Abstract
Background:
Cardiac surgery–associated acute kidney injury (AKI) is an adverse outcome that increases morbidity and mortality in patients undergoing cardiac surgical procedures. To date, the use of serum creatinine levels as an early indicator of AKI has limitations because of its slow rise and poor predictive accuracy for renal injury. This delay in diagnosis may lead to prolonged initiation in treatment and increased risk for adverse outcomes.
Objective:
This pilot study explores serum and urine matrix metalloproteinases (MMPs)-2 and MMP-9 and their association, and potentially earlier detection of AKI in patients following cardiopulmonary bypass (CPB)–supported cardiac surgery. We hypothesize that increased activity of serum and urine levels MMP-2 and/ or MMP-9 are associated with AKI. Furthermore, MMP-2 and/ or MMP-9 may provide earlier identification of AKI as compared with serum levels of creatinine.
Methods:
During the study period, there were 150 CPB-supported surgeries, 21 of which developed AKI according to the Kidney Disease Improving Global Outcomes criteria. We then selected a sample of 21 matched cases from those patients who went through the surgery without developing AKI. Primary outcomes were the measurement via gel zymography of the serum and urine activity of MMP-2 and MMP-9 drawn at the following intervals: pre-CPB; 10-minute post-CPB; and 4-hour post-CPB time points. Secondary variables were the measurement of serum creatinine, intensive care unit (ICU) fluid balance, and length of ICU stay.
Results:
At the 10-minute and 4-hour post-CPB time points, the serum MMP-2 activity of AKI patients were significantly higher as compared with non-AKI patients (P < .001 and P = .004), respectively. Similarly, at the 10-minute and 4-hour post-CPB time points, the serum MMP-9 activity of AKI patients was significantly higher as compared with non-AKI patients (P = .001 and P = .014), respectively. The activity of urine MMP-2 and MMP-9 of AKI patients was significantly higher as compared with non-AKI patients at all 3 time points (P = .004, P < .001, P < .001), respectively.
Conclusion:
Although the pilot study may have limitations, it has demonstrated that the serum and urine levels of activity of MMP-2 and MMP-9 are associated with the clinical endpoint of AKI and appear to have earlier rising levels as compared with those of serum creatinine. Furthermore, in depth, exploration is underway with a larger sample size to attempt validation of the analytical performance and reproducibility of the assay for MMP-2 and MMP-9 to aid in earlier diagnosis of AKI following CPB-supported cardiac surgery.
Introduction
Up to 36% of patients undergoing cardiopulmonary bypass (CPB)–supported heart surgery develop cardiac surgery–associated acute kidney injury (AKI), which is associated with increasing mortality, morbidity, and health care costs.1-3 AKI is characterized by a sudden, sustained, and functionally significant decrease in renal function with insufficient elimination waste products.4,5 Although nephrotoxins have been implicated, 2 the exact mechanism of this condition is unknown with most evidence pointing toward CPB.6-8 The use of serum creatinine (SCr) as an early indicator of AKI has limitations because of its slow rise and poor predictive accuracy for renal injury. 9 This delay in diagnosis may lead to a late initiation in treatment and increased risk of adverse outcomes. 10 Recent research investigating the early detection of AKI has been promising, but lack of a reliable predictor or early detector remains. 11 This research investigates serum and urine activity of matrix metalloproteinases (MMPs) MMP-2, and MMP-9, and their association with, and earlier detection of, AKI compared with the current “Gold Standard” levels of SCr.
MMP-2 and MMP-9 are proteolytic enzymes that play important roles in a variety of physiological and pathological processes, and are best known for degradation of extracellular proteins and remodeling of the extracellular matrix and collagens.12,13 Research with an animal model of myocardial infarction demonstrated that MMPs are released throughout the continuum, from ischemia, through injury, to infarction, and exacerbate injury to the myocardium. 14 Human atrial tissue biopsies reveal increased MMP activity following CPB. 15 Moreover, levels of MMP-2 and MMP-9 are increased in renal ischemia/reperfusion (I/R) injury in animal models.16,17 Our research employing a machine cold perfusion rat kidney model shows that MMPs are involved in kidney preservation injury and that MMP-2 inhibition with either doxycycline or MMP-2 small silencing ribonucleic acid (siRNA) as an inhibitor results in markedly decreased injury markers.18,19 However, research demonstrating an association between MMP activity and AKI has been studied sparsely. 20 Taken altogether, the literature suggests that I/R may promote increased circulating activity of MMPs, which may have a significant association with tissue injury. Increased activity of serum MMP-2 and MMP-9 are hypothesized to be associated with AKI. Furthermore the measurement of, MMP-2 and/or MMP-9 may provide earlier detection of AKI as compared with levels of serum creatinine.
Materials and Methods
Patient Sample
The University of Saskatchewan Biomedical Research Ethics Board approved the study protocol, and the Provincial Health Authority granted operational approval. Written consent for study participation was obtained and the study period ranged from December 2018 to December 2019. This research was a prospective, proof-of-concept, pilot study, consisting of 150 CPB-supported surgeries where 21 patients developed AKI according to the Kidney Disease Improving Global Outcomes (KDIGO) criteria. 21 Of the remaining 129 non-AKI patients, we used a matched based design to select a sample of 21 patients (non-AKI) matched by age (± 10 years), sex, body mass index (BMI; ± 5 kg/m2), EuroScore (± 10), baseline (pre-bypass) estimated glomerular filtration (eGFR; ± 5 mL/min/1.73 m2), and type of surgery with the AKI patients, rendering a final sample consisting of 42 patients. Demographic data and clinical characteristics were collected pre-, peri-, and post-operatively.
The inclusion criteria consisted of both sexes, 18 to 85 years of age, undergoing elective or urgent cardiac surgery with a hemoglobin (Hgb) ≥100 g/L. The exclusion criteria included patients for emergent surgery, pre-existing chronic kidney disease (eGFR ≤ 30 mL/min/1.73 m2) on dialysis or prescribed nephrotoxic mediations (fractionated heparins, opiates, penicillin-based antibiotics, and acyclovir). 22
Cardiopulmonary Bypass
Anesthesia followed standard cardiac surgical protocol. Heparin dosing was determined by a hemostasis management system, 2017 (HMS) (Medtronic, Minneapolis, MN). Cardiopulmonary bypass was initiated after attaining an activated clotting time (ACT) of ≥ 480 seconds.
The CPB system used a Sorin S5 fifth-generation modular heart-lung machine, which provided flows (LivaNova, London, United Kingdom, 2015) from 2.4 to 2.8 L/min/m2. Circuit priming consisted of 2 L of Plasma-Lyte A (Baxter, Mississauga, Canada), 50 mEq of sodium bicarbonate, 100 mL of 25% albumin, 2.5 mL/kg of 20% mannitol, and 10 KU of heparin. Following cannulation, the prime volume was reduced with retrograde autologous priming, allowing the replacement of the circuit crystalloid prime with the patient’s blood. 23 Specifically, the arterial and venous lines were carefully drained into the venous reservoir and the crystalloid pumped into a recirculation bag to be used, if required, for fluid replacement during CPB. Activated clotting times during CPB exceeded 480 seconds, and heparin concentrations were ≥ 300 units/kg. Following decannulation, heparin was reversed by an HMS-determined protamine dose and return to the baseline ACT was confirmed. Patients were transported to intensive care unit (ICU) on calibrated weight measuring beds.
