Abstract
Rationale:
Nocturnal enuresis is a common symptom of nephrogenic diabetes insipidus (NDI) in children. Published reports on the treatment of nocturnal enuresis in this population are scarce. Presenting concern of the patient: 2 brothers, aged 8 and 9 years, presented for outpatient pediatric nephrology follow-up. Despite being medically stable on their current medication regimen, they both experienced significant distress due to primary nocturnal enuresis, including decreased self-esteem and social stress.
Diagnoses:
The brothers had primary nocturnal enuresis related to their high urine output from NDI. Interventions: we describe a case of 2 brothers with NDI in whom indomethacin was added to their pharmacologic treatment specifically to address enuresis.
Outcome:
Both brothers experienced a significant decrease in the frequency of nocturnal enuresis and improvement in their perceived quality of life.
Teaching points:
Nocturnal enuresis is a bothersome symptom of NDI with adverse psychological effects. Indomethacin can improve nocturnal enuresis in some patients. Treatment with nonsteroidal anti-inflammatory drugs carries a risk of gastrointestinal and kidney side effects. We advocate for a patient-centered approach to the treatment of NDI which includes optimizing both the medical and the psychological needs of the patient.
Introduction
Nephrogenic diabetes insipidus (NDI) is a disease marked by polyuria causing compensatory polydipsia, due to an inability of the kidney to reabsorb free water in the collecting ducts to concentrate the urine. It can be inherited or secondary, the latter most commonly due to medications. In inherited forms of NDI, water reabsorption is impaired in the collecting ducts by a functional aquaporin deficiency, due to a mutation in the gene encoding either the arginine vasopressin receptor-2 (AVPR2) or the aquaporin 2 water channel. Arginine vasopressin (AVP) normally regulates water permeability in the collecting ducts by controlling the availability of aquaporin channels.
Treatment of NDI is aimed at reducing urine output, thereby allowing for adequate enteral water intake to maintain hydration and to allow for appropriate growth. Medication classes commonly used to treat NDI include diuretics and prostaglandin (PG) synthesis inhibitors, along with following a low-sodium diet. Thiazide diuretics are the most prescribed medications for treatment 1 and can significantly reduce urine output when paired with a low-sodium diet. 2 Potassium-sparing diuretics such as amiloride are often used in combination to counteract the urine potassium loss from thiazide diuretics. The main mechanism of action of diuretics in improving urine output is by promoting proximal tubule reabsorption of water, leading to less free water in the collecting ducts. 3 Indomethacin is the most frequently used nonsteroidal anti-inflammatory drugs (NSAIDs), working to reduce urine output and has an additive effect when used with thiazide diuretics. 4
Although access to free water, pharmacologic agents, and dietary modification can lead to normalized growth and development in NDI, they are not curative and most often these children still have significant ongoing polyuria and polydipsia. A retrospective study by Sharma et al highlighted nocturnal enuresis as a common complication of NDI and found that in their cohort of 36, 19 patients had resolution at the median age of 11.3 years. Most of their patients had their medications successfully discontinued during their teen years, either due to nonadherence without clinical deterioration or as advised by their physician. 5
Observational studies have found impaired self-esteem or discontent in children with monosymptomatic nocturnal enuresis, 6 which improved with effective treatment. 7 A study in Lebanon by Merhi et al 8 using the Short Form of the Pediatric Enuresis Module on Quality of Life 9 to assess the psychological impact of primary nocturnal enuresis on children found that 82.4% of children had a score indicating enuresis had a significant effect on their psychological well-being. Kanaheswari et al 6 found that low self-esteem was most common in adolescents, those with frequent wetting, and girls.
Presenting Concerns
We present 2 cases involving brothers with genetically confirmed X-linked NDI from a genetically proven AVPR2 gene mutation who suffered from primary nocturnal enuresis.
Clinical Findings
The eldest brother presented to hospital with failure to thrive, vomiting, and developmental delay at 6 months of age. A workup revealed hypernatremia and low urine osmolality that did not respond to desmopressin. He was then referred for nephrology evaluation. Treatment was initiated for NDI and genetic testing confirmed the diagnosis. The younger brother was diagnosed soon after birth due to hypernatremia and underwent genetic testing, which was positive for the same mutation as his brother. There were no other family members affected. Years later, both patients, aged 8 and 9 years, had well controlled disease with stable electrolytes and appropriate growth after treatment with hydrochlorothiazide (20 mg twice daily), amiloride (8 mg twice daily), and potassium chloride supplementation. They both had gastrostomy tubes which at this time were being used solely for medication administration. Their biggest concern in follow-up was that of ongoing primary nocturnal enuresis causing them psychological distress and social stress.
Diagnostic Focus and Assessment
Primary nocturnal enuresis was diagnosed clinically. To rule out structural abnormalities and monitor for the development of hydronephrosis, both boys had a kidney and bladder ultrasound.
Therapeutic Focus and Assessment
Indomethacin was added specifically as an attempt to improve enuresis. With treatment, enuresis improved without side effects and the dose was titrated from 25 mg orally nightly to 25 mg twice daily (approximately 1.5 mg/kg/d).
