Abstract
Background:
Current guidelines recommend that living kidney donors receive lifelong annual follow-up care to monitor kidney health. In the United States, the reporting of complete clinical and laboratory data for kidney donors has been mandated for the first 2 years post-donation; however, the long-term impact of early guideline-concordant care remains unclear.
Objective:
The primary objective of this study was to compare long-term post-donation follow-up care and clinical outcomes of living kidney donors with and without early guideline-concordant follow-up care.
Design:
Retrospective, population-based cohort study.
Setting:
Linked health care databases were used to identify kidney donors in Alberta, Canada.
Patients:
Four hundred sixty living kidney donors who underwent nephrectomy between 2002 and 2013.
Measurements:
The primary outcome was continued annual follow-up at 5 and 10 years (adjusted odds ratio with 95% confidence interval, LCLaORUCL). Secondary outcomes included mean change in estimated glomerular filtration rate (eGFR) over time and rates of all-cause hospitalization.
Methods:
We compared long-term follow-up and clinical outcomes for donors with and without early guideline-concordant care, defined as annual physician visit and serum creatinine and albuminuria measurement for the first 2 years post-donation.
Results:
Of the 460 donors included in this study, 187 (41%) had clinical and laboratory evidence of guideline-concordant follow-up care throughout the first 2 years post-donation. The odds of receiving annual follow-up for donors without early guideline-concordant care were 76% lower at 5 years (aOR 0.180.240.32) and 68% lower at 10 years (aOR 0.230.320.46) compared with donors with early care. The odds of continuing follow-up remained stable over time for both groups. Early guideline-concordant follow-up care did not appear to substantially influence eGFR or hospitalization rates over the longer term.
Limitations:
We were unable to confirm whether the lack of physician visits or laboratory data in certain donors was due to physician or patient decisions.
Conclusions:
Although policies directed toward improving early donor follow-up may encourage continued follow-up, additional strategies may be necessary to mitigate long-term donor risks.
Introduction
For eligible patients with end-stage kidney disease, transplantation is the preferred form of kidney replacement therapy due to its association with improved patient survival, better quality of life, and lower costs to the health care system.1,2 Living donor kidney transplants offer additional benefits compared with deceased donor transplants, including longer graft survival, potential to shorten or avoid dialysis (pre-emptive transplant), and reduced risk of delayed graft function due to less cold ischemia time. 3 The practice of living organ donation is grounded on the principle that donor nephrectomy has minimal perioperative and long-term risks with appropriate evaluation and care, and that well-informed donors choose to accept these risks based on an altruistic motivation to help patients in need. However, the paradigm of post-donation risk has evolved in recent years, as some studies have reported an increased risk of kidney failure, pregnancy complications, and gout in living kidney donors compared with healthy, matched nondonor controls.4 -9
To potentially mitigate post-donation risks, the Kidney Disease Improving Global Outcomes (KDIGO) “Clinical Practice Guideline on the Evaluation and Care of Living Kidney Donors” recommends that donors have lifelong annual physician follow-up care to monitor kidney health, including serum creatinine and albuminuria testing. 10 Despite this, many centers report that the majority of donors have not received annual follow-up care, possibly because the guideline recommendation was “not graded” due to a lack of evidence from eligible studies.11,12 In 2013, the Organ Procurement and Transplant Network (OPTN) in the United States mandated the reporting of complete and timely clinical and laboratory data for living donors at 6, 12, and 24 months post-donation. 13 Although this led to an improvement in the proportion of early follow-up data collected nationwide, 14 no mandatory regulations are in place for follow-up care beyond the first 2 years after donation in the United States. In Canada, there are no national standards for post-donation follow-up care, and it is unclear whether early mandated follow-up care results in improved long-term outcomes. To better inform this issue, we conducted a retrospective cohort study of living kidney donors using health care administrative databases in Alberta, Canada. Our primary goal was to evaluate the association between early post-donation guideline-concordant follow-up care with subsequent long-term follow-up care and outcomes.
