Abstract
Background:
Emerging data favor central blood pressure (BP) over brachial cuff BP to predict cardiovascular and kidney events, as central BP more closely relates to the true aortic BP. Considering that patients with advanced chronic kidney disease (CKD) are at high cardiovascular risk and can have unreliable brachial cuff BP measurements (due to high arterial stiffness), this population could benefit the most from hypertension management using central BP measurements.
Objective:
To assess the feasibility and efficacy of targeting central BP as opposed to brachial BP in patients with CKD G4-5.
Design:
Pragmatic multicentre double-blinded randomized controlled pilot trial.
Setting:
Seven large academic advanced kidney care clinics across Canada.
Patients:
A total of 116 adults with CKD G4-5 (estimated glomerular filtration rate [eGFR] < 30 mL/min) and brachial cuff systolic BP between 120 and 160 mm Hg. The key exclusion criteria are 1) ≥ 5 BP drugs, 2) recent acute kidney injury, myocardial infarction, stroke, heart failure or injurious fall, 3) previous kidney replacement therapy.
Methods:
Double-blind randomization to a central or a brachial cuff systolic BP target (both < 130 mm Hg) as measured by a validated central BP device. The study duration is 12 months with follow-up visits every 2 to 4 months, based on local practice. All other aspects of CKD management are at the discretion of the attending nephrologist.
Outcomes:
Primary Feasibility: Feasibility of a large-scale trial based on predefined components. Primary Efficacy: Carotid-femoral pulse wave velocity at 12 months. Others: Efficacy (eGFR decline, albuminuria, BP drugs, and quality of life); Events (major adverse cardiovascular events, CKD progression, hospitalization, mortality); Safety (low BP events and acute kidney injury).
Limitations:
May be challenging to distinguish whether central BP is truly different from brachial BP to the point of significantly influencing treatment decisions. Therapeutic inertia may be a barrier to successfully completing a randomized trial in a population of CKD G4-5. These 2 aspects will be evaluated in the feasibility assessment of the trial.
Conclusion:
This is the first trial to evaluate the feasibility and efficacy of using central BP to manage hypertension in advanced CKD, paving the way to a future large-scale trial.
Trial registration:
clinicaltrials.gov (NCT05163158)
Introduction
Chronic kidney disease (CKD) affects 4 million Canadians and is a major risk factor for cardiovascular (CV) disease, death,1 -4 and kidney failure.5,6 Hypertension is observed in almost all individuals with CKD,7,8 and blood pressure (BP) control represents a major pillar of therapy to prevent CV events and CKD progression.9,10 Considering the importance of BP management to prevent CV events, a reliable BP is one of the most important measurements in clinical medicine. 11 However, to this day, we assess BP solely based on a concept that was described more than 120 years ago. Riva-Rocci designed brachial cuff sphygmomanometry to provide a convenient surrogate of the true aortic BP, as the upper arm contains an accessible artery close to the aorta. 12 Even now, automated oscillometric devices are validated against auscultatory brachial cuff sphygmomanometry. 13 Only recently have we truly realized how this surrogate may be imperfect. A recent meta-analysis has shown that the mean difference between brachial cuff systolic blood pressure (SBP) and true aortic SBP is only 0.3 mm Hg (high accuracy) but with mean absolute difference of 8.0 mm Hg (poor agreement) indicating a very wide range of overestimation and underestimation. 14 The BP range where these differences appear to be the most important is 120 to 160 mm Hg. 15 Thus, while brachial cuff BP accurately estimates the true aortic SBP on a populational level, it can lack accuracy on an individual level. Highly accurate BP measures are primordial, as even BP variations as small as 5 mm Hg can significantly impact CV risk estimation, and result in misclassification and suboptimal treatment.16 -18 With these limitations, brachial cuff BP may not truly be the best way to manage the CV risk in high-risk populations where BP control is paramount.