Follow-up and Outcomes
After starting indomethacin in 2013, there was significant improvement in the enuresis, with less frequent wetting and much less volume. With the increased dose, both boys had essentially complete resolution of enuresis. The indomethacin was tolerated well without known gastrointestinal (GI) side effects. In 2015, both brothers experienced worsening enuresis despite increasing doses of hydrochlorothiazide. The dose of indomethacin was subsequently increased to approximately 2 mg/kg/d in both children, which again led to a significant improvement. In 2021, both boys were missing many doses of all their medications and remained well with stable laboratory parameters. We thus discontinued all medications successfully without recurrence of enuresis (Figure 1).
Timeline
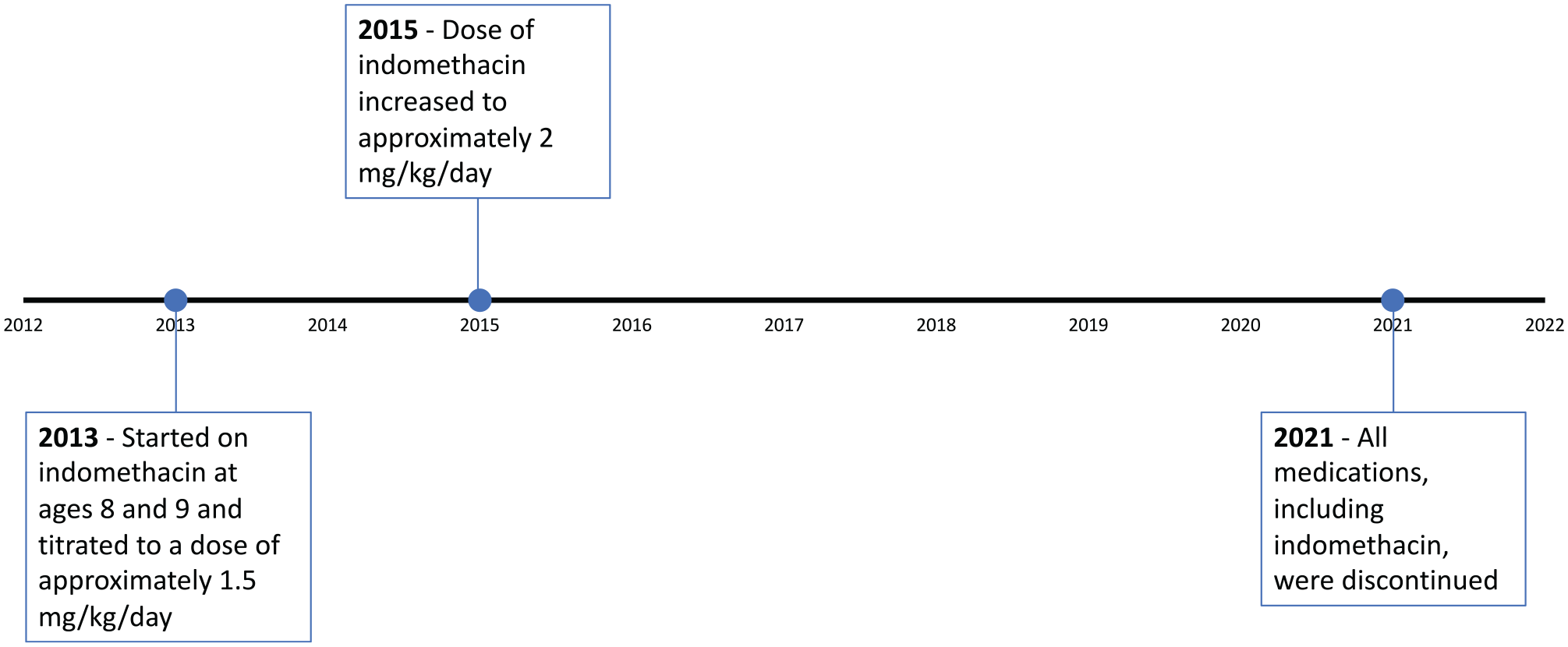
The timeline of treatment with indomethacin in the 2 brothers.
Patient Perspective
When I started taking indomethacin it seemed as if my life changed in a way. I rarely if not ever wet the bed, I was able to go over to friends houses and most of all I didn’t need to wear diapers to bed anymore, even during road trips I didn’t need to use the washroom nearly as much as I did before. (Brother 1) I was always ashamed of wearing diapers to bed and having to wake up in a wet mess. It riddled my self esteem. Starting Indomethacin, the bedwetting became less and less frequent, until it stopped completely. I didn’t need to wear diapers anymore, or worry about having to change my sheets every 2 days. I felt like a normal person. (Brother 2)
Discussion
After achieving adequate fluid balance, metabolic balance, feeding, and growth, the treatment of NDI should focus on optimizing the quality of life for patients. For the 2 young boys described above and for many children, addressing bedwetting is an integral part of patient-centered care. Although the vast majority of children are able to eventually achieve nighttime continence, some children, even after education about the symptoms and prognosis, would benefit from medical treatment to preserve psychological well-being. The addition of indomethacin, although not necessary for medical stability, had a significantly positive effect on the boys’ perceived health and happiness.