Methods
Design and Setting
We conducted a population-based, retrospective cohort study using linked health care databases within the Alberta Kidney Disease Network (AKDN). 15 More than 99% of Alberta residents are registered with Alberta Health and have universal access to hospital care and physician services. We followed guidelines for the reporting of observational studies (Supplemental Table S1) and a protocol approved by the research ethics boards at the University of Alberta and the University of Calgary, with a waiver of patient consent.
Data Sources
We ascertained baseline characteristics, covariate information, and outcome data from the AKDN database records (Supplemental Table S2). The Alberta Health database contains information on demographics, vital statistics, and diagnostic and procedural information for inpatient and outpatient physician services. We linked these data sources to a provincial laboratory repository via unique, encoded, patient identifiers. The serum creatinine measurements obtained in our databases have been standardized across provincial laboratories over time, reducing interlaboratory variation in measurements. 15 These databases have been previously used for research on health outcomes and services.12,16
Population
We identified all adult living kidney donors (≥18 years old) who underwent donor nephrectomy between May 1, 2002, and September 30, 2013, in Alberta, Canada (Supplemental Figure S1). Living kidney donors were identified using an algorithm that required the presence of 1 diagnostic code for kidney donation (International Classification of Diseases [ICD], Tenth Revision: Z52.4) and 1 procedural code for kidney procurement or excision (Canadian Classification of Health Interventions [CCI]: 1.PC.58, 1.PC.89, or 1.PC.91) (Supplemental Table S2). Similar codes have been used in prior studies to identify living kidney donors.12,16,17 We validated this algorithm and found it to have a sensitivity of 97% and a positive predictive value of 90% when compared with the gold standard of living kidney donor identification by the provincial tissue and organ agency, and verification through manual perioperative chart review. 18
We excluded out-of-province donors and a small proportion of donors (<3%) with missing data (eg, sex or date of birth). To avoid misclassification of patients who received a kidney from a living donor, we excluded anyone with evidence of prior dialysis, transplant, or an estimated glomerular filtration rate (eGFR) <60 mL/min/1.73 m2 prior to donation. We excluded donors who did not have an outpatient serum creatinine measurement recorded in our data sources in the year prior to donation. Donors were followed from 2.5 years after their donation date until the first of death, development of end-stage kidney disease (defined as receipt of maintenance dialysis or a kidney transplant), emigration from the province, or end of study period (March 31, 2019), ensuring that all donors had at least 3 years of potential follow-up time for outcome ascertainment.
Early Guideline-Concordant Follow-up Care
Living kidney donors were categorized based on their early follow-up status. Early guideline-concordant follow-up care was defined as evidence of annual outpatient physician visits, serum creatinine measurements, and albuminuria measurements at both the first- and second-year anniversary date (±6 months). We did not consider physician visits or laboratory values in the first 6 months after donation, as these are usually related to postoperative monitoring. Urinalysis, urine protein-creatinine ratio measurement, or urine albumin-creatinine ratio measurement qualified as albuminuria measurements in any given year.
Baseline Characteristics
Baseline demographics at the time of donation were determined from Alberta Health administrative data files. Postal codes were linked to the Canadian Census using the Postal Code Conversion file to determine median neighborhood household income quintile (level 5 being the highest) as well as rural versus urban location of residence and distance from the transplant center, as previously described. 19 The presence of 1 or more diagnostic codes in the 3 years prior to donation was used to identify comorbidities using validated ICD, Ninth Revision, Clinical Modification (ICD-9-CM) and ICD-10 coding algorithms applied to physician claims and hospitalization data (Supplemental Table S2). Demographic data were complete except for income quintile (<1% missing was imputed as middle income, that is, level 3).
Outcomes
We compared outcomes between donors with and without early guideline-concordant follow-up care. The primary outcome was evidence of continued guideline-concordant follow-up care annually (±6 months), beyond the first 2.5 years post-donation. We assessed trends in follow-up care over the first decade following donation. In additional analyses, we examined the components of the primary outcome (physician visits, serum creatinine measurements, albuminuria measurements) individually. We also evaluated the change in eGFR over time (in mL/min/1.73 m2 per year) using all available eGFR measurements from 2.5 years post-donation onwards. 16 The eGFR was estimated using the CKD-EPI (Chronic Kidney Disease-Epidemiology Collaboration) equation. 20 Finally, we compared all-cause hospitalizations between donors with and without early guideline-concordant care. For this analysis, we considered aggregate hospitalization counts (ie, each donor could have multiple hospitalizations if they were separated in time).
