Abstract
Background:
Thiazide diuretics are commonly prescribed to prevent kidney stones. However, it is unclear whether higher doses confer greater benefit.
Objective:
To determine whether lower doses of thiazide diuretics confer a similar protective effect against kidney stone events as higher doses.
Design:
Population-based cohort study.
Setting:
Linked health administrative databases in Ontario, Canada.
Patients:
Older adults newly prescribed a thiazide diuretic between 2003 and 2014 were separated into 2 groups based on daily dose: low dose (⩽12.5 mg hydrochlorothiazide/chlorthalidone, or ⩽1.25 mg indapamide) or high dose.
Measurements:
The primary outcome was time to a kidney stone event, using diagnosis and procedure codes. A secondary outcome was kidney stone surgery.
Methods:
An association between thiazide diuretic dose and a kidney stone event was estimated using Cox proportional hazards regression.
Results:
A total of 536 of 105 239 patients (0.51%) experienced a kidney stone event. We did not detect a difference in kidney stone risk in the high-dose relative to the low-dose group (adjusted hazard ratio, 1.10; 95% confidence interval, 0.93-1.31). Results were similar when analysis was restricted to the more specific outcome of kidney stone surgery. Neither a history of prior kidney stones nor the type of thiazide diuretic modified the effect of diuretic dose on outcome.
Limitations:
Patients were >65 years old and we were unable to adjust for some potential confounders such as dietary factors.
Conclusions:
Lower dose thiazide diuretics appear to confer a similar protective effect as higher dose thiazides against the development of kidney stones.
What was known before
Prior studies found that higher dose thiazide diuretics lower urinary calcium excretion to a greater degree than lower dose thiazides. Whether or not this translates into a difference in clinical outcomes is not clear as there are no data examining the relative ability of higher vs lower dose thiazide diuretics to prevent kidney stones.
What this adds
Using a large population-based administrative data set from people greater than 65 years of age we find that lower dose thiazide diuretics (⩽12.5 mg hydrochlorothiazide/chlorthalidone or ⩽1.25 mg indapamide) have a similar ability to prevent kidney stone formation as higher dose thiazides.
Introduction
Kidney stones are common and associate with significant morbidity and high health care costs.1,2 Hypercalciuria is the most common metabolic risk factor for the development of kidney stones. 3 Numerous studies (including several randomized trials)4-6 have shown the efficacy of thiazide diuretics for both lowering urinary calcium excretion and the subsequent likelihood of future kidney stone episodes.7-9 High doses of thiazide diuretics were used in the trials (ie, ⩾50 mg of hydrochlorothiazide or ⩾ 25 mg chlorthalidone daily), and such doses are associated with a greater frequency of side effects, including electrolyte disturbance, hyperglycemia, hyperlipidemia, and glucose intolerance. 10 In clinical practice, lower doses are commonly employed for hypertension and also for the treatment of hypercalciuria.11-13 However, whether lower doses of thiazides are equally as effective at preventing kidney stones is not known and a significant knowledge gap identified in guidelines from the American College of Physicians. 8
In this study, we determined whether initiating treatment with a low vs high dose of a thiazide diuretic is associated with an altered risk of kidney stones. We hypothesized that low doses of thiazide diuretics are as effective at preventing kidney stones as higher doses.
