Abstract
Summary
The COVID19 pandemic highlighted the need for remote diagnosis of cognitive impairment and dementia. Telephone screening for dementia may facilitate prompt diagnosis and optimisation of care. However, it is not clear how accurate telephone screening tools are compared with face-to-face screening. We searched Cochrane, MEDLINE, Embase, Web of Science, PubMed and Scopus for all English language papers published between January 1975 and February 2021 which compared telephone screening for dementia/ mild cognitive impairment and an in-person reference standard, performed within six-weeks. We subsequently searched paper reference lists and contacted authors if data were missing. Three reviewers independently screened studies for inclusion, extracted data, and assessed study quality using an adapted version of the Joanna Briggs Institute's critical appraisal tool. Twenty-one studies including 944 participants were found. No one test appears more accurate, with similar validities as in-person testing. Cut-offs for screening differed between studies based on demographics and acceptability thresholds and meta-analysis was not appropriate. Overall the results suggest telephone screening is acceptably sensitive and specific however, given the limited data, this finding must be treated with some caution. It may not be suitable for those with hearing impairments and anxiety around technology. Few studies were carried out in general practice where most screening occurs and further research is recommended in such lower prevalence environments.
Introduction
The progressive cognitive deterioration associated with dementia restricts an individual's capacity to complete routine tasks and retain novel information, and may cause behavioural change.1,2 Mild cognitive impairment (MCI) presents similar symptoms but, unlike dementia, does not significantly affect activities of daily living (ADL). Approximately half of patients with MCI develop dementia; both can be screened for using the same tests. 1
From 2015 to 2030, dementia cases are projected to increase globally by nearly 60%. 3 Prompt dementia diagnosis provides clarity for patients and the opportunity to optimise care, for example by commencing medications to delay progression or arranging power of attorney to help with financial planning. Despite these benefits, in England, nearly a third (31.3%) of cases are undiagnosed. 4
The COVID-19 pandemic has profoundly impacted dementia diagnosis. In England, diagnostic rates fell from 67.6% (January 2020) to 61.4% (January 2021) in those over 65 years. 5 Similarly secondary care referrals for dementia reduced: presumed reasons being delayed patient help seeking possibly through fear of infection, restructuring of general practice services (primarily offering remote consultation) and healthcare reprioritisation. 6
Screening is one approach to improve the timeliness of dementia diagnosis. Traditionally screening is carried out in a face-to-face general practice consultation using validated tools such as Mini Mental State Examination (MMSE), GPCOG and Montreal Cognitive Assessment (MoCA). However the COVID-19 pandemic has transformed interactions between clinicians and patients, with remote consultation for a time becoming the predominant model in the UK. Remote initial assessment for dementia requires rapid, comprehensive and effective telephone screening tools. Many of these have been available for several decades (e.g. the Telephone Interview for Cognitive Status (TICS) was developed in 1988), 7 but their validity in screening remains uncertain.
Remote consultation is likely to remain a routine part of healthcare beyond the COVID-19 pandemic. Home-based tele-assessment may be convenient, 8 time saving and less anxiety-inducing for patients, and may improve access for some patients where there are geographical, mobility and financial barriers to face-to-face clinic assessment.9,10
Despite these advantages, telephone screening is inappropriate for those without a telephone, or apprehensive using them or with hearing, speech or language problems. 9 Absence of non-verbal cues limits physicians’ capacity to assess wellbeing, comprehension and consider alternative aetiologies, 11 although video consultation may be more informative and personable. 12
Previous systematic reviews recognised that telephone screening has utility in identifying high-risk patients, but inclusion of a broad range of tools in the reviews limited their focus and some could be considered outdated due to literature growth.13–15 One review suggested that despite benefits in mitigating geographical inaccessibility, telephone screening was time-consuming and impacted by informational inconsistencies and patient technological engagement. 8
This review adds to previous reviews, providing appraisal of five telephone tools recommended for identifying dementia patients16–18: Telephone Mini-Mental State Examination (T-MMSE), Telephone Montreal Cognitive Assessment (T-MoCA), Telephone Interview for Cognitive Status (TICS) and modified (TICSm), and Tele-Test-Your-Memory (Tele-TYM).
Objectives
To assess the validity of telephone cognitive screening tools for MCI and dementia, compared to traditional in-person assessments.
Methods
Inclusion/ exclusion criteria
Studies were included if: the titles and abstracts indicated the evaluation of one of the named telephone screening tools (above) including administration within a test battery or of a culturally modified version; and the telephone screen was validated against a named in-person cognitive assessment, within a maximum timescale of six-weeks in a minimum of 95% of patients (to avoid clinically significant cognitive deterioration between assessments). Assessments were limited to adults but not to specific healthcare setting or participant numbers.