Statistical Analyses
We compared baseline characteristics of donors with guideline-concordant follow-up care versus donors without using χ2 or Fisher exact tests and Kruskal-Wallis tests. Associations of early and subsequent guideline-concordant follow-up care were assessed using mixed-effects logistic regression and included fixed effects for exposure (ie, donors with and without early guideline-concordant care) and follow-up year. We included a random, individual-specific intercept and adjusted for age, sex, socioeconomic status, residence, distance to transplant center, donation era, predonation eGFR, predonation albuminuria, hypertension, asthma, and chronic pulmonary disease (adjusted odds ratio with upper and lower confidence limits, LCLaORUCL). We modeled the change in eGFR over time using linear mixed-effects models (LCLeGFRUCL). We included a random, individual-specific intercept and slope with an unstructured covariance between the random effects to account for correlation among measurements within the same individual. We plotted eGFR over time using measurements that were within 6 months for the remaining timepoints (annually until year 10 post-donation). We used negative binomial regression to compare rates of hospital admissions (adjusted rate ratio, LCLaRRUCL) by including in each count model an offset term representing the log of the time at risk. Individuals were considered not at risk while hospitalized for the outcome of interest. A P value of <.05 was used to define statistical significance. Statistical analyses were performed using Stata MP 13.1 (Stata Corporation, College Station, Texas).
Results
Baseline Characteristics
Between 2002 and 2013, there were 523 living kidney donor nephrectomies performed in Alberta. Of these, 460 met study the inclusion criteria (Supplemental Figure S1). Less than half of donors received early guideline-concordant follow-up care (n = 187, 41%). The median follow-up time after donation for all donors was 10.7 (interquartile range [IQR], 7.6-13.5) years, with a maximum follow-up of 16.7 years. Compared with donors with early follow-up care, donors without early follow-up care were younger (median age, 40 vs 46 years; P < .001) (Table 1). Donors without early follow-up also lived farther from the transplant center (median distance, 55 vs 30 km; P < .001) and had a higher eGFR prior to donation (median eGFR, 103 vs 98 mL/min/1.73 m2; P = .01). There was no statistically significant difference in sex, socioeconomic status, predonation hypertension, or lung disease between donors with and without early follow-up care.
Baseline Characteristics at the Time of Donation of Living Kidney Donors With and Without Early Guideline-Concordant Care.

Note. Data are presented as median [interquartile range] or as count (%). eGFR = estimated glomerular filtration rate.
Continued Guideline-Concordant Follow-up Care
Five years after donation, evidence of continued complete follow-up care was present in 32% of donors without early guideline-concordant care and 64% of donors with early care. Donors without early guideline-concordant follow-up care were less likely to have subsequent follow-up care in adjusted analyses (Figure 1). The odds of having complete annual follow-up for donors without early care were 76% lower at 5 years after donation (aOR 0.180.240.32, P < .001) and 68% lower at 10 years after donation (aOR 0.230.320.46, P < .001) relative to donors with early care. Results were similar when assessing the individual components of follow-up care (physician visits, serum creatinine measurements, albuminuria measurements) (Table 2). Evidence of annual outpatient physician follow-up was consistently higher than annual serum creatinine and albuminuria measurements.