Physiologically, it is the aortic BP, not brachial BP, that directly affects the major organs, and the 2 can differ substantially. In recent years, various devices have been developed to derive the true aortic BP non-invasively though pulse wave analysis (herein referred to as “central BP”). Central BP, as a more precise surrogate of the true aortic BP.19 -21, provides several advantages compared to brachial cuff BP:
Central BP correlates better with CV events and end-organ damage.22 -31
Improvement of CV outcomes and surrogate markers correlate better with central BP lowering than brachial BP lowering.32 -34 The CAFE sub-study of ASCOT suggested that the CV benefit of amlodipine over atenolol was explained by a greater reduction of central BP, despite identical brachial BP in both groups. 33 A secondary analysis of SPRINT also shows the intensive treatment was superior to the standard treatment only when pulse wave velocity (PWV; a marker of aortic stiffness) decreases. As PWV is more strongly associated with central than brachial BP,22 -27,32 -34 this provides a clue that the benefits may have been driven by central BP lowering.
BP-lowering drugs differentially affect central and brachial BP, where certain drugs (e.g., beta-blockers) reduce disproportionally less central BP than brachial BP.32,35,36
In the general population, targeting central BP instead of brachial BP results in a lower use of BP-lowering drugs without detrimental effects on CV surrogate markers. 37
Up to 8% of the general population have central hypertension despite a normal brachial BP and this subgroup is at higher CV risk compared to individuals with normal brachial or central BP.38 -40
Considering all of this, several randomized controlled trials (RCT) have been launched to investigate whether targeting central BP improves CV outcomes beyond what is achievable by targeting brachial cuff BP.37,41,42 Importantly, these RCTs all exclude patients with CKD.
The brachial cuff SBP target in patients with advanced CKD remains controversial, with clinical practice guidelines recommendations ranging from <120 to <140 mm Hg. 10 ,43 -46 This stems from the lack of definitive RCT data in this population but may also reflect the shortcomings of brachial cuff BP. Individuals with increased arterial stiffness have a much higher aortic SBP than healthy individuals despite identical brachial cuff BPs.47,48 As such, in clinical conditions with elevated arterial stiffness, the brachial cuff BP loses accuracy against the true aortic SBP.47 -53 This is likely explained by the gradual loss of the normal aortic-to-brachial SBP amplification as arteries stiffen, where the brachial cuff further exaggerates the underestimation of the intra-arterial brachial SBP, decoupling the typical brachial cuff to true brachial to true aortic BP relationship.14,20 Subjects with advanced CKD are widely known to have a high degree of arterial stiffness, which has been shown to be predictive of CKD progression, kidney failure, CV events and mortality.49 -52 In patients with eGFR < 60 mL/min/1.73m2, Carlsen et al 48 showed that the accuracy of the brachial cuff SBP against the true aortic SBP decreases with lower eGFR. Very few observational studies investigated whether central BP measurements provide better risk stratification in advanced CKD patients, and the bulk focused on patients receiving dialysis.54 -56 In the Chronic Renal Insufficiency Cohort (eGFR range 20-70 ml/min/1.73m2), central BP was strongly linked to CV outcomes, but did not improve the prediction of CV outcomes compared with brachial cuff BP. 57 However, central BP was measured using an older Type I device, which is less accurate than more modern central BP devices and yield central BP measurements highly correlated with the brachial cuff BP.58,59 Thus, efficacy of central BP monitoring in advanced CKD remains unknown.
We hypothesize that targeting central BP instead of brachial cuff BP in patients with advanced CKD is more appropriate for titration of BP medication and thus beneficial for CV and kidney health. Ultimately, we will need to perform a suitably powered clinical trial aimed at examining the impact of such approach on CV events, CKD progression and patient-reported outcomes. However, feasibility data is needed before launching a large trial for the following reasons. First, it is important to determine if patients can be enrolled in such trial and if our approach can meaningfully alter treatment decisions. Second, the efficacy needs to be assessed using a recognized surrogate marker of CV event (i.e., aortic stiffness). Finally, preliminary efficacy and safety data needs to be collected.
To address all these points and provide the basis for a large-scale trial, we designed CENtral blood pressure Targeting: A pragmatic RAndomized triaL in advanced Chronic Kidney Disease (CENTRAL-CKD), a randomized double-blinded multicentre pragmatic pilot trial (Figure 1).