Statistical Analysis
Mean, standard deviation, and median were reported for continuous variables. Categorical variables were described by numbers and percentages. Continuous variables were evaluated for normal distribution and statistical analysis was performed accordingly. As appropriate, comparison of categorical variables between groups (AKI, non-AKI) used either a chi-square or Fisher’s exact test. A rank sums test was used for between-group comparisons. An independent-samples t test and Wilcoxon non-parametric test were used to compare normally and non-normally distributed continuous variables, respectively, between groups. For within-group comparison, a paired t test and Signed rank were used for normally and non-normally distributed continuous variables, respectively. A P value of less than .05 (2-sided) was considered as statistically significant. The data analysis for this manuscript was generated using SAS software. Copyright © 2017 SAS Institute Inc. SAS and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute Inc., Cary, NC, USA.
Classification of AKI
Due to lack of general agreement with the previous classifications of AKI,24,25 the KDIGO Acute Kidney Injury Network (AKIN) acknowledged that even smaller and more acute changes in SCr than those previously proposed resulted in adverse outcomes. 26 Post-operatively, patients were separated into categories based on 2012 KDIGO criteria for the diagnosis of AKI: Stage 1: an increase of SCr by ≥ 26.5 µmol/L within 48 hours; or an increase of SCr to ≥ 1.5 to 1.9 times baseline, which has occurred within 7 days; Stage 2: an increase of SCr by 2.0 to 2.9 times baseline which has occurred within 7 days and; Stage 3: increase of SCr serum creatinine to 3.0 times baseline or ≥353.6 μmol/L or initiation of renal replacement therapy. 27 Using the KDIGO criteria, the AKI patients in this study were classified as either Stage 1 or Stage 2.
For the purposes of this study, we did not use the urine volume criteria,21,28 eGFR were calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-Epi) formula.21,29 Our hospital laboratory uses the CKD-EPI equation for eGFR which employs a 2-slope linear spline to model the relationship between GFR and log serum creatinine, age, sex, and race. It has shown good performance for patients with all common causes of kidney disease. 29
Sample Collection Methods and Intervals
Specimens for MMP-2, and MMP-9, activity measurements were obtained at pre-CPB (prior to surgery), 10-minute post-CPB, and 4-hour post-CPB. Blood samples were transferred into 4.0-mL vacutainer serum tubes (EM Science, Merck KGaA, Darmstadt, Germany), allowed to clot for 20 to 30 minutes at room temperature and centrifuged at 3000 revolutions per minute for 15 minute at 8°. Next samples were divided, aliquoted, and storage at –80°C until assays were performed. 30 During sample analysis, thaw-refreeze cycles were avoided. Urine samples were collected into specimen cups and frozen at –80°C until sample analysis. Serum creatinine measurements were collected at the following points: baseline, 4-hour post-CPB, 8- and 18-hour ICU, and daily for 7 days.
Biochemical Measurements
Determination of MMP activity
Matrix metalloproteinases activity was assessed via gelatin zymography. 31 Serum and urine samples were applied to 8% polyacrylamide gel copolymerized with 2 mg/mL gelatin. After electrophoresis, gels were rinsed 3 times for 20 minutes in 2.5% Triton X-100 to remove sodium dodecyl sulfate (SDS). The gels were then washed twice in incubation buffer (50 mM Tris–HCl, 5 mM CaCl2, 150 mM NaCl, and 0.05% NaN3) for 20 minutes at room temperature and then placed in incubation buffer at 37°C for 24 hours. The gels were stained using 0.05% Coomassie Brilliant Blue G-250 in a mixture of methanol: acetic acid: water (2.5:1:6.5, v:v:v) and de-stained in an aqueous solution of 4% methanol and 8% acetic acid (v:v). Developed gels were scanned with a GS-800 calibrated densitometer (Bio-Rad, Hercules, CA, USA) and MMP-2 and MMP-9 activities were measured using Quantity One 4.6 software (Bio-Rad, Hercules, CA, USA).
Determination of Serum Creatinine
Serum creatinine was measured by the central hospital laboratory using a Roche Cobas 6000 analyzer (Roche Diagnostics, Indianapolis, IN).
Results
Demographics
Baseline demographics and clinical characteristics are shown in Table 1. The patients in the 2 groups (AKI vs non-AKI) were matched according to their baseline demographic characteristics. Therefore, there were no significant differences between the 2 groups preoperatively.
Demographics and Clinical Data.
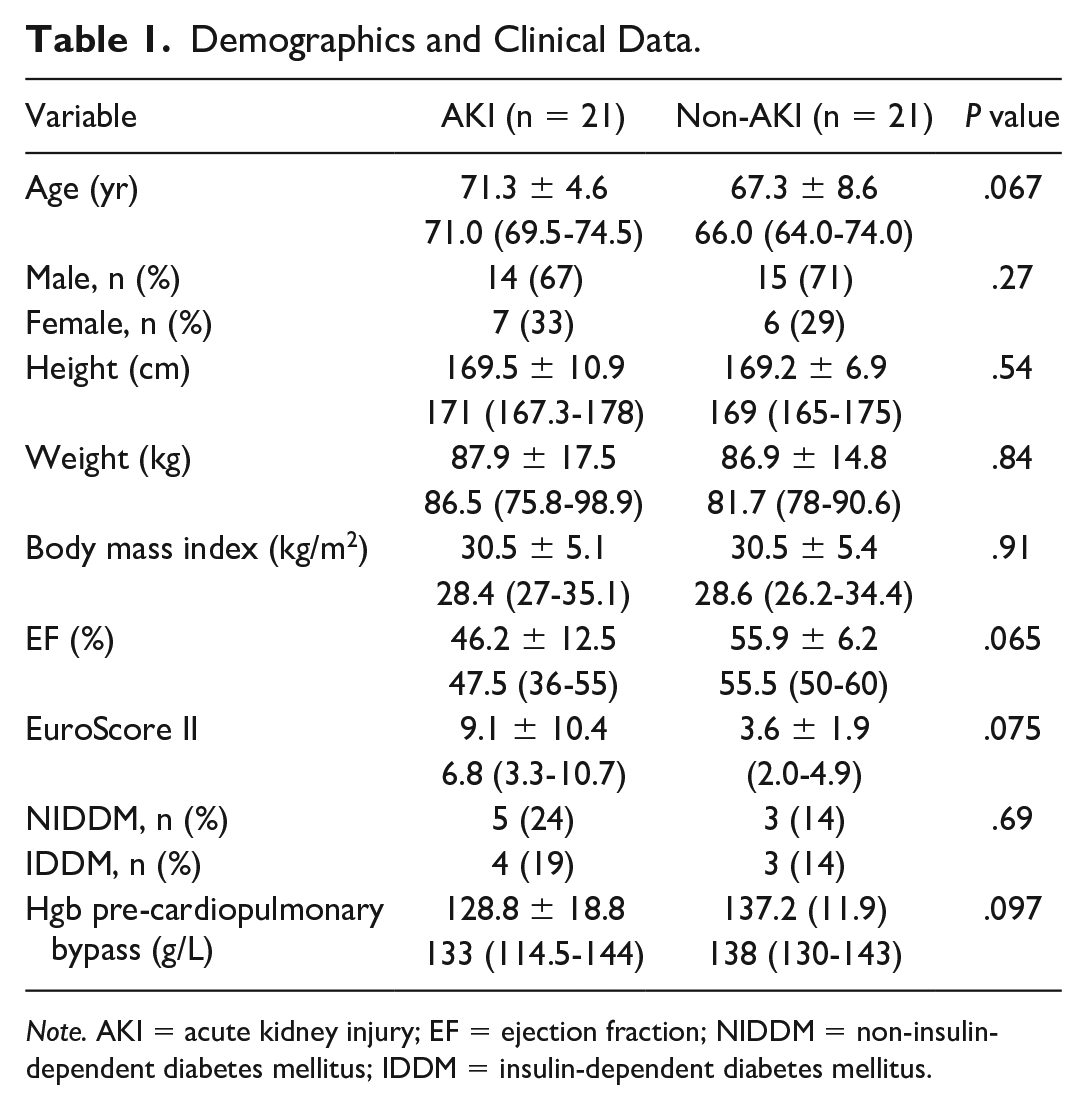
Note. AKI = acute kidney injury; EF = ejection fraction; NIDDM = non-insulin-dependent diabetes mellitus; IDDM = insulin-dependent diabetes mellitus.
