Abstract
Background:
Low-molecular-weight heparins (LMWH) replaced unfractionated heparin (UFH) in multiple indications. Although LMWH efficacy in hemodialysis was demonstrated through multiple studies, their safety remains controversial. The potential bioaccumulation in patients undergoing chronic hemodialysis raised the question of bleeding risk among this population.
Objective:
The aim of this study was to evaluate bleeding risk among patients with chronic hemodialysis receiving LMWH or UFH for the extracorporeal circuit anticoagulation.
Design:
We conducted a retrospective cohort study on data extracted from the Régie de l’assurance maladie du Québec (RAMQ) and Med-Echo databases from January 2007 to March 2013.
Setting:
Twenty-one hemodialysis centers in the province of Québec, Canada.
Patients:
Chronic hemodialysis patients.
Measurements:
Bleeding risk evaluated by proportional Cox model for time-dependent exposure using demographics, comorbidities, and drug use as covariates.
Methods:
Minor, major, and total bleeding events identified using International Classification of Diseases, Ninth Revision (ICD-9)/International Classification of Diseases, Tenth Revision (ICD-10) codes in the RAMQ and Med-Echo databases. Exposure status to LMWH or UFH was collected through surveys at the facility level.
Results:
We identified 5322 prevalent and incident patients with chronic hemodialysis. The incidence rate for minor, major, and total bleeding was 9.45 events/1000 patient-year (95% confidence interval [CI]: 7.61-11.03), 24.18 events/1000 patient-year (95% CI: 21.52-27.08), and 32.88 events/1000 patient-year (95% CI: 29.75-36.26), respectively. We found similar risks of minor adjusted hazard ratio (HR: 1.04; 95% CI: 0.68-1.61), major (HR: 0.83; 95% CI: 0.63-1.10), and total bleeding (HR: 0.90; 95% CI: 0.72-1.14) when comparing LMWH with UFH.
Limitations:
Potential misclassification of patients’ exposure status and possible underestimation of minor bleeding risk.
Conclusion:
LMWH was not associated with a higher minor, major, or total bleeding risk. LMWH did not increase the risk of bleeding compared with UFH for the extracorporeal circuit anticoagulation in hemodialysis. The convenience of use and predictable effect made LMWH a suitable alternative to UFH in hemodialysis.
What was known before
Low-molecular-weight heparins (LMWH) use as an anticoagulant of the extracorporeal circuit in chronic hemodialysis is known to be effective and this was demonstrated in multiple studies. However, their safety is still controversial and needs further investigation.
What this adds
This is the largest study aiming to evaluate the bleeding risk of LMWH compared with unfractionated heparin (UFH) for the extracorporeal circuit anticoagulation in chronic hemodialysis. This study shows that tinzaparin does not increase the risk of minor, major, and total bleeding among chronic hemodialysis patients.
Introduction
The first low-molecular-weight heparins (LMWH) have progressively replaced unfractionated heparin (UFH), considered the gold standard in multiple indications, for prophylaxis and therapeutic treatments.1,2 Not only does LMWH have a predictive dose-response relationship, but it also does not need monitoring and is easy to administer and is therefore attractive to health practitioners and nurses. 3 LMWH started to replace UFH in hemodialysis for the extracorporeal circuit anticoagulation especially in western Europe4,5 with the recommendations published in the European Best Practice Guidelines by the European Renal Association—European Dialysis and Transplant Association (ERA-EDTA). 6 However the transition toward LMWH in hemodialysis was controversial. Indeed, this new form of heparin is eliminated by the kidneys while UFH is eliminated mainly through the liver’s reticuloendothelial system. 3 Bioaccumulation studies in patients with impaired kidney function were conflicting: clearance rates vary from a study to another and between the different types of LMWH studied.7-10 Recently, a pharmacokinetic study showed that LMWH with higher molecular weight could be eliminated by the liver when the kidneys’ elimination route was compromised. 11 Despite existing clinical trials and reviews, the safety of LMWH in hemodialysis remains unclear. 12 Although almost all hemodialysis units in Europe chose LMWH as anticoagulant, it is quite the opposite in most of North America where UFH is still the gold standard. However, in the Canadian province of Québec, almost half of hemodialysis units made the choice to replace UFH by one of the available LMWH, therefore offering a unique opportunity to study this safety issue and providing strong evidence to physicians and decision makers.