Indomethacin is effective at decreasing urine output and has an additive effect when used along with thiazide diuretics 4 ; however, it is not without side effects. Indomethacin is known to cause significant GI complications, including gastritis, GI bleeds, abdominal pain, vomiting, and fistulae. 10 Nonsteroidal anti-inflammatory drugs have been known to cause kidney impairment, especially in those with additional risk factors and with long-term use due to their effect on kidney homeostasis. 11
Nonsteroidal anti-inflammatory drugs remain a common treatment option for children with NDI among care providers. D’Alessandri-Silva et al 1 surveyed pediatric nephrologist from the Midwest Pediatric Nephrology Consortium and found that of the 72 respondents, 55% reported prescribing NSAIDs as part of their usual treatment of NDI. The reasons for not prescribing indomethacin were GI and kidney side effects (43%).
The literature on treating nocturnal enuresis in the NDI population is sparse. Similar to our report, Monn describes a case of a 7-year-old boy with NDI where indomethacin 25 mg twice daily decreased both urine output and nocturnal enuresis within 1 day of starting the medication. 12 Desmopressin is the most used medication for the treatment of monosymptomatic nocturnal enuresis in the non-NDI population; however, mechanistically, this should not be effective in NDI due to the underlying defect causing the disease, although there are rare case reports describing its use. In a case report by Jonat et al, 13 a child with a confirmed hemizygous missense mutation of the AVPR2 gene had decreased nocturnal enuresis with desmopressin administration, although apparently no significant decrease in water intake or urine output. They suggest that AVP binding receptors in the central nervous system play a role in the pathogenesis of nocturnal enuresis. In one patient with partial NDI described by Sharma et al, 5 desmopressin resolved the nocturnal enuresis.
The advent of selective cyclooxygenase (COX)-2 inhibitors such as celecoxib provides an alternative treatment option to indomethacin, seemingly with less side effects. Nonsteroidal anti-inflammatory drugs exert their effect by inhibiting COX 1 and 2, leading to the suppression of PG synthesis. Both the kidney and the GI tract are important targets for NSAIDs due to the presence of COX-1 and COX-2. In the kidney, COX-1 has effects on the vasculature affecting renal hemodynamics which in turn affects the glomerular filtration rate (GFR). It is also thought to be the principal enzyme in the GI tract. Cyclooxygenase-2 has more effect on sodium and water excretion and is less prevalent in the GI tract. It regulates renin-angiotensin secretion thereby having some effect on GFR as well. In patients who are normotensive where renal hemodynamics is not dependent on the presence of PGs, neither nonselective NSAIDs nor COX-2-specific inhibitors (COXIBs) significantly affect renal function. However, in various disease states and renal hypoperfusion, both nonselective NSAIDs and COXIBs can lead to kidney damage. 14 Celecoxib has been used safely in children 15 and there exist case reports of it being used and effective in NDI. Soylu et al 16 describe a case where selective COX-2 inhibition with rofecoxib and celecoxib was more efficacious than both indomethacin and ibuprofen at controlling water diuresis and no adverse effects were noted. Cyclooxygenase-2-specific inhibitors have been shown to reduce the risk of symptomatic GI ulcers and serious GI complications when compared with nonselective NSAIDs in 2 systematic reviews,10,17 but studies show that kidney side effects remain. 18 A systematic review published in 2006 by Zhang found that although there was increased risk of kidney effects with rofecoxib, celecoxib led to a reduction in the development of hypertension and kidney dysfunction compared with controls. 19 A second systematic review by Ungprasert et al comparing different NSAIDs found a statistically significant elevated acute kidney injury risk for each of the traditional NSAIDs, including indomethacin, but not in COXIBs such as celecoxib. It should be noted, however, that the risk differences between traditional NSAIDs and COXIBs did not reach statistical significance, but they found that indomethacin had the highest risk ratio. 20 A COXIB was not considered for the treatment of enuresis in the cases described in this report because these drugs were not yet commonly used in the pediatric population.
These 2 cases highlight the treatment of nocturnal enuresis as an important aspect of patient-centered care in children with NDI. Unfortunately, there is little evidence to guide treatment choices of this bothersome symptom. We advocate for open discussions with the patients and caregivers about treatment options including possible adverse effects. In some situations, ameliorating the psychological distress caused by enuresis outweighs potential side effects of treatment with medication. We described 2 brothers that responded well to indomethacin by having significantly less frequent bed wetting and improved social and psychological well-being without side effects. Cyclooxygenase-2-specific inhibitors may offer another option for treatment with less risk of adverse GI outcomes.
Footnotes
List of Abbreviations
NDI, nephrogenic diabetes insipidus; NSAIDs, nonsteroidal anti-inflammatory drugs; GI, gastrointestinal; AVPR2, arginine vasopressin receptor-2; AQP2, aquaporin 2; AVP, arginine vasopressin.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Informed consent was obtained prior to publication.
Availability of Data and Materials
Not applicable, there are no data or materials used in this report except for patient charts and discussions with the patients.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
