortality among CKD and ESKD patients remains high. 2 This may be due in part to unique risk factors such as fluid overload that is commonly encountered in this population.3,4
Fluid overload has a prevalence of 40% to 50% among CKD and ESKD patients treated with maintenance dialysis.5-9 It is associated with systemic complications such as left ventricular hypertrophy and adverse cardiovascular outcomes.10,11 In general, volume status is assessed on clinical examination followed by appropriate diuretic prescription in the CKD population, or prescription of appropriate target weight in the population treated with maintenance dialysis. However, evidence has shown that clinical exam alone has limited sensitivity in assessing volume status.12,13 Complementary methods are emerging to improve our accuracy, including brain natriuretic peptide, echocardiography, lung ultrasound, and bioimpedance technologies. 7 Whether these additional measures will translate into improved clinical outcomes remains unknown. 14 Bioimpedance technologies, which have been extensively validated, are less prone to operator-dependent bias and allow volume measurements to be followed over time. Thus, bioimpedance adds another variable to the trend analysis (ie, clinical exam, blood pressure, proportion of treatments complicated by intradialytic hypotension, or failure to achieve target weight) that we use every day to make rational clinical decisions about volume status in our patients. 15 While there have been previous systematic reviews that have addressed the utility of bioimpedance in patients on maintenance dialysis, none have addressed the non-dialysis CKD population. In addition, there have been new studies published since previous reviews.
The purpose of this systematic review study is to compare bioimpedance-guided volume assessment with standard care in 2 populations (non-dialysis-dependent and dialysis-dependent patients) and to examine the effect of this strategy on important clinical outcomes including all-cause mortality.
Methods
The protocol for this systematic review and meta-analysis was prespecified and registered in the international prospective register of systematic reviews (http://www.crd.york.ac.uk/PROSPERO/CRD226687). Results are reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2009 checklist. 16
Search Strategy
A Medline search was conducted in PubMed and included articles published from January 1960 through August 2021 in the English language. The reference lists of selected studies and available meta-analyses were also reviewed. The following MESH terms were used in the search: ([Kidney Disease OR CKD OR renal disease OR renal failure OR End-stage renal disease OR ESRD OR chronic renal insufficiency OR non-dialysis dependent OR chronic dialysis OR dialysis dependent] AND (volume assessment OR volume management OR volume control OR volume overload OR fluid overload OR hypervolemia OR overhydration OR fluid status OR weight adjustments OR hydration status OR target weight OR dry weight OR goal weight OR ideal weight) AND (bioimpedance OR bioimpedance analysis OR BIA OR bioimpedance spectroscopy OR BIS OR Body Composition Monitor OR BCM OR lung ultrasound) AND (kidney function decline OR renal function decline OR glomerular filtration rate OR GFR decline OR doubling of serum creatinine OR kidney outcomes OR renal outcomes OR time to renal replacement therapy OR renal replacement therapy OR kidney failure OR renal failure OR renal progression OR time to dialysis OR time to end stage renal disease OR incident end-stage renal disease OR all-cause mortality OR hospitalizations OR major adverse cardiovascular events OR heart failure hospitalization OR left ventricular mass index OR heart failure admission OR cardiac death OR cardiovascular death OR cardiovascular events OR myocardial infarction OR stroke OR adverse outcomes OR adverse events)).
Study Selection
Two authors (L.H. and O.K.) independently screened the articles obtained from the MESH term search by first examining each title and abstract for eligibility. Selected articles then underwent full text review for inclusion based on the following eligibility criteria. The final group of selected studies was agreed upon by the 2 reviewers and a third author (C.W.) was available to resolve any discrepancies.