Methods
This study employed the thiazide diuretic users from a previously generated cohort. 9 In that study, we described in detail the population data sources, cohort entry criteria, baseline covariates, and the identification of kidney stone events. In brief, we employed linked administrative health data to create a retrospective, population-based cohort of individuals older than 65 years of age (a segment of the Ontario population where drug coverage is a universal benefit), with a new prescription for 1 of 4 classes of antihypertensives (angiotensin-converting enzyme inhibitors/angiotensin receptor blockers [ACEi/ARB], beta blockers, calcium channel blockers, and thiazide diuretics) between April 1, 2003, and March 31, 2014. The date of prescription served as the index date. We excluded patients with missing age or sex data, patients with previous antihypertensive use in the 180 days prior to index date (to ensure new use) or evidence of use of other drugs that might alter kidney stone risk (ie, furosemide, prednisone, or topiramate), patients with end-stage renal disease, and those with a hospital admission in the previous 90 days prior to index date. For the current study, we restricted to thiazide diuretic users only (hydrochlorothiazide, chlorthalidone, or indapamide), excluding patients prescribed the thiazide diuretic metolazone due to its small sample size (n = 254). A list of thiazide diuretic drug identification numbers is provided in Supplemental Table 1. We further divided thiazide diuretic users into a low-dose and a high-dose group. Individuals prescribed a low-dose thiazide diuretic were used as the referent category for all analyses. Low-dose hydrochlorothiazide was considered ⩽12.5 mg per day, low-dose chlorthalidone was considered ⩽12.5 mg per day, and low-dose indapamide was considered ⩽1.25 mg per day. The index date for our study (date where we started follow-up) was set as the date that the thiazide diuretic was prescribed. All doses higher than those detailed above were considered high dose.
We obtained vital statistics from the Registered Persons Database (RPDB). The Ontario Drug Benefit (ODB) database was used to acquire prescription drug data. 14 The Canadian Institute for Health Information Discharge Abstract Database (CIHI-DAD) was employed to identify baseline comorbidity data, hospital admissions, and kidney stone episodes. Emergency room visits were captured with the CIHI National Ambulatory Care Reporting System (NACRS) database. Physician billing claims from the Ontario Health Insurance Plan (OHIP) as well as diagnosis and procedural codes from the CIHI-NACRS and CIHI-DAD databases were employed to identify kidney stone episodes. Other relevant episodes requiring an interaction with the health care system and some baseline comorbidities were captured in the OHIP database. The data sets were linked using unique, encoded identifiers derived from health card numbers, and patient-level data were analyzed at the Institute for Clinical Evaluative Sciences (ICES). The reporting of this and our prior study followed the guidelines for observational studies (Supplemental Table 2).9,15
Baseline covariates included age and sex, obtained from the RPDB and socioeconomic status, which was determined based on neighborhood income quintile according to Statistics Canada. 9 Records in the 5 years prior to the index date were used to identify baseline comorbidities including alcoholism, chronic liver disease, dementia, diabetes, heart failure, history of kidney stones, human immunodeficiency virus, hypercalcemia, hyperparathyroidism, hypertension, inflammatory bowel disease, leukemia, peripheral vascular disease, and stroke/transient ischemic attack. Three years of hospitalization records prior to the index date were used to calculate the Charlson comorbidity index, which was calculated based on the adaptation by Quan et al. 16 Baseline demographic, comorbidity, and treatment data were reported for each dose group according to the index thiazide diuretic prescribed.
The primary outcome was first presentation with kidney stones to a primary care physician, emergency department, or hospital following index dose of thiazide diuretic prescribed. These outcomes were captured using OHIP, CIHI-DAD, and NACRS databases, employing a validated algorithm17-19 based on
Statistical Analysis
Variables were complete with no missing values except for income quintile (0.3% missing) and rural residence (<0.1% missing). Standardized differences were used to assess the balance in baseline variables between groups, with a standardized difference of more than 10% being considered potentially important. 20
Patients were followed from the time of the index thiazide diuretic prescription until the occurrence of the primary outcome, death, a prescription for a different antihypertensive (ACEi/ARB, beta blocker, calcium channel blocker), no subsequent refill prescription (stop), or the end of the study follow-up period (March 31, 2015), whichever event came first. Given the lack of an effect of other antihypertensive prescriptions on the risk of kidney stone presentation, we also repeated this analysis without censoring when another antihypertensive medication was started. A continuous user of a thiazide diuretic was defined by evidence of a repeat prescription within a period of time equivalent to 150% of the days supplied for the previous prescription. 21 Individuals were censored 365 days after their last prescription meeting this definition, in order to permit a formed kidney stone to become clinically apparent. Less than 0.5% of patients emigrate from the province each year, and this was a minor reason for not filling repeat prescriptions in the provincial drug database.
