Abstract
Purpose of Review:
To understand the impact of kidney disease in Canada and the priority areas of kidney research that can benefit from patient-oriented, precision medicine research using novel technologies.
Sources of Information:
Information was collected through discussions between health care professionals, researchers, and patient partners. Literature was compiled using search engines (PubMed, PubMed central, Medline, and Google) and data from the Canadian Organ Replacement Register.
Methods:
We reviewed the impact, prevalence, economic burden, causes of kidney disease, and priority research areas in Canada. After reviewing the priority areas for kidney research, potential avenues for future research that can integrate precision medicine initiatives for patient-oriented research were outlined.
Key Findings:
Chronic kidney disease (CKD) remains among the top causes of morbidity and mortality in the world and exerts a large financial strain on the health care system. Despite the increasing number of people with CKD, funding for basic kidney research continues to trail behind other diseases. Current funding strategies favor existing clinical treatment and patient educational strategies. The identification of genetic factors for various forms of kidney disease in the adult and pediatric populations provides mechanistic insight into disease pathogenesis. Allocation of resources and funding toward existing high-yield personalized research initiatives have the potential to significantly affect patient-oriented research outcomes but will be difficult due to a constant decline of funding for kidney research.
Limitations:
This is an overview primarily focused on Canadian-specific literature rather than a comprehensive systematic review of the literature. The scope of our findings and conclusions may not be applicable to health care systems in other countries.
Introduction
Chronic kidney disease (CKD) is a major health problem affecting approximately 4 million Canadians and 11% to 13% of the population globally.1,2 The prevalence of CKD was recently reported to be 71.9 per 1000 individuals by Bello et al 3 in the first Canadian estimation of CKD in primary care using data from 5 provinces across Canada. In 2020, over 50 000 Canadians were living with end-stage kidney disease (ESKD) (Figure 1).4,5 Excluding Quebec, there are more than 23 000 people starting renal replacement and over 1500 patients receiving a kidney transplant.4,6 The number of people receiving renal replacement therapy (RRT) worldwide exceeds 2.5 million and is expected to double to 5.4 million by 2030. 7
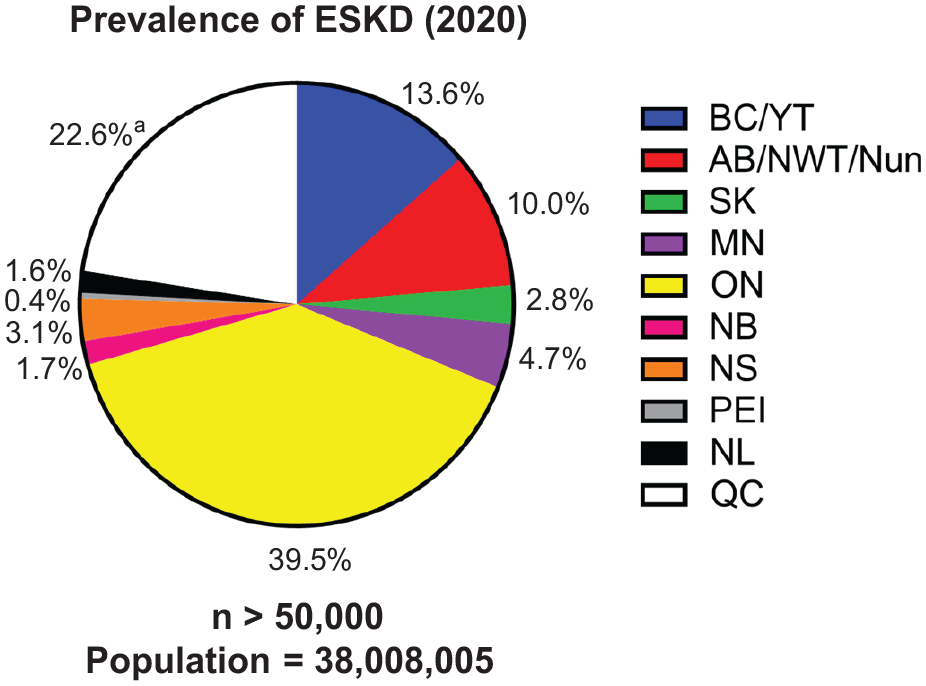
Canadian prevalence of ESKD in 2020.
