Abstract
Purpose of review:
Use of gadolinium-based contrast agents (GBCA) in renal impairment is controversial, with physician and patient apprehension in acute kidney injury (AKI), chronic kidney disease (CKD), and dialysis because of concerns regarding nephrogenic systemic fibrosis (NSF). The position that GBCA are absolutely contraindicated in AKI, category G4 and G5 CKD (estimated glomerular filtration rate [eGFR] < 30 mL/min/1.73 m2), and dialysis-dependent patients is outdated and may limit access to clinically necessary contrast-enhanced magnetic resonance imaging (MRI) examinations. This review and clinical practice guideline addresses the discrepancy between existing Canadian guidelines regarding use of GBCA in renal impairment and NSF.
Sources of information:
Published literature (including clinical trials, retrospective cohort series, review articles, and case reports), online registries, and direct manufacturer databases were searched for reported cases of NSF by class and specific GBCA and exposed patient population.
Methods:
A comprehensive review was conducted identifying cases of NSF and their association to class of GBCA, specific GBCA used, patient, and dose (when this information was available). Based on the available literature, consensus guidelines were developed by an expert panel of radiologists and nephrologists.
Key findings:
In patients with category G2 or G3 CKD (eGFR ≥ 30 and < 60 mL/min/1.73 m2), administration of standard doses of GBCA is safe and no additional precautions are necessary. In patients with AKI, with category G4 or G5 CKD (eGFR < 30 mL/min/1.73 m2) or on dialysis, administration of GBCA should be considered individually and alternative imaging modalities utilized whenever possible. If GBCA are necessary, newer GBCA may be administered with patient consent obtained by a physician (or their delegate) citing an exceedingly low risk (much less than 1%) of developing NSF. Standard GBCA dosing should be used; half or quarter dosing is not recommended and repeat injections should be avoided. Dialysis-dependent patients should receive dialysis; however, initiating dialysis or switching from peritoneal to hemodialysis to reduce the risk of NSF is unproven. Use of a macrocyclic ionic instead of macrocyclic nonionic GBCA or macrocyclic instead of newer linear GBCA to further prevent NSF is unproven. Gadopentetate dimeglumine, gadodiamide, and gadoversetamide remain absolutely contraindicated in patients with AKI, those with category G4 or G5 CKD, or those on dialysis. The panel agreed that screening for renal disease is important but less critical when using macrocyclic and newer linear GBCA. Monitoring for and reporting of potential cases of NSF in patients with AKI or CKD who have received GBCA is recommended.
Limitations:
Limited available literature (number of injections and use in renal impairment) regarding the use of gadoxetate disodium. Limited, but growing and generally high-quality, number of clinical trials evaluating GBCA administration in renal impairment. Limited data regarding the topic of Gadolinium deposition in the brain, particularly as it related to patients with renal impairment.
Implications:
In patients with AKI and category G4 and G5 CKD (eGFR < 30 mL/min/1.73 m2) and in dialysis-dependent patients who require GBCA-enhanced MRI, GBCA can be administered with exceedingly low risk of causing NSF when using macrocyclic agents and newer linear agents at routine doses.
What was known before
NSF is strongly associated with administration of GBCA in patients with category G4 or G5 CKD or on dialysis. Avoidance of GBCA in patients with renal impairment may be unnecessary due to important differences in incidence of NSF with macrocyclic and newer linear GBCA.
What this adds
This guideline acknowledges that NSF is exceedingly uncommon when macrocyclic or newer linear GBCA are used in patients with category G4 or G5 CKD improves access to GBCA-enhanced MRI when medically necessary and in immediate and anticipated reductions in outpatient screening of renal function prior to GBCA-enhanced MRI.