We used Cox proportional hazards regression with low-dose thiazide diuretics as the referent group, to estimate the hazard ratio (HR) and 95% confidence interval (CI) of a kidney stone event according to the dose of thiazide diuretic (low vs high) prescribed. Models were adjusted for the following factors, defined a priori: age (per 1 year), sex (male referent), number of primary care provider visits in the prior year (per 1 visit), location (urban referent), Charlson comorbidity score (per 1 unit increase), history of stroke, diabetes, hypertension, hypercalcemia, hyperparathyroidism, history of kidney stones, and inflammatory bowel disease. Supplemental Table 4 lists the specific codes used to identify these characteristics.
Several sensitivity analyses were performed. First, the outcome definition was restricted to a kidney stone requiring surgical intervention. We examined kidney stone risk as per the primary analysis, but without censoring for starting another antihypertensive. We examined kidney stone risk as per the primary analysis after excluding individuals receiving chlorthalidone. We examined the risk separately for those patients who remained on the same dose, compared with those who switched dose group in the first 90 days. This analysis, similar to others, examined stone events from the time of initial prescription, not from the time of switching dose. We also adjusted for a prior stone history. Consequently, for this analysis, experiencing a stone event should not influence dose adjustment. We used interaction terms to determine whether the presence or absence of a history of kidney stones in the 5 years prior to the index date, or whether the type of thiazide prescribed on the index date, modified the association between thiazide diuretic dose and the risk of kidney stones. Finally, we used an extended Cox model to assess the effect of thiazide dose, low vs high, on kidney stone events, allowing recurrent events. Kidney stone events separated by at least 45 days were considered distinct events and included in the model.
All analyses were conducted with SAS version 9.4 (SAS Institute, Cary, North Carolina, 2011) according to a prespecified protocol approved by the institutional review board at Sunnybrook Health Sciences Centre (Toronto, Ontario, Canada). Participant informed consent was not required for this study. Two-sided
Results
Cohort Characteristics
We identified 105 239 persons older than the age of 65 years who started a new prescription for a thiazide diuretic. In our prior analysis, individuals prescribed a thiazide diuretic had a significantly reduced risk of having a kidney stone event relative to those prescribed another type of antihypertensive medication (adjusted hazard ratio [HRadj] = 0.77, 95% CI: 0.69-0.85,
Baseline Characteristics.

Standardized differences are less sensitive to sample size than traditional hypothesis tests. They provide a measure of the difference between groups divided by the pooled standard deviation; a value greater than 10% is interpreted as a meaningful difference between the groups. 20
Rural residence was defined as a population <10 000 people.
Diabetes was considered to be use of any medication used to treat diabetes in the last 180 days preceding the index date (Supplemental Table 4).
Documented kidney stone event in the preceding 5 years.
Charlson comorbidity index 12 was calculated using 3 years of hospitalization data. “No hospitalizations” received a score of 0.
Baseline medication use in the 1 year preceding the index date.
Relationship Between Dose of Thiazide Prescribed and Kidney Stone Events
Of the 49 942 patients on a low-dose thiazide diuretic, 244 (0.49%) had a kidney stone event (2.95 events per 1000 person-years). This was very similar in the high-dose group, with 292 events (0.53%) from 55 297 persons receiving a new prescription (3.62 events per 1000 person-years). In unadjusted analyses, a high vs low dose of thiazide diuretic was associated with a higher risk of a kidney stone event (HRunadj = 1.19, 95% CI: 1.01-1.41), which was attenuated after adjustment (HRadj = 1.10, 95% CI: 0.93-1.31) (Table 2). Using the more specific outcome of stone-related surgery produced similar results (HRadj = 1.18, 95% CI: 0.96-1.46) (Table 2).
Association Between Thiazide Dose and Outcomes.

Models were adjusted for the following factors: age (per 1 year), sex (male referent), location (urban referent), number of primary care provider visits in the prior year (per 1 visit), Charlson comorbidity score (per 1 unit increase), history of stroke, diabetes, hypertension, hypercalcemia, hyperparathyroidism, history of kidney stones, and inflammatory bowel disease.
