Abstract
Background:
Childhood cancer survivors experience adverse drug events leading to lifelong health issues. The Applying Biomarkers to Minimize Long-Term Effects of Childhood/Adolescent Cancer Treatment (ABLE) team was established to validate and apply biomarkers of cancer treatment effects, with a goal of identifying children at high risk of developing cancer treatment complications associated with thrombosis, graft-versus-host disease, hearing loss, and kidney damage. Cisplatin is a chemotherapy well known to cause acute and chronic nephrotoxicity. Data on biomarkers of acute kidney injury (AKI) and late renal outcomes in children treated with cisplatin are limited.
Objective:
To describe the design and methods of the pan-Canadian ABLE Nephrotoxicity study, which aims to evaluate urine biomarkers (neutrophil gelatinase–associated lipocalin [NGAL] and kidney injury molecule-1 [KIM-1]) for AKI diagnosis, and determine whether they predict risk of long-term renal outcomes (chronic kidney disease [CKD], hypertension).
Design:
This is a 3-year observational prospective cohort study.
Setting:
The study includes 12 Canadian pediatric oncology centers.
Patients:
The target recruitment goal is 150 patients aged less than 18 years receiving cisplatin.
Exclusion criteria: Patients with an estimated glomerular filtration rate (eGFR) <30 mL/min/1.73 m2 or a pre-existing renal transplantation at baseline.
Measurements:
Serum creatinine (SCr), urine NGAL, and KIM-1 are measured during cisplatin infusion episodes (pre-infusion, immediate post-infusion, discharge sampling). At follow-up visits, eGFR, microalbuminuria, and blood pressure are measured and outcomes are collected.
Methods:
Outcomes: AKI is defined as per SCr criteria of the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines. CKD is defined as eGFR <90 mL/min/1.73m2 or albumin-to-creatinine ratio≥3mg/mmol. Hypertension is defined as per guidelines.
Procedure: Patients are recruited before their first or second cisplatin cycle. Participants are evaluated during 2 cisplatin infusion episodes (AKI biomarker validation) and at 3, 12, and 36 months post-cisplatin treatment (late outcomes).
Limitations:
The study has a relatively moderate sample size and short follow-up duration. There is potential for variability in data collection since multiple sites are involved.
Conclusions:
ABLE will provide a national platform to study biomarkers of late cancer treatment complications. The Nephrotoxicity study is a novel study of AKI biomarkers in children treated with cisplatin that will greatly inform on late cisplatin renal outcomes and follow-up needs.
What was known before
Kidney damage is a well-known adverse effect of cisplatin chemotherapy in children treated for cancer. Studies on biomarkers of acute kidney injury (AKI) and late renal outcomes in children treated with cisplatin are scarce.
What this adds
The pan-Canadian Applying Biomarkers to Minimize Long-Term Effects of Childhood/Adolescent Cancer Treatment Nephrotoxicity study is one of the largest studies evaluating late renal effects of childhood cancer therapy. This work will help determine whether novel urinary biomarkers are good AKI diagnostic tests, and evaluate their ability to predict long-term chronic kidney disease and hypertension.
Background
Late Effects of Cancer Treatment in Children
Childhood cancer survival is increasing, leading to a growing survivor population. 1 However, many childhood cancer survivors experience chronic health issues (late effects) as a result of treatment.2,3 Although two thirds experience at least 1 organ complication later in life, detailed characterization of these complications is lacking. 4 There is a need to understand how early identification of long-term risks aids in morbidity prevention. It is important to assess long-term effects and monitor their impact as they may have lasting consequences on quality of life and health care costs. 5 This work aims to describe and highlight the unique Applying Biomarkers to Minimize Long-Term Effects of Childhood/Adolescent Cancer Treatment (ABLE) study, a multi-disciplinary team project evaluating biomarkers of late cancer treatment effects, focusing on the ABLE Nephrotoxicity study to enhance future knowledge translation, knowledge dissemination, and collaborations.
ABLE Study Team and Infrastructure
ABLE is a multi-team project with an overall goal to evaluate and validate biomarkers predictive of late childhood cancer treatment complications. ABLE includes (a) 4 projects at different stages of biomarker development, including (1) chronic graft-versus-host disease (cGVHD), (2) deep vein thrombosis (DVT), (3) ototoxicity, and (4) nephrotoxicity, and (b) 3 core resources to support the projects, namely, (1) data management, (2) epidemiology, and (3) clinical trials. Projects and core principal investigators are described in Figure 1.

ABLE Study Organizational Structure.
