Abstract
Background:
Patients treated with hemodialysis and prescribed warfarin typically have lower time in therapeutic range (TTR) compared to the general population. This may result in less benefit or increased risk of over anticoagulation in these patients.
Objective:
To assess effectiveness of use of an electronic nomogram for the management of warfarin therapy in patients treated with hemodialysis.
Design:
Retrospective chart review.
Setting:
Adult patients treated with hemodialysis.
Patients:
Patients on hemodialysis receiving warfarin for the management of atrial fibrillation (AF) with therapy managed by nursing led electronic nomogram.
Measurements:
Time in therapeutic range (as fraction and Rosendaal).
Methods:
Retrospective chart review over 1 year of international normalized ratio (INR) results was completed, and TTR was calculated. Comparison of patients with TTR greater than 60% to those less than 60% was completed using chi-square analysis.
Results:
Of 43 patients with warfarin therapy managed by the nomogram, the mean TTR was 55.2% (calculated by fraction method) or 61.2% (calculated by Rosendaal method). More than half of the patients (63.5%) had moderate to good control, defined as TTR greater than 60%. Female sex, liver disease, or history of substance use and more medication holds were associated with lower TTR.
Limitations:
Small sample size and retrospective nature of review.
Conclusions:
The results of this review supports the use of an electronic, nursing-led nomogram for the maintenance management of warfarin therapy in stable patients treated with hemodialysis, as use results in TTR greater than 60% for more than half of patients.
Introduction
Oral anticoagulation has been shown to be effective in reducing the thromboembolic risk of atrial fibrillation (AF) in the general population. However, for patients treated with hemodialysis, the decision to provide anticoagulant medication for AF remains controversial due to a lack of robust and often conflicting evidence in this unique patient population. 1
For those that decide to pursue anticoagulation, international normalized ratio (INR)-adjusted warfarin is typically the agent of choice. 2 The time in therapeutic range (TTR) is a method of assessing the proportion of time the patient’s INR is maintained in the prescribed therapeutic window and is an accepted measure to assess the effectiveness and safety of warfarin. 3 In the general population, as well as in those with kidney dysfunction, TTRs above a threshold of at least 65% have been associated with reduced thromboembolic risk or reduction in risk of complications, such as intracranial bleeding.3,4
Locally, the INR of most patients on hemodialysis receiving warfarin are managed by nursing staff on the hemodialysis unit following an electronic warfarin nomogram. The nomogram uses the INR result and current warfarin dose to provide nursing with an order for warfarin dose adjustment (if appropriate) and time to next INR (Supplement 1). The nomogram also advises nursing with direction to hold warfarin (INR values greater than 4.1) or to contact a prescriber (INR values greater than 5.1 or signs of bleeding). Previous evaluation of the nomogram showed similar INR control compared to physician ordering. 5 However, it has been shown that the TTR of patients on hemodialysis are often much lower than the commonly accepted INR threshold of greater than 65%, even with physician management of warfarin dose adjustments, 6 which may contribute to the lack of benefit and potentially higher harm seen in many observational studies looking at anticoagulation use in patients treated with hemodialysis with AF. Specific evaluation of our nomogram effectiveness in terms of TTR had not been completed, and therefore, a quality assurance review was undertaken. The primary aim of this project was to determine the TTR of patients managed with warfarin for AF using our center-specific protocol.
Methods
Two independent individuals performed a retrospective chart review of patients receiving hemodialysis taking warfarin for AF with INR values drawn from January 2018 to January 2019 in Calgary, Alberta. Patient demographics and INR values were gathered from the Southern Alberta Renal Program patient information system (PARIS) database. 7 Patients with warfarin included in their medication profiles or identified as being followed on the warfarin nomogram in PARIS were identified and screened according to the inclusion criteria. All persons receiving hemodialysis in an outpatient setting for >3 months were screened for eligibility. Those above the age of 18 and receiving warfarin anticoagulation via our warfarin nomogram for the indication of AF were eligible for inclusion.