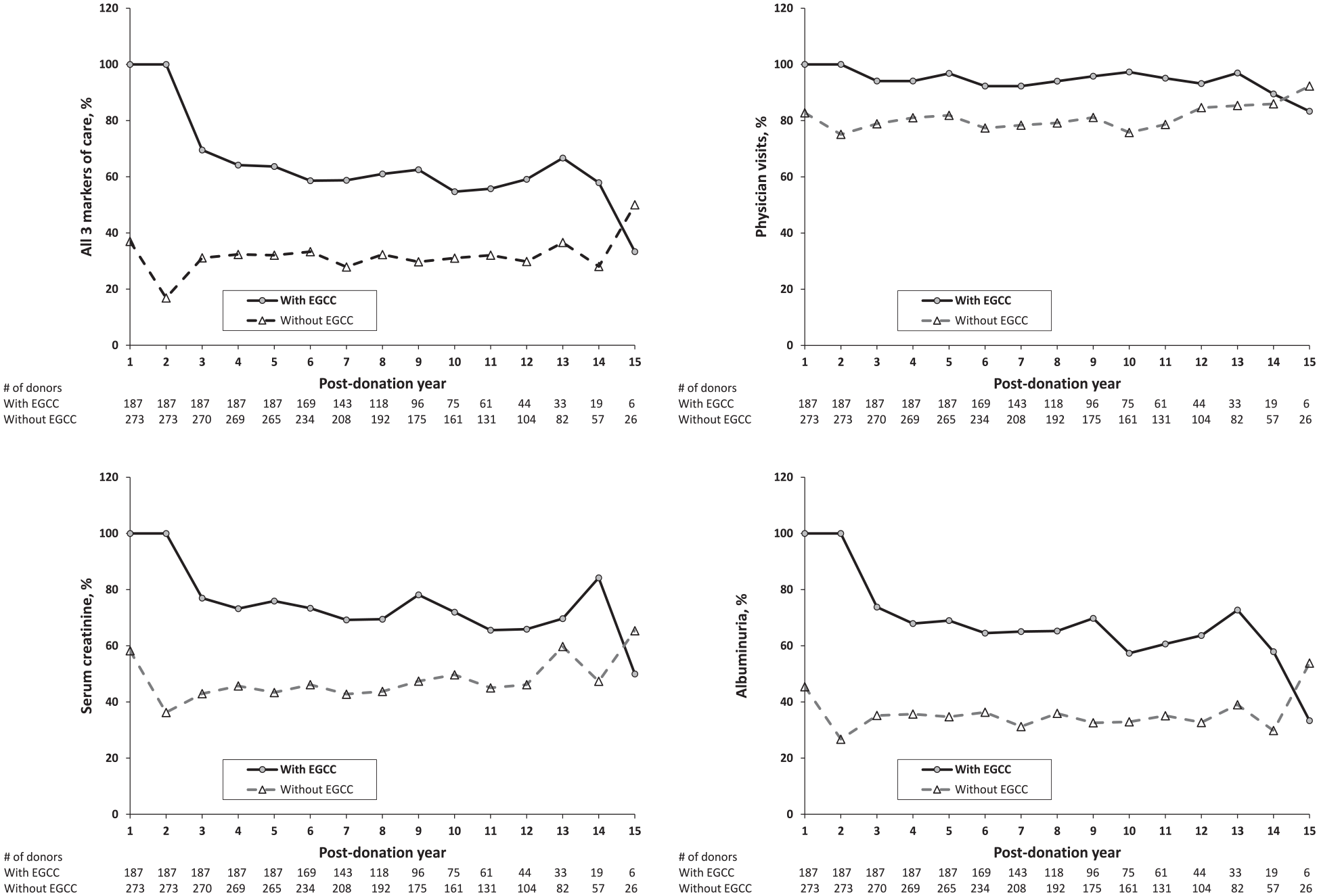
Proportion of living kidney donors with follow-up care during each post-donation year stratified by those with (solid line) and without (dotted line) EGCC.
Outcomes in Living Kidney Donors With and Without Early Guideline-Concordant Care.