Repeated analysis without censoring the follow-up period for the start of a different antihypertensive.
Our previous work failed to detect an effect of other antihypertensives on the risk of developing a kidney stone.
9
We therefore repeated these analyses without censoring when another antihypertensive medication was prescribed (Table 2). This longer follow-up period (median = 439 days, IQR = 365-1040) increased the proportion of individuals experiencing a kidney stone event in both groups to 378 (0.76%) in the low-dose group and to 424 (0.77%) in the high-dose group. This analysis also did not detect an altered risk in the high-dose group relative to the low-dose group (HRadj = 1.03, 95% CI: 0.89-1.18,
Sensitivity Analyses
To evaluate the potential effect of switching from a low- to a high-dose thiazide diuretic thereby biasing our results, we examined the risk of having a kidney stone in those that remained on either low or high dose and in those that switched to a different dose group within 90 days of initial prescription (Table 3). This analysis demonstrated an increased risk in the small number of patients (n = 1693), who started the study on a high dose then switched to a low-dose relative to those who remained on the low-dose throughout (HRadj = 2.19, 95% CI: 1.28-3.78,
Sensitivity Analysis, Examining the Primary Outcome by Dose Switch.
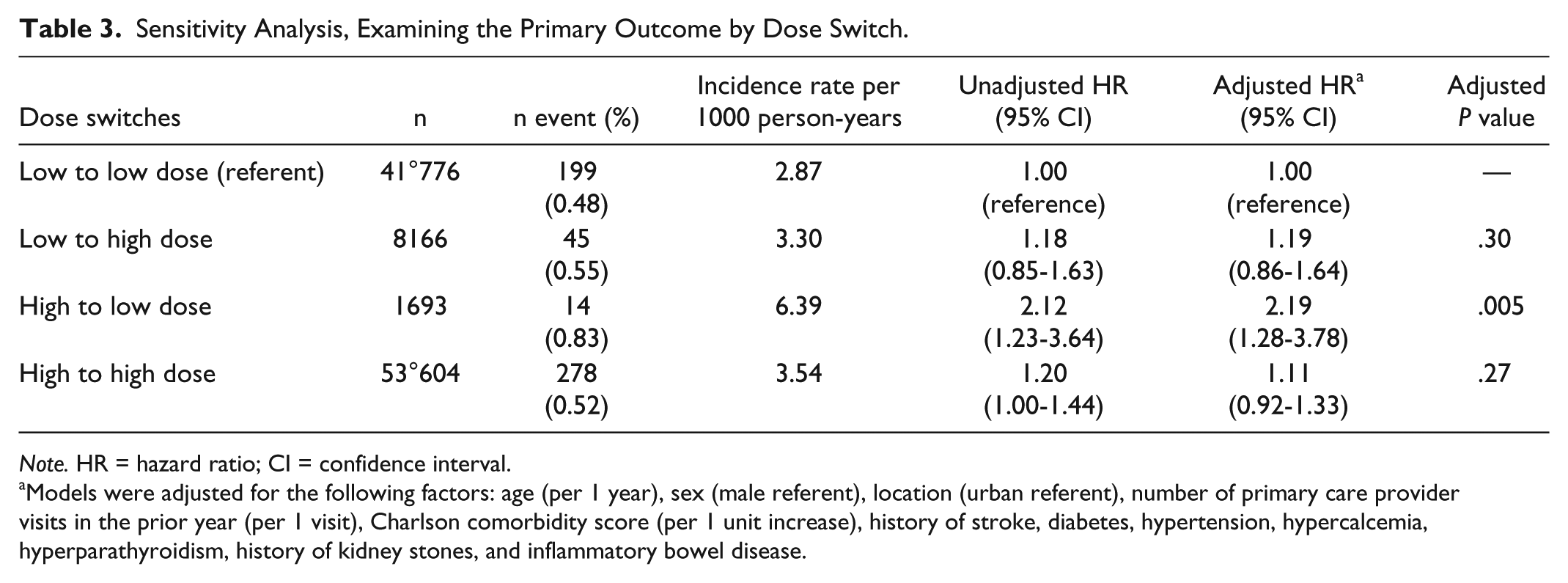
Models were adjusted for the following factors: age (per 1 year), sex (male referent), location (urban referent), number of primary care provider visits in the prior year (per 1 visit), Charlson comorbidity score (per 1 unit increase), history of stroke, diabetes, hypertension, hypercalcemia, hyperparathyroidism, history of kidney stones, and inflammatory bowel disease.
