Abstract
Background:
The use of small pediatric donors (age ≤ 5 years and body weight < 20kg) for adult transplant recipients is still regarded controversially in terms of early complications, long-term outcomes, and development of hyperfiltration injury due to body size mismatch.
Objective:
To investigate long-term outcomes of adult renal allograft recipients receiving a kidney from small pediatric donor (SPD) in terms of kidney function and early features of hyperfiltration injury such as histological changes and proteinuria.
Design:
Retrospective, single center study.
Settings:
Transplant center of the University Hospital of Basel, Switzerland.
Patients:
Adult renal allograft recipients receiving a kidney from a small pediatric donor at our center between 2005 and 2017.
Methods:
The outcome of 47 transplants from SPD were compared with 153 kidney transplants from deceased-standard criteria donors (SCD) occurring during the same time period. Incidence of clinical signs of hyperfiltration injury (eg, proteinuria) was investigated. According to our policy, surveillance biopsies were taken at 3 and 6 months post-transplant and were evaluated in terms of signs of hyperfiltration injury.
Results:
At a median follow-up of 2.3 years post-transplant, death-censored graft survival of SPD was comparable to transplants from SCD (94% vs 93%; P = .54). Furthermore, allograft function at last follow-up (estimated glomerular filtration rate–Modification of Diet in Renal Disease) was significantly higher in pediatric transplant (80 vs 55 ml/min/1.73 m2, P = .002). We found histological signs of early hyperfiltration injury in 55% of SPD. There was an equally low proteinuria in both groups during follow-up.
Limitations:
It is a single center and retrospective observational study with small sample size. The outcomes were investigated in a well-selected population of recipients with low body mass index, low immunological risk, and well-controlled hypertension and was not compared with equal selected group of recipients.
Conclusions:
Early histological and clinical signs of hyperfiltration injury in SPD is frequent. Despite the hyperfiltration injury, there is an equal allograft survival and even superior allograft function in SPD compared with SCD during follow-up. This observation supports the concept of high adaptive capacity of pediatric donor kidneys.
Introduction
The growing number of patients on the deceased donor renal allograft waiting list has prompted efforts to expand the criteria for acceptable organs, on both ends of the age scale. Presently, kidneys from pediatric donors as single organ or en bloc are frequently used for adult recipients. However, the literature analyzing the outcomes in pediatric donor transplantation reveals inconsistent results: some studies describe an elevated risk of early complications (ie, vascular and ureteral problems) and the development of hyperfiltration injury (ie, focal segmental glomerulosclerosis), whereas some studies report promising results.1,2 To overcome the risk of early surgical complications and early hyperfiltration injury, small pediatric kidneys donors (SPD) (defined as age
The aim of this retrospective study was to compare long-term outcomes of adult renal allograft recipients receiving a kidney from SPD (age
Materials and Methods
Patient Population
In this retrospective single center study, all pediatric kidney transplantations of donors age
To minimize the risk of early hyperfiltration injury in pediatric kidney transplantation, we very carefully selected recipients in terms of body weight and pre-existing hypertension. Only recipients with a body mass index (BMI) < 30 kg/m2 and a body weight < 100 kg were allowed to be transplantation with SPD. If there were several recipients suitable for pediatric kidney transplantation, the recipient with the lower BMI was preferred. Only patients without a history of hypertensive nephropathy and on 2 or less antihypertensive medication were suitable for transplantation with SPD. Even if there is no evidence in literature showing an increased immunologic risks in SPD transplantation, only recipients with a low immunological risk (ie, no donor-specific HLA-antibodies detected by single flow-beads and first kidney allograft) were preferred to minimize additional harm for SPD transplantation. The outcome of SPD was compared with the outcome in patients who received a graft from SCD during the same time period. SCD was defined as age < 50 years or age 50 to 59 years with only 1 of the following conditions: cerebrovascular cause of death, creatinine > 132 µmol/l, or hypertension. The local ethics committee of the University of Basel performed this retrospective single center study, after providing approval.