Operative and post-operative data are shown in Table 2. Transfusion of RBCs, platelets, and fresh frozen plasma was significantly higher in the AKI as compared with the non-AKI patients. In addition, the ICU fluid balance and length of stay (LOS) in ICU was significantly higher in the AKI as compared with the non-AKI patients. A comparison of the types of surgical procedures is shown in Table 3.
Operative and Post-operative Data.
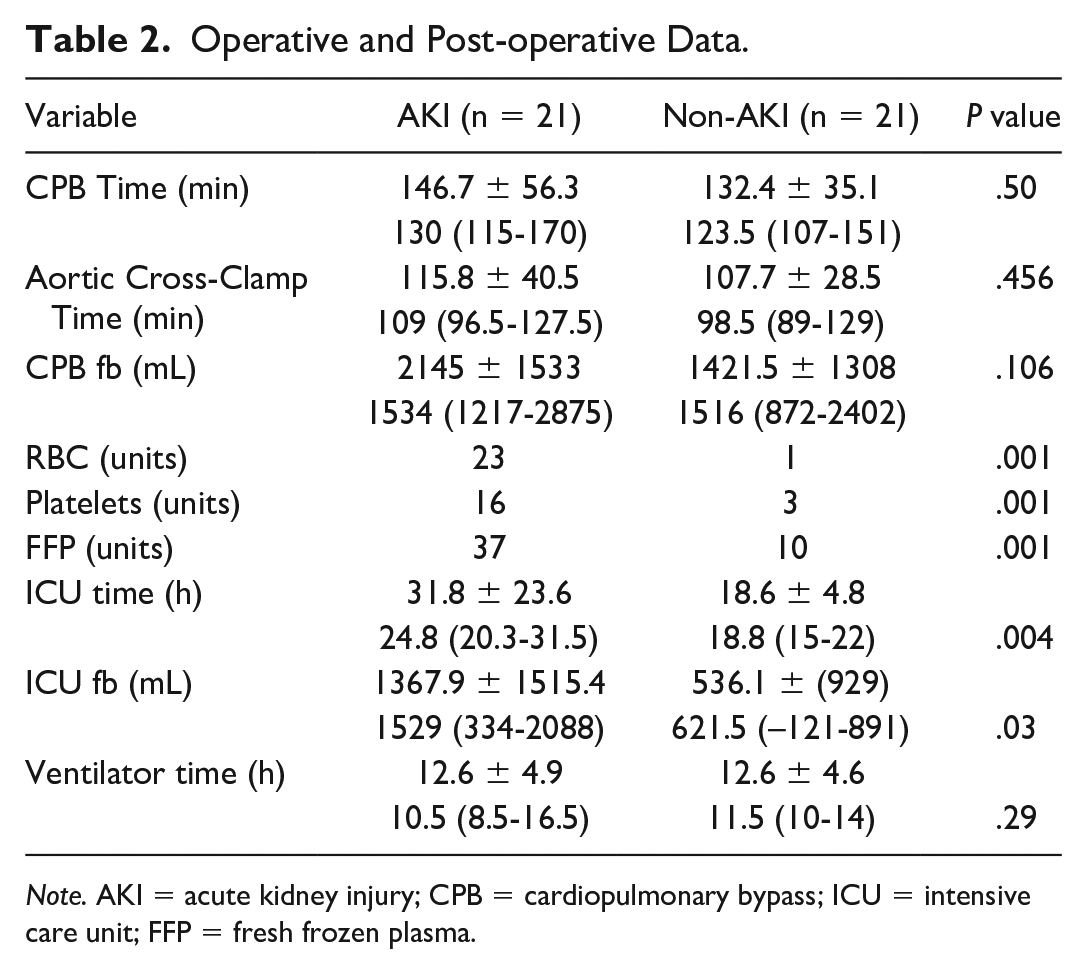
Note. AKI = acute kidney injury; CPB = cardiopulmonary bypass; ICU = intensive care unit; FFP = fresh frozen plasma.
Types of Surgical Procedures.
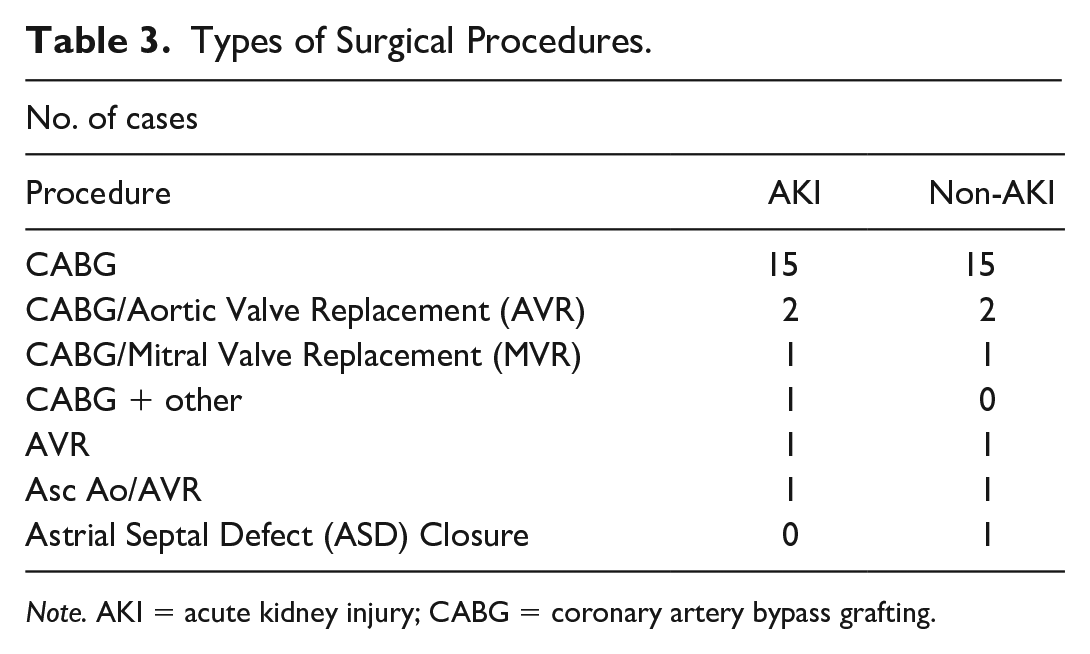
Note. AKI = acute kidney injury; CABG = coronary artery bypass grafting.