Chronic kidney disease is a growing problem with no curative treatments currently available. In the 2013 Global Burden of Disease study, which included the annual assessment from 188 countries, 956 200 people were estimated to have died from CKD, one of the largest rises among the top causes of death with an increase of 134% over 23 years. 8 With an aging population and no imminent therapies available, the burden of CKD and ESKD is approaching a threshold that will be difficult for the Canadian health care system to sustain. To better understand potential areas of integration of high-yield research directions, this narrative review will summarize the current landscape of kidney health and disease (Part 1) and potential avenues to advance kidney research (Part 2) in Canada.
Methods
A review of kidney disease in Canada and the patient-oriented research funding initiatives was conducted by use of search engines (PubMed, PubMed central, Medline, and Google) as well as cumulative perspective from our discussion with other health care professions, researchers, and patient partners. We also include our respective areas of expertise in hereditary and glomerular kidney diseases. Data on CKD prevalence and causes in Canada were generated from the Canadian Organ Replacement Register (CORR), which collects data from hospital dialysis programs, transplant programs, organ procurement organizations, and independent health facilities.
Review
Part 1: The Landscape of Kidney Health And Disease in Canada
Kidney disease is a “silent” disease with less public awareness compared with other diseases such as cancer or heart disease. In just 2 decades, CKD has increased in position from 18th to the 11th leading cause of death contributing to 1.2 million deaths worldwide.9,10 The 5-year mortality rate for hemodialysis (HD) patients remains unacceptably high at approximately 50%, rivaling the mortality rates of stage IV cancer.11 -13 Notably, the health-related quality of life (QOL) of patients on dialysis is reportedly lower than for other chronic diseases, such as cystic fibrosis, cancer, asthma, or sickle cell disease. 14 Patients experience a drastic change to their QOL affecting their physical and mental health.15 -19 Improving health-related QOL among incident dialysis patients has been slow and incremental over the past 2 decades.12,20 The majority of patients on HD continue to have 3 to 4 comorbid conditions with their ESKD, a high prevalence of cardiovascular disease, and polypharmacy averaging 8 to 10 medications daily. 12 Most patients benefit with an improved survival and QOL after receiving a kidney transplant.13,21 However, the median wait time to receive a kidney transplant in Canada is 3 years and 10 months. 22 There has been a push to improve the care of patients transitioning from CKD to ESKD. In the United States, the PREPARE NOW cluster randomized control trial has been completed to study the effectiveness of pragmatic health system intervention (“Patient Centered Kidney Transition Care”).22,23
Economic burden of kidney disease
The economic burden of chronic and end-stage kidney disease is staggering, costing the national health care system more than $40 billion per year in Canada. 1 Manns et al 1 estimated that up to $260 million is paid as disability benefit payments for patients with CKD. In a recent study by the same group, the mean unadjusted cumulative 1-year health care costs (ie, cost of drugs, emergency department visits, outpatient procedures, and hospitalizations) were on average $14 634 per patient. 24 The cost of providing dialysis treatment to patients with ESKD is one of the most expensive publicly funded medical treatments of modern medicine, totaling over $1.8 billion per year in Canada.25,26 In-center HD remains the most common form of RRT for ESKD in Canada accounting for 42.6% of the prevalent ESKD patients (Figure 2).25,27,28

Proportion of renal replacement modalities in Canada.
The annual cost of the patients on HD amounts to $100 000 per patient.29,30 In Manitoba, the reported cost of annual maintenance of HD was $64 214 per year compared with home modalities such as peritoneal dialysis (PD) and home hemodialysis (HHD) costing $38 658 and $39 236, respectively. Home modalities were significantly lower than reported by other studies. 27 By comparison, the initial cost for transplantation is approximately $100 000 per year for the first year of transplantation but tapers in subsequent years at approximately $20 000 per year, thus becoming substantially less costly to the health care system than dialysis. 31
Causes of kidney disease
Chronic kidney disease is multifactorial, systemic disease with contributions from multiple genetic and environmental risk factors. 32 Diabetes and hypertension remain among the most significant contributors to CKD in adults, while congenital anomalies are the leading cause of CKD in children (Table 1).
Causes of CKD and ESKD: Adult and Pediatric Populations.

Note. CKD = chronic kidney disease; ESRD = end-stage renal disease; CAKUT = congenital anomalies of the kidney and urinary tract.
