Abstract
Background:
Intravenous (IV) iron and erythropoietin stimulating agents (ESAs) are standard treatments for anemia in patients receiving maintenance hemodialysis. These medications are associated with significant costs to hemodialysis programs and patients. Recent trial evidence demonstrated that a high-dose IV iron protocol reduces ESA usage and improves cardiovascular outcomes. The cost of implementing a high-dose iron protocol within the Canadian public healthcare context remains unknown.
Objective:
Our primary aim was to estimate the costs of a high-dose IV iron protocol in a large Canadian hemodialysis program that currently uses a low-dose and reactive IV iron strategy. Our secondary aim was to estimate the reduction in ESA use required to maintain cost neutrality with a high-dose IV iron protocol.
Design:
In this modeling study of IV iron and ESA utilization from a regional hemodialysis program, changes in medication utilization were calculated based on observed effects from published trial data. Using data from a quality improvement audit of regional anemia management and medication utilization, we estimated potential cost differences under various modeling conditions.
Setting:
Four adult hospital-based and 9 community in-center hemodialysis units in the Alberta Kidney Care—South renal program during the observation period of September 1, 2018, to November 30, 2018.
Patients:
In total, data from 826 patients were included.
Measurements:
Mean monthly IV iron and ESA doses were obtained from routine audit data captured within an electronic medical record. Costs were determined from provincially negotiated medication prices.
Methods:
Current IV iron and erythropoietin dosages were aggregated at the hemodialysis unit level. We used the results from the PIVOTAL trial to estimate the expected increase in IV iron dose and reduction in ESA dose with a high-dose IV iron protocol. We assumed the split between various manufactures of IV iron and ESA were maintained in our cost model. Total medication costs were aggregated by hemodialysis unit, and the mean costs in each unit were used to estimate per-patient costs. Sensitivity analyses included models that assumed 100% IV iron sucrose usage, as well as models where community hemodialysis units and hospital-based hemodialysis units were examined separately. Finally, we calculated a break-even point for ESA dose reduction required to maintain cost neutrality.
Results:
Actual baseline IV iron and ESA dose utilization across 13 adult HD units were 118 mg/patient/month (95% confidence interval [CI]: 102-134 mg) and 20,764 IU/pt./mo. (95% CI: 18,104-23,424 IU), respectively. The mean combined cost of ESA and IV iron was $315/pt./mo. (95% CI: $274-$355). In comparison, using the results of the PIVOTAL trial and assuming a high-dose IV iron scenario, we estimated mean IV iron use of 215 mg/pt./mo. (95% CI: 187-243 mg/pt./mo.) and a reduction in mean ESA use to 15,923 IU/pt./mo. (95% CI: 13,883-17,962 IU/pt./mo.). This resulted in an estimated cost savings of $38/pt./mo. (95% CI: $33–$42/pt./mo.) and a total program savings of $370,000 per year (95% CI: $325,000-$420,000). Sensitivity analyses under various alternate conditions also showed potential cost savings. We estimated that a dose reduction of ESA of 10% would be required for cost neutrality with a high-dose IV iron protocol.
Limitations:
Our study is limited in its use of data from a single randomized controlled trial (RCT) to estimate cost savings rather than actualized utilization. Our models do not take into consideration anticipated reductions in transfusions and hospitalizations that could be realized from a high-dose IV iron protocol.
Conclusions:
Based on cost modeling, a high-dose IV iron protocol could be integrated in large Canadian regional hemodialysis program in a cost saving manner. Programs implementing such a protocol should monitor IV iron and EPO use prospectively to determine if the trial protocol as applied in a real-world setting translates into cost savings.
Introduction
Intravenous iron and erythropoietin stimulating agents (ESAs) are standard treatments for end-stage kidney disease (ESKD)-related anemia. Most patients treated with maintenance hemodialysis will require one or both medications to maintain a hemoglobin level within the target range specified by their dialysis program. 1 However, these medications are associated with significant costs to hemodialysis programs and patients. 2 Furthermore, high doses of ESA have been linked to poor cardiovascular outcomes. 3
The recently published Proactive IV Iron Therapy in Hemodialysis Patients (PIVOTAL) trial is the largest randomized study of IV iron dosing in hemodialysis patients. In the high-dose iron arm of this trial, ESA doses were reduced by 19.4% and red cell transfusions reduced by 21%. 4 The IV iron protocol from this trial offers an opportunity to simplify IV iron management in hemodialysis programs and possibly improve outcomes; however, the cost implications of adopting a similar protocol in Canadian hemodialysis programs remain unknown.