Introduction
Gadolinium-based contrast agents (GBCA) have been in clinical use for decades and have an integral role in magnetic resonance imaging (MRI) examinations. In general, GBCA have an excellent safety profile1-7; however, they have been identified as the causative agent in nephrogenic systemic fibrosis (NSF). 8 Due to the associations between NSF in patients receiving GBCA and renal impairment, the use of GBCA has been considered absolutely contraindicated in patients with acute kidney injury (AKI) and severe chronic kidney disease (CKD) and in those receiving dialysis.9-11 Nevertheless, the vast majority of documented NSF cases have occurred in patients who received linear nonionic or older linear ionic GBCA (often repeatedly and/or at higher-than-recommended dosage). 12 The incidence of NSF has substantially decreased over the past several years, and this is attributed mainly to physician awareness and avoidance of GBCA in at-risk patients, the use of newer macrocyclic and linear ionic GBCA, and avoiding repeat injections and greater than recommended dosing.12-14 In Canada, for example, to our knowledge the last officially documented case of NSF occurred in a 70-year-old patient who received an unspecified GBCA in 2011. 15
To address a disparity in Canadian guidelines regarding NSF and more recent literature evaluating NSF in macrocyclic and newer linear ionic agents, updated guidelines were developed by a joint working group involving members of the Canadian Association of Radiology (CAR) and the Canadian Society of Nephrology (CSN). The goals of the working group were to review the literature regarding the NSF safety profile of GBCAs that are currently approved for clinical use in Canada and to recommend clinical practice guidelines for physicians when considering the use of GBCA in at-risk patients, namely those with AKI, stage G4 or G5 CKD, or on dialysis (herein referred to simply as “at-risk” patients); see Appendix A. The purpose of these guidelines was not to exhaustively review the mechanisms of NSF, or the biochemistry of gadolinium chelates in general, or to address the recently-described phenomenon of gadolinium deposition in the brain (which is being reviewed with guidelines formulated by a separate working group commissioned by the CAR); however, these concepts were considered and are addressed briefly herein. Data on the risk of GBCA allergic adverse events are not reviewed and are beyond the scope of this guideline but have been described elsewhere. 16
Background
GBCAs have been used in conjunction with MRI since the 1980s and have an overall excellent cumulative safety record.17,18 The ideal GBCA has high relaxivity (to generate increased contrast on T1-weighted magnetic resonance images), would require a low, nontoxic dose, and is well-tolerated without any adverse immediate or long-term effects. GBCAs are derived and administered in a chelated form to minimize the amount of free gadolinium in the body.17-19 GBCAs used clinically should have high stability to prevent dissociation of gadolinium and should be rapidly cleared and excreted from the body.17,18 The stability of GBCAs depends on their chemical structure; GBCAs can be classified as (1) either linear or cyclic (note that GBCAs with a cyclic chelating ligand are commonly referred to as macrocyclic) and (2) electrically neutral (nonionic) or charged (ionic). Although the overall stability of a given GBCA is unique for each molecular structure and other factors that may influence stability that are beyond the scope of this article, 17 in general a macrocyclic structure confers greater stability than a linear structure, and to a lesser extent, ionic GBCAs have greater stability than nonionic structure.17-19 The rapidity of the clearance of GBCAs from the body is another key determinant of patient safety as the extended presence of GBCAs (due to delayed clearance) within the body increases the risk of release of dissociated free gadolinium ions. 17 All but one GBCA (ie, gadoxetic acid) are excreted almost exclusively by the kidneys: renal function is critical when considering risk of NSF.
NSF is a serious late adverse reaction associated with exposure to GBCAs that can occur in patients with severe renal impairment and for which there is currently no known specific or consistently effective treatment.12,18-21 Although the association between exposure to GBCAs and development of NSF is widely accepted, the mechanism is incompletely understood.12,18 The risk factors for NSF can be divided into patient-related factors and those related to the molecular structure and stability of the GBCA used. Based on current evidence, NSF occurs almost exclusively in patients receiving GBCAs who have category G4 (severe decrease in estimated glomerular filtration rate [eGFR], with or without other evidence of kidney damage; eGFR 15-29 mL/min/1.73 m2) or category G5 (established renal failure eGFR < 15 mL/min/1.73 m2 or on dialysis) CKD based on the Kidney Disease Outcomes Quality Initiative (KDOQI) from the National Kidney Foundation. 22 Therefore, severely reduced renal function is the most important patient-related risk factor predisposing to NSF.12,18,19,23 The degree of renal insufficiency is also important, with a much greater incidence of NSF in patients with category G5 CKD compared with category G4 CKD. Acute kidney injury is also considered a risk factor for NSF.12,19 A proinflammatory state in a patient with impaired renal function has also been reported as a risk factor.12,19,24,25 Despite initial concerns, severe liver disease is no longer considered a risk factor for NSF, as long as the patient has normal renal function.12,18,19,26-28 Because of renal immaturity in fetuses, neonates, and infants, this population (and consequently pregnant women because of the risk to the fetus) is considered potentially at risk.12,18,19,29,30 Regardless of the type of GBCA used, higher doses and multiple administrations, especially within a short period of time, have been reported as risk factors for the development of NSF.
Risk for NSF is also generally considered to be related to the molecular structure and stability of the GBCA used. The majority of unconfounded cases of NSF that have been reported are associated with 3 GBCAs: gadodiamide, gadopentetate dimeglumine, and gadoversetamide (see Table 1 and subsequent sections for a description of different GBCAs currently used clinically).19,31-33
Structure, Stability, Estimated Number of Global Injections, Number of Confounded and Unconfounded Reports of NSF, Classification by the European Medicines Agency, US Food and Drug Administration (FDA) and American College of Radiology, and Health Canada Guidelines for Patients With Severe Chronic Kidney Disease and Acute Kidney Injury Among Currently Approved Gadolinium-Based Contrast Agents for Clinical Use in Canada.

Note. NSF = nephrogenic systemic fibrosis; EMA = European Medicines Agency; FDA = Food and Drug Administration; ACR = American College of Radiology.