Eligibility Criteria
The following criteria were required for inclusion: (1) Study population: age >18 years, non-dialysis-dependent population defined as Kidney Disease Improving Global Outcomes (KDIGO) CKD stages 3 to 5; dialysis dependent ESKD as hemodialysis or peritoneal dialysis. (2) Intervention: volume assessment with bioimpedance analysis (BIA) or bioimpedance spectroscopy (BIS). Control: volume assessment by clinical exam alone or usual care. The initial protocol included lung ultrasound as an adjunct method for volume assessment. However, one author (T.M.) recently completed a meta-analysis on lung ultrasound to evaluate similar outcomes; thus, this technology was not included in this study. (3) Study design: randomized controlled trials and observational studies, including case control, cross-sectional, and cohort studies with prevalent and incident patients on maintenance dialysis were included. (4) Outcomes: at least one of the following outcomes should be reported: progression of CKD (estimated glomerular filtration rate slope of decline), incidence of ESKD, time to dialysis, renal death, blood pressure control and/or change in number medications, left ventricular mass index (LVMI), hospitalization for heart failure, all-cause hospitalizations, all-cause mortality, major adverse cardiovascular events (MACE; including cardiovascular death, myocardial infarction, and stroke), and residual kidney function.
Data Collection Process
Data collection was performed independently by the 2 authors using standardized digital spreadsheets. For repeated events that are neither binary nor continuous, we extracted the first event reported. When not reported, mean difference in blood pressure before and after the intervention was calculated for each study. In addition, the weight of each study was considered when calculating the mean of the means (eg, mean age between studies). The standard deviation of the mean difference was calculated as the square root of the sum of squares of the standard deviation prior to and after the intervention. One author did these calculations and a second author verified the results. 17 Initially, we set out to convert the blood pressure readings from weighted mean differences (WMD) into standardized mean differences (SMD) to solidify the foundations of our statistical methods. However, these calculations were difficult to interpret and thus we present the WMD, and have included the SMD, in the supplementary material.
Study Outcomes
Our initial primary outcome was hospitalization for heart failure. However, this outcome was not reported in any of the selected articles, thus we selected all-cause mortality as our primary outcome of interest. Our secondary outcomes are change in blood pressure, all-cause hospitalizations, MACE (including cardiovascular death, myocardial infarction, and stroke), LVMI, and residual kidney function. If an outcome was reported in an included study, we extracted the pertinent data, even if it was not a prespecified outcome of the individual article.
Risk of Bias
The risk of bias in the selected studies was assessed using the Risk of Bias (RoB) 2 tool for randomized controlled trials (RCT) 18 and RoB 1 tool for the observational studies. 19 Two authors (L.H. and O.K.) reviewed the articles to assess for risk of bias. The discrepancies were resolved by a third author (T.M.).
Statistical Analysis
The results are reported as mean ± standard deviation, median (interquartile range), or number (percentage). For continuous variables, the mean difference was pooled using a random effects model. In addition, SMD were pooled using the Hedges’ g test in a random effects model. Results were stratified by study design (RCTs or observational studies). For binary variables, the principal summary measure was the risk ratio (RR). The RR for each binary outcome was estimated using the Der Simonian-Laird random-effects model. To quantify heterogeneity, the I2 index was used. All analyses were performed in Stata (Version 17.0 SE, College Station, Texas).
Results
Our PubMed search revealed 819 citations. After initial screening, 32 articles were eligible for full text review. A total of 12 randomized clinical trials met inclusion criteria for the meta-analysis (Figure 1).20-31 In our original protocol, we aimed to include non-dialysis-dependent CKD patients and evaluate CKD-specific outcomes, however no CKD study met eligibility criteria and thus none were included in the final analysis.