Serum Creatinine
The SCr and eGFR values are shown in Table 4. Within the AKI group, there was no significant difference between the levels of SCr at the pre-CPB interval and the 10-minute post-CPB interval. However, at the 4-, 8-, and 18-hour intervals, the levels of SCr rose significantly as compared with the pre-CPB interval. Within non-AKI group, the levels of SCr were not significantly different between pre-CPB time points as compared with any of the subsequent intervals. Between groups, there were no significant differences seen in the AKI and non-AKI groups at baseline or the 10-minute post-CPB interval. There was a significant difference between the 4-, 8-, and 18-hour interval in the AKI group as compared with non-AKI group. Moreover, at 4-, 8-, and 18-hour post-CPB time points, the eGFR was significantly lower in the AKI group compared with the non-AKI group. Table 5 shows the level of SCr of each individual patient who met the criteria for AKI at their respective time points.
Serum Creatinine Levels and eGFR.

Note. AKI = acute kidney injury; CPB = cardiopulmonary bypass; eGFR = estimated glomerular filtration; SCr = serum creatinine.
Time Point to Meet Kidney Disease Improving Global Outcomes Criteria for AKI.

Note. CPB = cardiopulmonary bypass; SCr = serum creatinine.
MMP-2 and MMP-9 activity in serum
For all figures, the asterisk (*) denotes the P value; where P < .05 between groups (AKI vs non-AKI); whereas, the capital (T) denotes P < .05 within group. Serum MMP-2 and MMP-9 activities are shown in Figures 1 and 2, respectively. Within-group analysis of non-AKI patients demonstrated no significant differences in serum MMP-2 and MMP-9 activity in all measured time points. However, within-group analysis of AKI patients showed that the 10-minute post-CPB and 4-hour post-CPB activity of serum MMP-2 (P < .001, P = .043) and MMP-9 (P = .03, P = .005) were significantly higher as compared with the pre-CPB levels, respectively. Between groups, at the 10-minute post-CPB time point, serum activity levels of MMP-2 (P < .001) of AKI patients was significantly higher as compared with non-AKI patients. Between groups, at the 4-hour post-CPB time point, serum activity levels of MMP-2 (P = .004) of AKI patients was significantly higher as compared with non-AKI patients.
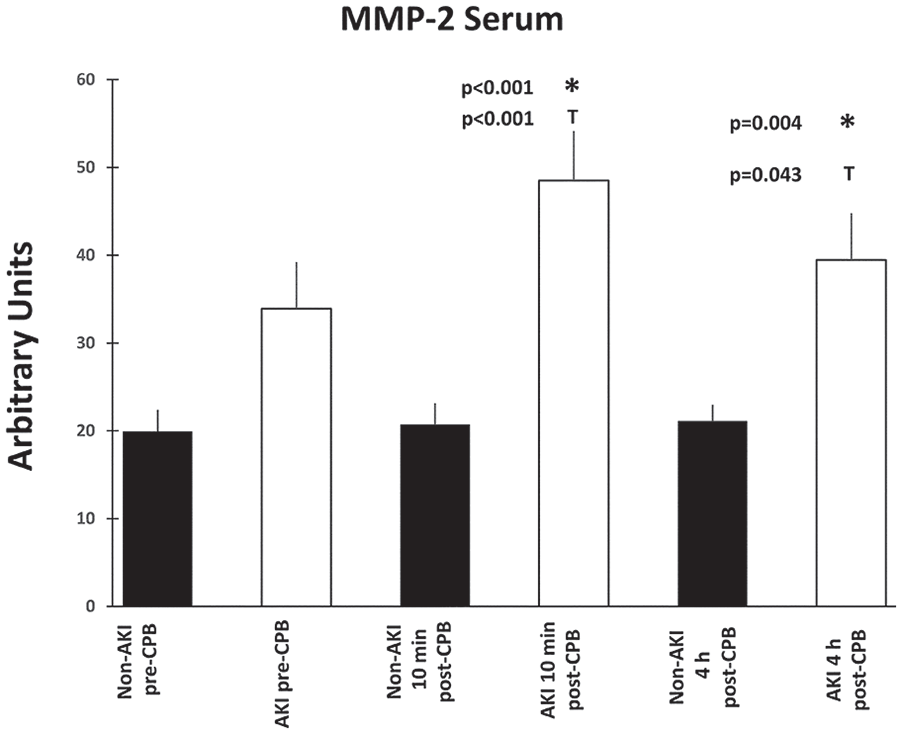
Serum MMP-2 activity in patients undergoing CPB at pre-CPB, 10-minute post-CPB, and 4-hour post-CPB time points.
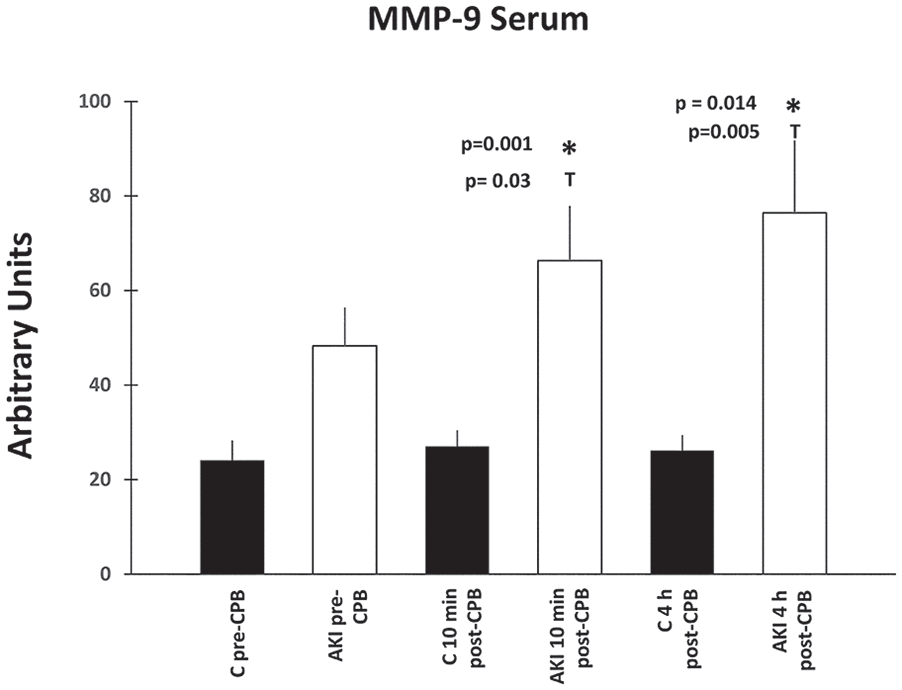
Serum MMP-9 activity in patients undergoing CPB at pre-CPB, 10-minute post-CPB, and 4-hour post-CPB time points.