Studies requiring particular software, video or virtual reality, or app based were excluded.
Search methods
We searched six databases: Embase, Cochrane, MEDLINE, PubMed, Scopus, and Web of Science, between 31/01/21–02/02/21. This was limited to English language papers published post-1975 (when the MMSE was developed). The same search strategy (Appendix A) was utilised throughout, conducted by the same researcher.
Title/ abstract screening
Titles and abstracts were initially screened by the first author (CR), and a random 20% of papers were considered by a second and third author (KF, BM), there were no discrepancies between reviewers on decisions to exclude.
Full-Text review
Of the papers which met the title/ abstract screening 25% had a full-text review by a second author (KF) to establish whether they continued to meet criteria, there were no discrepancies.
Data extraction
Study data were extracted and tabulated by CR and cross-checked by KF and BM. Where data was missing authors were contacted. The following data was extracted from each study (Table 2):
Author(s), date Sample size (included in study data analysis) Exclusion/ inclusion criteria Sample characteristics: age, educational level and sex (if provided) Telephone screening tool (including maximum score and language adaptations) The reference in-person diagnostic tool Time interval between screening and diagnostic tests Major findings Study limitations
Summary of included studies.

Statistical analysis
Studies were categorised by screening tool. Subsequently their scoring system, baseline characteristics, diagnostic criteria and relevant cognitive condition were compared to minimise clinical baseline and study heterogeneity.
The outcome measures - sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) - were extracted by study. Positive and negative likelihood ratios (LR) were calculated with fixed and random-effects models applied for comparison of heterogeneity. It was determined the studies were too heterogenous for meta-analysis.
Study quality
Quality and risk of bias were examined using an adapted and combined version of the Joanna Briggs Institute's critical appraisal tools. Checklists for diagnostic accuracy and cross-sectional studies were integrated for comprehensive analysis of quality (Appendix B).19,20 Critical analysis was conducted from 06/02/21- 04/03/21, with a quality assessment table produced (Table 1).
Joanna briggs institute quality assessment table.

Results
Search results
The search strategy returned 1371 papers: 327 Embase, 447 Cochrane, 150 MEDLINE, 147 PubMed, 119 Scopus, and 181 Web of Science. Papers were checked for retractions (none). Removing duplicates, 770 studies remained. A further 20 papers were included from the recommendations of experts within Memory Assessment Services, and references from prominent papers.
Subsequently, 790 records’ titles and abstracts were screened.
750 records were then excluded, not fulfilling criteria. Two other authors (KF and BM) checked 20% of these inclusions and exclusions and reported full agreement.
Of the 40 included papers, 25% were also screened by a second examiner (KF) without disputation.
Post full-text examination, a further 19 papers were excluded (Appendix C). Five papers had no in-person comparison test or diagnosis; nine were outside the time limit. Fourteen failed to mention a timescale between assessments; follow-up emails were sent, allowing three weeks to respond, resulting in exclusion of a further nine papers. Five responses were recorded, all meeting the six-week criteria.
In total, 21 papers were included for data-analysis and synthesis.
Quality of included studies
The quality of the 21 studies was appraised using a modified Joanna Briggs Institute checklist (Appendix B); each received a score out of ten (Table 1). Two studies achieved the optimum score of 100%, the lowest being 60%. Eight were deemed to have inappropriate exclusions and consequently high-risk of selection bias. Lack of clarity was also found regarding detection bias, with 11 studies failing to comment on blinding of assessors.
Findings
Of the studies included post-full-text review, three exclusively considered the TICS; six the modified TICS; two both the TICS and TICSm; seven the T-MMSE; one exclusively the T-MoCA; one the Tele-TYM. One study evaluated both the TICS and T-MoCA. 22
The tools were used to distinguish between a range of cognitive conditions. Four studies focussed on Mild Cognitive Impairment, nine on dementia (including Alzheimer's disease) and two considered both diagnoses. Five studies evaluated their use in detecting cognitive impairment post-stroke. One study examined unspecified cognitive impairment (Figure 1).
A mean of 144 individuals participated in each study, the majority recording a mean participant age between 70–75 years old. Female participation was higher than male, and there was high variability in years of education. These three variables are considered confounders, with elderly female uneducated patients being most at-risk of dementia. 42 Patients were recruited from several settings, nine studies enrolling patients from existing research, nine from hospital clinics and three using advertisements. The method for dementia diagnosis also varied. The majority used a well-established standard, such as The Diagnostic and Statistical Manual of Mental Disorders (DSM), or a neuropsychological testing battery. However, a small proportion referenced tests such as the MMSE as a ‘gold standard’, despite more limited accuracy. Follow-up time between screening and diagnostic tests varied, with the maximum falling at six-weeks and the minimum being the same-day; the majority (13 studies) occurred within a fortnight.