Immunosuppressive Regimen
The immunosuppressive regimen included induction treatment with basiliximab (Simulect, Novartis Pharma Schweiz AG, Bern, Switzerland). Maintenance immunosuppressive therapy changed over time due to different ongoing protocols but consisted of a triple therapy including tacrolimus and prednisone, as well as azathioprine or mycophenolate mofetil (MMF) or an mTOR-inhibitor (everolimus or sirolimus). Tacrolimus was started the day of transplantation, whereas mTOR-inhibitors were started at day 4 after transplantation. Tacrolimus target trough levels were 8 to 12 ng/ml during the first 3 months. Surveillance biopsies were performed at 3 and 6 months. In the absence of subclinical rejection, steroids were withdrawn and patients maintained on a dual therapy, which consisted of either tacrolimus (through level 4-8 ng/ml) and MMF or azathioprine, or an mTOR-inhibitor (trough levels 6-8 ng/ml) and MMF.
Surgical Technique and Perioperative Management
There were no specific changes in standard surgical technique for SPD. All transplantations (SPD and SCD) were only performed by vascular surgeons including anastomosis of the ureter to the bladder. Prophylaxis for vascular thrombosis with heparin 10 000 U per day was started immediately after transplantation if there were no major bleedings. This was later switched to aspirin 100 mg per day for 3 months. Recipients of a SCD received weight adapted low-molecular weight heparin as general thrombosis prophylaxis day 1 after transplantation and until discharge from hospital.
Clinical Outcomes
Currently, there is no knowledge of which formula provides the best accuracy to calculate the estimated glomerular filtration rate (eGFR) in SPD transplantation. Therefore, the Modification of Diet in Renal Disease (MDRD) equation was used to calculate the eGFR as 19 it provides at least a better accuracy of eGFR in adults to adults kidney transplant recipients than the Chronic Kidney Disease Epidemiology Collaboration equation.20,21 Proteinuria was evaluated by measuring the protein/creatinine-ratio (normal range < 11 mg/mmol) at 6 months after transplantation and annually thereafter. Blood pressure was measured at every visit and was recorded at specified time points. Kidney size was measured immediately after transplantation and 12 months later by ultrasound.
Evaluation of Renal Biopsy
Surveillance biopsies as well as clinical indicated biopsies were evaluated in terms of early hyperfiltration injury defined as mesangial matrix expansion, mesangial hypercellularity, focal segmental glomerulosclerosis or irregular lamination and splintering of the glomerular basement membrane (GBM) on electronic microscopy (EM) picture. 17 The pathological diagnosis of rejection of renal allograft was in accord with the most recent Banff criteria at the time of the study performance.22,23 The evaluation was performed unblinded to donor type.
Statistical Analysis
Statistical analyses were performed using STATA/MP v.11.0 (Stata Corp, College Station, TX). Discrete variables were expressed as counts (percentage). Non-normally distributed continuous variables were expressed as median and range. For categorical data, we used Fisher exact or Pearson chi-square test. To test equality of median between 2 groups, we used the K-sample equality-of-medians test. Graft survival rates were estimated using Kaplan-Meier analysis, and statistical comparisons of survival curves were done by log-rank test. A 2-tailed P value of < 0.05 was considered statistically significant and confidence intervals (CI) are 95% CI.
Results
Outcomes after SPD (n = 47) were compared with those after SCD (n = 153). Donor and recipient characteristics of both groups are summarized in Table 1. The median recipient weight and BMI were significantly lower in the SPD group compared with the SCD group (weight: 61 kg vs 76 kg, P < 0.001; BMI 22 kg/m2 vs 25 kg/m2, P < 0.001). Median kidney length was significant less in SPD group compared with SCD group (7.0 cm vs 11.3 cm, P < 0.001). Median cold ischemia time (CIT) was significant longer in the SPD compared with the SCD group because most of these grafts came from other surrounding European countries, due to lack of a transplant program for SPD into adults in these countries (13.9 h vs 8.5 h, P < .001). We observed a similar incidence of delayed graft function between the SPD (23%) and SCD (24%) (P = .9). The median follow-up time was 2.3 (5 days-12.1 years) years.
Baseline Characteristics of the SCD and SPD Group.

Note. SCD = standard criteria donors; SPD = small pediatric donor; BMI, body mass index; D/R BSA = Donor/Recipient body surface area ratio; cPRA = calculated panel reactive antibodies; CNI = calcineurin inhibitor.