TTR was calculated for each patient using Rosendaal method 8 and a fraction of INRs in range. Rosendaal method assumes linear progression of change in INR between results to provide an estimation of the number of days between laboratory values that were within target range. The calculations were performed using Microsoft Excel 2016. Patients were separated into 3 groups based on TTR: good control (>75%), moderate control (60%-75%), and poor control (<60%) based on the previous groups identified by Quinn et al. 6 Evaluating the effectiveness of the nomogram was done by comparing the TTR from our patient data set to the TTR goal of moderate to good control (TTR > 60%).
Medication lists were examined to determine drug interactions that may affect TTR. Interacting medications were defined as those that are deemed as highly probable or probable in causing a clinically significant drug interaction with warfarin 9 and then further defined as those that are known to affect INR (and therefore TTR).
Periods of times that warfarin was held were identified through the PARIS database and INR results for a period of 14 days postrestart of warfarin were excluded to allow time to achieve therapeutic steady state following the re-initiation of therapy. Indication for hold was determined based on concurrent INR and by review of patient’s hemodialysis chart. Indication for holds were classified as supratherapeutic, procedure, adverse effect, or other/unknown.
Comparison of patients with more than 60% TTR to those with less than 60% TTR was done using chi-square analysis as well as logistic regression (if sample size allowed) using STATA (www.STATA.com).
Results
A total of 193 patients were identified for screening, with 43 patients meeting inclusion criteria. Most of the patients excluded were excluded due to indications other than AF for anticoagulation (n = 75) or warfarin not being managed according to the nomogram (not on nomogram, n = 56) (Figure 1). Of the 56 patients that were classified as not on nomogram, 18 patients were managed by physician (5 due to physician preference but unable to confirm reason for 13 patients), 1 patient was managed by physician due to an INR target range outside of nomogram parameters and 2 patients self-adjusted warfarin doses. Ten patients appeared to have mixed management (management according to the nomogram and by physician with unclear distinction of times of nomogram management), and it was unclear if nomogram or physician-directed therapy was used for 5 patients. The remaining 20 patients were classified as not on nomogram but also met additional exclusion criteria (less than 90 days on dialysis, n = 5, indications other than AF, n = 7, unclear indication, n = 6, not ambulatory patients n = 2).