The aims of this study were to estimate the cost of implementing a high-dose IV iron protocol in a large regional hemodialysis center under various model conditions and to estimate the ESA reduction required to realize cost savings.
Methods
We conducted a modeling study using baseline data on IV iron and ESA use from an ongoing audit of the anemia protocol within 13 hospital-based and community adult, in-center hemodialysis units in a regional hemodialysis program, Alberta Kidney Care—South (AKC-S). Data on historical IV iron and ESA dosages during hemodialysis were collected from an electronic health record audit of actual delivered medications between September 1, 2018 and November 30, 2018 as part of a quality assurance process to understand baseline medication utilization trends. During the observation period, the AKC anemia management protocol (Supplement 1) followed a reactive iron strategy, similar though not identical to the PIVOTAL trial whereby loading iron doses would be administered if transferrin saturation fell below 20% and maintenance doses if transferrin saturation was between 20% and 40%.
We used local baseline data and the results from the PIVOTAL trial to estimate the expected increase in IV iron use and the expected reduction in ESA usage in our hemodialysis program. To achieve this, we calculated the mean differences realized in the trial and applied them to actual program usage to create the model estimates. Whereas the PIVOTAL trial used exclusively IV iron sucrose, our hemodialysis program used a combination of IV iron sucrose and IV sodium ferric gluconate. Therefore, multiple cost scenarios were analyzed. Our primary model assumed the same IV iron formulation distribution used in the AKC-S program. We assumed equal potency of IV iron formulations and a conversion ratio of darbepoetin alfa to epoetin alfa of 1 mcg:200 IU. 5 The cost of IV iron was derived from the drug cost paid by AKC-S in 2018 of $0.375/mg for IV iron sucrose and $0.274/mg for IV sodium ferric gluconate. The cost of ESA was based on Alberta Blue Cross retail drug formulary, with an estimated cost of $27.60 per 10 mcg darbepoetin alpha and $15.68 per 1000 IU epoetin alfa.
Sensitivity analyses included models with an assumption of 100% usage of each formulation of IV iron and ESA to reflect practices at other Canadian centers. Further analyses included models for hospital and community units separately. Finally, we modeled the costs in the PIVOTAL trial under each intervention arm using Alberta drug prices to estimate costs of replicating the trial protocol. Given that the reduction in ESA use from the trial may not be observed in clinical practice, we also estimated the ESA dose reduction required to achieve cost neutrality.
Results
To inform our modeling, we used data from 826 patients across 13 hemodialysis units in the AKC-S regional program. Overall, 96.1% of the study population was receiving IV iron and 80.5% was receiving ESA. Of those that required IV iron, 86% used IV sodium ferric gluconate (remainder used iron sucrose), and 85% of those that required ESA used darbepoetin alfa (remainder used epoetin alfa). The total yearly medication cost for ESA and IV iron across the AKC-S program ranged from $2.6 to $3.3 million dollars per year. The bulk of these costs were driven by ESA usage, which made up 89% of the medication costs, on average. Mean actual IV iron and ESA doses were 118 mg/patient/month (95% confidence interval [CI]: 102-134 mg) and 20,764 IU/patient/month (95% CI: 18,104-23,424 IU), respectively. The mean cost of ESA and IV iron was $315/patient/month (95% CI: $275-$355), of which $289/patient/month was attributed to ESA and $34/patient/month was attributed to IV iron.
In a high-dose IV iron scenario that maintained our program’s current iron sucrose/sodium ferric gluconate split, we estimated a mean IV iron use of 215 mg/patient/month (95% CI: 187-243 mg) and a mean ESA use of 15,923 IU/patient/month (95% CI: 13,883-17,962 IU). This resulted in estimated cost savings of $38/patient/month (95% CI: $34-$42) and total program savings of $370,000 per year (95% CI: $320,000-$420,000) (Table 1).
Comparison of Baseline Medication Usage to Predicted Usage Under Each Model Condition With Associated Savings (Values Represent Monthly Cost Per Patient).