Similarly, between groups, at the 10-minute post-CPB time point, serum activity levels of MMP-9 (P = .001) of AKI patients was significantly higher as compared with non-AKI patients. Between groups, at the 4-hour post-CPB time point, serum activity levels of MMP-9 (P = .014) of AKI patients was significantly higher as compared with non-AKI patients.
MMP-2 and-9 activity in urine
Urine MMP-2 and MMP-9 activities are shown in Figures 3 and 4, respectively. Within the AKI group, MMP-2 urine activity was significantly (P < .001) higher at the 10-minute post-CPB time point and the 4-hour post-CPB time point (P = .0002) compared with pre-CPB levels. Within the non-AKI group, analysis showed that MMP-2 urine activity was also significantly higher at the 10-minute post-CPB time point and the 4-hour post-CPB time points, respectively (P = .002; P = .002) compared with pre-CPB levels. Between groups, the data demonstrated that urine MMP-2 activity was significantly higher in AKI group as compared with the non-AKI patients at all 3 time points (P = .004, P < .001, P < .001), respectively.

Urine MMP-2 activity in patients undergoing CPB at pre-CPB, 10 minute post-CPB, and 4-hour post-CPB time points.

Urine MMP-9 activity in patients undergoing CPB at pre-CPB, 10-minute post-CPB, and 4-hour post-CPB time points.
Within the AKI group, the urine activity levels of MMP-9 showed that the 10-minute post-CPB (P < .0007) and 4-hour post-CPB (P = .002) time points were significantly higher as compared with the pre-CPB levels. Within non-AKI group, the 4-hour post-CPB (P = .025) urine MMP-9 activity level time point was significantly higher as compared with the pre-CPB levels. Between groups, the data demonstrated that urine MMP-9 activity levels were significantly higher in AKI group as compared with non-AKI patients at all 3 time points (P = .004, P < .001, P < .001), respectively.
Sensitivity and specificity of the MMPs
The ability of 10-minute post-CPB serum MMP-2 time point to predict AKI was assessed using a receiver-operating curve (ROC) (Figure 5). The area under the curve (AUC) = 0.88, 95% confidence interval (CI) 0.779-0.987, and the cut-off value was 23.6 arbitrary units with a sensitivity = 0.95 and a specificity = 0.77. The positive likelihood = 4.2; negative likelihood ratio = 0.065 with a Youden’s J statistic = 0.72. The ability of 10-minute post-CPB urine MMP-2 time point to predict AKI using a ROC (Figure 6) demonstrated an AUC = 0.915, 95% CI 0.831-0.999, and the cut-off value was 12 arbitrary units with a sensitivity = 0.95 and a specificity = 0.727. The positive likelihood = 4.2; negative likelihood ratio = 0.065 with a Youden’s J statistic = 0.723. The ability of the 10-minute post-CPB serum MMP-9 time point to predict AKI using a ROC (Figure 7) demonstrated an AUC = 0.76, 95% CI 0.603-0.91, cut-off value = 37 arbitrary units, sensitivity = 0.70, specificity = 0.64; positive likelihood = 1.9; negative likelihood ratio = 0.47; with a Youden’s J statistic =0.34. The ability of the 10-minute post-CPB urine MMP-9 time point to predict AKI using a ROC (Figure 8) produced an AUC = 0.95, 95% CI 0.831-0.999, cut-off value = 102 arbitrary units, sensitivity = 0.95, specificity = 0.86; positive likelihood = 7.0; negative likelihood ratio = 0.057; with a Youden’s J statistic = 0.81. The 10-minute post-CPB serum MMP-2 and MMP-9 time points combined to predict AKI are shown in Figure 9. Last, the 10-minute post-CPB urine MMP-2 and MMP-9 time points combined to predict AKI are shown in Figure 10.

The 10-minute post-cardiopulmonary bypass serum Matrix Metalloproteinase-2 time point to predict acute kidney injury.
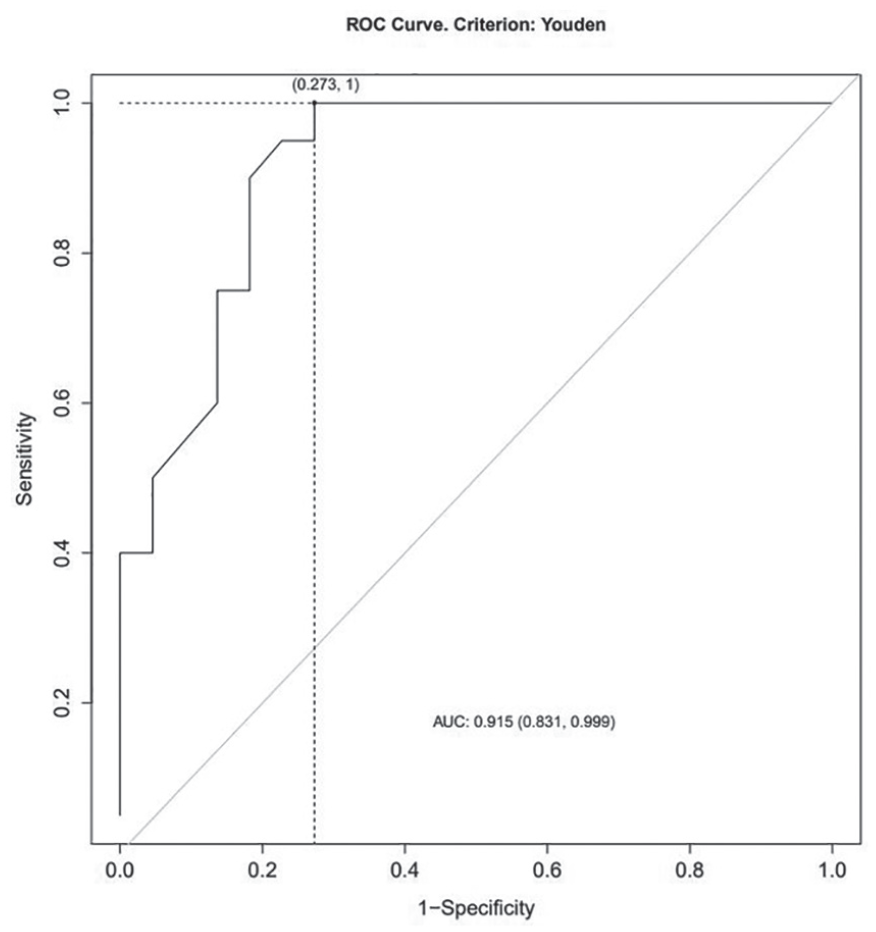
The 10-minute post-cardiopulmonary bypass urine Matrix Metalloproteinase-2 time point to predict acute kidney injury.