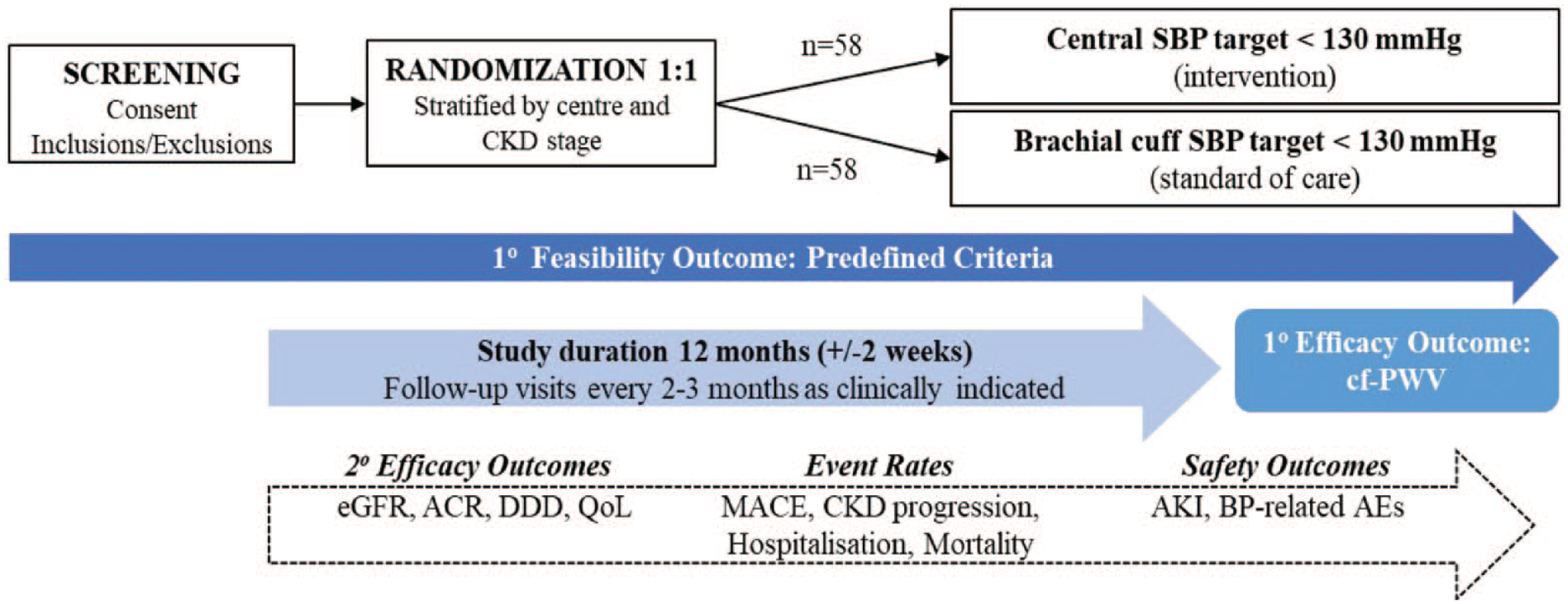
Study design and summary.
Methods
Setting
Participants will be enrolled in advanced CKD care clinics from 7 large academic centers across Canada.
Inclusion and Exclusion Criteria
The inclusion criteria are as follows:
Older than 18 years.
eGFR < 30 mL/min/1.73 m2 (within 30 days of screening).
Office brachial cuff SBP between 120 and 160 mm Hg (standardized measurements).
The trial exclusion criteria are as follows:
Already taking 5 or more antihypertensive medications.
Unwillingness to change antihypertensive medication by the attending nephrologist or patient.
Recent acute kidney injury (AKI; >50% increase in serum creatinine in preceding 30 days 60 ).
Previous kidney replacement therapy (kidney transplant, hemodialysis, or peritoneal dialysis).
Recent myocardial infarction, stroke, heart failure (in preceding 30 days).
Recent injurious fall requiring hospitalization (in preceding 30 days).
Concomitant major illness/comorbidity that may result in death in the next 6 months.
Participation in another study that is likely to affect BP levels.
Inability to provide consent due to cognitive impairment.
These criteria aim to optimize generalizability of the results to most patients with CKD G4-5 in whom the attending nephrologist is considering interventions toward BP reduction and where therapeutic inertia may be the lowest. This study population includes patients whose brachial cuff SBP is within the range where its reliability toward aortic SBP is the lowest 15 and thus is the group where clinical equipoise is the highest.