Despite over 600 genes now implicated in monogenic kidney diseases, there remains a significant proportion of patients for whom the underlying etiology cannot be established (~10%-15% of patients have CKD of unknown etiology).33,34 There remains a significant proportion of patients for whom the underlying etiology cannot be established (~10%-15% of patients have CKD of unknown etiology). The advances in molecular diagnostic medicine in recent years have helped identify an important monogenic contribution to CKD and ESKD for both children and adults. 35 While the individual genetic causes are rare, their combined disease burden is significant. With the currently available knowledge and technology, a genetic diagnosis can be established in around 20% to 30% of all cases of CKD before age 20 and in approximately 10% to 30% of adults.36,37 Although CKD and ESKD are fortunately rare occurrences in childhood, children with CKD face significant increases in morbidity and mortality that persist lifelong. The data on the epidemiology of pediatric kidney disease are limited and are likely an underestimation due to underdiagnosis and underreporting.38,39
The causes of CKD in adults are generally on the rise given the aging of the population. 40 Based on the Canadian Organ Replacement Registry (CORR), which does not include data from Quebec, the most common causes of CKD in Canada are diabetes, renovascular disease/hypertension, glomerulonephritis, and polycystic kidney disease (PKD), with prevalence of 38.0%, 12.2%, 10.2%, and 3.3%, respectively (Table 1). 41 Similar data from the Centers for Disease Control and Prevention for the United States show that the 2 most common causes are diabetes and hypertension, followed by glomerulonephritis, which account for more than two-thirds of CKD. 42
The search for genetic risk factors contributing to diabetic kidney disease (DKD), the major cause of CKD in adults, has been difficult due, in part, to factors such as heterogeneity in the type of variant, unknown number of causal variants within a gene, variant effect size, and the possibility of variants affecting protein coding or non-coding regulatory genes. 43 Recent studies have demonstrated genetic and epigenetic risks for developing DKD. The genetic susceptibility to diabetic nephropathy was reported for 2 sets of families in which both probands and siblings had diabetes mellitus now over 30 years ago. 44 Several studies have examined potential genetic factors using genome-wide association studies (GWAS) such as the Diabetic Nephropathy Collaborate Research Initiative which included almost 20 000 samples from participants with type 1 diabetes to determine the variants associated with DKD. The authors found 16 new DKD-associated loci at genome-wide significance. 45 The single-nucleotide polymorphism most significantly associated with DKD was a protective missense coding variant at COL4A3, a gene implicated in familial focal segmental glomerulosclerosis and Alport syndrome. 45 In a recent study utilizing genome-wide DNA methylation association analysis in the Chronic Renal Insufficiency Cohort study, a role of genetic variations in DNA methylation involved in mediating the genotype effect on kidney disease development, suggesting both genetic and epigenetic variations in DKD. 46
Pediatric causes of CKD are diverse and differ from those in adults (Table 1). 47 One of the major contributors to CKD in children are congenital anomalies of the kidney and urinary tract (CAKUT), which comprise embryonic disorders that arise during development and result in a spectrum of defects in the kidneys and outflow tracts, which include the ureters, the bladder, and the urethra.48 -50 The prevalence of CAKUT is estimated at 4 to 6 per 10 000 births depending on the registry, variation in sample size, method of diagnosis, and ethnic differences.51,52 Other causes include the hereditary nephropathies and glomerulonephritis, which include cystic kidney disease (autosomal dominant PKD and autosomal recessive cystic kidney disease) and the renal ciliopathies.38,53,54 As observed for adults, the etiology of CKD in children can vary by age, race, and geography, with children younger than 12 years more likely to have CAKUT, while glomerular-based disease, such as focal segmental glomerulosclerosis, is more likely to affect black adolescents. 38 Finally, in recent years, obesity has become a major new health concern in children worldwide, and studies identified early kidney dysfunction and an increased risk for CKD in these children.55,56 On the contrary, low birth weight and small gestational age are well-established risk factors for developing CKD and ESKD, as both have been linked to a reduced nephron number.57,58 Interestingly, it has been speculated that as the pediatric obesity problem rises and the low-birth-weight population ages, we may experience a potential shift in the epidemiological spectrum of pediatric CKD. 59
The longstanding paradigm for hereditary causes of CKD was that patients typically manifest early signs of CKD in childhood. However, recent work using whole-exome sequencing (WES) in adult CKD cohorts demonstrated that the genetic contribution to adult CKD is more extensive than previously thought. Groopman et al 37 used WES and diagnostic analysis in 2 cohorts, totaling more than 3000 adult CKD patients, and identified a genetic diagnosis in just under 10% of cases. This work is further corroborated by the work of Connaughton et al 36 who conducted WES in 114 families presenting to nephrology services in Ireland. A monogenic cause was identified in 36% of affected families with a positive family history of CKD, 69% of those with extra-renal features, and in 15% of those without a family history or extra-renal features. Notably, sequencing in this study was based on family history and presence of extra-renal features, and common monogenic causes of CKD (eg, autosomal dominant PKD, Alport syndrome, and MUC1/UMOD) were excluded. In Canada, Yao et al 60 performed WES in adults with focal segmental glomerulosclerosis