The 10-minute post-cardiopulmonary bypass serum Matrix Metalloproteinase-9 time point to predict acute kidney injury.

The 10-minute post-cardiopulmonary bypass urine Matrix Metalloproteinase-9 time point to predict acute kidney injury.
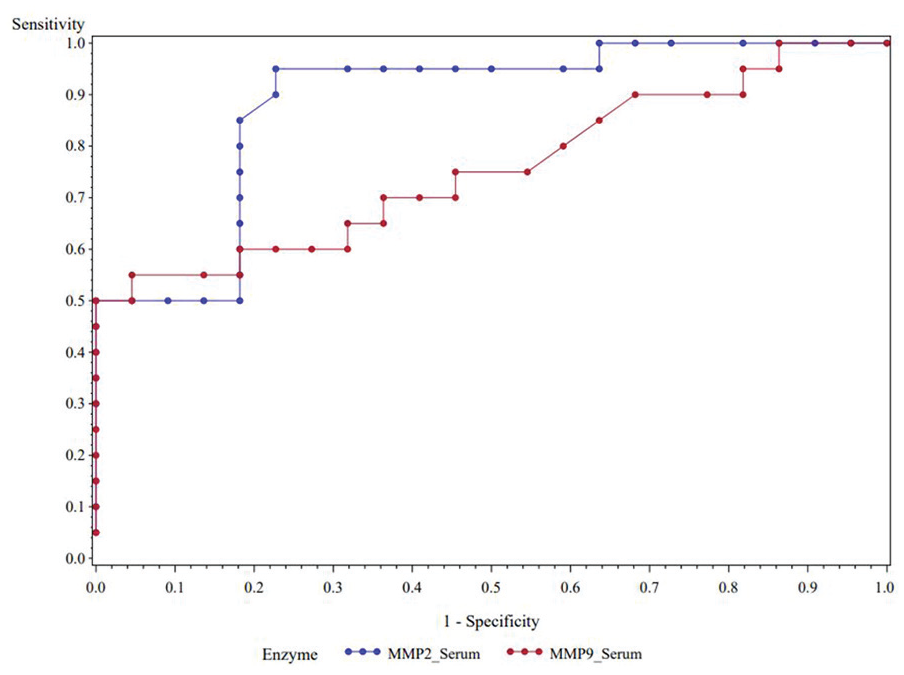
The 10-minute post-cardiopulmonary bypass serum MMP-2 and MMP-9 time points combined to predict acute kidney injury.
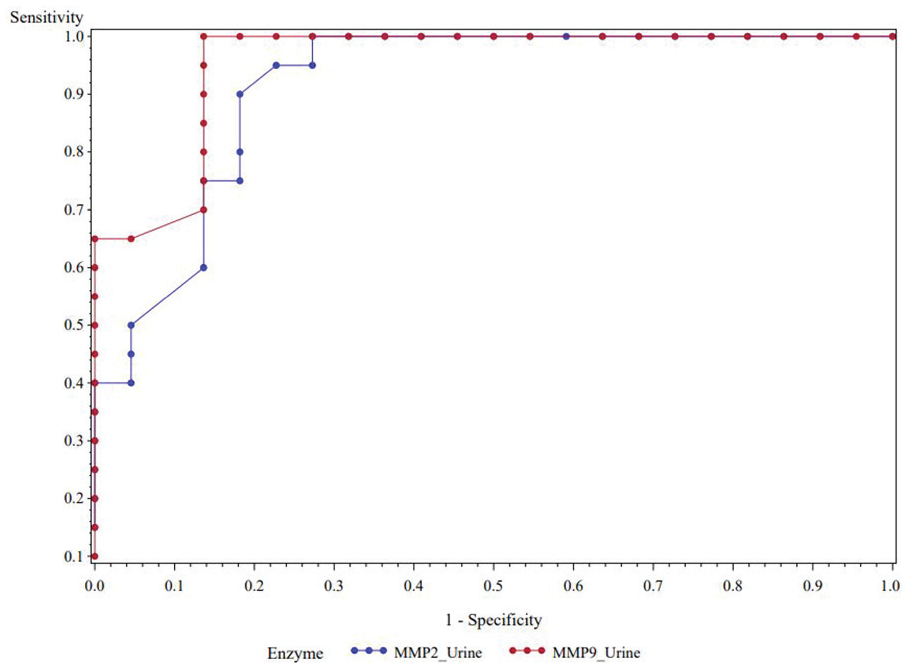
The 10-minute post-cardiopulmonary bypass urine MMP-2 and MMP-9 time points combined to predict acute kidney injury.
Pearson’s correlation
Figures 11 to 14 (supplementary material) demonstrate the correlation pattern between the highest SCr level and serum MMP-2 (Figure 11), urine MMP-2 (Figure 12), serum MMP-9 (Figure 13), and urine MMP-9 (Figure 14) in the AKI and non-AKI groups, respectively. The AKI patients had significant positive correlation between the 10-minute post-CPB serum MMP-9 levels of activity time point versus the highest level of serum creatinine over the post-operative period (r = 0.51, P = .022). Correlation between the highest level of serum creatinine and serum MMP-2 (r = 0.44, P = .05), urine MMP-2 (r = 0.44, P = .05), and urine MMP-9 (r = 0.21, P = .38) showed a positive correlation but did not achieve statistical significance in the AKI group (Figures 9-11). In the non-AKI group, none of the correlations achieved significance.
Linear regression model
The multivariable linear regression model shown in Table 6 (supplementary material), between highest SCr and serum MMP-9, includes multiple risk factors (covariates) that were adjusted for, such as age, sex, ejection fraction (EF%), BMI, EuroScore II, non-insulin-dependent diabetes mellitus (NIDDM), insulin-dependent diabetes mellitus (IDDM), pre-CPB hemoglobin, CPB time, aortic cross-clamp time, CPB fluid balance, ventilator time, ICU fluid balance, and ICU time.
We checked the interaction effect between serum MMP-9 and group. The data demonstrated that among the AKI group, for every 1-unit increase in serum MMP-9 activity, the highest SCr increased significantly by 2.48 units over the post-operative period (P < .0001). However, this phenomenon did not occur among non-AKI patients (P = .19). In addition, the rate of change of the highest SCr between the 2 groups (slope difference) was significantly different (P = .0005). The rate of changes was not significantly different between the 2 groups for serum MMP-2, urine MMP-2, and urine MMP-9.
Discussion
Despite recent advances in CPB, AKI and its associated adverse outcomes continue to occur in up to 36% of patients who undergo cardiac surgery.1,32 Our exploratory study has shown that MMP-2 and MMP-9 activity is significantly increased in the serum and urine of patients who developed AKI compared with those who did not following CPB. Furthermore, the rise in MMP-2 and MMP-9 in serum and urine is seen earlier than the rise in SCr, suggesting that MMP-2 and MMP-9 may be useful in the early detection of AKI. The MMP data gathered from the present study agree and are consistent with previous investigators, 33 and go beyond their investigations to report the association between MMP-2 and MMP-9 and AKI.