Trial Interventions
Participants will be randomized to a central BP target (intervention) or a brachial cuff BP target (standard care). The type of BP (central or brachial) will be blinded. Participants randomized to “central BP” will be treated to achieve a central SBP < 130 mm Hg, which is the threshold for central hypertension. 39 Participants randomized to “brachial cuff BP” will be treated to achieve a brachial cuff SBP < 130 mm Hg, as recommended by the American Heart Association for all patients with advanced CKD. 43 In advanced CKD, Hypertension Canada recommends either <130 mm Hg (diabetes) or <140 mm Hg (without diabetes), 46 both of which are based on non-standardized measurements, which yield SBP values 5 to 10 mm Hg higher than standardized measurements 61 (as will be used in this trial). Meeting the KDIGO target of <120 mm Hg, 10 as suggested in selected patients, will be at the discretion of the attending nephrologist.
As in SPRINT, 62 no guidance will be provided regarding diastolic blood pressure (DBP) because isolated diastolic hypertension is seldom encountered in CKD and central DBP is usually identical to brachial cuff DBP. 14 ,63 -65 Guidelines recommend caution when lowering SBP to target if diastolic BP is ≤60 mm Hg 66 although in SPRINT, even the participants in the lowest quintile of baseline DBP (mean 61 ± 5 mm Hg) benefited from an intensive SBP target.67,68 All other aspects of BP and CKD management, including BP-lowering drug-related decisions and clinical follow-up will be at the discretion of the attending nephrologist or nurse practitioner.
BP Measurements
Blood pressures will be measured using the WatchBP Office Central device (Microlife, Taiwan). 69 This device uses an upper arm cuff to first measure brachial cuff BP then uses pulse wave analysis with type II calibration 20 to provide an accurate estimation of the true aortic BP. It is validate for brachial cuff BP against the auscultatory method 70 and for central BP against invasive measurements. 69 Unpublished preliminary data suggest a high accuracy of central BP in regard to the invasive aortic BP in patients with CKD G4-5.
At the first visit, BP will be measured in both arms, and the arm with the highest value will be used throughout the study. If an arteriovenous fistula is present or planned, the contralateral arm will be used. All BPs will be logged as means of 3 consecutive measurements in a dedicated unattended room after 5 minutes of seated rest using an appropriate-sized cuff, in accordance with clinical practice guidelines (standardized BP).43,46 According to randomization, only the brachial or central BP value will be provided to the attending nephrologist on a standardized reporting form by a dedicated research staff, with a reminder to aim for the designated BP target (SBP < 130 mm Hg for both groups).
Cointervention Minimization
Letters describing the trial will be provided to each participant. These briefly explain the trial procedures to any doctor who may see the participant and will specify that BP drugs can be adjusted but that it is preferable to refer the patient to the nephrology clinic and trial staff (when safe and feasible). All BP drugs will be assessed at study visits to verify whether any change occurred. Home BP will not be prohibited, but the attending nephrologists will be encouraged to use the trial BP to inform decisions, as was done in the SPRINT trial. 65 If the attending nephrologist chooses to rely on Home BP for treatment decisions and does not adjust BP drugs despite a trial clinic BP >130 mm Hg, this will be considered as a therapeutic inertia event with the reason listed as “Home BP at target” on the report form.
Outcome Measures
The primary feasibility outcome is the feasibility of a larger trial which will be assessed using predefined criteria (Table 1). All criteria would need to be determined to be “Yes” or “Probable” to justify progress to a large-scale efficacy trial without major redesign of the study. Divergent treatment decision based on central BP compared to brachial BP will be established by determining whether there is a difference between the decision made based on central SBP and the decision that would have been made had the patient been in the brachial SBP arm. This will allow to document the degree to which the strategy drives treatment decisions. Therapeutic inertia is defined as the number of patients whose pharmacological treatment had not been modified, divided by the number of patients not reaching the target values at each visit. The reasons for therapeutic inertia target will be recorded in case report forms.
Feasibility Components to Be Assessed.

Note. BP = blood pressure.
The primary efficacy outcome is the difference in carotid-femoral PWV (cf-PWV) at 12 months. Aortic stiffness determined by cf-PWV has the advantages of being predictive of major adverse CV events (MACE), easily measured, highly reproducible, validated prospectively, and widely recognized in CV research.71,72 It will be measured by applanation tonometry with a SphygmoCor device (Atcor, USA).73,74 Measurements will be made in triplicate after a 10-minute rest in the supine position then averaged.