Gadoteric acid was not available for clinical use in Canada at time of most recent Health Canada update on NSF. 9
The incidence of NSF using newer linear and macrocyclic agents has decreased considerably. It is difficult to accurately estimate the prevalence of NSF, but it is now considered a rare entity. Reported incidences of NSF vary but when using standard dosing and macrocyclic or newer linear GBCAs are generally cited to be much less than 1% (with the denominator being administration to all patients); however, this may underestimate the true prevalence of disease given that for a time GBCA was avoided nearly universally in at-risk populations.12,18,19,33 For example, in at-risk populations, the prevalence can be much higher, between 3% and 7% for older linear GBCAs but remains case reportable for the newer linear and macrocyclic GBCAs.18,31 A discussion of criteria for the diagnosis of NSF, reporting bias or underreporting of NSF, and unconfounded versus confounded cases is beyond the scope of this article but has been described elsewhere.18,19,34
There is a long-standing recognition that gadolinium can replace calcium in the hydroxyapatite of bone 19 and more recently, there are increasing concerns about GBCA deposition in the brain.35-37 This is the topic of a separate CAR working group on GBCA use and safety and will not be further discussed here.
Currently Approved GBCA for Clinical Use in Canada
There are presently 8 GBCA approved for clinical use in Canada 9 ; these are summarized in Figure 1 and Table 1. The following section describes these agents in detail as they pertain to risk of NSF. Following published literature, “unconfounded” cases refer to the occurrence of NSF with prior exposure to only one GBCA; “confounded” cases refer to cases where more than one GBCA was administered, and hence causality is difficult to assign. The GBCAs are divided into 3 classes: (1) older linear agents associated with the highest number of unconfounded cases of NSF (gadodiamide, gadopentetate dimeglumine, and gadoversetamide), (2) newer linear agents associated with zero unconfounded cases of NSF (gadobenate dimeglumine and gadoxetate disodium), and (3) macrocyclic agents associated with an exceedingly low number of unconfounded cases of NSF. The American College of Radiology (ACR) manual on contrast media v 10.3 classifies the older linear GBCAs as class I, macrocyclic agents and gadobenate dimeglumine as class II, and considers gadoxetate disodium as a class III agent separate from gadobenate dimeglumine (due to limited data regarding NSF risk 38 ); however, in this document the panel suggested that gadoxetate disodium and gadobenate dimeglumine be considered together due to similar structure and number of NSF cases reported to date.
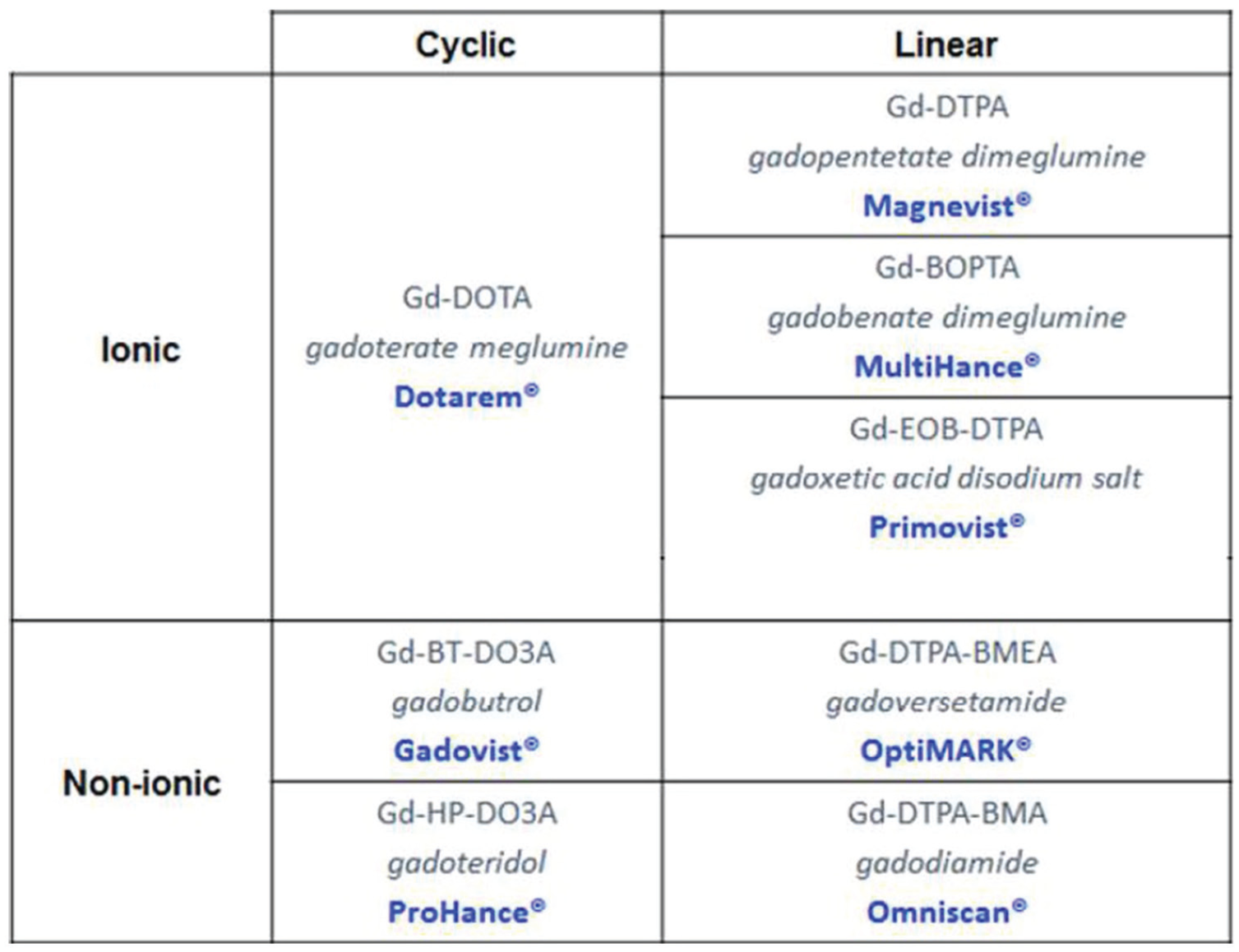
Depiction of the current gadolinium-based contrast agents approved for clinical use in Canada.