diagnosed between 1976 and 2017 in the Toronto Glomerulonephritis (GN) Registry and the genetic diagnostic rate was 11%. However, while this work certainly highlights the contribution of genetic causes to adult CKD, it is important to mention that there are limitations in how one can extrapolate these findings to the general population, since the studied cohorts appear to be not fully representative of the cohort of patients followed by nephrologists in the community. For example, the study by Groopman et al 37 included a significant proportion of patients with a clinical diagnosis of cystic kidney disease, likely an overrepresentation than what would be expected in the general population, while diabetes and hypertension appear to be underrepresented in their cohorts. Furthermore, it is worth mentioning that some individuals may have renal disease due to more than one etiology, genetic or non-genetic, since diabetes or hypertension are quite frequent in the general population and not all patients with CKD undergo a biopsy to clarify whether these non-genetic causes contributed to the progression of their renal disease.
Summary part 1
Chronic kidney disease is widespread in Canada, both affecting children and adults.
Chronic kidney disease exerts a large financial strain on the public health care system.
Causes in children differ from adults, with an increasing recognition of genetic factors for both cohorts.
Part 2: Advancing Research for Kidney Disease in Canada
The immense treatment costs of kidney disease combined with an expected increase in global cases highlight the need to simultaneously develop strategies to delay kidney disease progression while increasing access to currently available treatments. Understanding the genetic landscape of CKD, as well as its related disease pathways, offers the unique opportunity of early identification of individuals at risk while developing novel targeted and personalized treatment strategies. These treatments may eventually extend to other causes of CKD and ESKD, traditionally not considered as being genetic in their nature. Unfortunately, funding for basic research for nephrology continues to lag behind other research areas. 61 Despite the increasing number of patients with CKD and ESKD, the overall funding for kidney research remains significantly lower compared with other diseases. Also, the number of randomized control trials in nephrology remains at the lowest ranks compared with other specialties. 62 The majority of trials in nephrology are biased toward RRT and treatment of hypertension rather than progression of CKD or glomerulonephritis, which can partially be explained by the absence of reliable outcome measures or biomarkers, as well as influenced by biases in available industry support. 62
State of funding for health research
Over the past 20 years, the gross domestic spending on research and development (R&D) has been on a steady decline with 1.70% of Canada’s gross domestic product (GDP) allocated to R&D in 2020 compared with 2.02% of GDP in 2001. 63 By contrast, the United States has had a steady increase of their GDP to R&D with 3.45% of GDP in 2020 compared with 2.65% of their GDP to R&D in 2001. 63 In 2020, the total budget for medical research was approximately $41.7 billion compared to the approximately $1.1 billion the Canadian Institutes of Health Research (CIHR) budgeted toward health research of which 45% or $510 million was dedicated to biomedical research.64,65 The constant decline of funding for basic science research for kidney disease continues to be a major concern expressed by numerous Canadian kidney researchers over the past years.66 -68 Recently, in April 19, 2021, the multiyear spending plan announced by the Canadian Federal government allocated $2.2 billion for life sciences. 69 However, much of the money was geared toward enhancing biomedical applications and vaccine development rather than fundamental research. To address the rising prevalence of kidney disease and declining interest of trainees in kidney research, The KRESCENT (Kidney Research Scientist Core Education and National Training) Program was established in 2005. 70 The KRESCENT program continues to work in collaboration with the KFOC, Canadian Society of Nephrology, and the CIHR to enhance the research capacity of kidney research in Canada with a recent renewal of funding for KRESCENT 2.0 to expand and improve the innovative training platform. 71
Changing the established trends will require major shifts in health care funding and can only be achieved through public recognition of the current shortcomings. Currently, Canada is at the forefront of actively involving patient partners into the research funding process through initiatives such as the CIHR Strategy for Patient-Oriented Research (SPOR). 72 For CKD in particular, there has been a push toward dedicated funding for kidney research aimed specifically at facilitating patient-oriented research to integrate research into patient care such as the Can-SOLVE CKD (Canadians Seeking Solutions and Innovations to Overcome Chronic Kidney Disease). 73 The Can-SOLVE CKD is a network of Canadians supporting innovative and patient-oriented research to overcome CKD through a network of researchers and infrastructure. Moreover, the KFOC set forth priority research areas in the HORIZONS Strategic Research Framework for 2019-2024.62,73 -75 The priority research areas were developed by a collaboration of medical professionals, researchers, and allied health and patient partners in the HORIZONS 2022 Working Group (Table 2). The identified strategic kidney research priorities including Communication & Connectivity, Health System Approaches, Promoting Health and Quality of Life, Kidney Health/Wellness, and Treatment of Kidney Disease & Side effects set out the priorities most relevant to patients. While the outlined efforts are laudable, the stated priorities lack a clear recognition of the necessary funding of basic research initiatives that have already revolutionized other areas of clinical medicine.