The aim of this study was to evaluate the association between the use of LMWH in a context of extracorporeal anticoagulation, compared with UFH, and the risk of bleeding in a cohort of chronic hemodialysis patients.
Methods
Study Population and Data Sources
We conducted a retrospective cohort study to assess the association of the extracorporeal circuit anticoagulation with LMWH, compared with UFH, and bleeding risk among prevalent and incident chronic hemodialysis patients. Study data were obtained from the Régie de l’assurance maladie du Québec (RAMQ). This provincial single-payer health insurance plan provided to all residents of the Province of Québec, Canada, covers medical and hospital services. Information on all medical visits, diagnostic codes (using International Classification of Diseases [ICD]), medical procedures during in-patient and outpatient encounters, and hospital discharge summaries (Med-Echo) are provided by this administrative database. The Med-Echo database provides details on the date of admission and discharge, primary and secondary diagnoses, and the procedures performed during the hospital stay. Moreover, all individuals aged 65 years and older, individuals on welfare, and workers not insured by a private insurance company are covered by the provincial drug plan. Exposure to heparin is not recorded in the RAMQ drug plan and was collected at each of the 21 participating hemodialysis units in the province of Québec. The list of participating centers is provided in the supplementary appendix.
Study Cohort
We built a cohort of both prevalent and incident adult patients on maintenance hemodialysis between January 1, 2007, and March 31, 2013, identified in the RAMQ database. To be included, patients could not have a prior kidney transplant and should have at least 90 days of follow-up after hemodialysis initiation. Prevalent patients could not have started dialysis before January 1, 2001, to allow us to calculate vintage years (how many years they received chronic hemodialysis prior to cohort entry). The first hemodialysis code respecting the inclusion criteria was defined as the index date. End of follow-up corresponded to the date of kidney transplant, switch to peritoneal dialysis, end of study, or death, whichever occurred first. Moreover, only patients who received hemodialysis in one of our participating centers were kept. Patients followed in a participating center that did not provide exposure status or with an unclear exposure status were excluded.
Exposure Definition
The use of heparin as an extracorporeal anticoagulant during hemodialysis is defined at the center level. Each center has its own anticoagulation protocol and heparin is administered in hospital at every session. Therefore, exposure status could not be retrieved through the RAMQ drug coverage plan. The type of heparin used (tinzaparin, dalteparin, enoxaparin, nadroparin, UFH) was collected at the center level between January 1, 2007, and March 31, 2013. We recorded any changes in their respective protocols, including all changes of heparin type through the study period. Centers could have more than one heparin exposure period during the study time frame; however, transition periods from one heparin to another were removed from the analysis, as we could not segregate which form of heparin each patient received. Patients’ exposure status depended on the center where and when they were receiving hemodialysis. The exposure status for each patient changes every time: (1) the unit changes the protocol for a different heparin; and (2) the patient receives 2 or more hemodialysis sessions in a different unit.
Outcome Definition
All admissions for bleeding as a primary diagnosis on the discharge sheet during the study period were identified through the International Classification of Diseases, Ninth Revision (ICD-9) and International Classification of Diseases, Tenth Revision (ICD-10) codes (see Supplementary appendix for list of codes and their definition). Only the first bleeding event that occurs during the patient’s follow-up time was kept and was categorized as being a minor or a major bleeding.
Covariates
Covariates were evaluated at baseline through the RAMQ and Med-Echo and included age, gender, follow-up time, vintage time (time undergoing chronic hemodialysis for prevalent patients), cohort entry year, hospitalization in prior year, comorbidities, and drug use in the 6 months prior to cohort entry (see Table 1 for more details).
Patients’ Characteristics at First Exposure to LMWH and UFH.

Note. LMWH = low-molecular-weight heparin; UFH = unfractionated heparin; IQR = interquartile range; GERD = gastroesophageal reflux disease; NSAID = nonsteroidal anti-inflammatory drugs.
At cohort entry.
Statistical Analysis
Mean and standard deviation (SD) or median and interquartile range (IQR) were used to present descriptive baseline data where appropriate. Comorbidities are presented as a frequency expressed as a proportion (%).