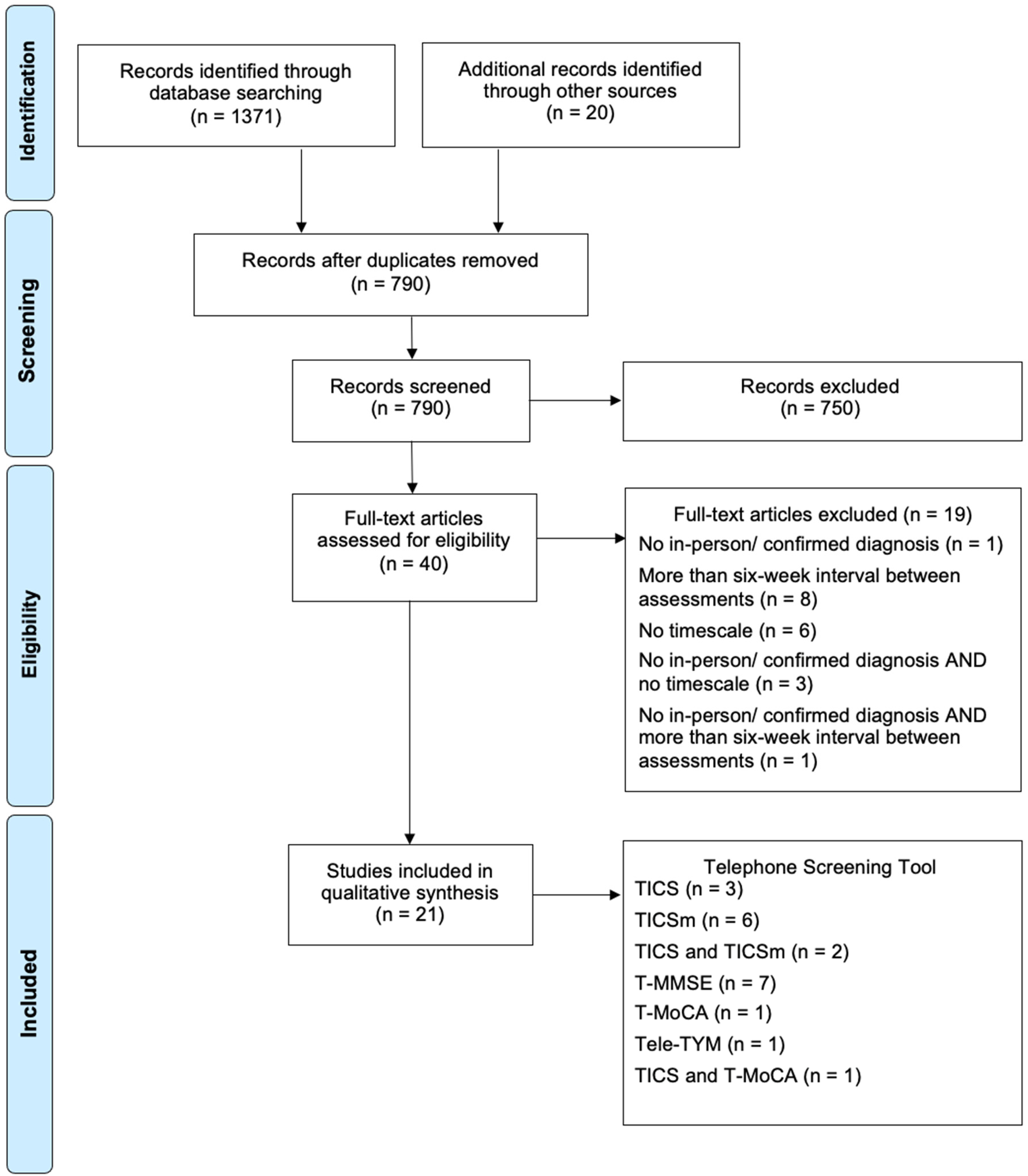
PRISMA flow diagram (Moher
Telephone Interview for Cognitive Status (TICS)
There is high variation between the six studies’ optimal cut-offs for dementia and MCI screening. For example three studies thresholds for dementia were 24/25,
25
28,
29
and 33.