Cardiopulmonary bypass–induced hemodilution may contribute to decreased post-operative levels of SCr, potentially delaying the detection of AKI. 34 In the present study, levels of SCr at the 4-hour post-CPB time point were significantly higher in patients who developed AKI as compared with the non-AKI patients. CPB-induced hemodilution may have led to falsely lowered SCr, delaying the diagnosis of AKI. We suggest that the levels of SCr in the patients who developed AKI may have been higher if not for hemodilution. Despite the rise in SCr levels at the 4-hour post-CPB time point, they were not high enough to meet the criteria for AKI. Thus, the protracted rise in SCr levels to meet AKI criteria may result in the loss of a critical time necessary for the treatment of AKI.
It also important to note that among the AKI group at the 10-minute post-CPB time point, the serum and urine levels of MMP-2 and MMP-9 were significantly higher as compared with the pre-CPB levels. In the same group, at the same time point, the levels of SCr remained relatively unchanged despite those patients eventually going on to develop AKI. In the patients who did not develop AKI, neither the levels of MMP-2 and MMP-9 nor the levels of SCr rose significantly from pre-CPB levels. This suggests that MMP-2 and MMP-9 may prove to be earlier rising indicators of AKI as compared with SCr.
As expected, the mean highest SCr post-CPB (5-7 days) was significantly higher for the AKI group as compared with the non-AKI group. Although it did not reach significance, our data also show a trend toward higher CPB fluid balance and significantly lower CPB hemoglobin levels in the patients who subsequently developed AKI compared with those who did not.
These data are consistent with other researchers who have reported that hemodilution 35 and lower hematocrits 36 are associated with increased risk of CPB-induced AKI. 37 In the present study, patients who developed AKI were transfused a significantly higher volume of RBCs compared with those who did not develop AKI, consistent with a report from Karkouti et al 38 who suggest increased RBC transfusion is associated with increased risk of AKI development. AKI patients in the present study also had a significantly higher transfusion of platelets and fresh frozen plasma as compared with the non-AKI patients. Furthermore, those patients who developed AKI also had a significantly higher ICU fluid balance compared with non-AKI patients, which is in agreement with Shen et al 39 who reported that cardiac surgical patients with a higher post-operative fluid balance were associated with a higher incidence of the development of AKI. Adverse outcomes of AKI have also been reported, such as prolonged hospital LOS leading to higher costs in ICU patient management.3,40 In the present study, the LOS in ICU was significantly longer in the AKI patients compared with the non-AKI patients.
Cardiac I/R and MMP Activity
During cardiac surgery, ischemia is artificially induced by aortic cross-clamping (ischemic period). Once surgery has been accomplished, the aortic cross-clamp is removed, and the restoration of blood flow to the previously ischemic myocardium results in I/R injury. 41 Despite myocardial preservation through maneuvers of arrest with modernized blood cardioplegic solutions, I/R injury still occurs.42,43 Although not completely understood, biochemical evidence suggests that the degradation of contractile proteins by proteolytic enzymes 43 particularly MMP-2,44,45 is a major contributor to this process. An abundance of evidence from animal studies implicate an increase in MMP-2 activity and its targeting of myocytes during I/R.42,46,47 Ali et al 45 provided a link to humans, showing that localized MMP-2 in the animal and human cardiac sarcomere is activated by I/R injury and contributes to contractile protein degradation. In the present study, we demonstrated that circulating serum MMP-2 activity is significantly higher at the 10-minute post-CPB time point in patients who developed AKI compared with non-AKI patients. In addition, the serum MMP-2 activity is significantly higher at the 10-minute post-CPB time point in AKI patients compared with the pre-CPB time point. The data we present are strongly supported by the results of Lalu et al, 48 who studied 15 patients with stable angina who underwent coronary artery bypass grafting (CABG) with CPB and demonstrated elevated circulating MMP-2 and MMP-9 activity 10-minute post-CPB compared with baseline, although they did not study the relationship between the circulating MMPs activity and the development of AKI. Proteins that are associated with the pathophysiologic process of kidney injury are probably released into the urine promptly after injury. The measurement of these urine protein concentrations may provide an earlier diagnosis of injury and evaluation of the extent of injury as compared with the late rising SCr biomarker. 49 The 10-minute post-CPB time point was chosen because it may represent an ideally sufficient time point for proximal tubule markers such as MMP-2 and MMP-9 to be present in the urine. To these authors’ knowledge, there are no other reports of a 10-minute post-CPB time point to measure MMP-2 and MMP-9 in association with AKI.20,50,51
At our institution, a small study of patients undergoing routine CABG demonstrated an increased cardiac tissue MMP-9 and serum MMP-9 activity post-cardiac I/R compared with baseline. 15 Data from the present study go a step further by demonstrating elevated serum activity of MMP-2 and MMP-9 at the 10-minute post-CPB and 4-hour post-CPB time points in patients who developed AKI compared with those who did not. These data suggest elevated MMP activity from cardiac I/R may be associated with increased activity of MMPs (-2 and -9) in the systemic circulation of patients who go on to develop AKI.
Renal I/R and MMP Activity
MMPs degrade extracellular matrix and are involved in ischemic organ injuries. 33 In a rodent model, MMP-2 and MMP-9 were increased in renal tubules and the interstitium following 1 to 3 days of reperfusion after 52 minute of ischemia. 16 Moreover, AKI characterized by damage to the glomerulus, cell adhesion molecules, tubular epithelium, and endothelial vascular permeability has been linked to MMP-mediated I/R.12,52 During cardiac surgery, several injury pathways contribute to the pathogenesis of AKI, such as renal hypoperfusion,53,54 renal atheroembolism, 55 tissue I/R, 56 inflammation, and nephrotoxic mechanisms. 57 The protective mechanisms of minocyclines in renal I/R are not completely understood, although the mechanism of inhibition of MMPs has been studied in an animal model.58,59 MMP-2 and MMP-9 involvement in acute and chronic renal injury along the spectrum from basement membrane damage, tubular atrophy, fibrosis, to renal failure have been reported.18,19,60 Minocycline, a semisynthetic tetracycline, provides a protective role by directly antagonizing the effect of MMP-2 and MMP-9 in renal microvascular leakage following I/R. 58 We have reported the presence of MMP-2 and MMP-9 in the perfusate of human kidneys for transplantation, suggesting MMP-2 and MMP-9 play a role in transplant kidney preservation injury, and that part of this injury can be prevented by the addition of doxycycline into the preservation solution.18,19 Research from the present study demonstrates a higher urine MMP-2 activity (10-minute post-CPB and 4-hours post-CPB) in patients who develop AKI compared with those who do not. Furthermore, the 10-minute post-CPB and 4-hour post-CPB urine MMP-2 activity was significantly higher than baseline in the patients who developed AKI. Moreover, the 10-minute post-CPB serum and urine MMP-2 time points to predict AKI had moderately high sensitivities and specificities, respectively. The 10-minute post-CPB urine MMP-9 time point had a high sensitivity and specificity as well.