Outcomes’ incidence rates were calculated by dividing the number of events (total bleeding, major bleeding, or minor bleeding) by the total patient-years (p-y) of follow-up and are presented as incidence rate per 1000 p-y. And 95% confidence intervals (CI) for rates were calculated using a Poisson distribution (inversed gamma formula).
The hazard ratio (HR) for the first event of each outcome was estimated using a time-dependent Cox proportional hazard model. It was adjusted for all the comorbidities presented in Table 1. All analyses were done using SAS 9.4 (Cary, North Carolina).
Sensitivity Analysis
LMWH are not interchangeable and should also be analyzed separately. We conducted the analyses using the same method but separating tinzaparin periods from dalteparin with UFH as the reference group. Moreover, bleeding risk was also evaluated by keeping only incident patients in the cohort.
Ethical Considerations
This study was approved by the Government of Québec ethics committee (Commission d’accès à l’information) and all hospitals ethics committees. Informed consent was waived.
Results
Our cohort included 5322 prevalent and incident patients on maintenance hemodialysis in one of the participating centers with at least one period with a known heparin exposure status, and represented 6079 patients when first exposed to UFH and LMWH (some patients were switched from one form of heparin to the other during the study period). Cumulative follow-up time under UFH was 7493 p-y, 3832 p-y for tinzaparin, and 189 p-y for dalteparin. Most patients (86%) were exposed to one type of heparin only, 12% switched once from one heparin to another, and the remaining (2%) switched more than once meaning that they switched back to their prior exposure.
Incident patients represented 70.6% of the cohort. Prevalent patients had a mean of 0.6 ± 1.3 vintage years at cohort entry. Mean age was 66.4 ± 14.0 years at cohort entry and 39.3% were women. Median follow-up time was 2.0 years (IQR: 0.8-3.6). Patients’ characteristics were overall similar between exposure groups (Table 1). However, in the LMWH exposed group, there were more incident patients, less hospitalizations in the prior year, and less prior bleeding.
A total of 403 bleeding events were identified from 12 255.41 p-y. The incident rate for total bleeding was 32.9 events/1000 p-y (95% CI: 29.75-36.26). The major bleeding and minor bleeding incidence rates were respectively 24.2 events/1000 p-y (95% CI: 21.52-27.08), with 300 events per 12 405.83 p-y, and 9.5 events/1000 p-y (95% CI: 7.61-11.03) with 117 minor events identified among 12 714.00 p-y. The two most frequent major bleeding codes were gastrointestinal hemorrhage and vitreous hemorrhage. For minor bleeding, the most frequent codes were hemorrhage and hematoma complicating a procedure as well as hemorrhage of anus and rectum. The total bleeding risk was similar (HR: 0.90; 95% CI: 0.72-1.14) for LMWH compared with UFH. An increased total bleeding risk of 36% for diabetes (HR: 1.36; 95% CI: 1.07-1.72) and 37% for malignancy (HR: 1.37; 95% CI: 1.05-1.77) was observed. Compared with UFH, the risk of major bleeding when using LMWH was comparable (HR: 0.83; 95% CI: 0.63-1.08) with diabetes increasing bleeding risk by 45% (HR: 1.45; 95% CI: 1.10-1.91). Minor bleeding risk did not increase with LMWH in comparison with UFH (HR: 1.04; 95% CI: 0.68-1.62). Malignancy and oral anticoagulants were statistically significant with an increased minor bleeding risk of 128% (HR: 2.28; 95% CI: 1.48-3.52) and 75% (HR: 1.75; 95% CI: 1.04-2.94), respectively. The complete results are presented in Tables 2, 3, and 4.
Total Bleeding Hazard Ratio for LMWH Compared With UFH.