The ABLE team is led by Dr Kirk Schultz, pediatric hematologist-oncologist at the University of British Columbia (UBC). ABLE is supported by the Canadian Institutes of Health Research (CIHR), C17 council, Canadian Cancer Society, Cancer Research Society, Garron Family Cancer Centre at the Hospital for Sick Children, Ontario Institute for Cancer Research, and Pediatric Oncology Group of Ontario. In response to a CIHR call for Team Grants to study late effects of child cancer treatment, ABLE and 3 other Teams were funded. ABLE funding was received by UBC in 2012.
ABLE principal investigators and core leaders make up most of the steering committee, providing multi-disciplinary scientific leadership to overall ABLE goals. Monthly, this committee discusses study progress at conference calls. Although the 4 projects outlined briefly below are different in topic and methodology, the committee allows project leads to discuss study issues and ensure a common ABLE team end-goal. ABLE has a core study coordinator (UBC) to manage common resource support for all 4 projects. Early in funding, ABLE also benefitted from guidance from an external advisory board composed of child cancer research experts.
Individual Projects and Principal Investigators
Core Groups and Leaders
All 4 ABLE projects interact with each other on a scientific level, use the same database, and share the same infrastructure, creating a unique multi-organ team in Canada.
Epidemiology Coordinating and Research (EPICORE) Centre, the data management core, provides centralized data management to all 4 projects, including design of case report forms (CRFs), data entry and quality management, and biostatistical support. Dr Ross Tsuyuki, Professor of Medicine at the University of Alberta, is director of EPICORE Centre. 12
The epidemiology core is led by Mary McBride, a cancer epidemiology researcher (UBC) with expertise in using BC health care administrative databases to study late childhood cancer treatment outcomes. 13 An overarching ABLE goal is to explore using data registries for linkage with clinical and biomarker data to create large-scale evaluations of associations of biomarkers and other factors with late complications. The epidemiology core’s role is also to encourage and enable large-data outcome studies in children treated for cancer, across all projects.
The clinical trial core is led by Dr Sylvain Baruchel (pediatric hematologist-oncologist, University of Toronto) and was established to facilitate potential clinical trials for each project, to ultimately better treat high-risk populations or prevent long-term effects based on predictive biomarkers. Most projects do not include a clinical trial within this grant per se; including a clinical trial core at this research stage enables the ultimate target goal of performing future trials based on knowledge gained. This core contributes in potential agent identification, clinical trial development discussions, and trial infrastructure.
ABLE Nephrotoxicity Study Protocol
Background and Rationale
Cisplatin is commonly used to treat solid tumors, and is well known to be nephrotoxic via several mechanisms, such as microvascular changes, inflammation, and direct tubular epithelial cell toxicity, often leading to renal cell death.14,15 Cisplatin can cause acute kidney injury (AKI), manifesting as an acute serum creatinine (SCr) rise or as renal tubular injury–associated electrolyte losses (mainly hypomagnesemia, hypophosphatemia, hypokalemia), acidosis, Fanconi syndrome, and polyuria.14,16,17 Most child AKI research has focused on intensive care unit populations, showing AKI associations with hospital morbidity and mortality.18-21 Although more established in adults, emerging child data suggest that AKI may also be associated with increased risk of long-term CKD and hypertension.22-24 Abnormal renal function is frequent 1 year post-cisplatin.25-27 A recent Cochrane systematic review on nephrotoxic childhood cancer treatments reported a 0% to 84% prevalence of long-term renal outcomes, reflecting inconsistent definitions used. 28 There is lack of knowledge on predicting chronic cisplatin nephrotoxicity risk and poor understanding of burden, phenotype, and morbidity. As a result, it is difficult to understand when long-term renal abnormalities become apparent, preventing comprehensive generation of appropriate follow-up guidelines. As CKD and hypertension are 2 important and treatable cardiovascular risk factors, it is important to understand the burden of AKI, CKD, and hypertension in this population and develop prevention and/or treatment strategies.29,30
SCr, the main diagnostic test for AKI, rises late in AKI pathophysiology, limiting ability to diagnose AKI in a timely fashion and test early interventions to mitigate injury progression. Many AKI biomarkers have been studied in the last 10 years to improve timing of AKI diagnosis, but almost none have been performed in the cancer setting. Most biomarkers are proteins measurable in urine in response to renal cell injury. 31 Two putative early AKI biomarkers well studied in non-cancer populations are neutrophil gelatinase–associated lipocalin (NGAL) and kidney injury molecule-1 (KIM-1).32-35 NGAL is a 25-kDa protein involved in injury and repair of renal tubular cells and iron transport.36,37 Animal studies suggest that urine NGAL may be an early cisplatin nephrotoxicity biomarker.38-40 In small adult studies, NGAL rises before SCr in cisplatin-AKI.41-43 KIM-1 is a proximal tubule trans-membrane protein involved in cell-cell interaction. 44 Studies in rats and adults demonstrate that KIM-1 discriminates for cisplatin nephrotoxicity.39,45-47 Both NGAL and KIM-1 are upregulated in proximal tubular cells,32,48 a primary site of cisplatin toxicity. As KIM-1 is a marker of proximal tubular damage and NGAL is a marker of proximal and distal tubule damage, we hypothesized that these urine biomarkers would likely be most diagnostic of cisplatin-induced kidney damage. The existence of non-cancer child data and the known kidney site from which these biomarkers originate provided our rationale to study NGAL and KIM-1. Because AKI biomarkers are indicative of renal cell injury, they may be useful to predict which patients will develop chronic tissue injury and ensuing CKD. This provided the basis to evaluate NGAL and KIM-1 not only as early diagnostic AKI biomarkers, but also as predictors of later CKD and hypertension. We will thus evaluate if NGAL and KIM-1 measured during cancer treatment might predict who will develop late CKD and hypertension (after cancer treatment end).