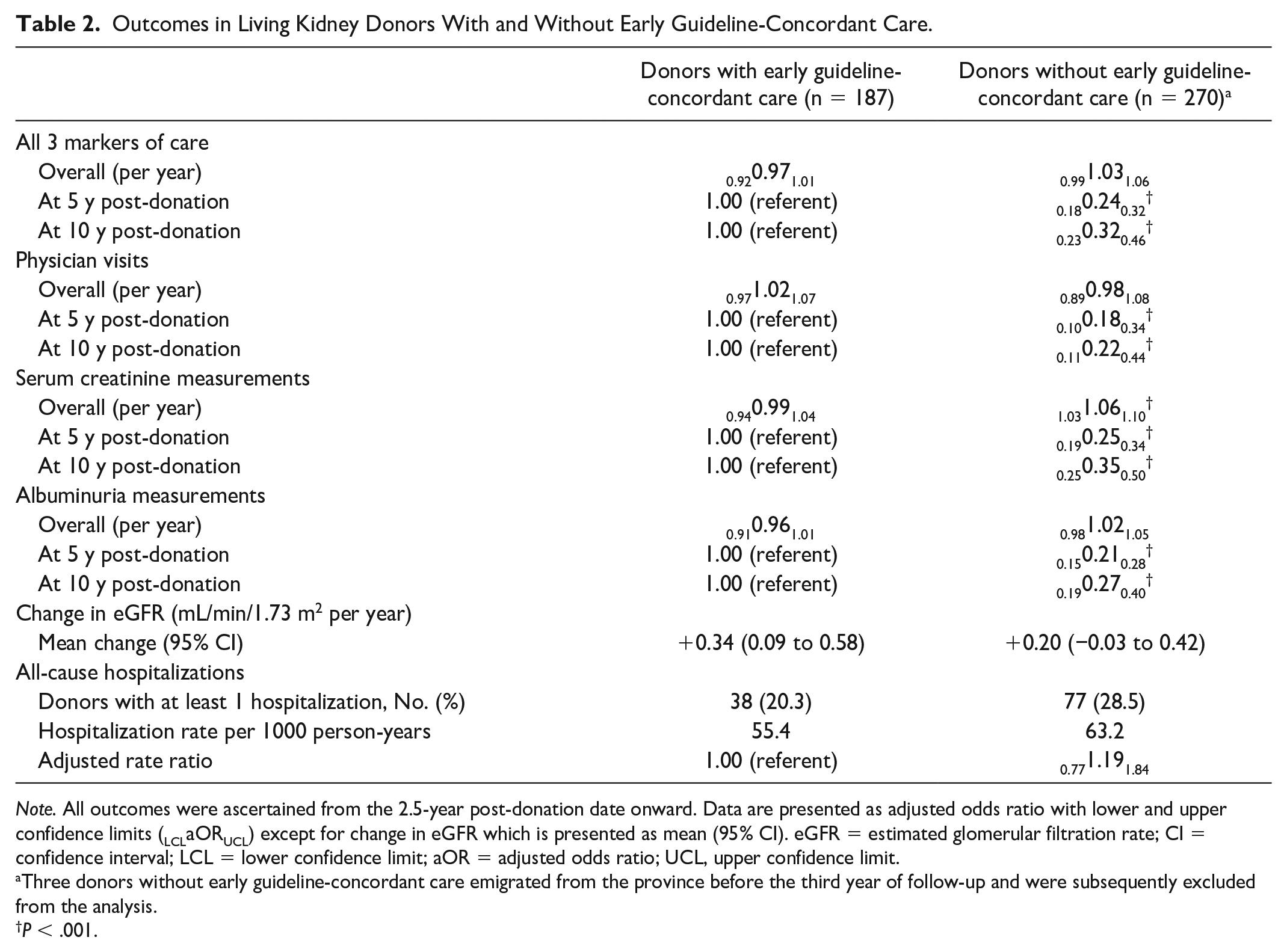
Note. All outcomes were ascertained from the 2.5-year post-donation date onward. Data are presented as adjusted odds ratio with lower and upper confidence limits (LCLaORUCL) except for change in eGFR which is presented as mean (95% CI). eGFR = estimated glomerular filtration rate; CI = confidence interval; LCL = lower confidence limit; aOR = adjusted odds ratio; UCL, upper confidence limit.
Three donors without early guideline-concordant care emigrated from the province before the third year of follow-up and were subsequently excluded from the analysis.
P < .001.
For donors with and without early guideline-concordant follow-up care, the proportion of donors with evidence of continued complete care remained stable over time. The odds of continuing follow-up decreased by 3% every year for donors with early follow-up (aOR 0.920.971.01, P = .2), and the odds of continued follow-up increased by 3% every year for donors without early follow-up (aOR 0.991.031.06, P =.1); neither was a statistically significant change (Table 2).