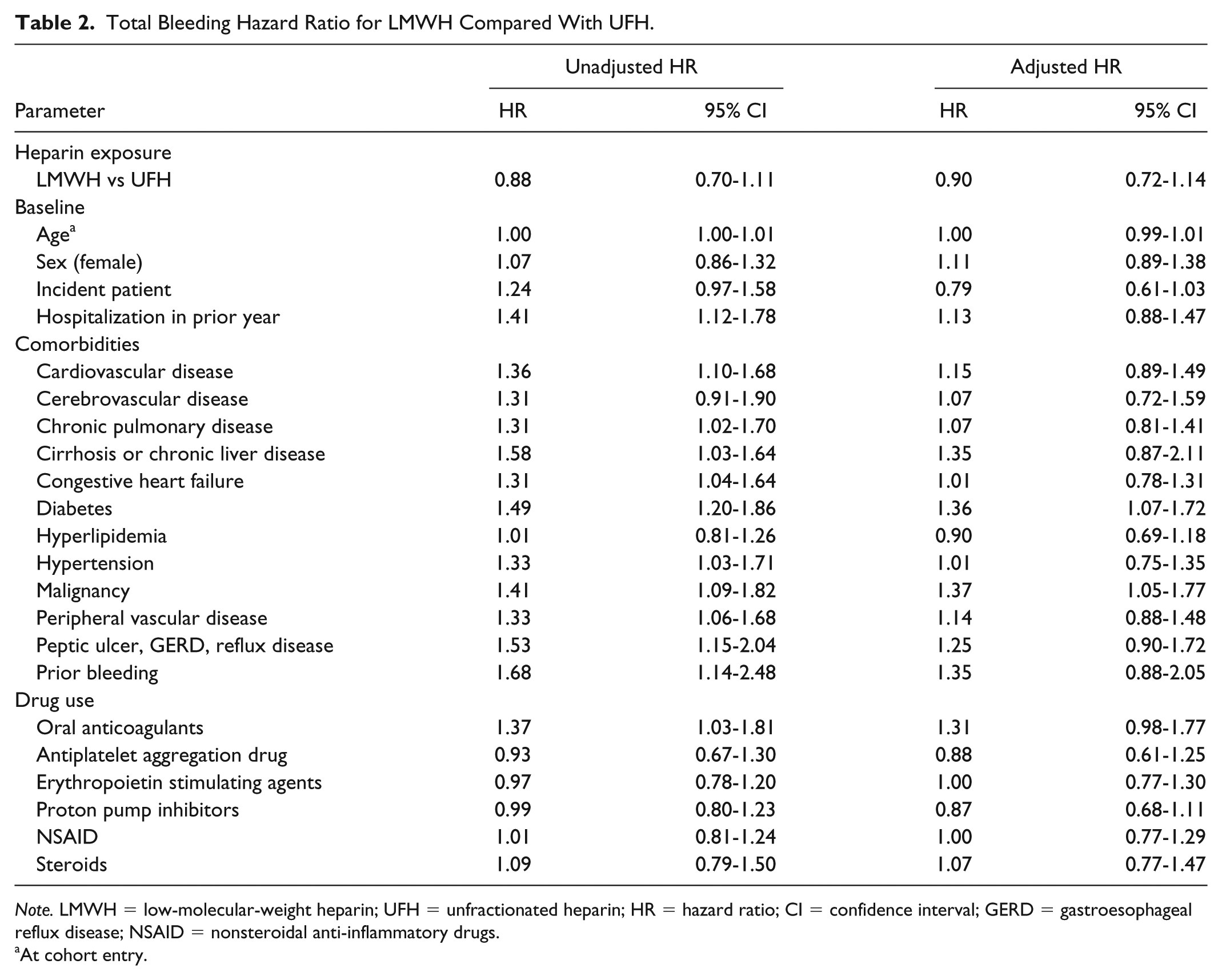
Note. LMWH = low-molecular-weight heparin; UFH = unfractionated heparin; HR = hazard ratio; CI = confidence interval; GERD = gastroesophageal reflux disease; NSAID = nonsteroidal anti-inflammatory drugs.
At cohort entry.
Major Bleeding Hazard Ratio for LMWH Compared With UFH.