Methods
ABLE Nephrotoxicity Hypotheses
An inherent objective is to also elucidate the specific phenotype and timing of CKD and hypertension post-cisplatin treatment. Because of funding restrictions, an original project hypothesis regarding proteomics for cisplatin-AKI biomarker discovery was deferred for future work.
Study Design and Setting
This is a 3-year multi-center prospective observational cohort study of 150 children treated with cisplatin. The study originally included 8 Canadian centers; 4 new Canadian sites have recently been approved, for 12 total planned sites (Table 1). Institutional ethics approval was first obtained at the overall ABLE-coordinating site (UBC) for all 4 projects. The Montreal Children’s Hospital Research Ethics Board approved the Nephrotoxicity study protocol. The Nephrotoxicity study was then approved sequentially at participating sites (Table 1). Each site has a pediatric oncologist which enhances recruitment and promotes future knowledge translation. Each site has at least 1 research coordinator who typically also coordinated other ABLE projects. Coordinators are trained via teleconference by the Nephrotoxicity project manager (McGill) and EPICORE Centre’s coordinator (Edmonton) before initiating study activities. They have monthly conference calls with the overall ABLE coordinator and all project managers.
Participating Sites of the ABLE Nephrotoxicity Study.

Note. ABLE = Applying Biomarkers to Minimize Long-Term Effects of Childhood/Adolescent Cancer Treatment; PI = principal investigator; CHU = Centre Hospitalier Universitaire; IWK = Izaak Walton Killam; REB = Research Ethics Board.
Of note, we combined recruitment into Nephrotoxicity and Ototoxicity projects to facilitate recruitment into both studies and enable investigation of common genes (Ototoxicity focus), which may also affect nephrotoxicity. In addition, this study can investigate the correlation between cisplatin ototoxicity and nephrotoxicity development.
Study Participants and Recruitment
Inclusion criteria were established to maximize sample size as there is little knowledge on long-term renal outcomes of cisplatin-treated children, suggesting that a natural history study is needed:
Aged less than 18 years at cancer diagnosis
Initiating cisplatin treatment, with at most 1 prior cisplatin cycle.
Exclusion criteria are as follows:
Pre-existing glomerular filtration rate [GFR] less than 30 mL/min/1.73 m2; or
kidney transplantation prior to cancer therapy initiation.
Site-specific characteristics were considered to enable participant identification and recruitment, including weekly oncology team meetings about new cancer diagnoses: collaboration with pharmacy and/or oncology staff. Most treatment protocols include 3 to 8 cisplatin cycles given every 3 to 6 weeks.49,50 Families are approached for participation ideally before the first cisplatin cycle. However, recruitment prior to second cycle is allowed to maximize recruitment. Cisplatin dose received is not considered when determining eligibility. Informed consent (or assent, as appropriate) is obtained for each patient.
Outcomes
AKI: Biomarker validation outcome
AKI is defined using SCr criteria of the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines (Table 2). 51 The KDIGO-AKI urine output criterion is not used because oliguria is not characteristic of cisplatin-AKI and feasibility of accurate urine output data on non-critical care wards is limited. AKI is ascertained with each cisplatin study infusion episode, using SCr values measured around the day of each respective study infusion episode (from routinely collected SCr values, or if missing, from measured study SCr values).
KDIGO-AKI Staging by SCr Criteria, Modified From the KDIGO Guidelines. 51

Note. AKI is staged into three severity levels. KDIGO = Kidney Disease: Improving Global Outcomes; AKI = acute kidney injury; SCr = serum creatinine; eGFR = estimated glomerular filtration rate.