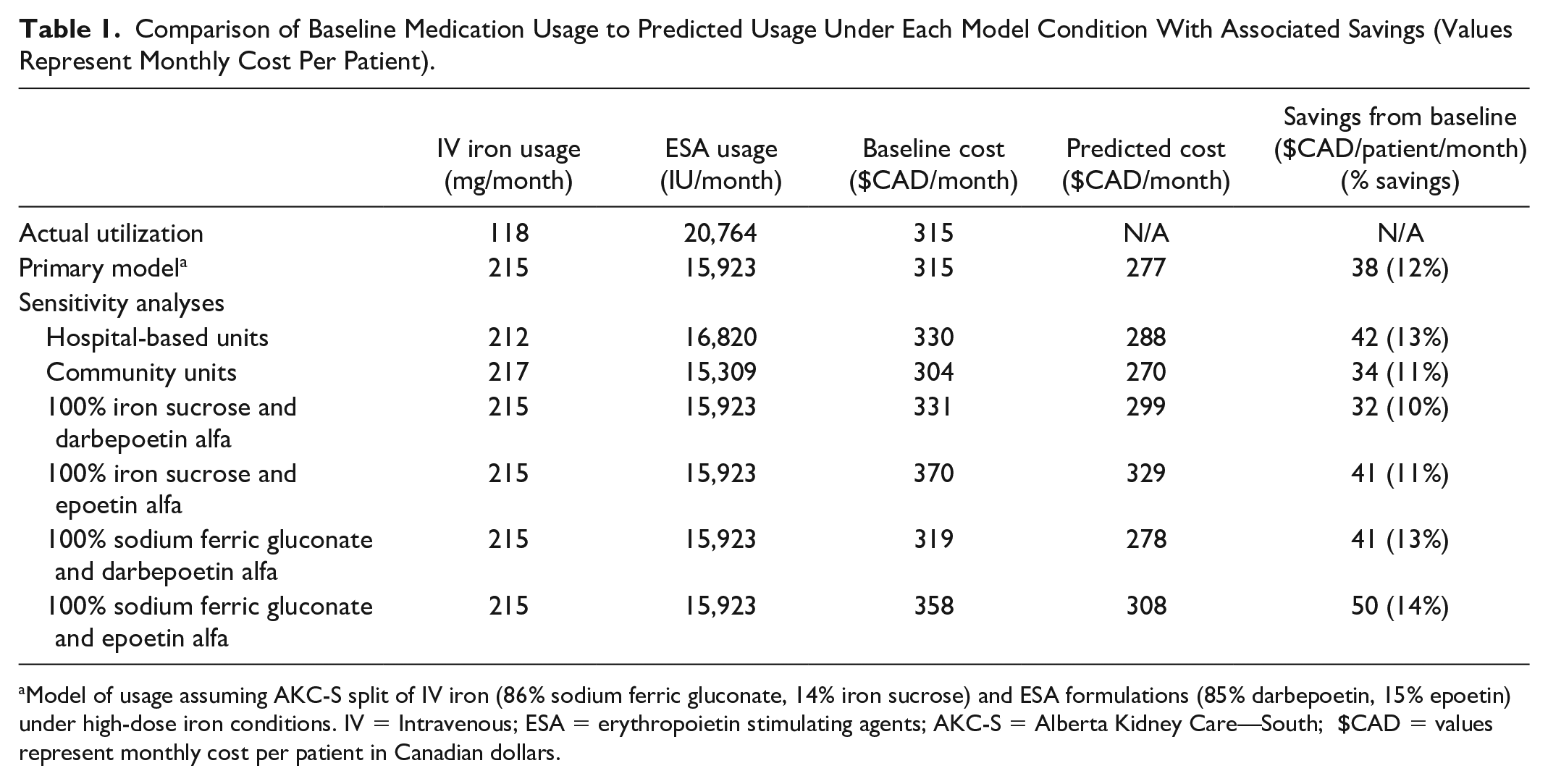
Model of usage assuming AKC-S split of IV iron (86% sodium ferric gluconate, 14% iron sucrose) and ESA formulations (85% darbepoetin, 15% epoetin) under high-dose iron conditions. IV = Intravenous; ESA = erythropoietin stimulating agents; AKC-S = Alberta Kidney Care—South; $CAD = values represent monthly cost per patient in Canadian dollars.