Rate of Change in Kidney Function
The proportion of donors with at least 1 serum creatinine measurement from 2.5 years post-donation was higher in donors with early care compared with those without (99% vs 89%) as was the median [IQR] number of measurements (8 [5-14] vs 6 [3-11]). Most of the serum creatinine measurements were done in the outpatient setting (82% for donors with early care vs 76% for donors without).
The mean eGFR measurements over time for donors with and without early follow-up care are presented in Figure 2. Overall, the eGFR increased slightly over time for donors with early follow-up care (0.090.340.58 mL/min/1.73 m2 per year, P =.007) and remained stable in those without early follow-up care (−0.030.200.42 mL/min/1.73 m2 per year, P = .08), although the latter was not statistically significant. Results were similar when we limited the analysis to donors with at least 3 eGFR measurements in subsequent follow-up (results not shown).

Mean eGFR over time in living kidney donors with (solid line) and without (dotted line) EGCC.
All-Cause Hospitalizations
Most donors did not have evidence of hospitalization beyond their second post-donation year (n = 345, 75%). Hospitalization rates did not differ significantly for donors without early guideline-concordant care compared with donors with early care (aRR 0.771.191.84, P = .4).
Discussion
In this retrospective cohort study, we compared long-term outcomes of 187 living kidney donors with early guideline-concordant follow-up care with 273 donors without early care over a median post-donation period of 10.7 years. We found that the odds of having annual follow-up for donors without early guideline-concordant care were 76% lower at 5 years and 68% lower at 10 years relative to donors with early follow-up care. The findings from our study suggest that early guideline-concordant care is associated with improved subsequent donor follow-up.
Many countries have attempted to improve follow-up care for living kidney donors through regulatory policies. The OPTN’s mandated policy led to an improvement in the proportion of early donor follow-up data collected nationwide from 33% pre-policy to 54% post-policy. 21 While there is no mandated policy for follow-up beyond the first 2 years post-donation, the Scientific Registry of Transplant Recipients has begun a registry for lifelong donor follow-up; this project is in an early stage and data on its feasibility and success are evolving. 22 In Switzerland, transplant centers organize lifelong follow-up for donors starting at 1 year and then biennially whereby each donor receives a package before each visit with a health questionnaire and a prepaid envelope with blood and urine tubes to facilitate laboratory collection. 23 Similarly, in the United Kingdom, transplant centers arrange immediate and lifelong follow-up for living donors with the donor’s transplant center, referring nephrologist, or primary care provider to maximize donor convenience. 24
The findings from our study fill an important knowledge gap in the current literature and may have useful implications for future policy development. There are currently no nationwide policies in Canada to standardize follow-up care for living kidney donors, in part, due to the lack of existing evidence of beneficial effect. Despite these uncertainties, many have argued that the medical community has an ethical obligation to protect these altruistic individuals through the provision of diligent long-term follow-up care to allow for the prevention and early detection of treatable medical comorbidities. 12 There are, however, barriers to enacting these policies that should be considered. Due to the lack of evidence that long-term follow-up improves outcomes, there is potential for harm to donors by identifying them as having a medical condition that requires regular follow-up and imposing inconveniences that do not necessarily provide benefit to their health and well-being. This especially holds true for donors of low socioeconomic status or those who live at farther distances from their transplant center for whom the inconveniences are more significant. Virtual and phone-based follow-up are becoming increasingly common to circumvent these barriers. 25 Donor attitudes regarding the necessity of follow-up also impact compliance and may be influenced by communication with transplant centers. 26 Finally, systematic restraints such as limited health care resources, infrastructure, and funds pose additional barriers to enacting these policies. 27
Although early guideline-concordant care was associated with improved follow-up at 5 and 10 years post-donation, we did not find meaningful differences in the change in eGFR over time or hospitalization rates between the 2 groups. From 2.5 years post-donation, there were small annual increases in eGFR for donors with and without early follow-up care (+0.34 and +0.20 mL/min/1.73 m2 per year, respectively). These results are similar to our previous study of 604 living kidney donors (2002-2016) in which we reported that from 6 weeks post-donation, the eGFR increased by +0.35 mL/min/1.73 m2 per year. 16 The annual change in eGFR over time was not significantly different in subgroups based on age, socioeconomic status, distance to the transplant center, predonation eGFR, or predonation hypertension history. Similarly, in a prospective US study by Kasiske et al 28 of 133 living kidney donors, the change in measured glomerular filtration rate (by iohexol clearance) from 6 months to 9 years post-donation was +0.02 mL/min/1.73 m2 per year, and the results were similar in the analysis that included only donors who completed follow-up visits at 9 years. Thus, rigorous follow-up, either early on or within the first decade of donation, does not appear to significantly affect the change in eGFR over time in living kidney donors.
