Abstract
Background:
The canagliflozin and renal endpoints in diabetes with established nephropathy clinical evaluation (CREDENCE) and dapagliflozin and prevention of adverse outcomes in chronic kidney disease (DAPA-CKD) trials have demonstrated significant kidney benefits with sodium-glucose cotransporter-2 (SGLT2) inhibitors. SGLT2 inhibitors are now standard of care for patients with diabetic kidney disease and have also been shown to be effective in those with albuminuric CKD with or without diabetes.
Objective:
We sought to determine how many patients in nephrology care in British Columbia, Canada, would have been eligible for those trials, to compare rates of outcomes, and to estimate cost avoidance arising from widespread use of SGLT2 inhibitors in this cohort.
Study design:
Retrospective cohort study.
Setting:
British Columbia, Canada.
Participants:
CKD patients followed in the Kidney Care Clinics in British Columbia.
Measurements:
We compared the outcomes of kidney failure, sustained estimated glomerular filtration rate (eGFR) <15 mL/min/1.73 m2, dialysis, transplant, death from any cause, and doubling of serum creatinine. We also compared the composite outcome of kidney failure and doubling of serum creatinine.
Methods:
The cohort was derived using a provincial database by combining the inclusion criteria of CREDENCE and DAPA-CKD trials. We included adult patients aged ≥18 years, urine albumin to creatinine ratio (UACR) ≥20 mg/mmol, and eGFR between 25 and 90 mL/min/1.73 m2, between April 1, 2014 and March 31, 2017. The primary outcome was compared with the outcomes experienced in the placebo arms of CREDENCE and DAPA-CKD. The composite outcome stratified by eGFR categories were compared in the British Columbia cohort and the CREDENCE trial. Cost avoidance was estimated based on the number needed to treat to prevent one instance of kidney failure.
Results:
A total of 17.5% (3138/17 963) of patients were eligible, resulting in a cohort with a mean age of 69.7 years and 38% women. The eGFR slope of the British Columbia cohort was −4.21 ± 0.47 mL/min. The mean eGFR was 37.0 mL/min/1.73 m2, median UACR was 55.3 mg/mmol, and use of renin-angiotensin-aldosterone system inhibitors was 56.6%. The British Columbia cohort experienced nearly double the outcomes of kidney failure, death from any cause, and doubling of serum creatinine than the placebo arms of CREDENCE and DAPA-CKD. When stratified by eGFR, the British Columbia cohort and the CREDENCE placebo arm had similar event rates for those with an eGFR <45 mL/min but there were still higher rates of outcome in the greater than 45 mL/min eGFR groups in the British Columbia cohort. Treating the British Columbia cohort with canagliflozin could lead to net cost avoidance of $2.31 million over 2.6 years.
Limitations:
The database only captures those referred to the Kidney Care Clinics by nephrologists, which may lead to selection bias of higher risk patients in the British Columbia cohort. The cost avoidance analysis was a limited high-level analysis.
Conclusions:
The British Columbia cohort represents a high-risk group in whom implementation of the use of SGLT2 inhibitors may well improve outcomes and reduce health care system costs.