Older Linear Agents Associated With the Greatest Number of Unconfounded Cases of NSF
Gadodiamide, gadopentetic acid, and gadoversetamide
Gadodiamide, marketed by GE Healthcare (Chicago, Illinois) as Omniscan, a linear nonionic GBCA (thermodynamic stability of 16.9), gadopentetic acid, marketed as Magnevist by Bayer Healthcare Pharmaceuticals (Berlin, Germany), a linear ionic GBCA (thermodynamic stability of 22.5), and to a lesser extent gadoversetamide, marketed as Optimark by Guerbet (Villepinte, France), a linear nonionic GBCA (thermodynamic stability of 16.6) are by far the most common GBCA associated with reported cases of NSF. 12 At the time of preparation of this document, 438 unconfounded cases of NSF have been attributed to gadodiamide, 135 unconfounded cases of NSF have been attributed to gadopentetic acid, and 7 unconfounded cases of NSF attributed to gadoversetamide. 12 By comparison to macrocyclic and newer linear agents, these 3 agents constitute the vast majority of reported cases of NSF. In cases of NSF confounded by the use of multiple agents, when 1 of these 3 agents had been administered (90 confounded cases of NSF with gadodiamide, 276 confounded cases with gadopentetic acid and 11 confounded cases with gadoversetamide 12 ), it is generally assumed that 1 of these agents was likely the causative GBCA which led to the development of disease, although it is not possible to prove which agent was ultimately responsible for causing NSF when multiple agents had been administered to the same patient. These agents are considered as high risk for causing NSF in at-risk populations by the European Medicines Agency 11 and categorized as Class I agents by the American College of Radiology Manual on Contrast Media v 10.3 (agents associated with the greatest number of NSF cases). 38 The use of any of these 3 agents in patients with AKI, on dialysis, or with severe CKD continues to be considered absolutely contraindicated by the US Food and Drug Administration (FDA), 10 European Medicines Agency, 11 and Health Canada. 9 The panel does not recommend using gadodiamide, gadopentetic acid, or gadoversetamide in at-risk patients under any circumstances and could not conceive of an instance when an MRI facility in Canada could not obtain an alternative macrocyclic or newer linear GBCA even with single vendor contrast agreements where in our opinion exemptions should be granted.
Newer Linear Agents Associated With Zero Unconfounded Cases of NSF
Gadobenate dimeglumine
Gadobenate dimeglumine, marketed as MultiHance by Bracco Pharmaceuticals (Milan, Italy), is a linear, ionic contrast agent. 39 It has been marketed in Canada since 2004, after receiving approval from Health Canada, 40 and is classified as a group II medical imaging agent by the FDA (approved in 2004) 41 and by the ACR. 42 The distribution of gadobenate dimeglumine is extracellular and its clearance is mostly renal (96%). 39 The biliary elimination pathway (4%) provides limited protection for patients with CKD. Gadobenate dimeglumine has a thermodynamic stability constant of 22.6. 39
According to a literature review performed by Bracco Pharmaceuticals, as of 31 October 2016, there are no medically confirmed, unconfounded cases of NSF following the sole administration of gadobenate dimeglumine. This document reports 32 confounded cases of NSF that arose following the administration of MultiHance and other GBCA, most often gadodiamide and gadopentetate dimeglumine. There are no known confounded cases of NSF where a patient received only gadobenate dimeglumine and gadoteridol in combination. 43 A summary of important studies evaluating NSF and gadobenate dimeglumine is provided in Appendix B.
Gadoxetate disodium
Gadoxetate disodium (marketed as Primovist in Europe and Canada and Eovist in the United States; Bayer Pharmaceuticals) is a linear ionic GBCA 39 distributed in Canada since 2010, after receiving approval from Health Canada. 40 The FDA approved it in 2008 44 and ACR 42 classifies it as a group III agent. The thermodynamic stability constant of gadoxetate disodium is 23.5 (37). Gadoxetate disodium has a dual-elimination pathway split evenly between renal (50%) and hepatobiliary (50%) pathways. 39 The extrarenal clearance could theoretically reduce the risk of NSF in patients with CKD, assuming normal liver function; however, empiric data are scant. Another potentially protective feature of gadoxetate disodium is the lower gadolinium dose, which is 25% that of other GBCAs at 0.025 mmol/kg, because a large proportion of NSF cases involved higher doses or repeated GBCA injections (see, for example, Zou and Ma, 45 where 245 out of 280 patients who developed NSF with other GBCAs had received a higher than standard dose 45 ); however, this is also unproven.