30
The extent of this discrepancy is witnessed through Seo
The TICS has been considered for identifying post-stroke MCI and dementia. Zietemann
Modified Telephone Interview for Cognitive Status (TICSm)
A modified 50-point version of the TICSm was examined by Cook
Other modified versions of the TICS have been examined. Baccaro
Telephone Mini-Mental State Examination (T-MMSE)
There are two versions of the MMSE, the 26-point T-MMSE and the 22-point telephone Adult Lifestyles and Function Interview MMSE (ALFI-MMSE). The difference is the addition of a three-step instruction and recall of the patient's telephone number in the 26-point version. 31
Naharci
The correlation between overall T-MMSE and in-person MMSE scores is significant, exceeding 0.85 in most studies, except Kennedy
Telephone Montreal Cognitive Assessment (T-MoCA)
Two papers examined the T-MoCA, both acknowledging its validity in identifying cognitive impairment post-stroke. They used alternative scoring systems, Wong
Telephone – Test – Your – Memory (Tele-TYM)
Brown
Discussion
Remote screening for dementia or cognitive impairment is established, with approximately twenty cognitive telephone tests available in clinical settings. 43 Telephone screening is considered useful for triage to Memory Assessment Services, particularly for people with access problems due to mobility problems or geographical isolation. This became important during the COVID pandemic to reduce disease transmission.
This review focusses on five tools deemed appropriate for dementia screening in the community, and is the first to consider these assessments in parallel.
Several studies compared the validity of telephone and in-person testing to determine if mode of administration influenced accuracy. Vercambre
There is disagreement regarding whether patients perform better in-person or over the telephone. Newkirk
Roccaforte
Hence, differences in the performance of telephone testing may be influenced by the setting and order of administration, in addition to cognitive status.
Optimal cut-off scores are not exclusively influenced by specific cognitive conditions, but also by population demographics, contextual setting and an institution's organisational structure. 46 A balance must be made between using high cut-offs and thus more false-positives (causing unnecessary distress, patient harm and financial burden), or low cut-offs with more false-negatives (resulting in under-diagnosis with subsequently more severe presentations).
Hearing impairment is of consideration when determining whether allowances in telephone screening cut-off scores should be made, especially where comprehensive issues could usually be gauged through physical cues in-person. Several studies considered whether hearing impairment negatively influences telephone scores, with no general consensus reached. Roccaforte
This review found no definitive hierarchy for the validity of telephone tools. Therefore, other factors such as training, completion time, and patient/ practitioner preferences should be gauged. Telephone assessment is double-edged in regard to patient accessibility. For example, necessity of travel may disadvantage those with mobility issues, but reliance on technological competency and hearing may be challenging for others. Therefore, the offer of telephone screening should be evaluated case-by-case.
Strengths and weaknesses
There were some methodological limitations. The order and timing of tests may influence results for example those studies with fewer than 72 hours between index and reference standard,23,39 risk participant learning-effect bias through memorisation of repeated questions. Of the 21 studies included for qualitative analysis, 15 failed to mention or apply blinding of assessors,22,24–26,28–31,33,35,36,38–41 risking detection bias. Harsh exclusion criteria (e.g. Lines
Home assessments are not standardised, which risks participants ‘cheating’ (e.g. by writing down questions), albeit unintentionally, and thus more false-negative results. Several studies excluded people with hearing problems and language barriers, where telephone consulting is challenging. 47 Lack of in-person contact risks some cognitive domains being unmeasured, such as motor skills, meaning some dementia subtypes, such as Parkinsonian dementia, are not effectively recorded.
The exclusion criteria narrowed the volume of data, in particular the six-week time limit. This decision could have disproportionally impacted larger studies where shorter timescales are unfeasible, such as Manly
Strengths of the review include a standardised search strategy for a range of electronic databases, facilitating reproducibility and accessing a wide scope of material. All papers used for qualitative analysis post-full-screen review were quality assessed, using pre-defined criteria, modelled from reputable Joanna Briggs Institute tools.19,20 Therefore, individual study bias could be determined enabling contextualisation in analysis of results. Cross-validation was conducted by a second or third author at each stage of exclusion, preventing selection bias.
Conclusion
Telephone screening for MCI and dementia appears useful in clinical practice in combination with subsequent diagnostic tests. Compared to existing in-person screening tools, telephone cognitive assessments perform well, with similar validities. Neither modality appears superior at a population level; however, suitability must be considered individually.
Most patients with dementia and cognitive impairment present in family practice and following screening are referred to specialist services. More quantitative research is required, looking at screening accuracy that focuses exclusively on this setting using larger sample sizes, with lower prevalence. Moreover, as patients often present to general practice with milder cognitive impairment, tools that can detect and distinguish these from dementia are important.