Introduction
More than 4 million Canadians suffer from chronic kidney disease (CKD) and it is the 10th leading cause of death, costing the health care system approximately $40 billion each year.1 -4 Chronic kidney disease, especially kidney failure, is also associated with poor quality of life and a markedly increased risk of cardiovascular (CV) disease.5,6 Diabetes is the leading cause of CKD in Canada and, as the prevalence of diabetes rises in an aging population, the burden of CKD is likely to grow. 7
Sodium-glucose cotransporter-2 (SGLT2) inhibitors are now standard of care for patients with diabetic kidney disease and have also been shown to be effective in those with albuminuric CKD of any cause. 8 Two landmark randomized controlled trials (RCTs) demonstrated kidney protective effects of SGLT2 inhibitors.9 -11 The canagliflozin and renal endpoints in diabetes with established nephropathy clinical evaluation (CREDENCE) trial demonstrated that, compared with placebo, canagliflozin reduced the risk of the composite outcome of kidney failure, doubling of serum creatinine, or kidney or CV-related death by 30% among patients with type 2 diabetes mellitus (T2DM) and albuminuric CKD. 12 The dapagliflozin and prevention of adverse outcomes in CKD (DAPA-CKD) trial found that dapagliflozin reduced the risk of kidney failure, a decline of at least 50% in estimated glomerular filtration rate (eGFR), or death from renal or CV causes by 39% in patients with albuminuric CKD, with or without diabetes. 8
In general, RCTs are conducted in a controlled environment with restrictive eligibility criteria that may not be representative of real-world CKD patients. 13 The degree to which the CREDENCE and DAPA-CKD study populations (as defined by their inclusion and exclusion criteria) represent the patients with CKD seen by Canadian nephrologists is unknown, yet this has important implications for patient outcomes and health service costs. The primary aim of this study was to identify the potential benefits and costs of SGLT2 inhibitor use in real-world CKD patients. To achieve this, we compared the CREDENCE and DAPA-CKD trial cohorts with patients meeting trial eligibility criteria from a contemporary cohort of CKD patients managed by nephrologists in British Columbia. We then estimated a high-level, preliminary cost implication for health care funders arising from the use of SGLT2 inhibitors in this real-world cohort.
Methods
Study Population
This retrospective cohort study was performed using data from the Patient Records and Outcome Management Information System (PROMIS), a population-based registry integrating laboratory and clinical data for all patients followed by nephrologists in BC, Canada, in provincial Kidney Care Clinics (KCC). In 2016, BC had a population of 4.6 million, 52% of whom resided in the Vancouver area. 14 The inclusion criteria for this cohort were derived combining eligibility criteria from both the CREDENCE and DAPA-CKD trials (Supplemental Table S1). The lower and upper range of the eGFR cutoff was from DAPA-CKD and CREDENCE, respectively, and the lower range of albuminuria cutoff was from DAPA-CKD to accomplish the goal of creating the most comprehensive cohort while maintaining similarity to the clinical trial participants. We aimed to have a pragmatic approach for the use of clinical trial data for all clinicians who are likely to consider treating patients with SGLT2 inhibitors if they fit either study inclusion criteria.8,12 We included adult patients (≥18 years of age) with a urine albumin to creatinine ratio (UACR) ≥20 mg/mmol and an eGFR between 25 and 90 mL/min/1.73 m2 on cohort entry date (index date). Patients with and without type 2 diabetes were eligible.
The study cohort was assembled from all eligible patients recorded in PROMIS at any point within the study period (April 1, 2014, to March 31, 2017). This period of 2014 to 2017 was selected as evidence for SGLT2 inhibitors were just emerging, which would minimalize the number of patients on an SGLT2 inhibitor, and setting the end date of enrolment to 2017 allowed for 3 years of follow-up time for those enrolled in 2017, permitting a similar median follow-up time to the CREDENCE and DAPA-CKD cohorts. Furthermore, BC public health insurance did not provide access to SGLT2 inhibitors until 2019, when empagliflozin was added as a third line agent in the management of diabetes.
The cohort consisted of prevalent patients (those entered in PROMIS prior to April 1, 2014) who were included if they had a UACR ≥20 mg/mmol in the prior 12 months and an eGFR measure between 25 and 90 mL/min/1.73 m2 within 3 months of the qualifying UACR. The date of study entry for such patients was set as April 1, 2014. Of note, a proportion of prevalent patients were found to have a change in eGFR and/or UACR within 3 months of April 1, 2014, such that they no longer met eligibility criteria. These patients were not considered in the primary cohort but were included in a sensitivity analysis. Incident patients were those who first met the study criteria after April 1, 2014, for whom the date of study entry was set to the first date at which all eligibility criteria were met. Patients with history of renal transplant; a diagnosis of autosomal dominant or autosomal recessive polycystic kidney disease, lupus nephritis, or antineutrophil cytoplasmic antibody; or type 1 diabetes were excluded. We further restricted our analyses to those who did not receive immunosuppressive therapy (ie, dispensed azathioprine, cyclophosphamide, cyclosporine, mycophenolate mofetil, mycophenolic sodium EC, prednisone, and tacrolimus) within 6 months prior to the index date. This study was approved by the research ethics board at the University of British Columbia (H20-03986).