The number of studies assessing the safety of gadoxetate disodium is very limited. In a 2009-2013 prospective multicenter study, in renally impaired patients scheduled for a liver imaging examination, Lauenstein et al reported no NSF events in 85 patients with category G4 or G5 CKD, including 39 on dialysis, who received gadoxetate disodium at full dose (0.025 mmol/kg). 46 The same study reported no NSF events in 193 patients with nondialysis category G3 CKD who received gadoxetate disodium at full dose. 46 In a 2011-2013 retrospective study, Song et al evaluated 40 patients at risk for NSF who underwent 2 liver MRI studies within a 6-month interval, using full and half doses of gadoxetate disodium; the eGFR at the time of the half-dose MRI ranged between 4.8 and 58.5 mL/min/m2. With the half-dose MRI, there was reduced hepatic enhancement in all but the arterial phase; however, there was no difference in diagnostic quality between the 2 doses or in any cases of NSF. 47
While no unconfounded or confounded cases of NSF have been reported using gadoxetate disodium, a limitation to evaluating its safety profile with respect to NSF is that, as a newer agent with specific indications, there have been substantially fewer injections of gadoxetate disodium as compared with other GBCAs. The use of gadoxetate disodium in clinical practice is generally restricted to the assessment of colorectal cancer liver metastases (CRCLM), differentiating hepatic focal nodular hyperplasia (FNH) from hepatocellular adenoma (HCA), and evaluating for biliary leaks. CRCLM detection is particularly crucial for surgical planning (ie, metastasectomy), and although alternative studies (contrast-enhanced computed tomography [CT] and positron emission tomography [PET]) are available, there is a higher detection rate using gadoxetate disodium–enhanced MRI compared with both CT and PET.48,49 Gadoxetate disodium–enhanced MRI is also more accurate when compared with conventional MRI and CT for confirmation of radiologic complete response of liver metastases (“disappearing metastases”). 50 The usefulness of gadoxetate disodium for diagnosis of FNH versus HCA has recently been found to be less than previously reported, 51 and the patient population for which these diagnoses are considered are generally young healthy female patients where AKI or severe renal impairment is not anticipated. Therefore, a clinical situation where gadoxetate disodium is required to evaluate for FNH versus HCA in AKI or severe renal impairment is unlikely. The use of gadoxetate disodium in cirrhotic patients for diagnosis of hepatocellular carcinoma is under active investigation and not currently considered the reference standard (compared with extracellular GBCAs). 52
Macrocyclic Agents Associated With an Exceedingly Low Number of Unconfounded Cases of NSF
Gadoterate meglumine
Gadoterate meglumine, marketed as Dotarem by Guerbet Pharmaceuticals (Villepinte, France) is a macrocyclic, ionic agent.53,54 This agent was approved by Health Canada in 2016 40 but has been in clinical use in Europe for many years. It is classified as a group II agent by the FDA, who approved it in 2013 55 and by the ACR. 42 Its distribution is extracellular, and its clearance is entirely renal. 39 The thermodynamic stability constant is 25.6. 39 Gadoterate meglumine has the highest thermodynamic stability of all the GBCAs, and it has been suggested that its structure may make it safer for use in patients at risk for NSF 53 although this is unproven.
The vast majority of events involving gadoterate meglumine are summarized in a 2015 article reviewing data from clinical study and patient safety databases, as well as postmarketing safety studies. 56 According to this article, as of September 2015, there have been 44 cases reported with a suspicion of NSF involving GBCAs; of these, administration of gadoterate meglumine was involved in 18 cases (of which 17 were multiagent cases). Upon further review, only 7 of these cases were deemed confirmed or consistent with NSF, and these were all confounded cases. 56 In our review, we found 1 potentially unconfounded case of NSF following administration of gadoterate meglumine in the literature; it was reported in 2006 in a nondialysis patient with CKD from Denmark who underwent 2 MRI scans and received a total dose larger than 30 mL. 57 It is unclear from that article whether the 2 MRI scans both used gadoterate meglumine and we attempted to contact the authors for clarification but were unsuccessful. Other important studies evaluating the safety of gadoterate meglumine with respect to NSF are summarized in Appendix C.