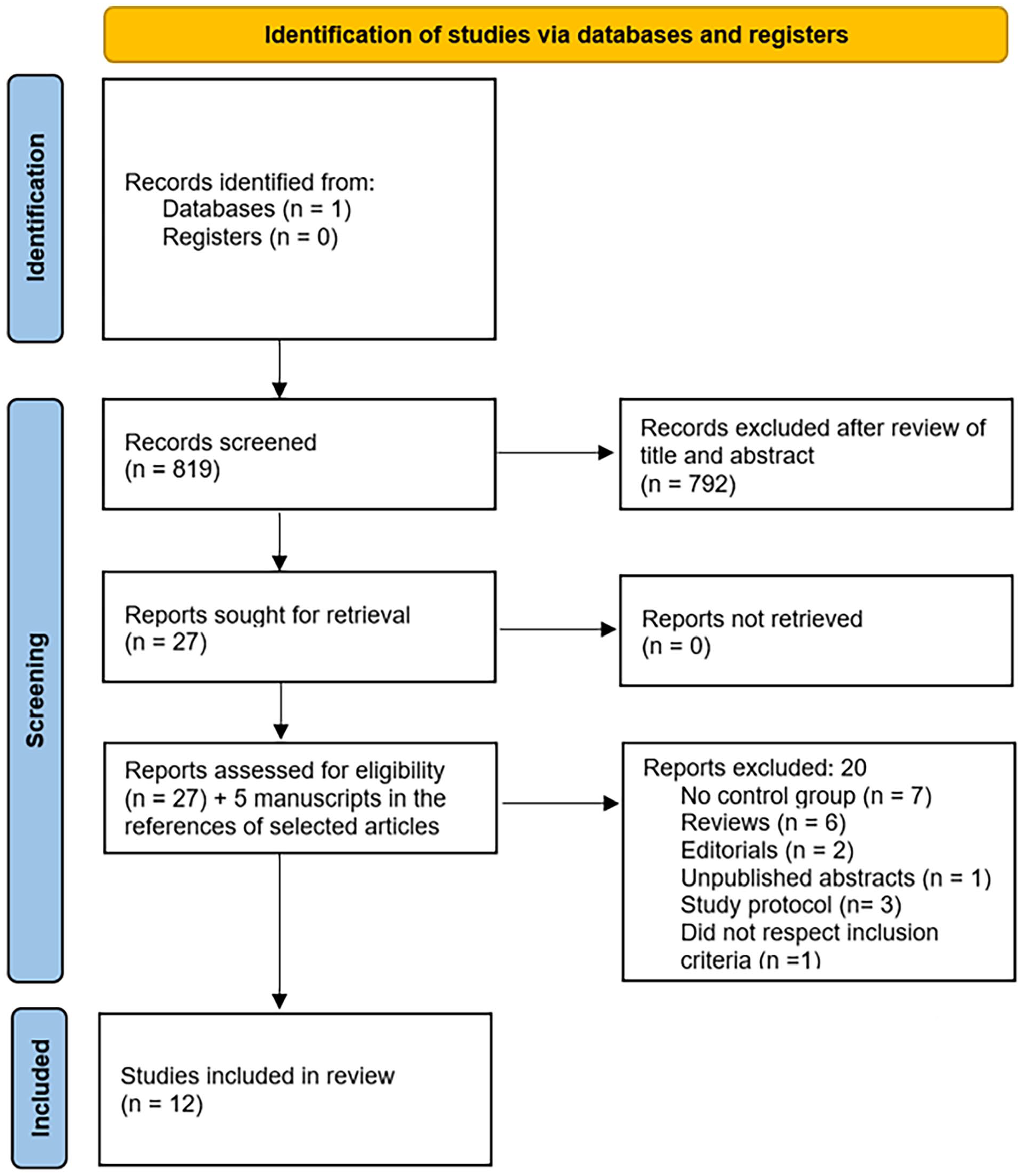
Flow diagram.
Selected studies were multinational (Europe and Asia) spanning the years 2011 to 2020 and included a total of 2420 patients. Most studies (N=7, 1374 patients) were hemodialysis patients and 5 studies were peritoneal dialysis patients (n=1046; Table 1). The average follow-up was 14 months, with a range of 3 to 30 months. The mean age was 55 years and dialysis vintage ranged from 13 to 106 months.