Our data regarding serum MMP-2 activity show a trend toward higher activity at baseline in patients with AKI as compared with baseline non-AKI, suggesting that there may be other mechanisms at play prior to cardiac or renal I/R. Perhaps MMP-2 activity may predict the development of AKI. However, more studies are needed to demonstrate whether elevated pre-CPB MMP-2 levels can predict the development of AKI.
Nevertheless, we foresee that earlier diagnosis of post-cardiac surgical AKI may affect how patients are treated by the following ways: (1) a significant decline in AKI development and progression to higher stages, which, in turn, could result in substantial improvement in overall outcomes; (2) improvement of risk stratification for severe AKI (Stage 2 or 3) in high-risk critically ill post-cardiac surgical patients; (3) in patients who have ongoing nephrotoxin exposure, early biomarker diagnosis has the potential to identify damage, resulting in the removal of agent before extensive injury has occurred; and (4) early detection biomarkers may separate kidney dysfunction from AKI. 11
Limitations of the Study
There are limitations to this study. Primarily, the sample size is low. However, this research was designed as a proof-of-concept pilot conducted prior to the main study with the purpose of enhancing the likelihood of success of the main study by searching for associations, reproducibility, and the avoidance of pitfalls that will be followed up in a subsequent larger scale study with modified design elements.
Next, it lacks the ability to discern the amount of MMPs originating from the heart and/or kidney. To quantitate the amount of serum MMPs from the heart would require sample aspiration from the coronary sinus via retrograde cannulation, while serum MMP samples from the kidney would necessitate cannulation of the renal artery. These additional invasive techniques increase the risk of perioperative complications. However, there has been an abundance of animal research demonstrating that these 2 organs produce large amounts of MMPs during I/R. Despite the limitations of gel zymography, 61 our data demonstrate the presence of increased serum and urine MMP-2 and MMP-9 activity levels in patients undergoing CPB-supported cardiac surgery who developed AKI.
This research represents a single center prospective study of adults with coronary artery disease (CAD) and/or valve disease undergoing elective and urgent CPB-supported cardiac surgery. Even though preliminary results showed clear statistical significance, the results will need validation in a larger prospective trial including adults with confounding variables and comorbidities that are prevalent with increasing age. Another limitation to our study is the lack of urine output measurements in our classification of AKI. We did not want to compromise the accuracy of our analysis because the use of urine output criteria for diagnosis and staging has been less well-validated. 27 In addition, patients transferred from the ICU to the post-operative ward routinely undergo Foley catheter removal, thus urine output is not closely measured. Furthermore, the need for clinical judgment regarding individual patient needs in the administration of angiotensin-converting enzyme inhibitors, regulation of fluid balance, and other factors must be included when using urine output for classification of AKI. Finally, the KDIGO criteria 27 require changes in serum creatinine or urine output, but not both, as do the previous RIFLE criteria. 26 Thus, we have only used SCr for classification of AKI using the KDIGO criteria.
Finally, our cohort consisted of mainly patients with normal kidney function at recruitment. To fill in the gaps of AKI, our finding needs to be confirmed in documented high-risk settings such as pre-existing dysfunction and nephrotoxic drug use.
Conclusion
AKI continues to be a common and important complication of cardiac surgery and is associated with increased mortality, complications, and length of hospital stay. The data from animal and human studies from our prior research, other researchers, and data from the present study suggest 2 consistent sources of MMP-2 and MMP-9 activity associated with I/R following CPB, the heart and the kidney. Damage induced by increased MMPs activity is also demonstrated in other human and animal studies. The MMP data gathered from the present study agree and are consistent with previous investigators, and go a step further to make the association between MMPs and AKI. Although this is part of a preliminary study, the data demonstrate that increased levels of MMP activity are associated with AKI. Moreover, the levels of MMP-2 and MMP-9 activity appear to rise earlier as compared with the levels of SCr in patients who develop AKI. Following this pathway of research, future clinical implications may show that MMP-2 and MMP-9 may be pharmacological targets for renal protection during cardiac surgery.
Supplemental Material
sj-docx-1-cjk-10.1177_20543581211019640 – Supplemental material for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery
Supplemental material, sj-docx-1-cjk-10.1177_20543581211019640 for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery by Erick D. McNair, Jennifer Bezaire, Michael Moser, Prosanta Mondal, Josie Conacher, Aleksandra Franczak, Greg Sawicki, David Reid and Abass Khani-Hanjani in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-tif-1-cjk-10.1177_20543581211019640 – Supplemental material for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery
Supplemental material, sj-tif-1-cjk-10.1177_20543581211019640 for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery by Erick D. McNair, Jennifer Bezaire, Michael Moser, Prosanta Mondal, Josie Conacher, Aleksandra Franczak, Greg Sawicki, David Reid and Abass Khani-Hanjani in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-tif-2-cjk-10.1177_20543581211019640 – Supplemental material for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery
Supplemental material, sj-tif-2-cjk-10.1177_20543581211019640 for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery by Erick D. McNair, Jennifer Bezaire, Michael Moser, Prosanta Mondal, Josie Conacher, Aleksandra Franczak, Greg Sawicki, David Reid and Abass Khani-Hanjani in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-tif-3-cjk-10.1177_20543581211019640 – Supplemental material for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery
Supplemental material, sj-tif-3-cjk-10.1177_20543581211019640 for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery by Erick D. McNair, Jennifer Bezaire, Michael Moser, Prosanta Mondal, Josie Conacher, Aleksandra Franczak, Greg Sawicki, David Reid and Abass Khani-Hanjani in Canadian Journal of Kidney Health and Disease
Supplemental Material
sj-tif-4-cjk-10.1177_20543581211019640 – Supplemental material for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery
Supplemental material, sj-tif-4-cjk-10.1177_20543581211019640 for The Association of Matrix Metalloproteinases With Acute Kidney Injury Following CPB-Supported Cardiac Surgery by Erick D. McNair, Jennifer Bezaire, Michael Moser, Prosanta Mondal, Josie Conacher, Aleksandra Franczak, Greg Sawicki, David Reid and Abass Khani-Hanjani in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
The authors would like to acknowledge the Perfusionists (Jo-Anne Marcoux, Jennifer Bezaire, Marnie Olson, Kyle Mikoliew, Tessandra Dmitrowicz, Victor Uppal, and Mark Rosin) and Cardiac Surgeons (Drs Taras Mycyk, Gregory Dalshaug, and Dorothy Thomson) of the Royal University Hospital. We would also like to thank Sharon Wandzura, Yolanta Sawicka, and Brooke Thompson for their contribution to this research.
Ethics Approval and Consent to Participate
The University of Saskatchewan Biomedical Research Ethics Board approved the study protocol, and the Provincial Health Authority granted operational approval. Verbal and written consent for study participation were obtained for each subject prior to enrollment. The study period ranged from December 2018 to December 2019. (ClinicalTrials.gov Identifier: NCT04387149)
Consent for Publication
All authors have participated in the revised final manuscript and have given their consent for publication.
Availability of Data and Materials
Data are available from the authors upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a University of Saskatchewan College of Medicine Research Award and the Royal University Hospital Foundation Grant.
Supplemental Material
Supplemental material for this manuscript is available on line.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