Strategic Kidney Research Priorities Set by the KFOC HORIZON 2022 Working Group.

Source. Adapted from The Kidney Foundation of Canada. HORIZONS—Strategic Research Framework 2019-2024. 74
Note. QOL = quality of life; CKD = chronic kidney disease.
Advocating for precision medicine in kidney research to improve personalized care
Advances in new technologies now provide feasibility and cost-effective use of multiomics (eg, genomics, proteomics, and transcriptomics) approaches to implement precision medicine for personalized care. Precision medicine is defined as medical care that is designed to optimize therapeutic efficiency or benefit for a particular group of patients, by molecular profiling using information at the DNA (genomics), RNA (transcriptomics), or protein (proteomics) level. This traditional bench-to-bedside concept involves using model systems and patient-derived samples to provide novel insights into the pathways that are deregulated in the respective disease of interest. Moreover, while relying on the state-of-the-art technology, precision medicine places the patient at front and center of its approach and considers the individual as an integral part to understanding and managing the underlying disease process. By doing so, precision medicine offers the opportunity to deeply involve patients and their families in their care that goes beyond the currently standardized one-fits-all treatment approaches.
Over the last 2 decades, precision medicine has already made considerable progress and fundamentally changed medical management for many different medical specialties, most notably in cancer treatment, 76 but also in other areas, such as neurology 77 and gastroenterology. 78 For example, whole-exome next-generation sequencing of patient-derived xenografts and the corresponding parental tumors allowed the identification of novel driver mutations and predicted treatment responses to immunotherapy more accurately than established biomarkers.79 -81 Moreover, the identification of common deregulated pathways and functional profiling among different types of cancers challenged our classic categorization of cancers by organ system. 82 Combining sequencing technology with novel gene delivery technologies offers the unique opportunity to treat disease by targeting patient-specific mutations. 83 Similar advances have led to the first Food and Drug Administration (FDA)-approved gene therapy for the treatment of spinal muscular atrophy, as well as to a landmark trial that showed a promising treatment response for treatment of transthyretin amyloidosis.83,84
There are now a growing number of examples in nephrology demonstrating the recent progress to our understanding of kidney diseases made possible by novel sequencing technologies. Whole-exome sequencing not only helped identify the underlying genetic etiology in many children and adults with CKD, but its use already had direct treatment implications for the management of patients with CKD.85 -87 For example, we have shown that combining sequencing data with a functional assay allowed to identify disease recurrence in children with steroid-resistant nephrotic syndrome after a renal transplant. 86 In patients with atypical hemolytic uremic syndrome, genetic testing is currently used to predict treatment response and risk for recurrence, while also enabling the identification of family members at risk. 88 Recent work by Mann et al 87 highlights the importance of having a genetic diagnosis for the management of kidney transplant recipients. The increased recognition of a significant genetic contribution to both pediatric and adult CKD highlights the importance of implementing novel sequencing technologies, such as WES/WGS, into the routine diagnostic work-up of patients with CKD. The evidence is particularly compelling in transplant recipients, where it may not only have direct treatment effects for the transplant recipients but also may inform the choice of family members as donors when a family member may be found to be at risk of CKD themselves.