Footnotes
Declaration
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix A: Search strategy

| Step in strategy | Search Query |
|---|---|
| 1 | TITLE(Dement* OR “cognitive disorder” OR “cognitive impairment” OR Alzheimer*) |
| 2 | ABSTRACT/TOPIC/TITLE(tele* OR remote OR phone OR virtual) |
| 3 | ABSTRACT/TOPIC/TITLE(TICS OR “telephone interview for cognitive status” OR “MMSE” OR “T-MMSE” OR “mini-mental state exam*” OR “MoCA” OR “T-MoCA” OR “Montreal cognitive” OR “test* batter*” OR TYM OR “test your memory”) |
| 4 | ABSTRACT/TOPIC/TITLE(“virtual reality” OR game OR monitor* OR rehab) |
| 5 | 1 AND 2 AND 3 |
| 6 | 5 NOT 4 |
| 7 | LIMIT 6 TO ENGLISH LANGUAGE |
Appendix B: JBI tool
Consecutive and random sampling are least likely to cause selection bias, studies should record their method of recruitment. This is inclusive of using advertisements whereby the patient then enrols themselves, contacting patients from a pool of participants in an ongoing/ previous study, or including all patients attending a clinic between two stated dates.
Inclusion/ exclusion criteria should be clearly stated, studies failing to meet this will be marked as unclear. Exclusion criteria are deemed acceptable only where there is cognitive impairment of an alternative aetiology for example brain tumour, an inability to communicate for example aphasia, incomplete data, or there is another factor that compromises testing procedures for example lack of a reliable informant if necessary.
Confounders include age, education, gender, and hearing impairment as these variables impact on telephone cognitive screening scores. At least one of these confounders should be mentioned with reference to baseline characteristics.
Confounders can be dealt with through design of studies and in statistical analysis. If there are two groups then confounders can be matched at baseline, statistical tests can also be performed to measure if there is a significant difference between the groups. Regression analysis is another tool for dealing with confounders through measurement of association.
Ideally the reference standard should be considered a ‘gold standard’ or meet established criteria such as DSM, but if not then it should be a reputable tool with high accuracy in determining cognitive diagnoses.
The index and reference standard should be completed by a minimum of 95% of the participants within a timeframe of six-weeks. This is to prevent significant cognitive deterioration between index and reference standard which would render the tests incomparable.
All patients should receive the same reference standard which is used for the comparison with the telephone screening test.
Generally, it is not feasible to blind patients to their diagnosis of cognitive impairment. To minimise detection bias assessors should be blinded to confirmed diagnoses or patient cognitive test scores. Where blinding is not mentioned studies should be marked as unclear.
Generally, it is not feasible to blind patients to their diagnosis of cognitive impairment. To minimise detection bias assessors should be blinded to confirmed diagnoses or patient cognitive test scores. Where blinding is not mentioned studies should be marked as unclear.
Loss to follow-up should be explained, with statistical measures completed to determine impact if >20% of the participants, post first cognitive test, withdraw as this threatens validity. Where studies are vague about losses, or only give partial reasoning, mark as unclear.
Appendix C: Excluded studies
Appendix D: Data Extraction Table
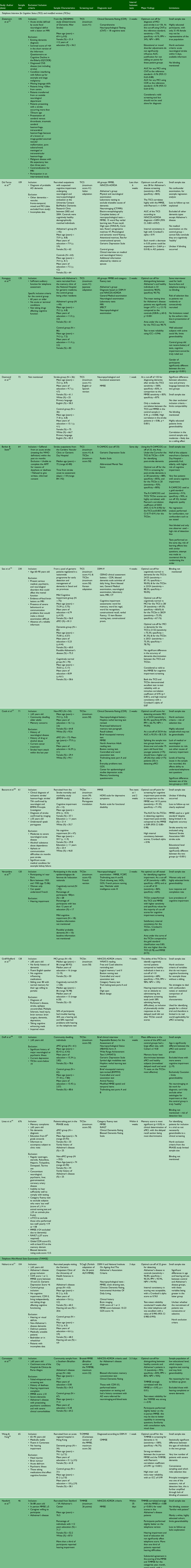
| Study: Author (date) | Sample size | Exclusion/ Inclusion criteria | Sample Characteristics | Screening tool | Diagnostic tool | Interval between tests | Major findings | Limitations |
|---|---|---|---|---|---|---|---|---|
| Zietemann |
105 | Inclusion:
Acute stroke defined by acute focal neurological deficit with a lesion on MRI Existing dementia diagnosis Summed score of >64 in the short version of the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE) Diagnosed CNS disease (not including stroke) Condition interfering with follow-up for example end stage malignancy Missing language skills Patients living >30km from centre Patients transferred from an outside neurological department Patients presenting with a stroke occurring more than 72hours ago Presentation of: cerebral venous thrombosis, traumatic cerebral haemorrhage, intracerebral haemorrhage because of a known or image-guided assumed vascular malformation, pure subarachnoid, meningeal or intraventricular haemorrhage Malignant disease with life expectancy less than three years Contraindication for MRI Participation in an interventional study |