Outcome
The primary outcome was a composite of the first occurrence of kidney failure (eGFR <15 ml/min/1.73 m2 demonstrated by at least 2 measurements separated by at least 90 days, chronic dialysis for ≥90 days or kidney transplantation), and sustained doubling of serum creatinine (demonstrated by at least 2 measurements separated by at least 30 days). As the CKD patients in this study cohort did not receive SGLT2 inhibitors, the outcomes were compared with the outcomes observed in the placebo arm of the RCTs. The composite outcome in the BC cohort was compared as a primary outcome, as well as the number of events stratified by eGFR categories of 25-29, 30-44, 45-59, 60-90, and >90 mL/min/1.73 m2 with the CREDENCE trial only as outcomes by eGFR category was not available for DAPA-CKD trial. 15 We compared this primary outcome with the composite outcome in the CREDENCE trial for kidney failure, doubling of serum creatinine, and renal death. In the placebo arm of the CREDENCE trial, only five renal deaths were recorded in the placebo arm, representing just 2.2% (5/224) of this composite outcome. 12 As such, we assumed that the effect of renal death on this secondary composite outcome was negligible when comparing with the BC cohort.
Estimated Glomerular Filtration Rate Slope Calculation
To compare the slopes between the BC cohort and the placebo arms of CREDENCE and DAPA-CKD, we followed the methods described in the supplemental material for the CREDENCE trial.12,16 We used a two-slope model with fixed effects of baseline eGFR, eGFR strata at baseline (25-30, 30-45, 45-60, and >60 mL/min/1.73 m2), continuous time (in years), time spline (with 1 knot at 3 weeks), with interaction terms of eGFR strata by time, eGFR strata by time spline, and the random effects of intercept, time, and time spline. The eGFR slope was estimated by restricted maximum likelihood. As patients from the BC cohort did not receive any treatment, variables involving “treatment” were not included in our model.
Cost Avoidance Analysis
Using the published data of event rates in the canagliflozin and placebo arm stratified by eGFR categories (15-29, 30-44, 45-59, 60-90, and >90 mL/min/1.73 m2) in the CREDENCE trial, we calculated the number needed to treat (NNT) values in each of the eGFR categories.15,17 We calculated the NNTs for the outcome of dialysis, transplantation, or renal death in each eGFR category as the reciprocal of the difference in event rates between the canagliflozin and placebo arms in the CREDENCE study. Supplemental Table S4 provides the event rates by eGFR category used to calculate the NNT, and Supplemental Table S5 provides the estimated NNTs.
The cost avoidance analysis was conducted by calculating the potential future costs minus the cost of the intervention. The potential future cost was attributed to the cost of dialysis initiation per patient, and the cost of intervention was the cost of treating all participants in the BC cohort for 2.6 years (the median follow-up of CREDENCE trial). We used the NNT of dialysis, transplant, or renal death for this calculation as this reflects the number of dialysis starts in the trial cohort. Although the cost of transplant in the first year is more expensive, this was assumed to be the same cost as a year of dialysis, for more conservative estimates. 18 The renal death was considered negligible in this calculation as absolute number of renal deaths were low in the trial (2.2%).
To explore the possibility that canagliflozin may not be as efficacious in the BC cohort as was demonstrated in the CREDENCE trial, we calculated cost avoidance, assuming canagliflozin was 20%, 30%, or 35% less effective in the BC cohort.
Currently, the cost of an SGLT2 inhibitor in Canada is approximately $2.92/day, which is $1,066/year. 19 However, costs can be mitigated by halving a higher dose pill of canagliflozin to match similarly the dose given in the major trials, thus reducing the cost to approximately $533/year. Dialysis costs were estimated to be $87,000/patient/year in BC when adjusted for inflation in 2021. 20
Statistical Analysis
Descriptive statistics were used to characterize eGFR, UACR, and serum creatinine. Continuous variables were reported as mean (± SD) or median (interquartile range [IQR]) as appropriate. Categorical variables were reported as frequency (percentage).