Patients meeting inclusion criteria.
Demographics of the included patients are shown in Table 1. Most patients were male with a mean age of 74.1 years. Patients may have had more than one cause of kidney disease recorded in the electronic health record and may have been included in more than one classification of kidney disease. Most patients had some component of diabetic and ischemic nephropathy contributing to their disease, with more than 60% of patients having at least one of these causes listed.
Baseline Characteristics of Patients Treated With Hemodialysis Taking Warfarin as Per Nursing Led Warfarin Nomogram.
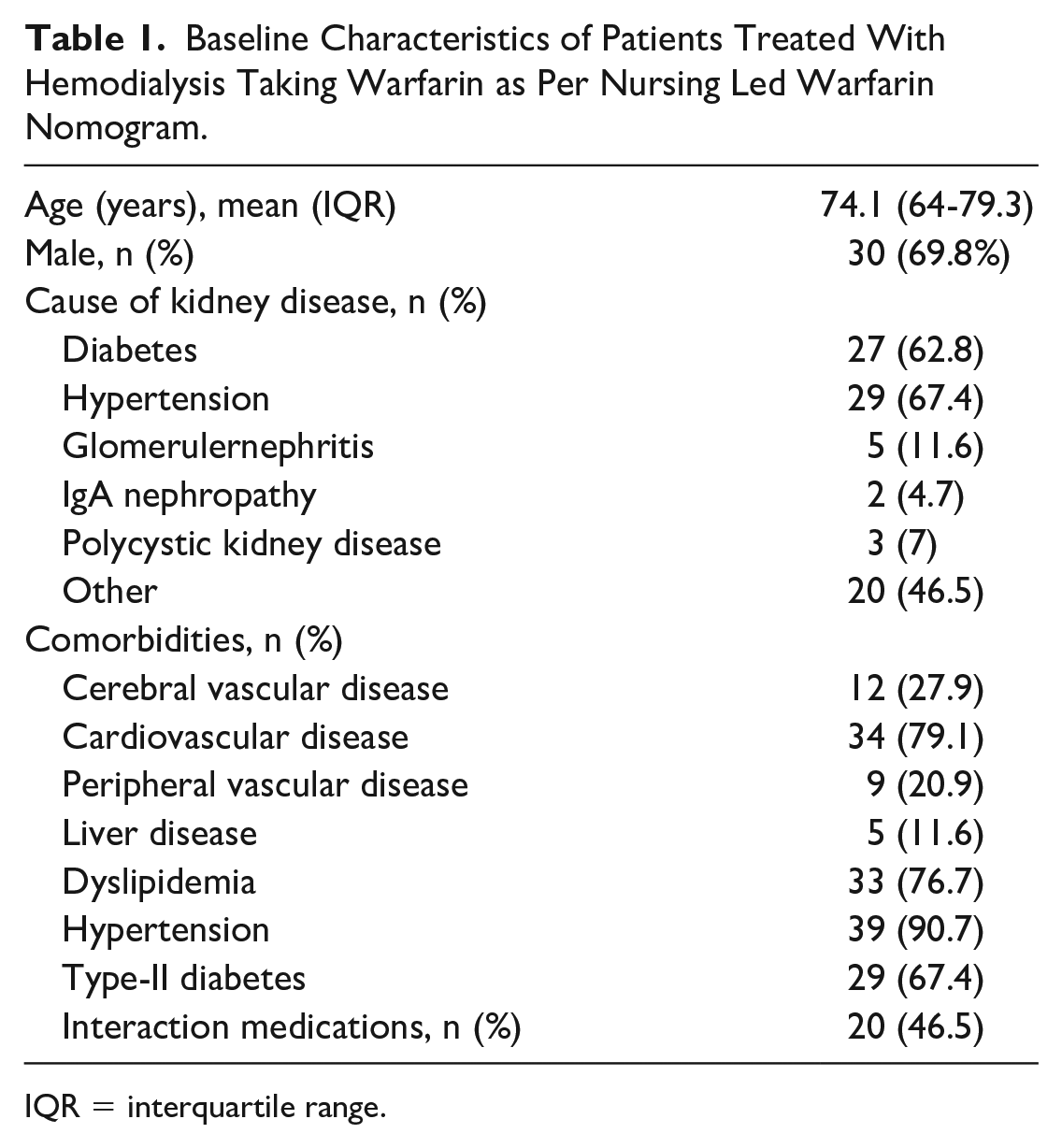
IQR = interquartile range.
Most patients had a history of cardiovascular disease, defined as documented history of coronary artery disease, myocardial infarction, angina, coronary artery bypass surgery, or congestive heart failure. About a quarter of patients had history of cerebral vascular or peripheral vascular disease defined as history of cerebral vascular event, transient ischemic attack (TIA), or carotid surgery or previous vascular surgery, claudication or ischemic ulcers, respectively.
Interacting medications were identified in 20 patients, with a total of 35 courses of therapy seen. Identified medications included metronidazole (n = 6), fluoroquinolones (n = 18), macrolides (n = 6), fluconazole (n = 1), and amiodarone (n = 4).
The mean TTR of patients was 61.2% (SD = 17.3) using Rosendaal method and 55.2% (SD = 16.8) when calculated using the fraction method. Out-of-range INR readings were subtherapeutic 23.1% or 21.9% of the time and supratherapeutic 18.8% or 11.2% of the time, calculated by fraction and Rosendaal, respectively (Table 2).
TTR of Patients Treated With Hemodialysis on Warfarin Managed by Warfarin Nomogram.

Note. TTR = time in therapeutic range; INR = international normalized ratio.
As per the predefined classifications of TTR, about 46.5% of patients had poor control of their warfarin therapy (TTR < 60%), whereas 23.3% had good control (TTR > 75%) and 30.2% had moderate control (TTR = 60%-75%) (Figure 2).

Percentage of patients with poor, moderate, and good control of INR.