Gadoteridol
Gadoteridol (marketed as ProHance; Bracco Pharmaceuticals) is a macrocyclic, nonionic agent, which was first approved by the FDA in 1992 and later by Health Canada in 1996. Currently, it is the only agent approved by the FDA for a cumulative dose of 0.3 mmol/kg and is classified as a group II agent by the ACR. 38 Gadoteridol is primarily eliminated by the kidneys, with over 94% excreted within 24 hours after injection. 58 Gadoteridol has one of the highest thermodynamic stability constants among the approved GBCAs at 23.8 39 and is one of the most kinetically inert. 59
As of 2014, a literature review of regulatory databases identified 22 of 382 histologically proven cases of NSF with either gadobenate dimeglumine or gadoteridol; however, all 22 cases involving these agents were confounded. 60 The review did not indicate which other GBCAs these patients were initially exposed to, or the number of times they were exposed to a particular agent. A different study cites that multiagent NSF cases involving gadoteridol were confounded by prior administration of either gadodiamide or gadopentetate dimglumine 61 ; however, it is unclear whether the patients from the latter study were included in the 2014 literature review. A letter dated June 2017 from Bracco Pharmaceuticals confirms that they are aware of a total of 37 confounded cases involving gadoteridol, mostly involving gadodiamide and/or gadopentetate dimeglumine. 43
A review from 2017 identified only 2 unconfounded cases of NSF with gadoteridol; however, we could not find the source for this to confirm. 39 Potentially, one of the cases was described in another study from MedWatch data, which reported that the patient had received 5 injections of gadoteridol in a 2-year period before the development of NSF. 62 A literature search by Bracco Pharmaceuticals acknowledges this as an unconfounded case of NSF but with 6 high doses of gadoteridol; however, they indicate that no other instances of single-agent NSF associated with gadoteridol administration have been reported. 43
There have been both retrospective and prospective, multicenter studies focused on gadoteridol use in relation to CKD and NSF development. Even with some patients receiving higher doses for magnetic resonance angiography studies, there have been no reports of NSF developing within 2 years of observation post-injection for patients with category G3-5 CKD.25,63
Gadobutrol
Gadobutrol, marketed by Bayer Healthcare Pharmaceuticals (Berlin, Germany) as Gadovist (a macrocyclic, nonionic GBCA) was approved by Health Canada in 1999 and by the FDA in 2011. It is classified as a group II by the ACR 38 and has a thermodynamic stability constant of 21.8. Several clinical trials and postmarketing surveillance studies have indicated that gadobutrol is well-tolerated among diverse patient populations.64-67 After injection, gadobutrol is rapidly distributed into the extracellular space; elimination is primarily by the kidneys, with 100% excreted within 72 hours in healthy adults. 68 Likewise, for patients with mild or moderate CKD (eGFR between 30 and 80 mL/min/1.73 m2), a standard dose of gadobutrol was renally cleared within 72 hours; however, in patients with severely impaired renal function (≤30 mL/min/1.73 m2 but not requiring dialysis), recovery in the urine was approximately 80% even after 120 hours. 69 However, all gadolinium could be recovered in patients with severe renal insufficiency over a 6-day period with dialysis, 70 suggesting minimal, if any, gadolinium dissociation over this time frame.
As of December 2015, more than 5.7 million injections have been given to patients, with 12 reports of patients experiencing NSF-like symptoms after injection. 71 Of these, only 3 cases were found to be unconfounded. One patient when first exposed to gadobutrol apparently had category G3 CKD (limitations pertaining to the reported stage of renal disease in this patient are discussed in the guideline section of the article); another had insulin-dependent diabetes and was on hemodialysis when exposed to gadobutrol, 72 and the third patient had end-stage renal failure when exposed to gadobutrol for the first time, 73 although it is unclear whether this was a true case of NSF. 74 For multiagent cases of NSF, the patient was previously exposed to gadopentetate dimeglumine within 6 months prior to gadobutrol. 75 No details could be found regarding the other 8 cases.
Recently, the results of the prospective, multicenter “GRIP” (Gadobutrol in Renally Impaired Patients) study were released 67 ; 927 patients with category G3-5 CKD were enrolled (category G3 586/927 and category G4 or G5 284/927), given 0.1 mmol/kg gadobutrol and followed for 2 years post-injection for signs of NSF. Similar to other studies, no incidences of NSF were reported.
Screening for Compromised Renal Function to Identify At-Risk Patients for NSF
The documented increased risk for development of NSF following GBCA administration in patients with AKI, with severe CKD, and on dialysis24,76,77 makes screening for renal disease important prior to GBCA administration. Traditionally, screening for renal dysfunction prior to GBCA administration was considered of paramount importance to reduce the incidence of NSF by identifying at-risk patients. The eGFR, which can be calculated from serum creatinine concentration, age, ethnicity, and gender, is a better measure of renal function compared with serum creatinine alone. 78 Routine screening for CKD in asymptomatic adults before enhanced MRI is now considered controversial.79-82 Obtaining blood samples to determine eGFR on all patients referred for GBCA-enhanced MRI leads to considerable expense for an anticipated minimal benefit, particularly considering the exceedingly low risk of developing NSF with macrocyclic and newer linear GBCA, and furthermore may not identify patients with AKI.83,84 Another approach would be to ask patients whether they have kidney disease or use a combination of blood work and surveys 85 ; however, patient awareness of their renal function is poor, and directly asking patients whether they have CKD is likely not an effective approach to screening.86,87
A questionnaire developed by Choyke et al 88 has been shown to effectively stratify patients by risk of NSF 89 and includes 6 questions (Table 2). Using these 6 questions at time of MRI scheduling and scanning to predict eGFR < 30 mL/min/1.73 m2 in 665 patients, Sena et al found that answering “no” to all questions both times the questionnaire was administered had a sensitivity of 100% (95% confidence interval 59%-100%). 89 In this same study, only 2 patients with eGFR < 30 mL/min/1.73 m2 would have been missed if the questionnaire was only administered at time of MRI scheduling. In 2015, Too et al also found the Choyke questionnaire to have 100% sensitivity for detection of eGFR less than 30 mL/min/1.73 m2 in a study of 1361 outpatients when serum creatinine was measured at time of scanning. 90 The Choyke questionnaire was not found to be very specific, with specificities ranging from 44% and 74% in these 2 studies89,90; however, the extremely high sensitivity is the desired outcome when applying the questionnaire to identify patients with possible renal dysfunction. Choyke et al did not find age to be a significant predictor of renal function. 88 In addition to the questions included in the Choyke questionnaire, dialysis-dependent patients must clearly also be identified before MRI.