The incidence rates of the primary and the secondary outcomes were calculated per 100 and 1000 person-years of follow-up, for overall study cohort and within each eGFR category. We compared the number of outcomes between this study cohort and the CREDENCE and DAPA-CKD trials, and the primary outcome by eGFR categories between the study cohort and the CREDENCE trial. The incident rate per patient-year was compared with the clinical trial rates under the assumption that both outcome events follow a Poisson distribution. To test equivalency of the two rates, we used the uniformly most powerful unbiased test where the test statistic follows a binomial distribution.
We conducted a sensitivity analysis including patients who were excluded from the primary analysis for not meeting the UACR or eGFR criteria within 3 months after the index date. All analyses were carried out using SAS Version 9.4 (SAS Institute, Cary, North Carolina).
Results
Cohort Derivation
The PROMIS database included 17 963 patients, of whom 3138 (17.5%) met cohort entry criteria (Figure 1). In addition, 1533 patients who met study criteria in the 12 months prior to April 1, 2014, but who did not have a UACR ≥20 mg/mmol and/or eGFR 25 to 90 mL/min/1.73 m2 within 3 months of this date were identified for inclusion in a sensitivity analysis.

Cohort derivation.
Table 1 presents the overall characteristics of the study cohort in comparison with the placebo arm of the CREDENCE and DAPA-CKD trials. On average, the BC cohort was older with a mean age of 69.7 (±13.5) years, and 38% were women. Approximately, 56.2% were white, and 8.3% were Asian. The majority cohort had a diagnosis of diabetes (65%) and hypertension (86%). Around half of the cohort (57%) were on an angiotensin-converting enzyme (ACE) inhibitor or angiotensin II receptor blocker (ARB). The median duration of follow-up for the BC cohort was 4.1 years (range = 3.0-5.3). The mean eGFR was 37 mL/min compared with 56 and 43 mL/min in CREDENCE and DAPA-CKD trials, respectively. The median UACR in the BC cohort was 55.3 mg/mmol (range = 29.7-135.4) in comparison with 105.2 and 105.5 mg/mmol in CREDENCE and DAPA-CKD, respectively. Over half of the cohort (65.9%) had UACR 30 to 300 mg/mmol, and 8.9% of the cohort had a UACR ≥300 mg/mmol.
Demographic and Clinical Characteristics of the BC Cohort at Index Date.

Note. Plus-minus values are means ± SD. BC = denotes British Columbia; CREDENCE = canagliflozin and renal endpoints in diabetes with established nephropathy clinical evaluation; DAPA-CKD = dapagliflozin and prevention of adverse outcomes in chronic kidney disease; ACE = inhibitor angiotensin-converting enzyme inhibitor; ARB = angiotensin II receptor blockers; IQR = interquartile range; eGFR = estimated glomerular filtration rate; ACR = urine albumin to creatinine ratio.
Demographic and clinical characteristics of the participants in the placebo arms.
Race or ethnic group was reported by the patients. 13.1% of the cohort did not have a defined or an unknown race.
Hypertension is the combination of hypertension diagnosis and receiving medication for hypertension. Patients with diabetes are defined as those with renal diagnosis, comorbidity condition, use of glucose lowering drugs/insulin, or with either glycated hemoglobin (A1C) ≥7% and/or fasting glucose (fasting blood sugar [FBS]) ≥7 mmol/L.
From the CREDENCE trial supplemental appendix.
Observations of the Distribution of Patients by eGFR Category Between the BC Cohort, CREDENCE, and DAPA-CKD
Table 2 presents the proportion of CKD patients in each of the eGFR categories observed in the BC cohort, CREDENCE, and DAPA-CKD trials. There is a higher proportion of patients with lower eGFR in the BC cohort, with around 80.7% with an eGFR less than 45 mL/min, with 28.9% having an eGFR <30 mL/min. In contrast, the CREDENCE trial had 4% of participants with eGFR <30 mL/min, (those who at the time of screening had an eGFR >30 mL/min and at randomization fell to below <30 mL/min) and the DAPA-CKD cohort had 15.4% of participants.