Our study has several strengths including a provincial analysis of the impact of early guideline-concordant care of living kidney donors on long-term outcomes. There are, however, limitations to this study that are worth noting. First, this was a retrospective, observational study, and the possibility of residual confounding must be considered. In addition, our results may not be generalizable to other countries or regions that do not have a similar universal health care system. We also lacked data on certain donor characteristics that may affect follow-up, including smoking, blood pressure control, and body mass index, as well as transplant-related characteristics, such as donor-recipient relationship; however, we were able to identify and control for important demographics and comorbidities commonly associated with follow-up care. We also looked at outpatient physician visits as a surrogate for blood pressure measurements and other markers of care, including preventive health delivery. We are unable to confirm whether the lack of physician visits or laboratory data in certain donors was due to physician or patient decisions. Finally, we can only describe associations and cannot conclude that interventions aimed at increasing follow-up care will improve donor outcomes.
The results from our study show that early post-donation follow-up care is associated with a higher likelihood of subsequent follow-up, but does not have a meaningful impact on long-term eGFR or hospitalization rates. Therefore, mandating early follow-up could serve as a useful policy to encourage continued follow-up, but may not sufficiently mitigate long-term risks. Further research is needed to determine the impact of guideline-concordant follow-up care on long-term kidney function and adverse outcomes.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581231158067 – Supplemental material for Long-Term Outcomes for Living Kidney Donors With Early Guideline-Concordant Follow-up Care: A Retrospective Cohort Study
Supplemental material, sj-docx-1-cjk-10.1177_20543581231158067 for Long-Term Outcomes for Living Kidney Donors With Early Guideline-Concordant Follow-up Care: A Retrospective Cohort Study by Anisha Dhalla, Anita Lloyd, Krista L. Lentine, Amit X. Garg, Robert R. Quinn, Pietro Ravani, Scott W. Klarenbach, Brenda R. Hemmelgarn, Uchenna Ibelo and Ngan N. Lam in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
This study is based in part on data provided by Alberta Health and Alberta Health Services. The interpretation and conclusions contained herein are those of the researchers and do not necessarily represent the views of the Government of Alberta or Alberta Health Services. Neither the Government of Alberta nor Alberta Health or Alberta Health Services express any opinion in relation to this study.
List of Abbreviations
AKDN, Alberta Kidney Disease Network; LCLaORUCL, adjusted odds ratio with lower and upper confidence limits; LCLaRRUCL, adjusted rate ration with lower and upper confidence limits; CCI, Canadian Classification of Health Interventions; CKD-EPI, Chronic Kidney Disease-Epidemiology Collaboration CI, confidence interval; EGCC, early guideline-concordant care; eGFR, estimated glomerular filtration rate; LCLeGFRUCL, estimated glomerular filtration rate with lower and upper confidence limits; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification; ICD-10, International Statistical Classification of Diseases, Tenth Revision; IQR, interquartile range; KDIGO, Kidney Disease Improving Global Outcomes; OPTN, Organ Procurement and Transplant Network.
Ethics Approval and Consent to Participate
This study was approved by the institutional review board at the University of Alberta and the University of Calgary.
Consent for Publication
All authors consent to the publication of this study.
Availability of Data and Materials
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Canadian Institutes of Health Research (CIHR) Project Grant (391688). K.L.L. was supported by the Mid-America Transplant/Jane A Becham Endowed Chair in Transplantation. A.X.G. is the Ontario Renal Network Provincial Medical Lead for Access to Kidney Transplantation and Living Kidney Donation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