To avoid the potentially confounding effect of different thiazide diuretics, we repeated the analysis after excluding patients receiving chlorthalidone. This did not alter the primary outcome. Persons on a higher dose thiazide diuretic had a similar risk of kidney stones (HRadj = 1.12, 95% CI: 0.94-1.33, p = 0.21). We also examined whether our primary analysis was modified by history of prior kidney stone and did not find an effect modification (Table 4,
Subgroup Analysis.

Models were adjusted for the following factors: age (per 1 year), sex (male referent), location (urban referent), number of primary care provider visits in the prior year (per 1 visit), Charlson comorbidity score (per 1 unit increase), history of stroke, diabetes, hypertension, hypercalcemia, hyperparathyroidism, history of kidney stones, and inflammatory bowel disease.
Recurrent Event Analysis.

Discussion
In a large population of persons receiving a new prescription for a thiazide diuretic, we found that lower dose thiazide diuretics confers a statistically similar protective effect compared with higher dose diuretics. Our results were robust to the more specific definition of requiring kidney stone surgery and were not modified by having a history of prior kidney stones. This work provides support for using lower dose thiazide diuretics to prevent recurrent kidney stone episodes.
Our inability to find an effect of thiazide dose on kidney stone risk could be explained by the fact that thiazide diuretics reduce kidney stone risk independently of their urinary calcium lowering effect or that thiazides have a similar ability to reduce calciuria at a range of doses. However, the limited evidence available supports that higher doses of hydrochlorothiazide have a greater urinary calcium lowering effect.13,22 The nature of our cohort is such that we do not have information about urinary calcium excretion or the influence of thiazide dose on calciuria. However, in several trials examining the protective effect of thiazide diuretics, a protective effect was demonstrated even though hypercalciuria was not an inclusion criterion.5,23,24 Consistent with this, we did not find an effect modification by history of kidney stones in our current or prior analysis (albeit the confidence intervals were wide), consistent with thiazide diuretics being protective regardless of the presence of this metabolic abnormality. 9
The median follow-up of our cohort was 365 days. This is a relatively short time as it may take considerably longer for a kidney stone to form and become clinically apparent. Nonetheless, in a prior study employing this cohort we demonstrated a clear reduction in the risk of forming a kidney stone when an individual was prescribed a thiazide diuretic in comparison to another antihypertensive. 9 This is consistent with the possibility that thiazide diuretics might have another effect on kidney stone formation/presentation, such as stabilizing calculi in situ, lowering urinary oxalate excretion or urinary pH, independent of lowering urinary calcium excretion.25,26
A recent meta-analysis by Fink et al clearly highlights the utility of thiazide diuretics in preventing further kidney stone events in persons with a history of kidney stones. 8 The trials included in this review were completed in the 1980s and early 1990s when much higher doses of thiazide diuretics were utilized than are currently employed in clinical practice for the treatment of hypertension.23,24,27 Higher doses of thiazide diuretics can lead to side effects such as hyperglycemia, hyperlipidemia, and glucose intolerance, which may increase the risk of cardiovascular disease. 28 Thus, for the treatment of hypertension, practitioners have used progressively lower doses. Fortunately, lower doses have a similar effect on reducing blood pressure and cardiovascular risk. 29 One study suggests that practitioners have adopted these lower doses for the prevention of kidney stones without any evidence that they remain effective for the prevention of recurrent kidney stone episodes. 13 Consequently, there is currently a clinical trial examining this. 30 We provide evidence that lower dose thiazide diuretics confer a similar protective effect to high-dose thiazide diuretics on the risk of having a kidney stone episode.