Note. eGFR = estimated glomerular filtration rate; BC = denotes British Columbia; CREDENCE = canagliflozin and renal endpoints in diabetes with established nephropathy clinical evaluation; DAPA-CKD = dapagliflozin and prevention of adverse outcomes in chronic kidney disease.
Comparison of the eGFR Slope Between the BC Cohort, CREDENCE, and DAPA-CKD
The eGFR slope for the BC Cohort was −4.21 ± 0.47 mL/min, while the eGFR slope for the placebo arms of CREDENCE and DAPA-CKD were −4.71 ± 0.15 and −3.79 ± 0.11 mL/min, respectively, as demonstrated in Table 3.8,12
Note. eGFR = estimated glomerular filtration rate; BC = denotes British Columbia; CREDENCE = canagliflozin and renal endpoints in diabetes with established nephropathy clinical evaluation; DAPA-CKD = dapagliflozin and prevention of adverse outcomes in chronic kidney disease.
Comparison of the Outcomes Experienced Between the BC Cohort, CREDENCE, and DAPA-CKD
The BC cohort experienced significantly higher rates of outcomes than the placebo arms of the RCTs as demonstrated in Tables 4 and 5. The BC Cohort experienced significantly higher number of kidney failure events than both CREDENCE and DAPA-CKD placebo arms. However, this outcome was heavily weighted with nearly double the number of sustained eGFR less than 15 mL/min in the BC cohort. The placebo arms of the clinical trials experienced higher rates of the composite outcome of kidney transplant and dialysis starts at 17.7 events per 1000 patient-years and 2.4 events per 100 patient-years in the CREDENCE and DAPA-CKD trials, respectively, compared with 12.4 events per 1000 patient years and 1.2 events per 100 patient years in the BC cohort.
Comparison of Outcomes Between BC Cohort and Placebo Arms of CREDENCE. 12