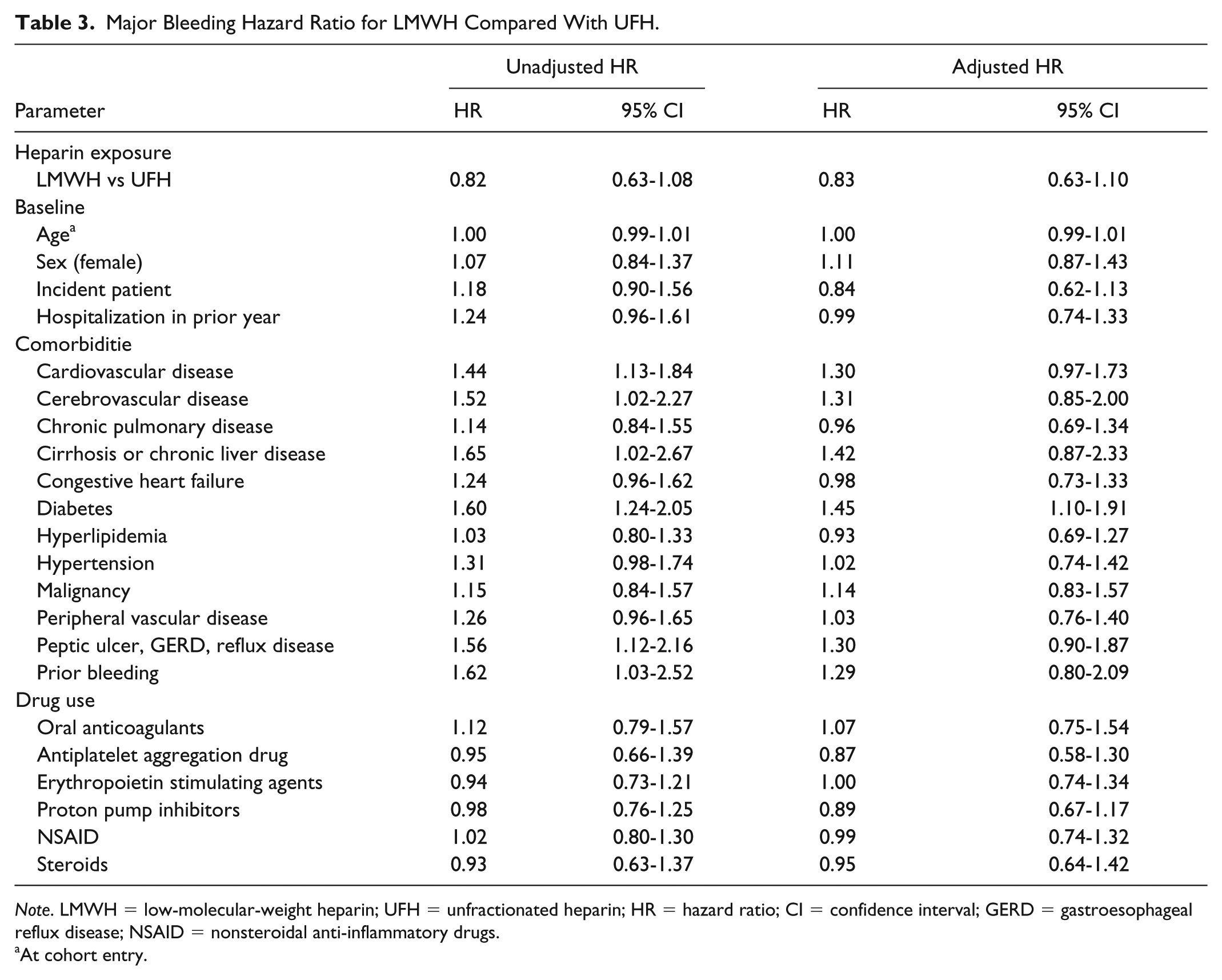
Note. LMWH = low-molecular-weight heparin; UFH = unfractionated heparin; HR = hazard ratio; CI = confidence interval; GERD = gastroesophageal reflux disease; NSAID = nonsteroidal anti-inflammatory drugs.
At cohort entry.
Minor Bleeding Hazard Ratio for LMWH Compared With UFH.