Screening Questionnaire to Be Administered to the Outpatient Population to Identify Renal Disease at Time of MRI Scheduling (for Institutions Using Macrocyclic Agents and Newer Linear GBCA) and Also Immediately Before MRI for Institutions Which Use Gadodiamide, Gadopentetate Dimeglumine, and Gadoversetamide.

Note. Pregadolinium screening questionnaire for identifying patients at risk of poor renal function, adapted from Choyke et al. 88 MRI = magnetic resonance imaging; GBCA = gadolinium-based contrast agents.
Not included in the original Choyke survey but important to clarify prior to administration of GBCA. The panel suggests the question be asked to the patient at time of MRI scheduling and at time of MRI.
Given the exceedingly low risk of NSF when using standard doses of macrocyclic and newer linear GBCA, the panel unanimously favored that in outpatients, screening for renal disease with the questionnaire at the time of MRI scheduling only when using these agents was sufficient and that it was not necessary to repeat the questionnaire on the day of the test13,63,67,91,92; however, the patient should be asked again at time of MRI scanning whether they are presently receiving dialysis. At institutions that continue to use high-risk agents (gadodiamide, gadopentetate dimeglumine, and gadoversetamide), 42 the panel suggests using the pregadolinium screening questionnaire both at time of MRI scheduling and immediately prior to MRI scanning to maximize sensitivity for detection of renal impairment.88-90 Institutions which use high-risk agents should also consider switching to a macrocyclic or newer linear agent for patients approaching category G4 CKD, and in patients with category G4 or G5 CKD or on dialysis, high-risk agents are absolutely contraindicated. For outpatients answering “yes” to the questionnaire, a serum creatinine is required for estimation of eGFR. The panel suggests obtaining a serum creatinine, if not available or older than 3 to 6 months from the date of administration of the screening questionnaire. For inpatients, the panel endorses what has been recommended by the ACR, namely obtaining an eGFR within 48 hours of anticipated GBCA-enhanced MRI. 42 Inpatients should also be assessed for potential AKI regardless of their eGFR, because eGFR is not always representative of renal function in this setting. Several helpful clinical tools include the Acute Kidney Injury Network (AKIN) and Kidney Disease Improving Global Outcomes (KDIGO) criteria.93-95
We acknowledge the current position of the ACR which suggests optional screening of renal function when using “class II” agents (macrocyclic GBCA and gadobenate dimeglumine) due to the “few if any” unconfounded cases of NSF attributed to these agents and that dialysis would not be administered to a patient not already on dialysis when receiving a class II GBCA even with AKI or severe CKD. 42 The panel in general agreed with this strategy; however, at this time unanimously suggested an intermediate approach using less rigid screening parameters than what has been suggested previously by the CAR. For institutions using macrocyclic agents and newer linear GBCA, we do not consider screening with the questionnaire at time of MRI scheduling to be particularly onerous and plan to revisit the need for routine outpatient screening when reevaluating the guideline at our next audit.
In conclusion, this document reviews the relevant literature regarding NSF in an era of increased physician and patient awareness of disease and associated risk factors (eg, renal impairment, increased and repeated dosing of GBCA, and dialysis) and specifically reevaluates the role of GBCA-enhanced MRI using newer macrocyclic and linear agents. Guidelines were developed by a panel of radiologists representing the CAR and nephrologist members of the CSN based on the best available evidence and may provide a framework for use of GBCA in renal impairment in clinical practices in Canada. In summary, the risk of developing NSF in patients with AKI, with severe CKD, and on dialysis when macrocyclic or newer linear GBCAs are used is exceedingly low when standard dosages are prescribed and short-term repeat injections are avoided. We recommend screening for renal impairment with the 6-question Choyke questionnaire at time of MRI scheduling in facilities using macrocyclic and newer linear agents and also advise that the questionnaire should be repeated at time of MRI for facilities using gadodiamide, gadopentetate dimeglumine, and gadoversetamide. In patients with AKI or severe CKD, alternate diagnostic tests should be considered first; however, if GBCA-enhanced MRI is considered medically necessary, it can be performed using macrocyclic or newer linear agents; gadodiamide, gadopentetate dimeglumine, and gadoversetamide remain absolutely contraindicated. Patient informed consent is recommended and can be obtained by a physician or their delegate. Dialysis should only be performed in patients who are already dialysis dependent, and there is no reliable evidence to suggest that initiating dialysis, switching from peritoneal to hemodialysis, or altering dialysis regimens reduces the risk of developing NSF.