The KDIGO-AKI definition was not developed with nephrotoxicity phenotypes in mind. There are validity issues with this definition for defining cisplatin-AKI, which is characterized by polyuria and electrolyte disturbances (phosphate, potassium, magnesium), sometimes with no significant SCr rise. Moreover, patients typically receive large amounts of fluids to promote cisplatin excretion, which may affect SCr concentrations. Thus, renal tubular dysfunction may be the most important clinical feature of cisplatin-AKI. There are no currently validated renal tubular dysfunction definitions. Dr Carleton’s team recently proposed a cisplatin nephrotoxicity definition that combines SCr and electrolyte criteria. 52 In order to truly understand the complex cisplatin-AKI phenotype, we think a separate electrolyte-based definition would be useful and allow evaluating SCr-based GFR reduction on its own (KDIGO AKI), electrolyte-based renal tubular dysfunction on it’s own and the composite outcome of SCr rise and electrolyte abnormalities (both outcomes combined). After literature review, we identified a simple nephrotoxicity definition by the National Cancer Institute (NCI) Common Terminology Criteria for Adverse Events, which classify serum electrolyte abnormalities into 5 grades (Table 3). 53 We will define AKI by (1) KDIGO-SCr definition, (2) NCI definition (hypokalemia, hypomagnesemia, or hypophosphatemia [≥NCI Grade 1]), and (3) both.
NCI Common Terminology Criteria for Adverse Events (Version 4.0) for Electrolyte Abnormalities.

Note. NCI = National Cancer Institute; LLN = lower limit of normal for age.
CKD: Long-Term outcome
As per the KDIGO definition of CKD, CKD is defined as an estimated GFR (eGFR) <90 mL/min/1.73 m2) or a urine albumin-to-creatinine ratio ≥3 mg/mmol measured from a random urine sample (Equivalent to ≥Grade 2 CKD, as shown in Tables 4 and 5). CKD is ascertained at each follow-up period described below.
KDIGO GFR Categories in CKD.
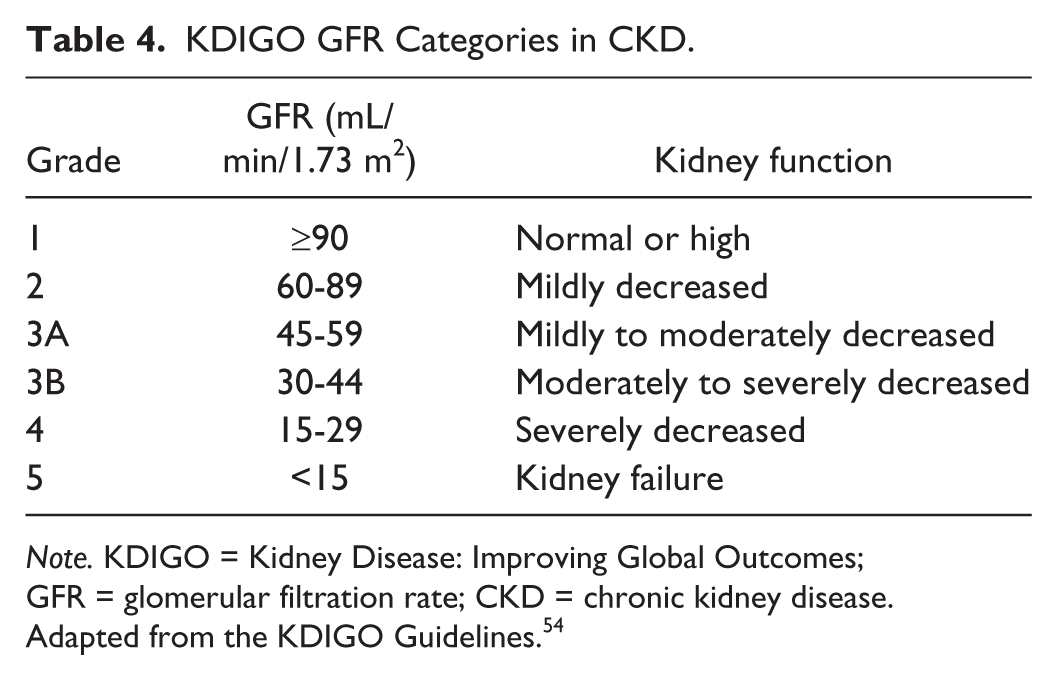
Note. KDIGO = Kidney Disease: Improving Global Outcomes; GFR = glomerular filtration rate; CKD = chronic kidney disease.
Adapted from the KDIGO Guidelines. 54
KDIGO Albuminuria Categories in CKD. 54

Note. KDIGO = Kidney Disease: Improving Global Outcomes; CKD = chronic kidney disease.
GFR is estimated utilizing a combined SCr-and cystatin C-containing equation 55 : GFR (mL/min/1.73 m2) = (507.76 × e0.003 × Height)/(Cystatin C0.635 × SCr0.547[µmol/L]), for highest accuracy.56,57 GFR is also estimated by the updated CKD in Children (CKiD) formula 58 : GFR (mL/min/1.73 m2) = 36.5 × (Height [cm]/SCr [µmol/L]), as currently this equation is most frequently used in pediatric CKD studies.