The secondary outcomes include (1) eGFR decline; (2) change in albuminuria; (3) Daily Defined Doses (DDD) of BP medication 75 ; and (4) quality of life (KDQOL-SF questionnaire).76,77 All values collected in the 12 months follow-up will be evaluated to determine whether the intervention results in decreases eGFR decline with or without changes in albuminuria and/or DDD (a way to assess cumulative drug changes over time, with dosage considerations), and whether titrating BP medication with central BP measurements improves quality of life. These will be used as pilot efficacy data for the larger trial.
Several adverse events will be collected:(1) MACE (CV mortality, myocardial infarction, stroke, heart failure requiring hospitalization, peripheral artery disease requiring revascularisation); (2) progression toward kidney failure (sustained eGFR loss ≥ 40%, ESKD or death from renal failure) 80 ; (3) all-cause hospitalization; and (4) all-cause mortality. These will be collected as preliminary data to inform the larger trial, as MACE will be its primary outcome and the others, important secondary outcomes.
Finally, 2 important safety outcomes will also be collected: (1) symptomatic orthostatic hypotension, dizziness, light headedness, injurious falls, syncope, or any unexpected event attributed to the intervention and (2) AKI (transient >50% increase in serum creatinine 60 ). These will be used as pilot safety data for the larger trial.
Randomization and Blinding
Participants will be randomized in a 1:1 ratio via a web-based randomization system, with stratification by center and CKD stage [G4 (eGFR 15-30 mL/min) or G5 (eGFR < 15 mL/min)] and random permuted blocks (sizes 2 and 4). The WatchBP device can be programmed to mask all BP readings to the patient, allowing blinding. Only one dedicated research staff will have access to the randomization module and all BP readings on a connected laptop. Both the attending nephrologist and the participant will remain blinded to whether the provided BP value represents central or brachial BP throughout the study. The trial coordinator, other health care providers, outcome adjudicators, including trial statisticians, data analysts, and all investigators will remain blinded to allocation.
Duration and Frequency of Follow-Up
The trial duration is 12 months. This study is designed as a pragmatic trial to reflect the “real-world” impact of the intervention (Table 2). All participants will be assessed by their attending nephrologist at every 2 to 4 months, based standard local practices. The only obligatory follow-up visit will at the end of the trial (12 months ± 2 weeks) to collect all outcomes data.
Visit Schedule.
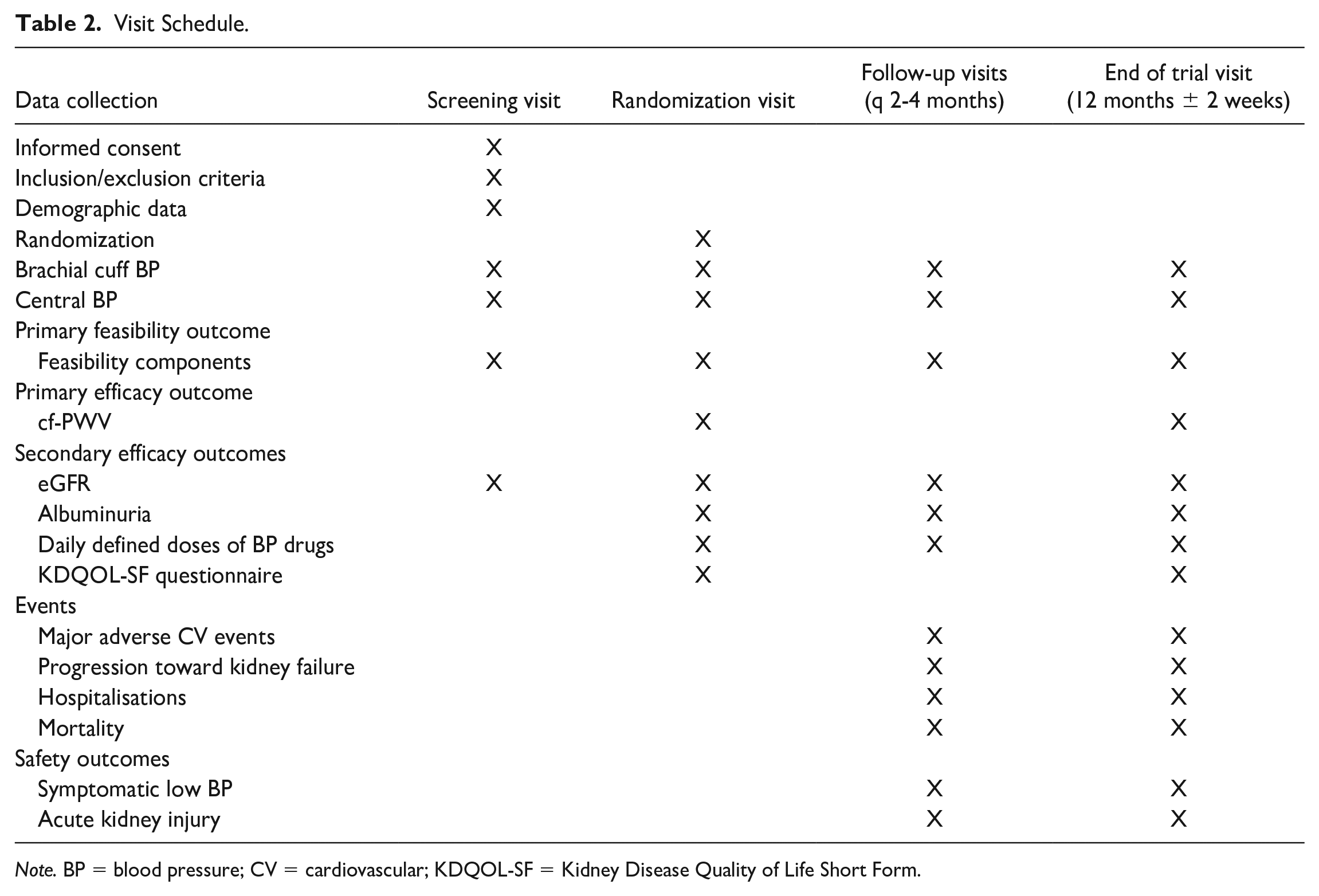
Note. BP = blood pressure; CV = cardiovascular; KDQOL-SF = Kidney Disease Quality of Life Short Form.
Sample Size