Footnotes
Appendix A
Appendix B
Several studies have reported a 0% incidence of NSF after administration of gadobenate dimeglumine, some using restrictive measures on patients with renal dysfunction.13,23,118-120 In a large, 1999-2009 retrospective 2-center study in the United Kingdom, in patients undergoing GBCA-enhanced MRI, Chrysochou et al reported no NSF events in 74 patients with category G5 CKD without dialysis who received gadobenate dimeglumine at the standard dose (0.1 mmol/kg). 23 In the same study, Chrysochou et al reported no NSF events in 272 patients with category G4 CKD without dialysis who received gadobenate dimeglumine at full dose. 23 Similarly, in a large 2000-2008 retrospective study collecting data from 2 American academic centers, Altun et al reported no NSF events in over 25,000 patients, including 549 dialysis or at-risk patients (eg, category G4 or G5 renal disease), who received gadobenate dimeglumine at half dose. 118 In a 2007-2008 retrospective single-center study, Martin et al reported no NSF events in 784 patients on dialysis who received gadobenate dimeglumine at a mean dose of 0.11 mmol/kg (0.05-0.75 mmol/kg). 119 In a 2002-2010 retrospective single-center study, in patients undergoing contrast-enhanced MRI according to the 2007 restrictive GBCA guidelines of the Massachusetts General Hospital, Wang et al reported no NSF events in 36 patients with category G4 or G5 CKD, including 4 with dialysis at the moment of the MRI procedure. 13 The same study found no cases of NSF in 6454 patients with category G3 CKD who received gadobenate dimeglumine and/or gadopentetate dimeglumine (Magnevist). 13 In a 2007-2013 retrospective single-center study, in patients sustaining gadolinium-enhanced MRI examinations before liver transplantation, Shaffer et al reported no NSF events in 64 CKD patients (eGFR < 60 mL/min/1.73 m2) who received gadobenate dimeglumine at half-dose (0.05 mmol/kg). 120 A 2010 single-center study by Nandwana et al retrospectively evaluated 401 renally impaired patients, including 246 on hemodialysis and 57 on peritoneal dialysis, and in which 93% of patients had category G4 or G5 CKD. Patients received an off-label, increased dose of 0.15 mmol/kg of gadobenate dimeglumine; however, no cases of NSF were detected. 121
In a 2008-2013 prospective multicenter cohort study sponsored by Bracco Pharmaceuticals, Soulez et al reported no NSF events in 45 patients with category G4 or G5 CKD, including 20 on hemodialysis and 6 on peritoneal dialysis, who received gadobenate dimeglumine at a mean dose of 0.11 ± 0.04 mmol/kg. 63 Finally, a 2008-2014 retrospective single-center study found no cases of NSF in 3819 patients with severe CKD who received gadobenate dimeglumine. 91 Patients were administered a mean dose of 23 mL (range, 3-45 mL), which corresponded to 0.05 mmol/kg for MRI and 0.1 mmol/kg for Magnetic Resonance Angiography (MRA). 91
Appendix C
Several studies have evaluated the safety of gadoterate meglumine in patients with acute or chronic kidney disease.23,122-125 In the FINEST study, a 2005-2006 retrospective multicenter study from France, with a cohort of 232 patients undergoing contrast-enhanced MRI, a majority of whom (176/232) received gadoterate meglumine, Janus et al reported no patients with NSF events in 96 patients with CKD category G5 who received gadoterate meglumine. 122 The same study reported no patients with NSF events among 39 patients with category G4 CKD who received gadoterate meglumine. 122 In a large, 1999-2009 retrospective bicenter study in the United Kingdom, in patients undergoing GBCA-enhanced MRI, Chrysochou et al reported no NSF events in 23 patients with category G4 CKD without dialysis who received gadoterate meglumine at the standard dose (0.1 mmol/kg). 23
In the RESCUE trial, a 2008-2011 prospective European multicenter study, Deray et al reported no NSF events at 3-month follow-up in 70 patients with category G3 or G4 CKD who received gadoterate meglumine at full dose (0.1 mmol/kg). 123 Furthermore, in the 2009-2011 Pro-FINEST prospective multicenter trial, Amet et al reported no patients with NSF events in 255 end-stage renal disease (category G5 CKD) patients undergoing long-term dialysis who received gadoterate meglumine at variable doses. 124 Most recently, in the results from the 2008-2013 SECURE study—an international, multicenter, prospective observational study evaluating the safety profile of gadoterate meglumine—Soyer et al found no cases of NSF among 476 post-MRI patients with category G3-5 CKD who received gadoterate meglumine at full dose; however the follow-up duration was less than 3 months in 160 of 476 patients. 125
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
All authors consented for publication
Availability of Data and Materials
Publicly available
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