Population Characteristics.

Note. CAD = coronary artery disease; HF = heart failure; DM = diabetes mellitus; HTN = hypertension; PD = peritoneal dialysis; BIS = bioimpedance spectroscopy; HD = hemodialysis; BIA = bioimpedance analysis.
For the trials examining all-cause mortality, 2 were deemed at high-risk bias. One of these studies had a high risk of missing outcome bias and some concerns for randomization bias. 24 This study also accounted for 18.61% of the weight of data toward all-cause mortality. The other article was considered having a high risk of missing outcome data. 22 Otherwise, the other studies that had some concerns were used to assess blood pressure control. Many of the included articles were considered to low risk of bias. The risk of bias is presented in Figure 2.

Risk of bias for randomized controlled trials (traffic light plot).
All-cause mortality was a study outcome in 7 of the 12 RCTs (n=1630 patients; Table 2). Among these RCTs, there was a statistically significant difference in all-cause mortality between the 2 management strategies of bioimpedance versus usual care: relative risk (RR) 0.64 (95% confidence interval [CI]: 0.41, 0.99, I2 = 22.7%; Figure 3).
Outcomes Reported in Each Included Study.
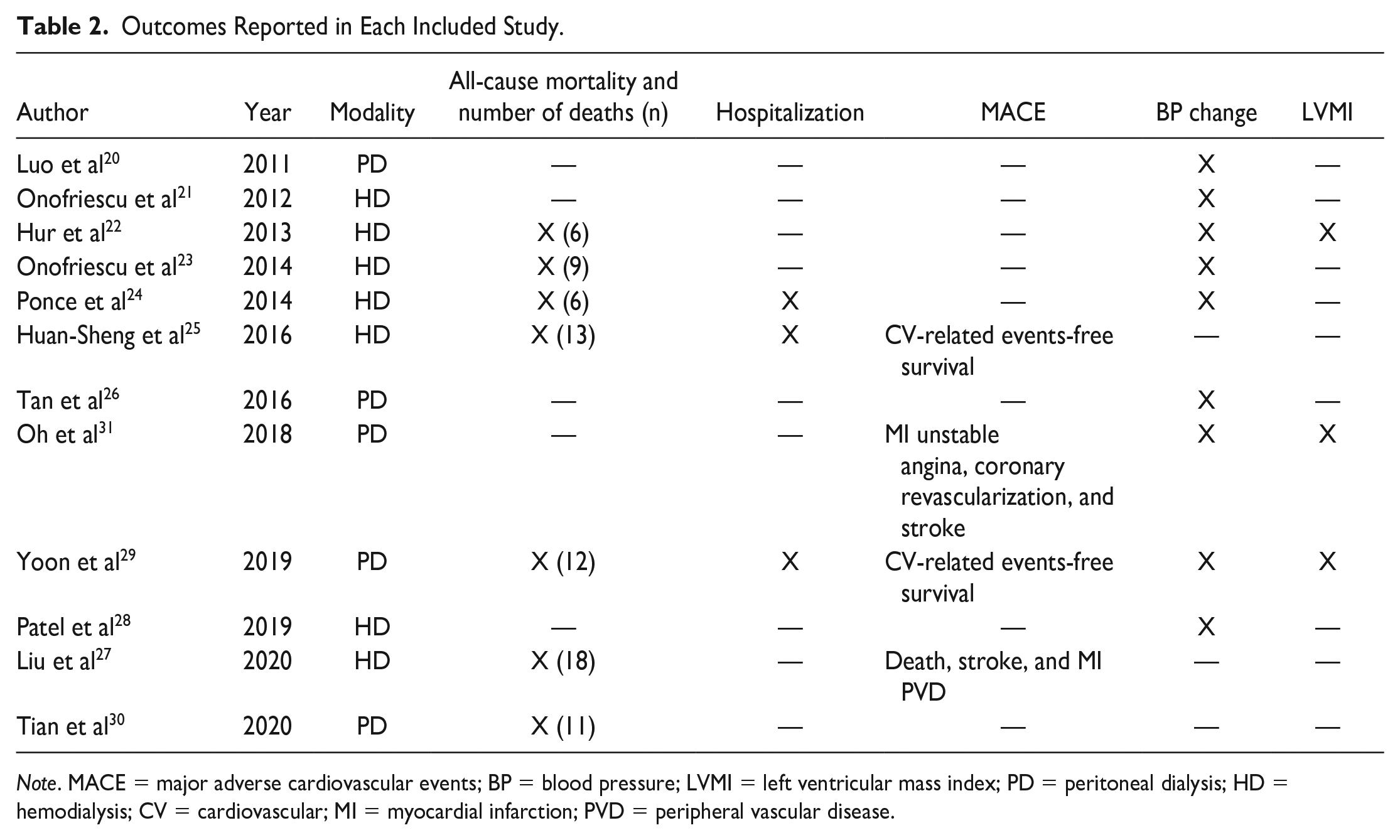
Note. MACE = major adverse cardiovascular events; BP = blood pressure; LVMI = left ventricular mass index; PD = peritoneal dialysis; HD = hemodialysis; CV = cardiovascular; MI = myocardial infarction; PVD = peripheral vascular disease.

All-cause mortality.
Upon reviewing effects on blood pressure, both systolic blood pressure and diastolic blood pressure (DBP) were significantly lower when using bioimpedance-guided volume assessment over clinical exam alone: WMD −3.13 mm Hg (95% CI: −5.73, −0.53 mm Hg, I2 = 0.2%) and WMD −2.50 mm Hg (95% CI: − 4.36, −0.64 mm Hg, I2 = 23.9%), respectively (Figures 4 and 5).

Change in systolic blood pressure.

Change in diastolic blood pressure.
The incidence of all-cause hospitalizations was neither different between the 2 groups (RR 0.99, 95% CI: 0.84, 1.17, I2 0.4%; Figure 6) nor was the incidence of MACE in the meta-analysis (RR 0.61, 95% CI: 0.38, 1.00, I2 15.4%; Figure 7).
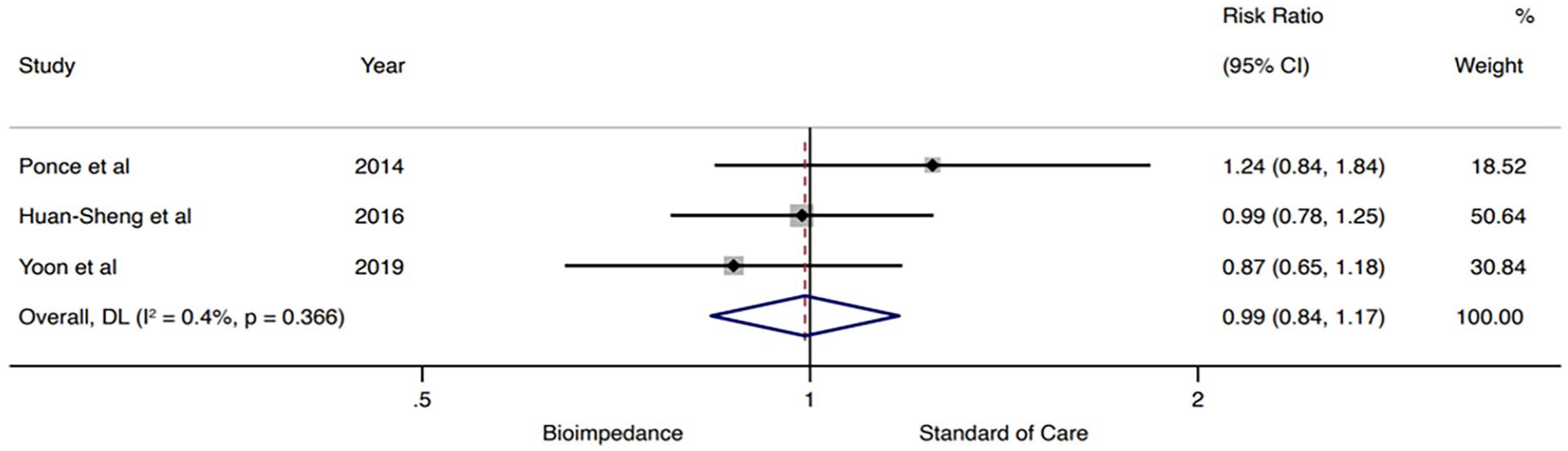
All-cause hospitalizations.