Overall, death-censored graft survival and patient survival were equal between the SPD and SCD group (P = .28 and P = .09, respectively). Compared with the SCD group, median graft function according to the MDRD formula at 1 year was numerically higher in the SPD group (eGFR of 69.7 versus 60.2 ml/min/1.73m2, P = .15), and during follow-up (eGFR of 80 ml/min/1.73m2 versus 55 ml/min/1.73m2, P = .002) (Figure 1). In a subgroup analysis comparing recipients of SPD with recipients of SCD with the same selection criteria in terms of BMI < 30 kg/m2 and at least 1 year follow-up, we still found a numerically higher median graft function in the SPD group at 1 year (eGFR of 69.9 versus 59.7 ml/min/1.73m2, P = .053) and during follow-up (eGFR of 91 versus 57 ml/min/1.73m2, P < .0001).
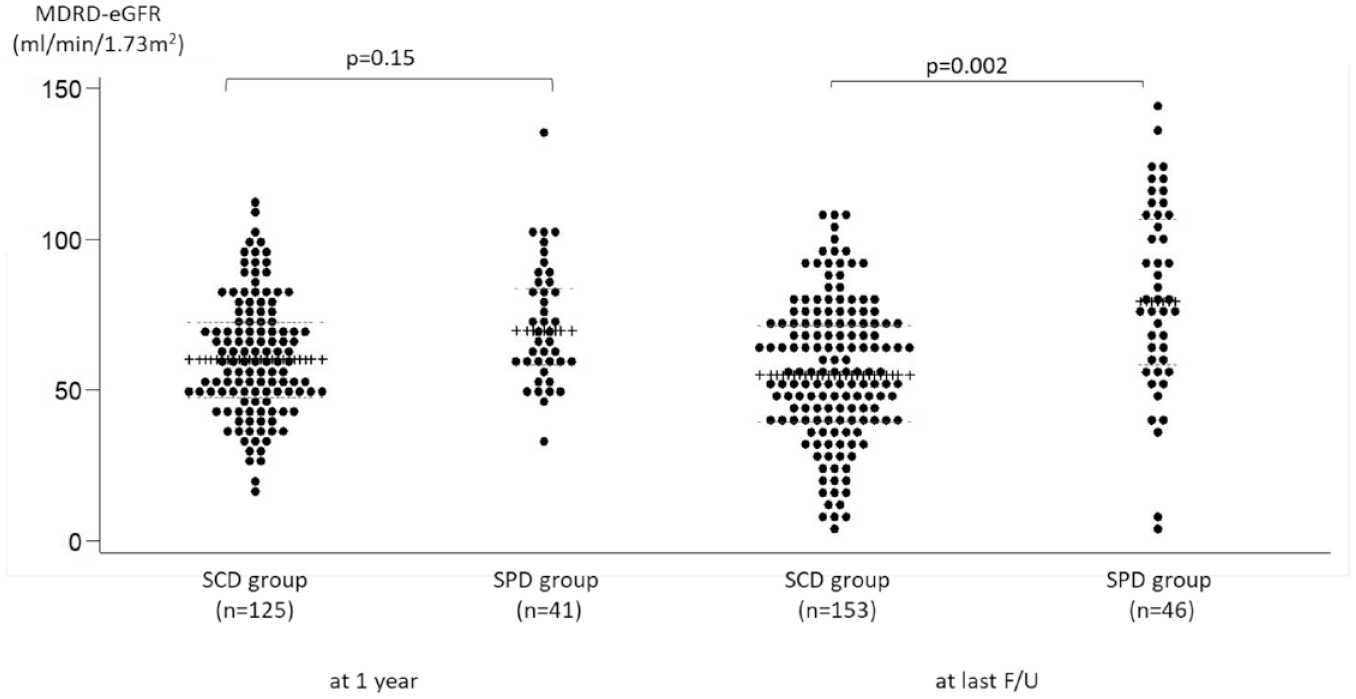
Graft function defined by the MDRD formula at 1 year and end of follow-up within the 2 groups.
In an another subgroup analysis comparing SPD with recipients of SCD with an age <50 years (n = 98), we still found a higher eGFR at the end of follow-up (eGFR 80 ml/min/1.73 m2 versus 63 ml/min/1.73 m2, P = .005, respectively, data not shown).