TTR was calculated for the subset of patients that were excluded due to being managed by physician for INR monitoring and warfarin dose adjustment but that met other inclusion criteria (n = 19). Mean TTR was 48.6 or 46.9 (Rosendaal and fraction). The number of patients with TTR > 60% was 4 (21%) or 5 (26.3%) (Rosendaal and fraction, respectively).
There was a total of 150 times that warfarin was held in the 43 patients. Supratherapeutic INR (n = 106) was the most common indication for therapy interruption, with the remaining 35 holds due to procedures and 9 times due to adverse effects, such as bleeds (n = 2) or undocumented reasons.
Comparison of patients showed that those with a TTR of less than 60% were more likely to be female gender (P = .033), have liver disease (P = .031), substance use (P = .007), or have a higher number of therapy interruptions (P = .040). Regression analysis was not carried out due to small sample size and large number of potential variables.
Discussion
The nomogram used within Alberta Kidney Care-South was previously shown to result in similar INR control to physician-controlled management, with 77.4% of INR results between 2 and 3 for patients managed with the nomogram over a 5-month period (versus 82.2%). 5 However, many studies have shown that INR control in patients treated with hemodialysis is suboptimal, regardless of physician assessment and prescribing of therapy. 6 This quality assurance study showed that patients receiving hemodialysis with AF that were managed by a nurse-driven electronic nomogram for monitoring and dosing of warfarin therapy had mean TTR of 61.2 ± 17.3 and 55.2% ± 16.8% as calculated by Rosendaal and fraction methods, respectively. Although this study showed that the TTR is not meeting the generally accepted goal of greater than 65%, similar to other studies in patients treated with hemodialysis,6,10 it appears that the use of the nomogram may result in warfarin control close to this target and to the estimated average in the general population (~63%-67%).11,12
At our center, 53.5% of patients managed using a nomogram had good to moderate control of INR, which was defined as TTR greater than 60%. When compared to similar studies in hemodialysis populations, Quinn et al showed only ~15% of patients with moderate to good control when using physician management for INR. While this finding favors the use of nomograms in the hemodialysis population, it is confounded by the possibility of selection bias. That is, at our center, patients that have difficult to manage INR, or those on warfarin doses not covered by the nomogram (>10 mg daily) are converted to physician management. Therefore, only those patients with less labile INR values remain managed by nomogram. In contrast, of the small subset of patients in this cohort that had warfarin therapy managed by physician, 26% and 21% of patients had TTR greater than 60% when TTR was calculated as fraction and Rosendaal, respectively, which is similar to the results from Quinn et al. 6 Both the mean TTR and number of patients with good to moderate control using a nomogram, compared within our own cohort as well as to other hemodialysis populations, is important because it shows the utility of a nomogram in this patient population and further confirms the efficacy of the electronic nomogram. However, the comparison is limited by exclusion of the subset of patients where it was unclear if the nomogram was followed for a period of time and then discontinued, as well as those who had mixed management (both physician and nomogram). The exclusion of these patients from the nomogram may have been due to the medical complexity of the patient or lability of INR results, reducing the nomogram’s generalizability. Unfortunately, the retrospective nature of this study limits the ability to ascertain reasons why nomogram was not used in these patients. A randomized controlled evaluation would be useful in determining the nomogram’s generalizability and efficacy regardless of patient complexity or baseline characteristics. Interestingly, Quinn et al noted that subtherapeutic INR was more likely than supratherapuetic INR, perhaps showing a more conservative approach of prescribers in this patient population. 6 The use of the nomogram showed approximately equal possibility of supra or subtherapeutic INR and, although not a main outcome of this review, only 2 bleeding events were documented during the 1-year period (1 possible gastrointestinal [GI] bleed and 1 eye bleed). This may imply that although these patients have an increased bleed risk, a more conservative approach in warfarin therapy is not required and that use of an electronic nomogram does not result in an increased incidence of bleeds. However, these conclusions are limited by the small study population (N = 43) and retrospective study design.