Chronic renal tubular dysfunction at follow-up visits is defined as need for potassium, magnesium, or phosphate replacement, or any of these electrolytes below lower limit of normal for age.
Hypertension: Long-term outcome
Although hypertension is not a CKD criterion per se, it is a crucial component of CKD evaluation. Hypertension may result from CKD, be an early manifestation of kidney fibrosis or glomerular loss, and is a relevant late AKI outcome in children. Blood pressure is measured using one of the following devices: GE Carescape Dinamap V100, Pro 400 V2, Procare 100, Procare 300 (USA); Welch Allyn Vital Signs 300 series, 6000 series, Connex (USA); or Hadeco Minidop ES-100VX (Japan). For feasibility, site clinic-specific monitors were utilized as follow-up study visits mostly occur during routine clinic visits. Blood pressure is measured 3 times, seated, using age-appropriate cuffs at 1 sitting at each follow-up and is expressed using percentiles and z scores for age, gender, and height. 59 Hypertension is defined as average systolic blood pressure (SBP) and/or diastolic blood pressure (DBP) ≥95th percentile. Child pre-hypertension is defined as average SBP or DBP ≥90th and <95th percentile. Pre-hypertension in adolescents is defined as blood pressure ≥120/80 mmHg (even if <90th percentile). 59 For participants who reach adult age during follow-up, hypertension is blood pressure ≥140/90 mmHg.
Study Time Points: Acute Visits and Follow-Up Visits
Participants undergo 5 study visits (Figure 2).
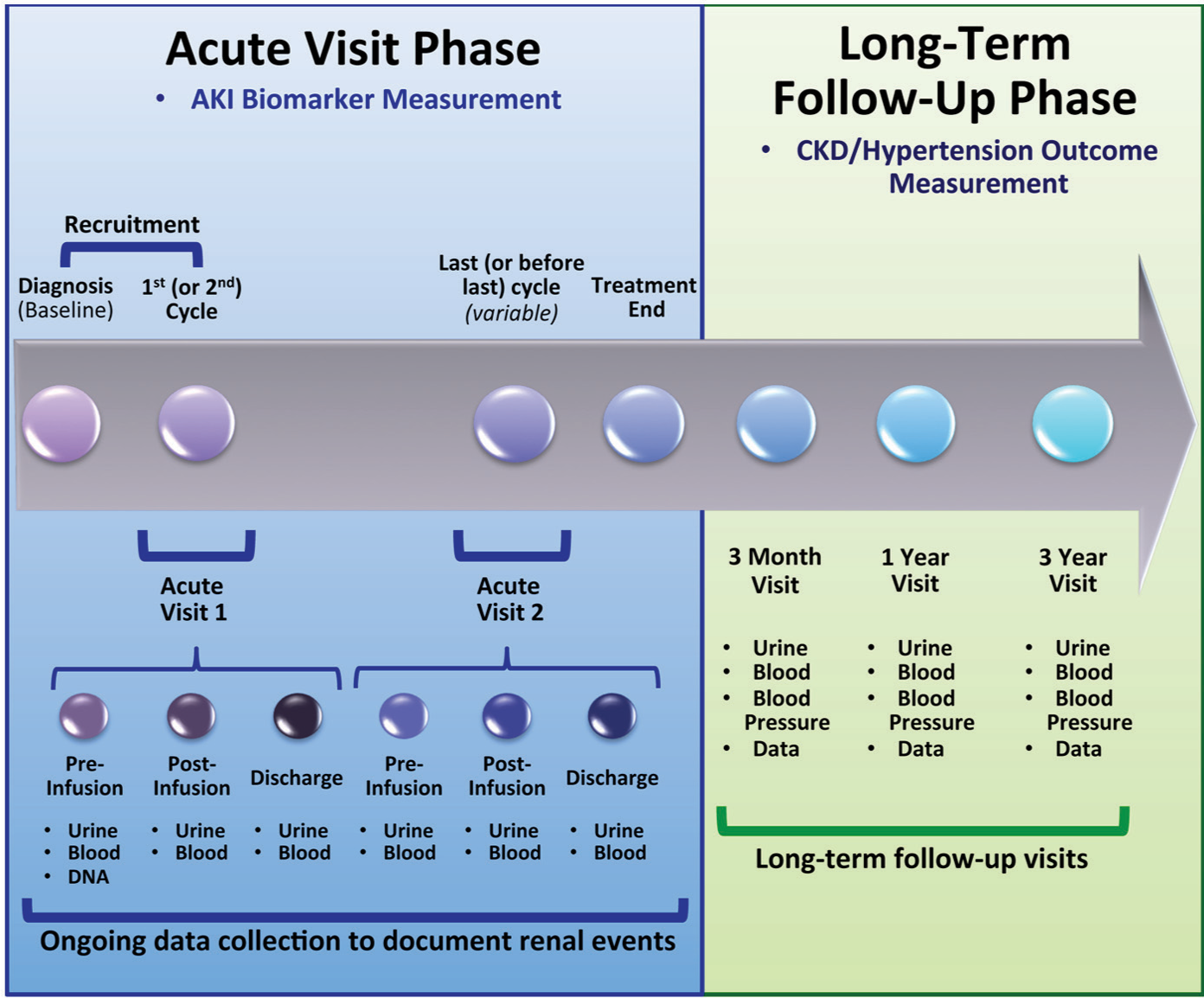
Summary of the ABLE Nephrotoxicity Study Time Points.