Despite employing a large cohort of patients prescribed a new thiazide diuretic, this work has limitations. Our outcome required the person to seek medical care for a kidney stone, and therefore we could not detect asymptomatic kidney stones or events for which an individual did not seek medical attention. This also prevented us from determining whether an event was due to a single stone or multiple and whether a recurrent event was due to a prior or new kidney stone. Although we adjusted for several confounders, including important comorbidities, we were unable to adjust for other potential confounders altering kidney stone formation risk, such as dietary factors. Furthermore, individuals were not randomly assigned a thiazide diuretic dose. Consequently, there is a risk of residual confounding. Our follow-up period was short; nonetheless, this time frame was sufficient to detect a difference in risk of stone presentation between different classes of antihypertensives. 9 Finally, our cohort only included individuals older than 65 years of age, and whether or not our results are valid in a younger population needs to be confirmed.
Conclusions
We employed a large cohort of individuals receiving a new prescription for a thiazide diuretic to examine the relative risk of presenting with a kidney stone based on the dose (low vs high) of drug prescribed. We showed no difference in the likelihood of an event in those prescribed low- vs high-dose thiazide diuretics. These results are consistent with lower dose thiazide diuretics preventing kidney stones to the same degree as higher doses.
Supplemental Material
supplemental_material-_R1Black – Supplemental material for Thiazide Diuretic Dose and Risk of Kidney Stones in Older Adults: A Retrospective Cohort Study
Supplemental material, supplemental_material-_R1Black for Thiazide Diuretic Dose and Risk of Kidney Stones in Older Adults: A Retrospective Cohort Study by R. Todd Alexander, Eric McArthur, Racquel Jandoc, Blayne Welk, Daniel G. Fuster, Amit X. Garg and Robert R. Quinn in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
R.T.A. is the Canada Research Chair in Renal Tubular Transport Physiology. A.X.G. was supported by the Dr. Adam Linton Chair in Kidney Health Analytics and a Clinician Investigator Award from the Canadian Institutes of Health Research (CIHR). D.G.F. was supported by the Swiss National Centre of Competence in Research NCCR TransCure and the Swiss National Science Foundation (grants 31003A_172974 and 33IC30_166785). This study was supported by the ICES Western site. The analysis was conducted by members of the Institute for Clinical Evaluative Sciences Kidney, Dialysis and Transplantation (ICES KDT) team at the ICES Western facility, who are supported by a grant from CIHR. The study design and conduct, opinions, results, and conclusions reported in this article are those of the authors and are independent of the funding sources. No endorsement by ICES, Academic Medical Organization of Southwestern Ontario (AMOSO), Schulich School of Medicine and Dentistry (SSMD), Lawson Health Research Institute (LHRI), CIHR, or the Ontario Ministry of Health and Long-Term Care (MOHLTC) is intended or should be inferred. Parts of this material are based on data and/or information compiled and provided by CIHI. However, the analyses, conclusions, opinions and statements expressed in the material are those of the authors, and not necessarily those of CIHI. Authors also thank IMS Brogan Inc for use of their Drug Information Database.
Ethics Approval and Consent to Participate
This study was approved by the institutional review board at Sunnybrook Health Sciences Centre (Toronto, Ontario, Canada). Participant informed consent was not required for this study.
Consent for Publication
We have the authors consent for publication.
Availability of Data and Materials
The materials are provided in detail in the supplemental materials section. Please contact the corresponding author for Data queries.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Canadian Institutes of Health Research (CIHR) operating grant (MOP# 136891) to R.T.A. and by the Stollery Children’s Hospital Foundation through the Women and Children’s Health Research Institute as well as the Institute for Clinical Evaluative Sciences Kidney, Dialysis and Transplantation (ICES KDT) Program. ICES is a nonprofit research corporation funded by an annual grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC). Core funding for ICES Western is provided by the Academic Medical Organization of Southwestern Ontario (AMOSO), the Schulich School of Medicine and Dentistry (SSMD), Western University, and the Lawson Health Research Institute (LHRI).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