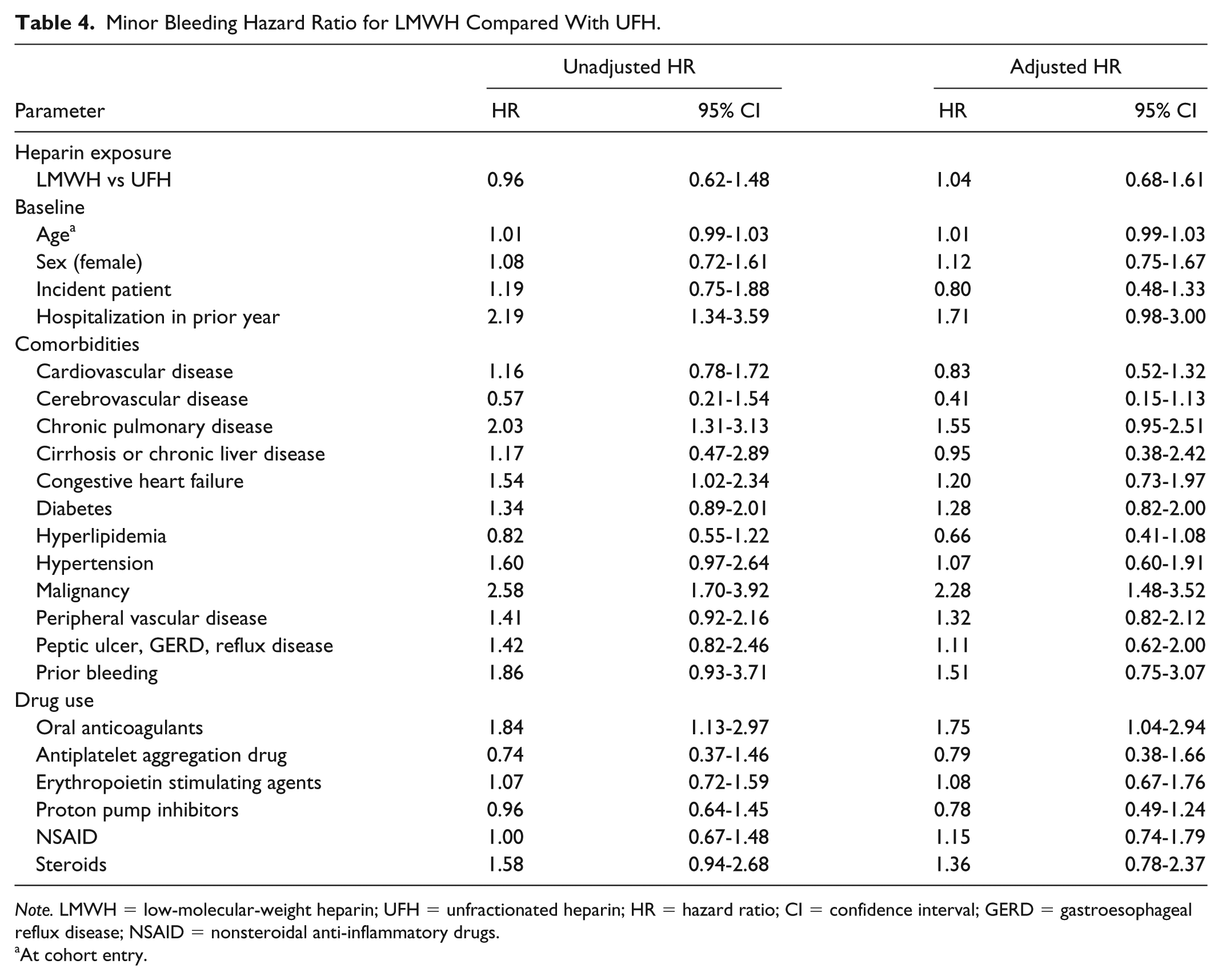
Note. LMWH = low-molecular-weight heparin; UFH = unfractionated heparin; HR = hazard ratio; CI = confidence interval; GERD = gastroesophageal reflux disease; NSAID = nonsteroidal anti-inflammatory drugs.
At cohort entry.
Sensitivity Analysis
Bleeding risk was also evaluated by type of LMWH using UFH as the reference. From the total LMWH follow-up time, tinzaparin accounted for 95% of the time and the remaining 5% was under dalteparin (189 p-y). For both tinzaparin and dalteparin, when compared with UFH, there was no statistical difference for total and major bleeding. The total bleeding risk with tinzaparin was similar to UFH (HR: 0.96; 95% CI: 0.76-1.22) and dalteparin (HR: 0.26; 95% CI: 0.06-1.05). Major bleeding risk was also comparable with UFH for both tinzaparin (HR: 0.88; 95% CI: 0.66-1.16) and dalteparin (HR: 0.34; 95% CI: 0.08-1.38). No event was recorded for minor bleeding with dalteparin and tinzaparin was not statistically significant compared with UFH for the same outcome (HR: 1.12; 95% CI: 0.71-1.76). Confidence intervals were larger for dalteparin because of a shorter exposure time.