Acute visits
The first 2 visits, called acute visits (AVs), occur during 2 separate cisplatin infusion episodes. AV1 occurs at the first (or second) cisplatin cycle; AV2 happens during the last (or before last) cycle. At each AV, blood and urine samples are collected: (1) pre-cisplatin infusion (study day 1), (2) morning after infusion (post-infusion/study day 2), (3) at hospital discharge.
We chose to study participants during 2 infusions for several reasons. First, obtaining urine biomarkers early in cisplatin treatment (ie, AV1) is important to understand baseline biomarker concentrations and kidney function. However, we surmised that later cisplatin cycles might be most likely associated with AKI risk (exposure to risk factors throughout treatment), providing the highest AKI event rates to evaluate cisplatin-AKI biomarkers. Also, 2 separate infusions allow comparing biomarker performance early and later during treatment and evaluating within-subject validity. Multiple pre-post cisplatin time points allow plotting biomarker excretion patterns surrounding nephrotoxic injury.
At each time point (pre-infusion, post-infusion, discharge) at each AV, blood is measured for SCr, cystatin C, magnesium, potassium, and phosphate; urine is measured for creatinine, electrolytes, NGAL, and KIM-1. To ensure standardization, specimen collection kits are prepared by the central site (McGill) and shipped. Saliva is obtained to extract DNA for the pharmacogenetic studies (Ototoxicity study primarily). Large amounts of clinical data are collected during AVs, including sociodemographic and diagnostic data, medical (renal, non-renal)/family history (eg, prior hypertension, treatment with antihypertensives, family history of hypertension, acute dialysis needed, congenital renal problems, urinary tract infection, etc), routinely collected renal imaging/function, nephrotoxins, cisplatin treatment details (eg, total cisplatin dose), renal complications (AKI, abnormal electrolyte levels, acid-base disturbances, need for electrolyte supplements), dialysis requirement, intensive care unit admission, infections. In addition, routine care SCr values are collected throughout treatment to capture any AKI episodes that may have occurred during treatment, outside the AVs of the project; this information is useful when examining the overall effect of AKI episodes during the whole cisplatin therapy on later renal outcomes.
Follow-up visits
Three months, 1 year, and 3 years after cancer treatment end, study visits are performed to document long-term renal outcomes and CKD and hypertension risk factors. Urine (for albuminuria, electrolytes, and creatinine measurement); blood (for SCr, cystatin C, and electrolytes); and clinical data, blood pressure, height, and weight, are collected. Additional data collected include GFR test results performed as part of routine cancer care (eg, Technetium-99m [99mTc]–diethylenetriaminepentaacetic acid (DTPA), 99mTc–mercaptoacetyltriglycine (MAG3), 51 Cr–ethylenediamine tetraacetic acid (EDTA), iohexol GFR, 24-hour creatinine clearance, or others), nephrotoxic medications, cisplatin-specific complications, presence of renal disease, and potential outcome risk factors (eg, sepsis, intensive care unit admission). Urine and serum are banked for potential future studies of chronic renal or other biomarkers.
Data management
We developed CRFs iteratively, obtaining detailed feedback from site investigators and coordinators on feasibility and relevance. After approximately 15 participants were enrolled, final CRF modifications were made. We developed detailed CRF instructions and coordinators receive CRF training, crucial to ensuring multi-site standard data collection. CRFs are sent to EPICORE Centre for data entry into the Research electronic data capture (REDCap) database, constructed by EPICORE Centre with our guidance, 60 with regular study site queries. EPICORE Centre devised an ABLE-specific Web site containing all study documents and where online participant enrollment occurred. 61
The Nephrotoxicity coordinating site also developed an online specimen tracking system using REDCap. 60 Each specimen tube has a unique barcode number, which is scanned using a barcode-reading device and logged into REDCap. Specimen-related activities are monitored in near–real time by the Nephrotoxicity coordinator (from collection to shipment), allowing for regular and immediate quality assurance checks with site feedback, limiting sample losses.