Median proteinuria (protein/creatinine ratio) was equal between the SPD group (11 mg/mmol) and SCD group (13 mg/mmol) at the end of follow-up (P = .7) (Figure 2). In the SPD group, only 4 patients developed macroalbuminuria during follow-up. Two developed macroalbuminuria due to recurrence of native kidney disease, 1 patient revealed severe focal segmental glomuerlosclerosis (FSGS), and 1 patient suffered from poorly controlled hypertension. In total, we performed 105 renal biopsies in 42 of the SPD group (ie, 78 surveillance biopsies and 27 clinical indicated biopsies).

The protein to creatinine ratio (mg/mmol) at the end of follow-up stratified by the 2 groups.
We found mesangial matrix expansion in 23/42 (55%) patients, mesangial hypercellularity in 13/42 (31%) patients, and FSGS in 3/42 (7%) patients. In patients with clinical suspicion of significant hyperfiltration injury whenever possible, EM was performed (n = 29). Eight of these (28%) showed splintering of GBM (Table 2). There were 4 patients with recurrence of native kidney disease (3 with an IgA nephritis and 1 with oxalosis due to short bowel disease). Other pathologies were de novo nephrocalcinosis (n = 5) and de novo thrombotic microangiopathy (n = 2). In summary, a total of 23 patients (55%) showed histological signs of early hyperfiltration injury.
Type and Incidence of Histological Signs of Hyperfiltration Injury in Allograft Biopsies in SPD.

Note. FSGS = focal segmental glomuerlosclerosis; EM = electronic-microscopy; GBM = glomerular basement membrane.
Incidence per patient with allograft biopsy and available.
In 135 of SCD patients with an allograft biopsy, 16 (12%) showed histological signs of early hyperfiltration injury, namely 6% (8/135) FSGS, 5% (7/135) mesangial matrix expansion, 3% (4/135) mesangial hypercellularity, and none splintering of GBM.
The median blood pressure was 127/74 mmHg at month 3, 131/78 mmHg at months 6, 132/75 mmHg at months 12, and 133/78 mmHg at last follow-up. The patients were on a median of 1.78 blood pressure medications (range: 0-5). There were 77% of patient on a RAAS-Inhibition. Only single agent was used for RAAS-Inhibition.
The overall risk of any clinical or subclinical rejection was equal between the SPD and SCD group (P = .15 and P = .85, respectively). Also the type of rejection episodes (antibody mediated rejection and T -cell mediated rejection) were equal (P = .37 and P = .23, respectively, data not shown). During follow-up, there was a continuing growth of kidney size observed in the SPD group with a median length growth by 28.6% from 7.0 cm up to 9.0 cm. We observed equal incidence of surgical complications within the first year postoperative (P = .94, data not shown).
Discussion
In our single center retrospective study, we found an equal graft function at 1 year with a steady rise of the GFR during follow-up leading to a significantly higher eGFR in the SPD group compared with the SCD group (eGFR of 80 ml/min/1.73 m2 versus 55 ml/min/1.73 m2, P = .002). This observation reinforces our strategy to use single kidney from pediatric donors for transplantation even in very small donors. In the past, there have been concerns about a reduced long-term allograft function caused by hyperfiltration injury in SPD, with possible low or insufficient nephron mass. To reduce the main risk factor for hyperfiltration injury namely, the donor recipients weight mismatch, we selected only recipients for SPD if the BMI was < 30 kg/m2 and a maximum body weight of 100 kg. Consequently, the BMI in the SPD group was significant lower (22 kg/m2 vs. 25 kg/m2, P < .001). But even in the subgroup analysis with SCD with the same selection criteria in terms of BMI < 30 kg/m2 and a follow-up of at least 1 year, we found a significantly higher eGFR in the SPD group compared with the SCD group at 1 year (eGFR of 69.9 versus 59.7 ml/min/1.73m2, P = .053), and during follow-up (eGFR of 91 versus 57 ml/min/1.73m2, P < .0001). In this subgroup analysis, despite the same selection criteria of BMI < 30 kg/m2, BMI in the SPD group still remained significant lower (22.3 kg/m2 vs 24.7 kg/m2, P = .05). In addition, we also selected recipients without history of severe hypertension and on
Allograft adaption with allograft hypertrophy is a well-known phenomenon in SPD. Basiri et al 25 showed that there is a substantial growth in kidney size during the first 12 months with a lower additional growth between months 12 to 18 in SPD. They observed a median kidney length growth of 37 mm during the first 12 months, with further rise to 43 mm until months 18. We found also an allograft hypertrophy in our SPD group, but the median kidney growth was slightly lower with 20 mm during the first 12 months. This hypertrophic adaption in single kidney transplantation has also seen in kidneys from adult donors but it is significant less in these patients and the adaption is seen only in the first few months after transplantation, whereas in pediatric donors the adaptive capacity is seen up to 6 years after transplantation. 26 In the literature, there are several data reporting poorer outcomes in transplantation with significant body weight mismatch.10,11,27 This may not hold true in pediatric donors since the adaptive capacity is able to compensate the size mismatch. 28 In fact, the adaptive capacity of pediatric kidneys is most probably the main factor for the comparable results in recipient of SPD with even higher BMI (>30 kg/m2) or body weight > 100 kg. 29 The permitted upper limit of the recipient’s BMI or body weight is still not defined and requires further investigations.