Knowledge of the exact underlying genetic etiology has also led to the development of promising novel targeted treatment strategies.85 -87,89 For example, small interfering RNAs have been developed and used in clinical trials for the treatment of primary hyperoxaluria. 90 Treatment with anti-microRNA-21 oligonucleotides in a murine model of Alport nephropathy has proven to prevent disease progression by stimulating metabolic pathways that enhance mitochondrial function, which in turn reduced the production of reactive oxygen species and thus preserved tubular functions. 91 Moreover, over the recent years, it has been shown that the many different genetic causes converge on common kidney disease pathways, which offer hope for the development of treatments allowing targeted modulation of a kidney disease pathway relevant to several different genetic causes. It is also conceivable that some of these treatments will show benefits for patients with CKD not due to a genetic cause but with dysregulation of similar disease pathways. This notion highlights the importance to fully elucidate the underlying disease pathways, and their contributory role to the different causes of CKD, for the successful development of novel targeted therapeutic strategies.
The combined use of novel technologies such as proteomics with bioinformatic approaches and genomic information are expected to identify new potential treatment targets. 92 Patient-derived induced pluripotent stem cells (iPSC) combined with CRISPR/Cas9 gene editing technology have tremendous potential for disease modelling and drug testing. We and others have gained new insight into the mechanisms contributing to kidney diseases such as APOL1 nephropathy, PKD, and nephrotic syndrome using iPSC-derived kidney organoids.93 -100 CRISPR/Cas9 edited iPSC and the use of patient-derived iPSC are beginning to shed light on the kidney developmental processes and mechanistic pathways that contribute to disease. These particularly powerful approaches will eventually enable us to study patient-specific disease effects that will lead to the personalization of treatment strategies in the future.101,102 In Table 3, we summarize some of the most promising personalized technologies, both diagnostic and therapeutic, with potential high impact on research areas of CKD that were identified by patient partners.
Novel Technologies With Potential Impact on High-Relevance Research Areas.

Note. CKD = chronic kidney disease.
Summary part 2
Public funding of R&D for CKD has been on a constant decline over the last 2 decades in Canada.
Patient-oriented research initiatives are promising, but mainly focus on clinical and educational research initiatives.
Allocation of resources and funding toward existing high-yield personalized research initiatives has the potential to significantly affect patient-oriented research outcomes through the development of novel and innovative treatments.
Conclusion
Chronic kidney disease poses a major public health threat to both children and adults in Canada. Much of the disease burden in CKD is associated with increasing obesity rates of recent decades; however, a significant genetic contribution has also been recognized. The study of specific CKD genes and their disease pathways enables the development of novel treatments and therapeutic strategies. While these novel technologies offer hope for the future of the treatment of CKD, their successful clinical implementation will require active engagement of patients and other stakeholders. Canadian researchers are motivated to adopt novel technologies in high-relevance research areas to promote patient-oriented research. However, declining funding for kidney research will be an ongoing issue for the foreseeable future. Funding agencies for kidney research such as the CIHR and the KFOC will be much needed to support revolutionary basic research initiatives required to advance precision medicine. The lessons drawn from patient-oriented research programs may provide particularly useful to enhance the availability and outreach of new treatment programs to vulnerable communities. Developing research programs that work in collaboration with already existing clinical services, as well as early involvement of local patient partners, may eventually increase accessibility to novel treatments and prevention programs for those individuals and communities who are most at risk of CKD.
Footnotes
Acknowledgements
The authors would like to thank our patient partners Mary Beaucage and Hans Vorster for helpful discussions. Special thanks to Dr Sunny Hartwig, Dr Todd Alexander, Dr Adeera Levin, and Dr Leanne Stalker for critical reading of the manuscript. Members of the HORIZONS Working Group 2022 (Dr Tomoko Takano and Elisabeth Fowler), Melanie Taslon, and Elizabeth Myles provided useful information for the manuscript.
Ethics Approval and Consent to Participate
No ethics approval or consent to participate was required for this publication.
Consent for Publication
All authors have reviewed and approved the final version of the manuscript.
Availability of Data and Materials
Data used to generate graphs are publicly available from the websites and references cited.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: T.M.K and J.C. are supported by KRESCENT New Investigator Awards funded by the Canadian Institutes of Health Research and the Kidney Foundation of Canada.