When only incident patients were included in the cohort, there was no difference in total (HR: 0.85; 95% CI: 0.64-1.13), major (HR: 0.78; 95% CI: 0.55-1.09), and minor (HR: 1.03; 95% CI: 0.61-1.72) bleeding risk.
Discussion
The controversy around LMWH’s bleeding risk in hemodialysis has been lingering despite several published clinical trials attempting to answer this question. In this retrospective cohort study, we tackle the problematic of total bleeding, major bleeding, and minor bleeding associated with LMWH as a group and individually for in-hospital hemodialysis anticoagulation.
Regardless of the bleeding category we looked at, LMWH showed to be as safe as UFH for bleeding risk. Major bleeding accounts for most of all observed bleeding events. The most likely explanation is that we had only access to hospitalization data and even if we had an exhaustive list of both minor and major bleeding codes, it is less likely to have a hospitalization triggered by a minor bleeding. It would be reasonable to think that due to their nature, in our study’s context, minor bleedings would be underestimated. However, we are confident that major bleedings were adequately captured. Among hemodialysis patients, bleeding incidence rates published previously varied highly based on the study’s context and the type of bleeding considered as the outcome. Holden et al reported an incidence rate for major bleeding of 2.5 events/100 p-y and could range from 3.1 to 6.3 events/100 p-y depending on patients’ use of aspirin and/or warfarin. 13 Another study evaluating the incidence of moderate to severe bleeding events among nondialysis patients receiving therapeutic doses of UFH and LMWH estimated the rate to 3.5 events/100 p-y. 14 We estimated a major bleeding incidence rate of 2.42 events/100 p-y, which is in the lower range of what was previously published.
Results were similar when comparing separately tinzaparin and dalteparin with UFH. Tinzaparin proved to be as safe as UFH for total, major, and minor bleeding. No minor bleeding events were recorded with dalteparin making it impossible to evaluate the risk of minor bleeding. However, dalteparin appears as safe as UFH for major and total bleeding risk. Tinzaparin did not represent a higher bleeding risk compared with UFH in hemodialysis. Dalteparin did not seem to present a higher bleeding risk either but we had fewer data compared with UFH and tinzaparin with a shorter follow-up time.
As LMWH were introduced as a potential replacement to UFH for extracorporeal circuit anticoagulation in hemodialysis, numerous studies were conducted and published looking at their efficacy and safety. Although efficacy was thoroughly covered and demonstrated, LMWH safety status remained unclear. 12 Bioaccumulation risk was always a concern with LMWH with the assumption that they are exclusively eliminated by the kidneys, which is problematic in patients undergoing hemodialysis. 15 Multiple studies were published measuring the possible bioaccumulation of different types of LMWH. The results were as different as LMWH’s pharmacokinetic profiles differ from each other. Bioaccumulation studies were conflicting; some showed an accumulation of dalteparin in patients with severe renal failure, 7 while there was no bioaccumulation in other studies in the same population.8,9 Tinzaparin was also evaluated and was not found to accumulate with severe renal failure. 10 A study published by Johansen and Balchen 11 highlighted the fact that not all LMWH were exclusively eliminated by kidneys and some could be eliminated by the liver when their molecular weight was higher, and therefore would not accumulate in patients in hemodialysis. Tinzaparin is the heavier form of LMWH. A recent single center observational study evaluated the risk of major bleeding in hemodialysis comparing LMWH with UFH and found no difference between both groups. 16 These findings were consistent with our own results.
Diabetes was a statistically significant risk factor for both major and total bleeding. In other studies, diabetes was identified as an independent risk factor for major bleeding events.17,18 Malignancy was also identified in our study as a factor increasing the risk of minor and total bleeding. In a study comparing bleeding risk in patients receiving anticoagulants with or without cancer, the former group had a higher risk of bleeding. 19 The more advanced cancer’s stage is, the higher the risk of hemorrhage. 20 As for the increased risk of minor bleeding with oral anticoagulant, there is no clear answer in previously published studies. 21 A study published by Limdi et al 22 and evaluating complications with warfarin by kidney function stage showed an increased risk of first bleeding event (HR: 2.33; 95% CI: 1.44-3.75) in patients with an estimated glomerular filtration rate lower than 30 mL/min/1.73 kg/m2.