As per our Primary Feasibility Outcome criteria, we have established that a minimal recruitment of 54 participants in 24 months (0.5 per site per month) must be achieved to prove feasibility. This would allow us to estimate a consent rate of 60% (95% CI: ±6%), a randomization rate of 80% (95% CI: ±6%), and a completion rate of 60% (95% CI: ±5%), as calculated using the Wilson method with continuity correction. 79
Our Primary Efficacy Outcome is the cf-PWV after 12 months of intervention. In advanced CKD, an increase of 1 m/s coincides with a 18% increase in CV events 49 while in dialysis, a decrease of 1 m/s with BP medication is linked to a 39% reduction of mortality. 80 Therefore, a 1 m/s reduction of cf-PWV over 12 months represents a reasonable and highly clinically significant endpoint. Considering a mean PWV of 9.5 ± 1.7 m/s found in a local representative CKD G4-5 cohort (unpublished data), a sample size of 92 participants (46 in each group) will have an 80% power to detect this difference with a 2-sided alpha of 0.05. While failure to demonstrate such difference in cf-PWV would not necessarily demonstrate a lack of efficacy toward reducing CV events, we will consider the intervention apparently futile if the difference is less than the arbitrary cut-off of 0.3 m/s. Therefore, taking into consideration a projected loss to follow-up 20% (due to drop-out, death, or kidney replacement therapy initiation), we will enroll 116 participants to assess both feasibility and primary efficacy.
To ensure appropriate sex and gender representations, we aim to randomize a minimum of 40% females and 40% individuals self-identifying as women. This will be achieved by verifying the percentage of enrolled females and women once 50%, 75%, and 90% of the targeted trial sample is reached, to determine whether enrollment needs to be enriched with individuals of an underrepresented sex or gender.
Statistical Analyses
The statistical plan was designed with the help of an experienced statistician. All analyses will follow the intention-to-treat principle. A larger trial will be considered feasible without major redesign if all components of the Primary Feasibility Outcome are either in the “Yes” or “Probable” ranges. The Primary Efficacy Outcome will be tested using an analysis of variance (ANOVA) for repeated measures, with the group as a between-subject factor and the time as a within-subject factor to compare the cf-PWV between intervention groups, and an estimation of the mean difference between groups at 12 months will be provided using a 95% CI. Secondary Efficacy Outcomes will be analyzed with mixed regression models to account for repeated measures. Events and Safety Outcomes will be analyzed through Kaplan-Meier estimators and log-rank test. Planned subgroup analyses include sex, self-identified gender, baseline CKD stage (G4 vs G5), baseline CV disease, and baseline type II diabetes status.