Major adverse cardiovascular events.
Five of the 12 studies reported on residual kidney function, all of which were studies in the peritoneal dialysis population. However, there were small numbers of patients with multiple missing data points and therefore no statistical analysis was performed. Finally, 3 studies in 464 patients (n=234 intervention group) reported on change in LVMI, which demonstrated no statistically significant difference WMD −6.68 g/m2 (95% CI: −15.79, 2.43 g/m2, I2 = 98.6%; Figure 8).

Change in LVMI.
Discussion
To our knowledge, this is the largest meta-analysis examining the use of bioimpedance-guided volume assessment in the maintenance dialysis population. We found that the use of bioimpedance technologies, in addition to clinical exam, lowers all-cause mortality and improves blood pressure control. We did not observe a difference in all-cause hospitalizations, MACE, or change in LVMI. Overall, there was only mild heterogeneity as evaluated by the I2 index concerning all-cause mortality and blood pressure control. Although we had set out to study the non-dialysis-dependent population, no study in the CKD population met inclusion criteria. This highlights a knowledge gap as to the possible benefit that bioimpedance technologies may have in this population.
Our study showed a significant difference in all-cause mortality between bioimpedance-assisted volume management versus clinical exam alone (0.64, 95% CI: 0.41, 0.99, Figure 3). This is not in keeping with the results of 2 previous meta-analyses that found no difference in all-cause mortality.32,33 The discrepancy in results can be explained by the inclusion of newer studies in our meta-analysis, specifically Yoon et al 29 and Liu et al, 27 and a different case-mix. For example, our article included studies that had n=1374 on maintenance hemodialysis and n=1046 patients on peritoneal dialysis. In comparison, Beaubien-Souligny et al 32 had n=1053 patients on maintenance hemodialysis and n=910 on peritoneal dialysis, whereas Covic et al 33 had n=844 patients on maintenance hemodialysis and n=766 patients on peritoneal dialysis. As risk of mortality is usually lower in patients with residual renal function, the inclusion of more studies in peritoneal dialysis may have led to this significant difference.
Our study also showed a significant decrease in blood pressure with the bioimpedance intervention as compared with clinical exam alone, with the strongest effect on systolic blood pressure (MWD −3.13 mm Hg, 95% CI: −5.73, −0.53 mm Hg) as compared with DBP (MWD −2.50 mm Hg, 95% CI: −4.36, −0.64 mm Hg). This is in keeping with previous reports. Beaubien-Souligny et al 32 reported a systolic mean blood pressure difference of −3.14 mm Hg (CI: −5.89, −0.38), Covic et al 33 reported a systolic mean blood pressure difference of −2.73 mm Hg (CI: −5.00, −0.46), and Scotland et al 34 reported a systolic mean blood pressure difference of −2.46 mm Hg (CI: −5.07, 0.15). These authors did not show a significant difference in DBP. The reasons for these differences are not entirely known; however, our study included more contemporary trials and, if summative blood pressure data were not reported, we calculated the mean difference of blood pressure (see “Methods” section), thus maximizing data points to analyze. This latter fact could explain why heterogeneity in the blood pressure arms was lower than previously reported.28,29,35
Our study demonstrated no difference in other clinical outcomes such as MACE or all-cause hospitalization. Our findings are similar to the meta-analysis of Beaubien-Souligny et al 32 that included 6 trials comprising 1254 patients and a RR 0.88 (CI: 0.63, 1.21) for all-cause hospitalizations. Concerning MACE, although not statistically significant, the results are quite similar to all-cause mortality even if these outcomes were not reported within the same study groups. This is not surprising, given the reduced morbidity of the sample population and, hence, reduced all-cause mortality.