Biospecimen processing and storage
Sites centrifuge blood at 1000 g for 10 minutes at room temperature. Serum is aliquoted and frozen at −80°C. Urine is divided into 2 tubes and stored at −80°C. NGAL and KIM-1 are stable when uncentrifuged urine was frozen at −80°C. 62 Specimens are shipped approximately biannually to McGill for −80°C storage. There, urine is thawed in a water bath and centrifuged 10 minutes at 1000 g. Urine supernatant is aliquoted for measurements and long-term storage. Urine aliquoting occurs in Montreal to reduce site sample manipulations and processing burden, enhancing procedure compliance. Saliva is shipped to Vancouver (Ototoxicity main site).
Laboratory Measurements
NGAL and KIM-1 are measured using validated enzyme-linked immunosorbent assay (ELISA) in duplicate and blinded at Dr Devarajan’s biomarker laboratory (CCHMC).33,35 Serum cystatin C is measured by nephelometry (Prospec II; Siemens Healthcare Diagnostics, Marburg, Germany) at CCHMC. To avoid inter-laboratory variations, all other measurements are performed at the central Nephrotoxicity site (McGill). SCr is measured using isotope dilution mass spectrometry (IDMS)–traceable assay. Genetic analyses are performed at Vancouver’s Canadian Pharmacogenomic Network for Drug Safety Laboratory.
Study Progress
Because of slow recruitment at study start, mainly due to the late initiation of the overall study at larger sites, 4 sites were added to the initial 8 to boost recruitment. For the 4 new sites, follow-up duration was modified to 1 year. However, we plan to use alternative funding to complete 3-year visits.
Analysis, Sample Size, and Power Considerations
Main planned analyses are described in Table 6. Two issues guided our sample size decision: feasibility of recruitment after querying all sites before grant submission and ensuring adequate power for main aims. Moreover, it was difficult to confidently use literature-derived estimates of outcome prevalence as data were variable and largely unavailable. Thus, we assumed that all 150 participants would be available for both AVs and about 10% lost to follow-up by 3 years (n = 135). Loss to follow-up is minimized because almost all Canadian tertiary oncology centers are included, allowing 1 site to perform study visits for another site should participants move cities. This sample size allows estimating a 15% prevalence of each long-term renal outcome (likely an underestimation) with ±6.9% level of precision. For biomarker diagnostic characteristics, assuming 150 patients from the AVs, we will have more than 80% power to estimate 85% sensitivity or specificity with ±8% precision level for NGAL or KIM-1 to diagnose AKI. Our sample size and high-expected AKI event rates will enable comfortably including 6 to 8 variables within regression models. We plan to control for the effects of age, gender, total cisplatin dose, other nephrotoxins (yes/no) during treatment, and pre-cisplatin GFR in multivariate analyses. Other potential variables that we may consider to include in multivariate analyses will be guided by univariate examination of the data.
Summary of the Main Exposures, Outcomes, and Primary Analyses for the 3 Study Aims.

Note. NGAL = neutrophil gelatinase–associated lipocalin; KIM-1 = kidney injury molecule-1; AKI = acute kidney injury; SCr = serum creatinine; KDIGO = Kidney Disease: Improving Global Outcomes; NCI = National Cancer Institute; AUC = area under the curve; CKD = chronic kidney disease; GFR = glomerular filtration rate.
Discussion
The ABLE Nephrotoxicity study is a unique longitudinal study addressing several knowledge gaps in child AKI and late cancer outcomes. This study has several strengths. First, it includes almost all Canadian childhood cancer treatment centers, with positive implications for generalizability and future knowledge translation. Study protocols balance multi-center feasibility with validity and rigor. Our evaluation of specific potential cisplatin-AKI phenotypes will elucidate utility of the current international standard KDIGO-AKI definition, which has not been studied in this population. The choice of biomarkers to evaluate was rational, based on the presence of both human and animal data specific to cisplatin, hopefully increasing the likelihood of being useful cisplatin-AKI tests. If these biomarkers are effective early AKI diagnostic tests, we may pursue the next step of incorporating them in AKI-reducing interventions. These may include pharmacological interventions,63-67 which will likely take time given concerns of testing new drugs in cancer patients; however, interventions may also include AKI-risk reduction, including active avoidance of other nephrotoxic medications, increasing fluid administration, and ultimately active dosage reassessment for future cisplatin infusions. The inclusion of 2 study cisplatin infusions is also a strength. Biomarkers useful at diagnosing AKI during an earlier cisplatin infusion may not necessarily be useful with a later infusion, once there may already be chronic renal damage from past cisplatin infusions. Finally, our specimen collection strategy is highly amenable to new biomarker validation and discovery. We plan on imminently doing discovery work on cisplatin nephrotoxicity biomarkers.