CIT was significant longer in SPD group (13.9 h vs 8.5 h, P < .001). Kayler et al 30 found a very small but significant impact of CIT on graft survival in kidney transplant from small pediatric donors only if the CIT exceeded 18 hours with a HR of 1.37 (CI 1.04-1.81). Therefore, they suggested that ischemic injury is likely a reversible lesion especially in pediatric kidneys. In adult donors, the effect of CIT on the outcome in terms of graft survival and delayed graft function is more pronounced and starts already if CIT exceeds 14 hours. 31 Similarly, Hansson et al 32 observed an increase of 8% of graft failure per any hour of increase in CIT.
Pediatric kidney transplantation is associated with elevated risk of surgical complications due to small vessel and ureter size with the risk of thrombotic complications, renal artery stenosis and urinary leaks.33,34 We found an equal low surgical complications rate in our SPD compared with SCD.
This study has some limitations. First, it is a single center and retrospective observational study with small sample size. Second, the good clinical outcome was achieved in a well-selected population of recipients with low BMI, low immunological risk, and well-controlled hypertension and was not compared with equal selected group of recipients, especially in terms of BMI. So, there is a uncertainty whether the observed equal death-censored graft survival and allograft function would be observed in a well-matched control group. Third, the transplantations were performed by dedicated vascular surgeons with improved knowledge and handling of technical complications. Fourth, the evaluation of the allograft biopsies was not blinded to donor type, so there is a risk of overdiagnosis of hyperfiltration injuries in these recipients. As we did not perform an allograft biopsy at the time of transplantation, there might be an overestimation of hyperfiltration injury in SCD as we could not rule out pre-existing hyperfiltration injury, whereas it is very unlikely in pediatric donors. Fifth, there might be a lower risk of rejection episodes leading to better allograft outcomes in SPD compared with SCD due to selection of low immunological risk in these recipients, although the SCD recipients happened to have the same low immunological risks (cPRA ≥ 10% was not different between the groups, P = .2).
Whether these good results could be achieved in an unselected population and not experience transplant team is unknown.
In conclusion, early clinical and histological signs of hyperfiltration injury is frequent in SPD, even in a well-selected recipient population. Despite the hyperfiltration injury, we observed an excellent long-term allograft survival and superior long-term allograft function in SPD compared with SCD. Our observation supports the concept of high adaptive capacity of pediatric kidneys and the importance of early rigorous blood pressure control in these patients.
Footnotes
List of Abbreviations
BMI, body mass index; CIT, cold ischemia time; eGFR, estimated glomerular filtration rate; EM, electronic microscopy picture; FSGS, focal segmental glomerular sclerosis; GBM, glomerular basement membrane; HLA, human leukocyte antigen system; MDRD, modification of diet in renal disease; MMF, mycophenolate mofetil; mTOR, mammalian target of rapamycin; RAAS, renin angiotensin aldosterone system; SCD, standard criteria deceased donor; SPD, small pediatric deceased donor.
Ethics Approval and Consent to Participate
This retrospective study was approved by the ethic committee of Northwestern and Central Switzerland (Project ID 2018_01571).
Consent for Publication
Not applicable.
Availability of Data and Materials
Data will be available upon request from the corresponding author.
Author Contributions
Participated in the research design: F.B., Y.H., S.S., and P.A. Participated in the writing of the paper: F.B., Y.H., and P.A. Participated in the performance of the paper: F.B., Y.H., A.G., C.W., P.H., G.H., H.H., L.G., J.S., S.S., and P.A. Participated in the data analysis: F.B., Y.H., and P.A.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