Note. Outcome event rates in per 1000 patient-years. BC = denotes British Columbia; eGFR = estimated glomerular filtration rate; CREDENCE = canagliflozin and renal endpoints in diabetes with established nephropathy clinical evaluation.
Comparison of Outcomes Between BC Cohort and Placebo Arm of DAPA-CKD. 8

Note. Outcome event rates in per 100 patient-years. BC = denotes British Columbia; DAPA-CKD = dapagliflozin and prevention of adverse outcomes in chronic kidney disease; eGFR = estimated glomerular filtration rate.
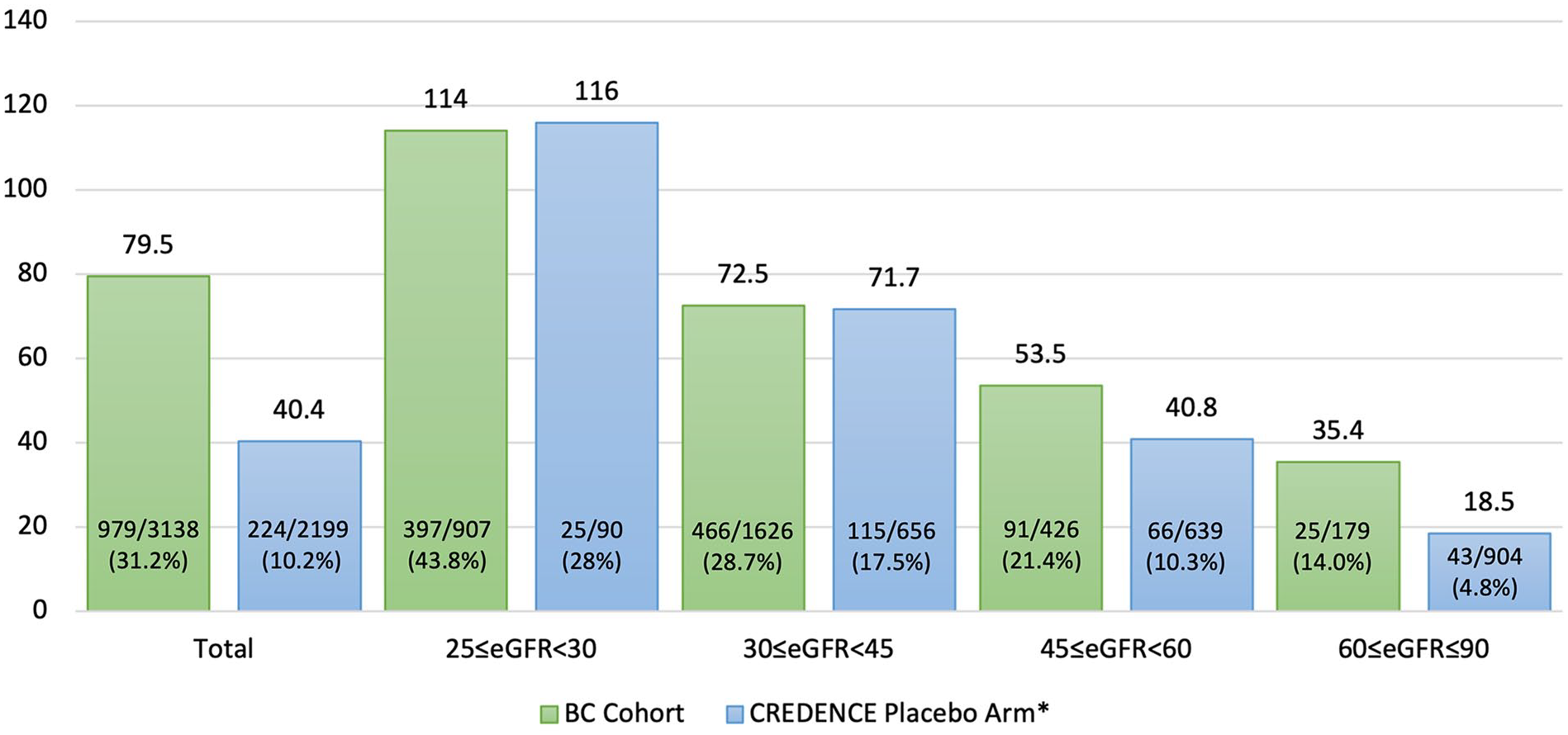
Figure 2 demonstrates the composite outcome of kidney failure and doubling of serum creatinine. A total of 979 events of kidney failure and doubling of serum creatinine (31.2%) occurred, for 79.5 events per 1000 person-years. The kidney event rates of kidney failure and doubling of serum creatinine were similar in the BC and CREDENCE cohorts for those with eGFR <45 mL/min. However, there were still higher rates of outcome in the 45 ≤eGFR <60, and 60 ≤eGFR <90 groups, 53.5 versus 40.8 per 1000 person-years and 35.4 versus 18.5 per 1000 person-years, in the BC cohort and CREDENCE cohort, respectively.
Sensitivity Analysis
Sensitivity analysis was performed including the 1,533 patients who did not have a UACR ≥20 mg/mmol and an eGFR 25 to 90 mL/min/1.73 m2 within 3 months after the index date. There were no significant changes in the distribution of patients in the eGFR group in the BC cohort as demonstrated in Supplemental Table S2. The percentage does not add up to 100% due to missing eGFR values within the time frame of the analysis. The composite of kidney failure and doubling of serum creatinine had 1629 events and 87.5 events per 1000 person-years. The sensitivity analysis demonstrated higher event rates in comparison with the primary analysis. The outcomes stratified by eGFR categories are demonstrated in Supplemental Table S3.