Current local policy related to anticoagulation and the use of the warfarin nomogram directs nursing to contact a prescriber for assessment and ordering of anticoagulation following therapy interruption, as the nomogram has only been established for use in maintenance warfarin therapy. As such, a 14-day period was arbitrarily used in this review to establish maintenance dosing following a hold of therapy. It is unclear from the retrospective nature of this review if maintenance dosing was established prior to the 14-day period (and if use of the nomogram was initiated prior to day 14, resulting in more time that the nomogram could have been assessed) or if the nomogram was used (inappropriately) following medication holds. It is also an important factor when considering the use of the nomogram in a patient with frequent therapy interruptions because the results of this review only confirms the use of the nomogram for maintenance dosing.
Most of the medication holds (106/150) were due to high INR and, interestingly, holds were associated with TTR less than 60% despite censoring of INR results from analysis for a 14-day period after therapy interruption and re-initiation. It is possible that the use of the nomogram following therapy interruption may have contributed to the number of supratherapeutic INR results seen, as well as the association of holds to lower TTR. Further work is ongoing to evaluate how warfarin is managed peri-medication hold in patients managed by the nomogram. This has included further development of the nomogram to include direction following therapy interruption and assessment of nursing education and policy development.
Female sex, substance use, and liver dysfunction were also associated with lower TTR. Due to the association of coagulopathy with liver disease and a potential affect of metabolism with liver dysfunction, it is not surprising that history of liver disease was associated with worse control of anticoagulant therapy. Substances such as alcohol and smoking can interact with warfarin metabolism, potentially contributing to lability of INR result with use and possibly explaining the association between substance use and lower TTR.
The association of female sex with lower TTR in patients treated with hemodialysis is interesting, as the same association has been shown in patients without renal dysfunction, possibly contributing to the higher stroke risk shown in females with AF in the general population.13,14 The similar association in this cohort may imply the same trend is seen in the hemodialysis patient population.
The incidence of stroke, thromboembolic events, or bleeds were not looked at specifically in this review. Low TTR is just one of the potential factors that has been postulated to contribute to the lack of benefit and increased risk seen with anticoagulation use for the indication of AF in patients with end-stage kidney disease on hemodialysis.1,15 The question of benefit of anticoagulation for AF in the hemodialysis patient population still requires robust randomized controlled trials.
Conclusions
This quality assurance study further supports the use of an electronic warfarin anticoagulation nomogram administered by nursing for maintenance management of INR results in stable patients who receive hemodialysis. TTR was 55.2% and 61.2% calculated by fraction and Rosendaal, respectively, and more than half of the patients (53.5%) had a TTR greater than 60%. Previous evaluation of the nomogram showed a significant reduction in the frequency of INR testing without an increase in bleeding or thrombotic events. The results of this review were used to validate and improve the protocol policy and nursing education regarding the utilization of the nomogram, especially around therapy interruptions. The use of a nomogram for complex patients with multiple interruptions in therapy or with labile INRs has not been validated. Further assessment of use of the nomogram in patients with difficult to manage INR, factors associated with low TTR and, ultimately the benefit of anticoagulation for AF in patients on hemodialysis are required.
Ethical Considerations
A Project Ethics Community Consensus Initiative (ARECCI) is a program that helps identify and mitigate ethical risks for program evaluations, quality improvement initiatives, or knowledge translation projects. Prior to data collection and analysis, an ARECCI ethics screening tool and questionnaire were completed, and the protocol was discussed with an ARECCI reviewer. Given the quality improvement nature of the project, formal research ethics were not sought but ethical considerations in the collection, use, and storage of personal health information were identified. Strategies were implemented to mitigate potential risk to patients.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581211046079 – Supplemental material for Time in Therapeutic Range Using a Nomogram for Dose Adjustment of Warfarin in Patients on Hemodialysis With Atrial Fibrillation
Supplemental material, sj-docx-1-cjk-10.1177_20543581211046079 for Time in Therapeutic Range Using a Nomogram for Dose Adjustment of Warfarin in Patients on Hemodialysis With Atrial Fibrillation by Kimberly Defoe, Jenny Wichart and Kelvin Leung in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors would like to acknowledge Katina Woo, BScPharm, PharmD, for her assistance with data collection and Dr. Daniel Dutton, PhD, for his statistical guidance.
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Materials
Patient data cannot be made available due to health information disclosure and use restrictions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