Modeling of medication usage as reported in the PIVOTAL trial with Alberta medication costs showed cost savings of $75/month with a high-dose IV iron protocol assuming AKC-S split of ESA (85% darbepoetin alfa and 15% epoetin alfa). Sensitivity analysis using various combinations of IV iron and ESA formulations all resulted in savings between 10% and 14%. Finally, analyses examining costs according to outpatient hemodialysis unit location showed estimated cost savings of $42/patient/month in hospital-based units and $34/patient/month in community units with a high-dose IV iron protocol (Table 1). We estimated a 10% reduction in ESA dose was required to active cost neutrality with high-dose IV iron.
Discussion
To our knowledge, this is the first study to report the potential cost impact of a high-dose IV iron protocol in a large hemodialysis program. This work shows that high-dose IV iron might reduce costs through its impact on lowering the use of ESAs, as was observed in the PIVOTAL trial. Challenges with the implementation of a program such as this include the multiple payers involved in medication coverage in Canada. Specifically, in Alberta, the budget for IV medications (including IV iron) is often covered by the AKC-S renal program, whereas ESAs are covered by provincial drug plans or private insurance as an outpatient medication. Therefore, hemodialysis programs with similar medication funding models to ours may be disincentivized to implement a high-dose IV iron protocol as they may not realize the cost savings directly. We found this to be a limitation in implementation within our regional setting — administrators within Alberta Health Pharmacy Services were hesitant to approve a protocol that would increase direct costs to the program without proven costs savings that could be derived elsewhere. Given the reduced rates of cardiovascular events in the high-dose IV iron group in PIVOTAL, this protocol may be considered standard of care in the future. However, currently these recommendations have not yet been incorporated into international or Canadian practice guidelines and would likely require confirmation of findings in additional trials to ensure robust results. 6 This barrier could be overcome with more research on the direct cost savings health care systems might realize if reductions in cardiovascular disease-related hospitalizations or transfusions are seen in real-world settings. Furthermore, there may be local hesitation related to implementing a protocol that delivers significantly more IV iron without long-term safety data. Longitudinal safety studies on populations that have implemented a high-dose iron protocol would be beneficial in overcoming this.
Future directions of this work including implementing and testing a high-dose IV iron protocol on prevalent dialysis patients in the Canadian dialysis context. Metrics of interest, including hospitalizations, blood transfusions, actual medication usage, and rates of iron toxicity, should be collected to better appreciate the economic consequences of a high-dose iron protocol. Further study is also needed regarding the potential impact of a high-dose IV iron protocol on patient-relevant outcomes, such as symptoms, quality of life, and care experiences.
Our study is limited by its use of data from a single trial to estimate cost savings rather than actualized utilization. Furthermore, estimates are based on Alberta drug costs and may change with different provincial pricing elsewhere. Our models do not take into consideration likely reductions in transfusions and hospitalizations that could be realized from a high-dose IV iron protocol, nor do they consider potential costs from iron overload, which are challenging to classify clinically in a dialysis population and therefore measure. 7
Conclusions
In this modeling study, we estimated that a high-dose IV iron protocol may be cost saving among adults receiving in-center hemodialysis in Southern Alberta. Important next steps will be to adapt and implement a similar protocol in practice, track iron and ESA usage prospectively, and determine whether trial results are mirrored in the real world through actual cost savings.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581211063984 – Supplemental material for Evaluation of the Cost of a High-Dose Intravenous Iron Protocol in a Regional Hemodialysis Program: Research Letter
Supplemental material, sj-docx-1-cjk-10.1177_20543581211063984 for Evaluation of the Cost of a High-Dose Intravenous Iron Protocol in a Regional Hemodialysis Program: Research Letter by Adam Papini, Braden J. Manns and Meghan J. Elliott in Canadian Journal of Kidney Health and Disease
Footnotes
Ethics Approval and Consent to Participate
An REB exemption was provided by the Conjoint Health Research Ethics Board at the University of Calgary due to the focus of this project being quality improvement.
Consent for Publication
All authors read and approved the final version of this manuscript.
Availability of Data and Materials
This study is based in part on data provided by Alberta Health Services. We are not able to make our dataset available due to restrictions on sharing confidential patient-level data in the setting of waived consent of participants, and patients could be identifiable with our sample size. Questions about the data can be directed to the authors directly.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