As expected, the proportion of patients by year of cohort entry slightly decreased through time for UFH whereas it went in the opposite direction for LMWH. The later started being used in hemodialysis units in 2007 and the number of units switching from UFH kept on increasing year after year leading to more patients, therefore more exposure time, receiving LMWH.
Our study was the first multicenter cohort study evaluating minor, major, and total bleeding risk with LMWH for the extracorporeal circuit anticoagulation in hemodialysis on a large scale with a long follow-up. The use of administrative data from our provincial health care insurance allowed including all eligible patients undergoing hemodialysis in participating centers and linking all the available patients’ information to our collected exposure data at the center level. The universal health care insurance provided in the province of Québec offers systematic care to dialysis patients limiting selection bias. The use of RAMQ and Med-echo data allowed us to collect multiple covariates including drug exposure and therefore minimizing confounding. By defining our exposure as being time-dependent and allowing patients to switch exposure during their follow-up, we avoided the introduction of an immortal time bias.
Our study has some limitations. The exposure was measured at the center level, meaning we cannot be certain that all patients receiving hemodialysis in these units were on the regular anticoagulation protocol. There was a potential misclassification bias for exposure status; however, the risk would be similar for both UFH and LMWH. Also, we could not adjust based on individual doses since that information was not available. Moreover, identification of bleeding events could not be done by chart reviews because of the multicenter nature of our study. We had to rely on ICD codes reported on hospital discharge sheets. Since only events leading to hospitalization could be identified, we most likely could not capture all minor bleeding events. Although we had access to many covariables to introduce them in the model, confounding remains possible. Finally, only a limited number of follow-up time were under dalteparin, which made the bleeding risk estimation for this form of LMWH less reliable.
In conclusion, our large retrospective cohort study showed that LMWH, more specifically tinzaparin, is as safe as UFH for minor, major, and total bleeding risk when used for the extracorporeal circuit anticoagulation in hemodialysis. With LMWH replacing UFH for multiple indications and their convenient use, practitioners and policy makers needed a clear evidence of their safety since efficacy was already proven. Ongoing clinical randomized studies, like Use of Tinzaparin for Anticoagulation in Hemodialysis (HEMO-TIN) trial, are comparing specific forms of LMWH to UFH in hemodialysis. Results from those studies will add to the body of evidence and, combined to our study, will offer a better understanding of bleeding risk associated with LMWH. Multiple studies demonstrated that when considering the product’s cost, material and nursing time, the cost of both forms of heparins was similar. Tinzaparin is a safe and simple alternative to UFH in hemodialysis. Heparin is known to cause other side effects and whether LMWH has the same impact is still a pending question.
Supplemental Material
Supplementary_table_1 – Supplemental material for Association Between Low-Molecular-Weight Heparin and Risk of Bleeding Among Hemodialysis Patients: A Retrospective Cohort Study
Supplemental material, Supplementary_table_1 for Association Between Low-Molecular-Weight Heparin and Risk of Bleeding Among Hemodialysis Patients: A Retrospective Cohort Study by Hind H. Lazrak, Emilie René, Naoual Elftouh and Jean-Philippe Lafrance in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
We would like to thank all our collaborators in the participating centers who made exposure data collection, and this study, possible in their respective hemodialysis units: Dr Pierre Cartier, Dr Karine Tousignant, Dr Carole Pichette, Dr Gérard Désaulniers, Dr Jean Proulx, Dr Sacha Deserres, Dr Paul Montambault, Dr Jean-François Cailhier, Dr George Soltys, Dr Damien Bélisle, Dr Marc Ghannoum, Dr Jean-Philippe Rioux, Dr Sylvain Brunet, Dr Marie-Josée Dion, Dr Paul Barré, and Dr Simon Desmeules.
Ethics Approval and Consent to Participate
This study was approved by the Government of Québec ethics committee (Commission d’accès à l’information) and all hospitals ethics committees. Informed consent was waived.
Consent for Publication
Not applicable.
Availability of Data and Materials
The datasets cannot be made available due to privacy restrictions of the Commission d’accès à l’information du Québec.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