Cost Avoidance Analysis
Figure 3 demonstrates the costs associated with treating the cohort, and potential cost avoidance if canagliflozin was assumed to be equally effective, or 20%, 30%, or 35% less effective in the BC cohort in comparison with CREDENCE trial. The cost of treating CKD patients in BC cohort with an SGLT2 inhibitor for 2.6 years was approximately $4.35 million, and to recuperate this investment, at least 50 dialysis starts needed to be prevented. When assuming canagliflozin is equally efficacious in the BC cohort, we estimate this will result in approximately 77 fewer dialysis starts, translating to a net cost avoidance of $2.31 million. If canagliflozin is 20% less effective in the BC cohort, the net cost avoidance from preventing dialysis starts may range around $0.98 million. Even when assuming 35% less efficacy in our BC cohort, this may not accrue additional health care costs but, by avoiding 50 patients commencing dialysis, would still provide net benefit.

Hypothetical cost avoidance analysis by treating everyone in the BC Cohort with an SGLT2 inhibitor.
Discussion
This is the first study that assessed the applicability of the RCT inclusion criteria in a population-based cohort being cared for by nephrologists in a universal health care system, demonstrating that 17.5% of patients met criteria for enrolment. The BC cohort experienced greater number of kidney outcomes than both placebo arms of CREDENCE and DAPA-CKD trials.8,12,15,17 When stratified by eGFR, the BC cohort experienced similar rates of kidney outcomes relative to the placebo arms in the eGFR <45 mL/min group, but higher event rates when eGFR ≥45 mL/min to the placebo arm of the CREDENCE trial. A hypothetical cost avoidance calculation was performed, using conservative estimates, which exhibited potential for cost avoidance when simply considering prevention of initiation of dialysis after treating the CKD patients with an SGLT2 inhibitor.
As a selective group of patients with an elevated baseline risk for CKD deemed to require multidisciplinary care by a nephrologist, those with relatively high or normal eGFR in the BC cohort may not necessarily demonstrate similar progression as the general population. Although the BC cohort at baseline was older, had lower average eGFR, and only around two thirds on an ACE inhibitor or ARB, there were similar rates of kidney failure and doubling of serum creatinine observed with the eGFR <45 mL/min groups. This could potentially be explained by the higher level of albuminuria in the CREDENCE cohort, as this has been associated with more rapid decline in kidney function, regardless of baseline eGFR. 21 The eGFR slopes between the BC cohort and the two trials were comparable, but the BC cohort experienced far greater number of event rates in sustained eGFR <15 mL/min and doubling of serum creatinine, which may be explained due to the lower average eGFR in the BC cohort. The use of ACE inhibitors or ARBs in the BC cohort is concordant with other reports in CKD populations. 22
The challenge in extrapolating clinical trial findings to clinical practice is twofold: the eligibility criteria are often restrictive, and significant differences remain in the baseline characteristics between the trial cohort and a population-based cohort even when the inclusion criteria are met. Combining the two inclusion criteria from CREDENCE and DAPA-CKD trials to establish the most inclusive cohort, the derived BC cohort was in keeping with other studies assessing generalizability of SGLT2 inhibitor CV outcome trials to a population database.23 -27 The inherent differences between trial cohorts versus population cohorts derived by those meeting eligibility criteria is well established, demonstrating that inclusion criteria may be insufficient for identification of individuals who may derive similar benefits exhibited in the trial. 28
The cost avoidance analysis demonstrated that for an investment of $4.35 million over 2.6 years, the reduction in dialysis starts could result in a potential for cost savings of approximately $2.31 million (assuming SGLT2 inhibitors exhibit similar efficacy as demonstrated in CREDENCE). Canagliflozin has demonstrated consistent benefits over a range of kidney function and CV risk profiles.15,29 However, to construct the most conservative estimates of cost savings, we explored different efficacy thresholds. We found that even if canagliflozin is 35% less efficacious, it will not cost additional health care dollars spent. Possible further cost savings may incur from avoiding other outcomes, such as preventing hospitalizations for heart failure, which we did not have data to explore. This attempts to quantify the impact in a universal health care system with a single government payer, where savings in one area (avoidance of dialysis cost) can offset the investment in a different area (treating non-dialysis CKD patients with SGLT2 inhibitors). These data may provide reference in determining specific group of patients to target for treatment, such as the eGFR <45 mL/min group, where there is a high risk for disease progression and complications, and focus management where the treatment is demonstrated to be the most effective. Similar “theoretical” analyses examining delay in time to dialysis in those treated with canagliflozin and resultant cost savings of the CREDENCE trial have presented comparable significant health care reductions. 30