From the DEDEMAS study (Determinants of Dementia After Stroke) |
TICS (maximum score 41) | Clinical Dementia Rating (CDR) |
2 weeks | Optimum cut off for diagnosis of MCI post-stroke is at <36. At this cut-off using CNT as the reference standard, sensitivity = 73%; specificity = 61%; PPV = 34%, NPV = 89% |
Small sample size |
| Dal Forno |
109 | Inclusion:
Diagnosis of probable AD dementia Other dementias – vascular, fronto-temporal, mixed and MCI (data excluded from study) Incomplete data |
Recruited outpatients with suspected cognitive impairment undergoing neurological and neuro-psychological evaluation at the University Campus BioMedico Dementia Research Clinic between 2002 and 2004. Controls were cognitively healthy demographically matched individuals. |
TICS (maximum score 41) |
All groups: MMSE, NINCDS-ADRDA |
Less than 6 weeks | Optimum cut-off score was 28 for Alzheimer's disease screening (sensitivity = 84%; specificity = 86%) |
Small sample size |
| Konagaya |
135 | Inclusion:
Sufficient auditory function for telephone assessment 60 years or older No acute or terminal conditions Not taking drugs affecting cognitive function |
Patients with Alzheimer's were recruited from the memory clinic of the National Hospital for geriatric medicine, the control group being urban residents |
TICS (maximum score 41) |
All groups: MMSE and category fluency test |
2 weeks | Optimal cut off for distinguishing between Alzheimer's and healthy individuals is 33 (sensitivity 98.0%; specificity 90.7%) |
Same interviewer used for both face-to-face and telephone testing – no blinding |
| Desmond |
72 | Not mentioned | Stroke group (N = 36): |
TICS (maximum score 41) |
Neuropsychological and functional assessment |
1 week | At a cut-off of <25 for diagnosing dementia after stroke the TICS sensitivity = 100%; specificity = 83%, in comparison to the MMSE sensitivity = 83%; specificity = 87% |
Significant difference between education, race and primary language between the two groups |
| Barber & Stott 39 | 64 | Inclusion: • Suffered from an acute stroke (meeting the WHO definition) within the past six months Exclusion • Unable to complete the AMT for reasons of dysphasia or deafness • Refused to give written, informed consent | Stroke outpatients from the Cerebro Vascular Clinic or Geriatric Day Hospital |
TICS (maximum score 41) & TICSm (maximum score 50) | R-CAMOG (cut off 33) |
Same day | Using the R-CAMCOG cut off of 33, the Area Under the Curve for the TICS & TICSm = 0.94 for identifying post-stroke dementia |
Small sample size |
| Seo |
230 | Inclusion:
Age 60–90 years old Present serious medical, psychiatric, and neurological disorders that could affect the mental function Evidence of focal brain lesions on MRI Presence of severe behavioural or communication problems that would make a clinical examination difficult Absence of a reliable informant |
From a pool of geriatric patients registered in a nationwide programme for early detection and management of dementia in Seoul and six provinces |
TICS (maximum score 41) & TICSm (maximum score 50) |
DSM-IV |
4 weeks | Optimal cut-off for cognitively normal vs dementia for the TICS is 24/25 (sensitivity = 87.1%; specificity = 90.0%) & for the TICSm is 23/24 (sensitivity = 88.2%; specificity = 90.0%) |
Blinding not mentioned |
| Cook |
71 | Inclusion:
≥65 years old Community dwelling older adults Memory concerns History of neurological disease History of drug or alcohol abuse Current cancer treatment Stroke/ heart attack within the last year |
Non-MCI (N = 54): |
TICSm (maximum score 50) | Clinical Dementia Rating (CDR) |
2 weeks | Optimal cut-off score for detecting amnestic MCI is at 33/34 (sensitivity = 82.4%; specificity 87.0%; PPV = 66.7%; NPV = 94.0%) |
Small sample size |
| Baccaro |
61 | Inclusion:
Clinical diagnosis of ischaemic stroke/ haemorrhagic stroke/ TIA confirmed by neurologist and EMMA Principle Investigator Diagnosis of stroke confirmed by imaging ≥35 years old Understand/ speak portuguese Moderate to severe neurological acute disease Alcohol/ substance abuse dependence Aphasia or communication difficulties six months post stroke Significant acute medical condition for example kidney failure |