Notwithstanding the exciting potential of novel AKI biomarkers, likely one of the most important aspects of this study is the long-term follow-up. By study completion, we will know what renal measures are abnormal within the first 3 years after cisplatin therapy, when they become apparent and if they improve over time and in what patients (ie, identifying risk factors). This information is currently unknown. Our multi-disciplinary team nature and links with American cancer centers enhance the likelihood of quickly disseminating knowledge gained, and hopefully striking or joining committees that guide late child cancer follow-up to devise rational and specific follow-up strategies.
Our study also has limitations. The multi-center nature, though a strength, also increases possibility of variability in data collection, despite our best efforts. Feasibility played a role in our methodology. For example, after discussions with site investigators before study start, we realized that obtaining urine specimens multiple times per day with specific timing would be particularly difficult. Therefore, early biomarker rises immediately post-cisplatin infusion will not be captured. We also allowed for patients to be recruited before their second cisplatin cycle (as opposed to only cisplatin naïve patients), after discussion with oncology site investigators. There are many challenges to recruiting patients before their first cycle of cisplatin (ie, at cancer diagnosis). In fact, thus far, about 50% of our participants were recruited before their first cycle, the other half, before the second cycle. Although this is a limitation in terms of evaluating biomarkers in cisplatin naïve patients, it is also a strength. The reality is that if biomarkers are to be useful in these patients in the future, they should be useful at all cisplatin infusions, not just the first one. Thus, we will be able to surmise the extent to which biomarker performance differs, depending on whether they are being evaluated in cisplatin naïve, versus non-naïve patients. Ideally, we would measure gold standard GFR methods, collect first morning urine during follow-ups for albuminuria (to reduce risk of postural proteinuria), and perform 24-hour ambulatory blood pressure monitoring (gold standard test for hypertension). However, due to feasibility issues, for the current study, we will be limited to eGFR, random urine for albuminuria, and office BP. Should our results demonstrate a high CKD or hypertension incidence (even in the whole cohort, regardless of AKI associations), this would warrant a 5-year follow-up, wherein we could consider performing gold standard measures of renal outcomes assessment. At a potential 5-year follow-up, we would have the added advantage that children would be older and more likely to be able to perform these gold standard measures (eg, first morning urine, 24-hour ambulatory blood pressure monitoring). Another feasibility-driven issue is the use of site-specific blood pressure machines. Initially, we planned to send the same device to all sites. However, it became apparent that the only way to ensure complete data collection was to incorporate blood pressure measurement within routine clinical care visits. Thus, some concessions to ascertainment were made to ensure data completeness and feasibility. Despite this being one of the largest and most comprehensive pediatric AKI prospective longitudinal follow-up studies, sample size will limit controlling for many potential confounders or effect modifiers of AKI–late outcome associations. We also acknowledge that 3 years is a relatively short follow-up duration. If we find evidence of substantial CKD or hypertension, this will warrant longer follow-up.
Potential future directions of the Nephrotoxicity study include performing genetic, proteomic and metabolomic studies to initiate new AKI and CKD biomarker discoveries and stimulate collaborations to evaluate long-term cardiovascular effects of CKD and hypertension. Moreover, we would like to link with clinical pharmacologists/pharmacists to begin exploring how cisplatin injury biomarkers may be potentially incorporated in determining nephrotoxicity risk and early nephrotoxicity identification within clinical care. Ultimately, we plan to evaluate effects on practice change from knowledge translation interventions and knowledge dissemination and treatment interventions on short-term and long-term renal outcomes.
Footnotes
Acknowledgements
We would like to acknowledge the work of Mary L. McBride, epidemiology core leader, as well as the work performed by all the study site coordinators for data collection, the site investigators not listed in the author list, Drs Eric Bouffet, Sunil Desai, Conrad Fernandez, Adam Fleming, Paul Gibson, Caroline Laverdière, Victor Lewis, Bruno Michon, Raveena Ramphal, Christine Sabapathy, and the study participants and their families.
Ethics Approval and Consent to Participate
Research is conducted according to the World Medical Association Declaration of Helsinki. Written informed consent (or assent) is obtained from research participants. The McGill University Health Centre Pediatrics Research Ethics Board approved the Nephrotoxicity study protocol (12-301-PED).
Consent for Publication
Consent for publication was obtained from all authors.
Availability of Data and Materials
There is no data to share at this stage of the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by a Team Grant from the Canadian Institutes of Health Research. In addition, this work is also supported by a Fonds de recherche du Québec - Santé (FRQS) Doctoral Training Scholarship awarded to Kelly McMahon.