The strength of this study includes the use of referred CKD population-level health database of patients in a real life, nephrology practice network in BC to accurately calculate specific outcomes of interests to compare directly with event rates experienced in the trials. An important limitation of this study is that the PROMIS database, although a large sample size of around 18 000 patients, only captures patients who have been referred to the KCC and it does not consider patients managed in the private nephrologist offices, or those not referred. This may have led to a selection bias in the development of a more high-risk cohort. In addition, the medication list used for this analysis is derived at the time the patient enters the cohort, and thus medications changed or started during the follow-up years, such as increasing number of prescriptions for RAS blockade, will not be captured. Nonetheless, this represents real-world treatment of CKD patients, and it is not likely that the addition of higher use of RAS blockade would substantially change the outcomes of interest. This preliminary cost analysis highlights the scope of potential benefits that could accrue with more widespread adoption of SGLT2 inhibitors; however, it is limited by estimating only use of SGLT2 inhibitors for 2.6 years and estimates of costs of treating kidney failure for a single year. We attributed the cost of preventing kidney failure (ie, eGFR <15 mL/min) equal to the cost of starting dialysis, although patients may reach the definition of kidney failure but may not necessarily start dialysis until later. Moreover, additional benefits of SGLT2 inhibitor use, such as reductions in CV disease, reduced use of other diabetes treatments, lower hospitalization costs, and the improvements in quality of life that may accrue have not been considered. 31 Comprehensive modeling is required to better define the cost implications of SGLT2 inhibitor use, with this study emphasizing the potential for substantial gains.
In conclusion, the BC CKD cohort is a high-risk group of patients in whom implementation of the use of SGLT2 inhibitors may well improve outcomes and reduce health care system costs. Efforts to increase utilization of these agents should consider concentrating on this cohort. The cost-effectiveness in primary care settings requires further study.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581221145068 – Supplemental material for Findings of Sodium-Glucose Cotransporter-2 Inhibitor Kidney Outcome Trials Applied to a Canadian Chronic Kidney Disease Population: A Retrospective Cohort Study
Supplemental material, sj-docx-1-cjk-10.1177_20543581221145068 for Findings of Sodium-Glucose Cotransporter-2 Inhibitor Kidney Outcome Trials Applied to a Canadian Chronic Kidney Disease Population: A Retrospective Cohort Study by Tae Won Yi, Mohammad Atiquzzaman, Yuyan Zheng, Brendan Smyth, Meg Jardine and Adeera Levin in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
This study was approved by the research ethics board at the University of British Columbia (H20-03986).
Consent for Publication
All authors provided their consent for publication.
Availability of Data and Materials
The de-identified data underlying this article will be shared on reasonable request to the corresponding author.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: No funding was provided for this work. B.S. is supported by The Jacquot Research Establishment Fellowship from the Royal Australasian College of Physicians. A.L. was a member of the Steering committee for canagliflozin and renal endpoints in diabetes with established nephropathy clinical evaluation (CREDENCE) and has received grant support from Janssen. M.J. is responsible for research projects that have received funding from Amgen, Baxter, CSL, Dimerix, Eli Lilly, Gambro, and MSD; has received Advisory, Steering Committee and/or speaker fees from Akebia, Amgen, Astra Zeneca, Baxter, Bayer, Boehringer Ingelheim, Cesas Lynx, Chinook, Medscape, CSL, Janssen, MSD, Roche, and Vifor; with any consultancy, honoraria, or travel support paid to her institution.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: T.W.Y. is supported by the University of British Columbia Clinician Investigator Program. B.S. is supported by a Jacquot Research Establishment Fellowship from the Royal Australasian College of Physicians.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