Recruited from the Stroke mortality and morbidity study (EMMA study) |
TICSm (maximum score 39) |
MMSE |
Two tests at 1 and 2 weeks | Optimal cut-off point for screening for cognitive impairment post-stroke in comparison to the MMSE was 14/15 points (sensitivity = 91.5%; specificity = 71.4%) |
Small sample size |
| Vercambre |
120 | Inclusion
Participating in/ near Paris Born between 1925 and 1930 (age 72-86) Women only Can speak/ understand French Hearing impairment |
Participating in the etude epidemiologique de femmes de l’education nationale |
TICSm (maximum score 43) |
Neuropsychological examination - MMSE, FCSRT, Trailmaking test A and B, Clock drawing test, IADL, GDS, French picture naming test, Wechsler adult intelligence scale III | 2 weeks | No optimal cut off stated for identifying cognitive impairment. At a cut-off of 30 (sensitivity = 68%; specificity = 89%) and at a cut-off of 33 (sensitivity = 86%; specificity = 60%) |
Small sample size |
| Graff-Radford |
128 | Inclusion:
≥85 years old No family history of dementia Fluent English speaker No cognitive influencing medications Sibling over 80 with normal memory for their age willing to partake Diagnosis of Parkinson's disease, stroke, epilepsy uncontrolled, Multiple Sclerosis, head injury, brain tumour, brain surgery, dementia, depression, Taking cognitive enhancing meds Impaired vision |
MCI group (N = 8): |
TICSm (maximum score 50) | NINCDS-ADRDA criteria |
4 weeks | The ability of the TICSm to identify cognitively normal patients compared to healthy individuals is best at a cut-off of <31 (sensitivity = 68%; specificity = 75%; PPV = 98%; NPV = 13%) |
Small sample size |
| Duff |
123 | Inclusion:
≥65 years old Significant history of major neurological or psychiatric illness Current depression TICSm score below 19 |
Community dwelling individuals |
TICSm (maximum score 50) | Brief clinical interview |
1-3 weeks | Mean difference in the scores of the aMCI and control groups had a large effect size, with Cohen's d >0.8 |
Small sample |
| Lines |
676 | Inclusion
Memory complaints ≥65 years old No dementia diagnosis Education from 8th grade onwards Informant to accompany subject to clinic visits Regular oestrogen, steroids, Raloxifene, Heparin, Ticlopidine, Donepezil, Tacrine use Concomitant neurological, psychiatric, liver, gastrointestinal, coronary artery disease Inability to hear sufficiently well to comply with testing Category fluency test to exclude subjects who were ‘too well’ (score of ≥14 in animal naming test and ≥25 on animals plus fruits) mTICS to exclude those who performed too well/ poorly <19 or >38 MMSE (<24 excluded due to dementia) RAVLT (≤37 score required) Global CDR score 0.5, with at least 0.5 on the memory domain Blessed dementia rating scale score >3.5 |
Patients taking part in the PRAISE study |
TICSm (maximum score 50) | Category fluency test |
Within 6 weeks | Memory score is most significant (p < 0.05) in clinical determination of aMCI, with the delayed recall section being most discriminative |
For identifying patients for inclusion in a trial so not necessarily generalisable to a clinical screening |
| Naharci |
104 | Inclusion:
≥65 years old Alzheimer's disease group inclusion: CDR score 1 or 2, MMSE score between 10 and 23, Geriatric Depression Score <6 Control group inclusion: No cognitive impairment, CDR 0, living independently, not taking drugs affecting cognitive functioning Hearing or visual deficits Non-Alzheimer's disease dementia Delirium patients Medically unstable patients Bedridden or in wheelchair Patients with incomplete data |
Recruited people visiting the Geriatric Outpatient Clinic of the University of Health Sciences in Ankara |
T-CogS (Turkish adaptation of the 26 point ALFI-MMSE) | DSM-V and National Institute On Ageing And The Alzheimer's Association Criteria |
3 days | Optimal cut-off of 22 given for detecting Alzheimer's disease vs controls (sensitivity = 96.8%; specificity = 90.2%; PPV = 93.9%, NPV = 94.9%) |
Small sample size |
| Camozzato |
133 | Inclusion:
≥60 years old Catchment area of the Hospital de Clinicas de Porto Alegre Failed whispered voice screening test History of deafness Hearing impairment complaint MMSE score ≤10 Severe dementia Alzheimer's disease with preexisting psychiatric conditions and with severe clinical comorbidities |
Community sample from a Southern Brazilian City |
Braztel-MMSE (Brazilian version of the T-MMSE, maximum score 22) | NINCDS-ADRDA criteria used for Alzheimer's disease diagnosis |