There are several limitations to our study. First, while we set out to study dialysis-dependent and non-dialysis-dependent populations, no pre-dialysis studies met eligibility criteria. The differences in the methodological use of bioimpedance between studies could have influenced the outcomes and led to bias of the individual study results. In addition, the analysis of cardiovascular and renal events was limited by the low number of trials reporting these outcomes. Furthermore, although blood pressure reduction was demonstrated, the reliance on dialysis measurements as compared with ambulatory measurements is known to be suboptimal. In addition, the methodology and frequency of BP measurements was different between the included studies. One key outcome that was not followed in many studies was the risk of hypovolemic complications such as intradialytic hypotension. Thus, it is unclear whether an aggressive approach aimed at achieving euvolemia will translate into meaningful benefits for all patients and whether these benefits ultimately outweigh the risks.
In conclusion, among a younger, heterogeneous population of maintenance dialysis patients, the use of bioimpedance-guided volume assessment demonstrated a decrease in all-cause mortality and better blood pressure control. There is inconclusive evidence concerning the outcomes of hospitalization, MACE, and LVMI. Future studies should aim to investigate bioimpedance technologies for volume management in the dialysis and non-dialysis CKD population, using a protocolized approach and appropriate duration of follow-up.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581231185433 – Supplemental material for Bioimpedance-Guided Monitoring of Volume Status in Patients With Kidney Disease: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cjk-10.1177_20543581231185433 for Bioimpedance-Guided Monitoring of Volume Status in Patients With Kidney Disease: A Systematic Review and Meta-Analysis by Laura Horowitz, Oliver Karadjian, Branko Braam, Thomas Mavrakanas and Catherine Weber in Canadian Journal of Kidney Health and Disease
Footnotes
Data Availability Statement
Data available on request.
Authors’ Note
A part of this work was presented in the form of a poster at the 2022 American Society of Nephrology Kidney Week in Orlando, Florida, on November 5, 2022.
Author Contributions
Study collection from Medline search: O.K. and L.H. Study selection via abstract: O.K. and L.H. Study inclusion based on full text review: O.K. and L.H. Discrepancies upon full text review: C.W. Data collection: O.K. and L.H. Discrepancies upon data collection: O.K. and L.H. Data conversion to compare the same outcome that was presented differently within included manuscripts: O.K. and T.M. Statistical analysis: T.M. Risk of bias: O.K. and L.H. Discrepancies with risk of bias: T.M. Tables and figures: O.K., L.H., and T.M. Data interpretation: O.K., L.H., T.M., and C.W. Writing the manuscript: O.K. and L.H. Review of the manuscript: T.M., C.W., and B.B. Editing the manuscript: O.K., L.H, C.W., and T.M.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author T.M. received speaker honoraria from Daiichi Sankyo, BMS Canada, Janssen, Astra Zeneca, and Pfizer; has served on advisory boards for Boehringer Ingelheim, Bayer, GSK, and Servier; and has received an unrestricted research grant from Astra Zeneca outside the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Author B.B. is supported by the Translational Kidney Health Research Chair by the Faculty of Medicine, Department of Medicine, Division of Nephrology. Author T.M. is supported by a Fonds de Recherche Santé Quebec (FRSQ) Junior 1 Clinician Scholar award. The FRSQ had no influence on the choice of topic, the results, or conclusions. He is also supported by an operational grant from the Quebec Society of Nephrology.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