Expected Challenges and Mitigation Strategies
A challenge that could arise in this trial is distinguishing whether central BP is truly different from brachial BP to the point of significantly influencing treatment decisions. Preliminary data obtained from patients that would be eligible to this trial suggests that on average, central SBP exceeds brachial SBP by a mean of 10 mm Hg in this population. If this is also the case in trial participants and if we achieve similar level of success in terms of meeting the target BP for each intervention arm, we can foresee up to 10 mm Hg difference in brachial SBP between groups. It is important to note that brachial SBP lowering per se has been shown to reduce CV outcomes, most notably in SPRINT (though not in this population). We recognize that this may lead to a difficulty in interpreting our findings as results may be attributed to simply targeting a lower brachial BP and not targeting central BP.
Our approach to mitigate this is as follows:
This pilot, and the larger trial, examine the effects of different strategies, not a different target. Preliminary data show high and unpredictable interindividual variation in the difference between central and brachial SBP. The range of the difference between central and brachial SBP is wide, and almost 20% of individuals have central SBP lower than brachial SBP, proving heterogeneity and poor correlation. Targeting central BP may allow selection of individuals in which brachial cuff BP can be safely intensively lowered.
For each treatment decision, we will document whether there is a difference, and its direction, between the decision made based on central SBP and the decision that would have been made had the patient been in the brachial SBP arm. The proportion of treatment decisions that differ according to assignment will be assessed to document the degree to which the strategy drives treatment decisions.
We anticipate that a significant proportion of treatment decisions will differ. If the proportion of different treatment decisions is <10%, we will regard the intervention as indistinguishable from brachial SBP lowering. We also recognize that there may be heteroskedasticity in the relationship between central and brachial SBP, although our preliminary data argue against this. Nonetheless, if this is the case, then in patients whose SBP is controlled close to target, the difference in treatment decisions may be reduced. Identifying how different actual treatment decisions are in the 2 arms is an important aim of the feasibility assessment.
Planned Full Trial
CENTRAL-CKD is designed as a pilot trial aimed at providing the framework and justification to proceed to a large-scale trial with adequate power to detect the impact of the proposed intervention on clinically important outcomes. Unless major redesigns are warranted, the trial procedures will remain as described. The primary outcome will be time to first MACE (composite of CV mortality, myocardial infarction, stroke, heart failure requiring hospitalization, peripheral artery disease requiring revascularisation). Secondary outcomes will be chosen from the parameters assessed in this pilot trial and using inputs from various stakeholders and patient-partners. The full trial will be monitored by an independent DSMB. Our pilot trial will allow to properly assess the full trial sample size.
Progress to Date
CENTRAL-CKD is endorsed by the Canadian Nephrology Trials Network, a group of trialists established to improve nephrology clinical trials and is listed on clinicaltrials.gov (NCT05163158). It is funded by a Kidney Health Research Grant (grant no. 852040-21KHRG) from the Kidney Foundation of Canada. The project is approved by the research and ethics boards at all 7 trial sites. The first patient was enrolled at the Hôpital du Sacré-Coeur de Montréal on May 24, 2022. As of December 2022, 36 participants have been randomized. Enrollment is anticipated to be completed by May 2024, with the final study visit by May 2025.
Conclusions
The CENTRAL-CKD trial will be the first to evaluate the feasibility and efficacy of using central BP measurements to guide management in patients with advanced CKD. This study could provide the basis and justification to conduct a large scale with clinically meaningful endpoints and patient-reported outcome measures, and has the potential to ultimately change how we manage hypertension in this important high-risk population.
Footnotes
Ethics Approval and Consent to Participate
The study received approval from all appropriate Research Ethics Boards. Informed consent was obtained from all participant.
Consent for Publication
All authors reviewed the final manuscript and provided consent for publication.
Availability of Data and Materials
Data may be available upon written request to the corresponding author and subject to Research Ethics Board approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial is funded by a Kidney Health Research Grant from the Kidney Foundation of Canada (grant # 852040-21KHRG).