2-3 days | Optimal cut-off for distinguishing between healthy controls and Alzheimer's patients is 15 (sensitivity = 95%; specificity = 84%; PPV = 85%; NPV = 93%) |
Sample population of low educational level, which impacts cognitive test scoring and may limit generalisability |
| Wong & Fong 26 | 65 | Inclusion:
65–95 years old Medically stable Fluent in Cantonese No hearing impairment Head injuries Brain tumour Acute delirium Psychiatric illness Those taking medications that affect cognitive function |
Recruited from an acute regional hospital in Hong Kong |
T-CMMSE (Cantonese version of the MMSE, maximum score 26) | Diagnosed according to DSM-IV |
1 week | Optimal cut-off for the T-MMSE in screening for dementia is 16 (sensitivity = 100%; specificity = 96.7%) |
Small sample size |
| Newkirk |
46 | Inclusion:
56-88 years old In person MMSE ≥5 Caregiver willing to participate Alzheimer's disease |
Recruited from Stanford / VA Alzheimer's Center |
T-MMSE (maximum 26 points) | NINCDS-ADRDA criteria |
Within 35 days | TMMSE correlated strongly with the MMSE (r = 0.88 p < 0.001) for total scores in the Alzheimer's disease patients |
Small sample size |
| Metitieri |
104 | Exclusion:
Deafness Severe aphasia Acute conditions Advanced dementia |
Recruited from the Alzheimer's unit at the IRCCS San Giovanni de Dio |
Itel-MMSE (Italian version of the T-MMSE, maximum score 22) | Known diagnosis |
1 week | Inter-rater reliability high = 0.82–0.90 |
Small sample size |
| Roccaforte |
100 | Inclusion:
Community based patients |
Recruited by attendance to the University of Nebraska Geriatric Assessment centre |
ALFI-MMSE (maximum 22 points) | DSMIII |
Mean of 8.7 days | No significant differences in the scores of the 22 items of the in-person and T-MMSE, with a trend towards higher in-clinic scores |
Small sample size |
| Kennedy |
402 | Inclusion:
African American and non-Hispanic white adults ≥75 years old Living independently in the community Able to schedule study appointments and answer questions by themselves |
Participants of the University of Alabama Birmingham Study of Ageing II |
T-MMSE (maximum 22 points) | Diagnosis in medical records/ patient has been told they have dementia |
(95%) within 6 weeks | For the 22 common questions in the T-MMSE and the MMSE there was 0.688 (p < 0.001) correlation in scores |
Process of identifying dementia may lead to under-reporting (especially in milder forms) |
| Zietemann |
105 | Inclusion:
Acute stroke defined by acute focal neurological deficit with a lesion on MRI Existing dementia diagnosis Summed score of >64 in the short version of the Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE) Diagnosed CNS disease (not including stroke) Condition interfering with follow-up for example end stage malignancy Missing language skills Patients living >30km from centre Patients transferred from an outside neurological department Patients presenting with a stroke occurring more than 72 hours ago Presentation of: cerebral venous thrombosis, traumatic cerebral haemorrhage, intracerebral haemorrhage because of a known or image-guided assumed vascular malformation, pure subarachnoid, meningeal or intraventricular haemorrhage Malignant disease with life expectancy <3 years Contraindication for MRI Participation in an interventional study |
From the DEDEMAS study (Determinants of Dementia After Stroke) |
T-MoCA (maximum score 22) | Clinical Dementia Rating (CDR) |
2 weeks | Optimum cut off for diagnosis of MCI post-stroke is at <19. At this cut-off using CNT as the reference standard, sensitivity = 81%; specificity = 73%; PPV = 45%, NPV = 94% |
Small sample size |
| Wong |
104 | Inclusion:
Stroke/ TIA patients Pre-stroke dementia Moderate to severe dementia patients (CDR ≥2) Inadequate sensorimotor and language capacity |
Participants from the STRIDE study |
T-MoCA 5 min protocol (maximum score 30) Cantonese version | MMSE |
4 weeks | Scores were on average 1.8 points higher on the T-MoCA 5 min protocol than on the in-person MoCA |
Small sample size |
| Brown |
81 | Exclusion:
Parkinson's disease History of stroke Epilepsy |
Recruited patients due to be seen at a specialised memory clinic |
Tele-TYM (maximum score 50) | Addenbrooke's cognitive examination (ACE-R) |
2 weeks | Optimal cut-off at ≥43 for screening for cognitive impairment (sensitivity = 78%; specificity = 69%) |
Unclear recruitment strategy |
